Upper Extremity Burn & Contracture Reconstruction: An Intraoperative Masterclass
Key Takeaway
This masterclass guides you through acute and reconstructive surgical techniques for upper extremity thermal and electrical injuries. We'll meticulously cover comprehensive anatomy, preoperative planning, real-time intraoperative execution of escharotomy, fasciotomy, and complex tissue rearrangements like the "jumping man" Z-plasty. Emphasis is placed on neurovascular preservation, managing compartment syndrome, and optimizing functional outcomes for these challenging cases.
Comprehensive Introduction and Patho-Epidemiology
Welcome to the operating theater, colleagues. Today, we confront one of the most demanding and critical arenas in orthopaedic and reconstructive surgery: the acute management and secondary reconstruction of thermal and electrical injuries to the upper extremity. These devastating injuries transcend mere cutaneous defects; they represent a profound challenge to the intricate form and function of the hand and forearm. If mismanaged, they lead inexorably to debilitating contractures, neurovascular compromise, and the permanent loss of prehension. Our overarching objective is the meticulous restoration of vital hand and forearm mechanics, aggressively intercepting the tragic sequelae of untreated or undertreated burn injuries.
Before we contemplate our first incision, we must cultivate a profound understanding of the underlying pathogenesis. Burns and electrical injuries to the upper extremity present a vast spectrum of surgical pathology, ranging from acute, limb-threatening compartment syndromes to chronic, rigid, and disabling contractures. The epidemiology of these injuries skews toward working-age adults, often occurring in industrial settings, which underscores the profound socioeconomic impact of functional loss. High-voltage electrical injuries, in particular, carry a disproportionately high rate of major amputation and permanent disability compared to isolated thermal burns, necessitating a highly specialized, multidisciplinary approach to limb salvage.
Electrical Burns: The Hidden Damage
High-voltage electrical injury, classically defined as involving a power source exceeding 1,000 volts (though some literature cites >600 volts), is uniquely insidious. Unlike pure thermal burns where the zone of coagulation is readily apparent on the skin surface, the visible cutaneous injury in an electrical burn frequently belies massive, "hidden" deep tissue destruction. This deep myonecrosis occurs primarily through two distinct but synergistic mechanisms: Joule heating and electroporation. As electrical current traverses the upper extremity—often entering through the hand and exiting elsewhere—tissue resistance generates intense heat, causing direct thermal denaturation of proteins along the current's path. Tissues with high electrical resistance, notably skin and bone, generate the most heat. Consequently, the periosteal blood supply is often obliterated, and the deep musculature immediately adjacent to the bone suffers the most profound thermal destruction, a phenomenon known as the "core-heating" effect.
Simultaneously, the tissues are subjected to electroporation, a phenomenon characterized by cellular membrane damage induced directly by the extreme electric field. The severity of electroporation is heavily influenced by the target cell's size and its resting transmembrane potential. Myocytes and neural tissues, possessing large surface areas and complex ion channels, are exquisitely vulnerable to this non-thermal injury. Furthermore, myocytes situated near the diaphysis of long bones experience amplified transmembrane potentials due to architectural and field-orientation factors, exacerbating deep muscle necrosis. This lethal combination of periosseous thermal injury and widespread electroporation rapidly produces extensive deep tissue edema and muscle necrosis, setting the stage for a fulminant acute compartment syndrome.
Thermal Burns and the Pathogenesis of Contractures
Thermal burns, particularly those that are circumferential or near-circumferential and full-thickness, precipitate acute compartment syndrome through a mechanical restriction mechanism. The deeply burned, denatured dermis transforms into an inelastic, leathery eschar. As the underlying soft tissues succumb to massive obligatory edema—driven by the systemic inflammatory response and aggressive fluid resuscitation—this eschar acts as a rigid tourniquet. It rapidly compromises venous outflow, subsequently obliterates capillary perfusion, and ultimately halts arterial inflow, leading to distal tissue ischemia. The inelasticity of the eschar means that compartment syndrome can occur even without the involvement of the deep fascial envelopes.
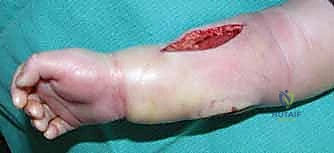
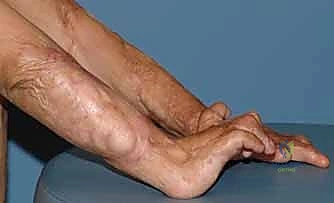
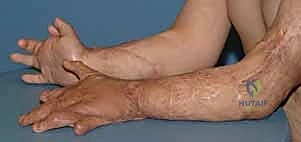
In the long term, both thermal and electrical injuries frequently culminate in severe, rigid contractures. The normal, highly organized, and pliable dermal architecture is replaced by dense, hypertrophic scar tissue. This process involves the excessive and disorganized deposition of type I and type III collagen fibers, which cross-link and actively contract via myofibroblast activity. During the acute and subacute phases, intense inflammation, pain, and persistent edema encourage the patient to hold the limb in the "position of comfort." Unfortunately, in the hand, the position of comfort is invariably the "position of deformity": wrist flexion, metacarpophalangeal (MCP) joint extension, proximal interphalangeal (PIP) joint flexion, and thumb adduction. This rapid soft-tissue shortening produces the classic "claw hand" deformity, effectively destroying the hand's functional arches and prehensile capabilities despite aggressive acute care and therapy.
Detailed Surgical Anatomy and Biomechanics
Mastery of the intricate, constrained anatomy of the forearm and hand is the absolute prerequisite for safe and effective surgical intervention in these patients. The upper extremity is composed of tightly bound fascial spaces; any pathological increase in volume—whether from burn edema, hematoma, or myonecrosis—will rapidly compromise the enclosed neurovascular structures. Surgical release must be anatomically precise to decompress these spaces without causing iatrogenic injury to the very structures we are attempting to salvage.
Forearm Compartments and Neurovascular Topography
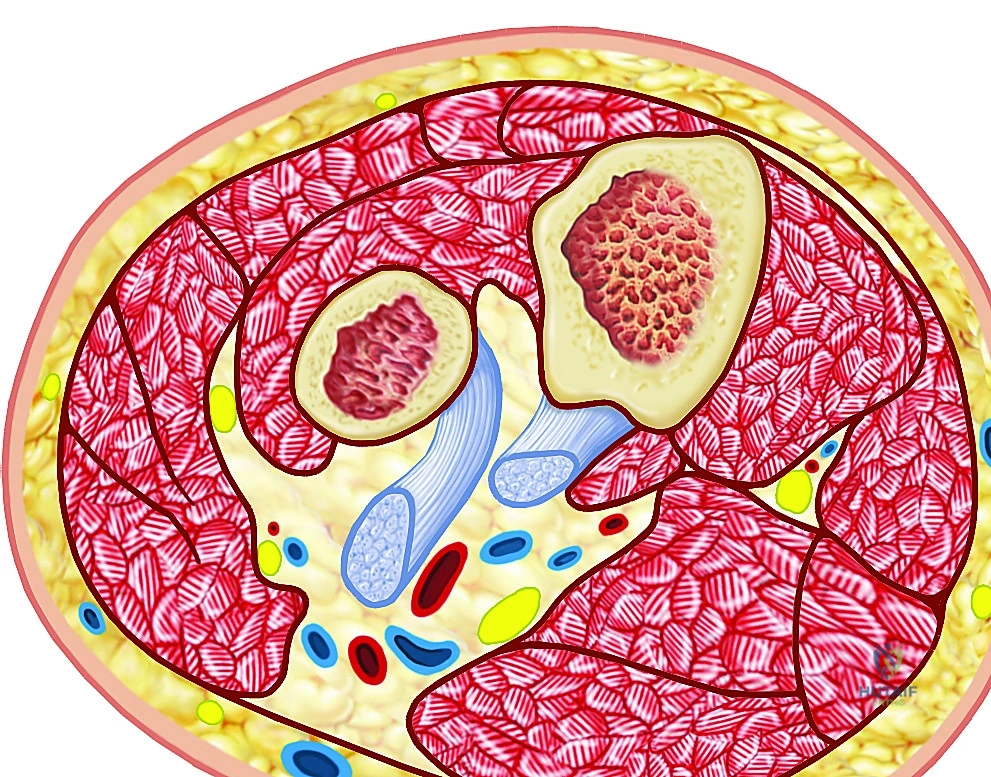
The forearm is meticulously partitioned by the antebrachial fascia and intermuscular septa into three primary compartments, each harboring specific muscle groups and critical neurovascular bundles. The volar compartment is the largest, most complex, and most frequently implicated in compartment syndrome. It is subdivided into superficial and deep spaces. The superficial muscles (pronator teres, flexor carpi radialis, palmaris longus, flexor digitorum superficialis, flexor carpi ulnaris) and deep muscles (flexor digitorum profundus, flexor pollicis longus, pronator quadratus) are tightly packed. The median nerve is highly vulnerable here, running between the FDS and FDP proximally before emerging superficially. The ulnar nerve and artery travel medial to the FCU. Ischemia in this compartment leads to catastrophic loss of digital flexion and intrinsic hand function.
The dorsal compartment houses the extensor musculature (extensor digitorum, extensor digiti minimi, extensor carpi ulnaris, and the deep outcropping muscles of the thumb). The posterior interosseous nerve (PIN), a branch of the radial nerve, courses through the supinator to innervate these structures. While less commonly the primary site of compartment syndrome compared to the volar aspect, the dorsal compartment must be routinely evaluated and released, as isolated dorsal compartment syndrome can cause irreversible loss of finger and thumb extension.
The mobile wad, or lateral compartment, comprises the brachioradialis, extensor carpi radialis longus, and extensor carpi radialis brevis. The superficial sensory branch of the radial nerve runs deep to the brachioradialis. This compartment is often decompressed simultaneously with the volar or dorsal compartments, but its distinct fascial envelope means it can harbor isolated elevated pressures, particularly in localized electrical contact injuries.

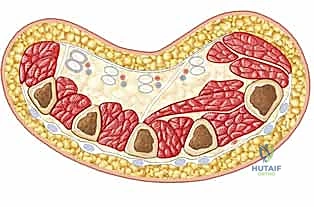
Intrinsic Compartments of the Hand
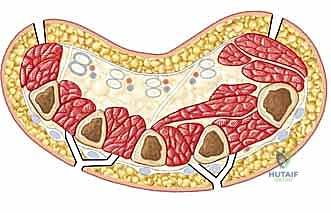
The hand itself is a highly compartmentalized structure, divided into ten distinct intrinsic fascial spaces that must be individually addressed during a thorough fasciotomy. There are four dorsal interosseous compartments and three volar interosseous compartments situated between the metacarpals. These house the interossei muscles, which are vital for digital abduction and adduction, and are innervated by the deep branch of the ulnar nerve. Ischemia here leads to devastating intrinsic-minus deformities.
The thenar compartment contains the abductor pollicis brevis, flexor pollicis brevis, and opponens pollicis, primarily innervated by the recurrent motor branch of the median nerve. The hypothenar compartment houses the abductor, flexor, and opponens digiti minimi, supplied by the ulnar nerve. Finally, the adductor compartment, containing the adductor pollicis, lies deep in the palm. Failure to decompress any of these ten compartments during acute burn management will result in localized muscle necrosis and subsequent severe, localized contractures that are notoriously difficult to reconstruct.
Biomechanics of Burn Contractures
The biomechanics of burn contractures in the hand are dictated by the imbalance of forces acting across the joints once the skin and fascial envelopes lose their elasticity. In a healthy hand, the intrinsic muscles (lumbricals and interossei) flex the MCP joints and extend the IP joints. Following severe burns, dorsal hand edema forces the MCP joints into hyperextension. If the intrinsic muscles become ischemic or if dorsal scar tissue contracts, the collateral ligaments of the MCP joints shorten in this extended position.
Simultaneously, the PIP joints are drawn into flexion due to the mechanical advantage of the extrinsic flexors over the weakened or compromised extensor mechanism. This results in the classic intrinsic-minus or claw hand. Furthermore, contracture of the first web space adductor musculature and overlying skin obliterates the thumb's ability to oppose, reducing the hand to a non-functional hook. Understanding these deforming forces is critical for both acute splinting (which must enforce the intrinsic-plus position: MCPs flexed to 70-90 degrees, IP joints fully extended, thumb abducted) and for planning secondary reconstructive releases.
Exhaustive Indications and Contraindications
Surgical intervention in the burned upper extremity is broadly divided into acute, limb-saving procedures (escharotomy and fasciotomy) and delayed, function-restoring procedures (contracture release and reconstruction). The decision-making process requires a nuanced understanding of tissue viability, systemic patient stability, and precise timing.
Acute Phase Interventions
The absolute indication for acute surgical intervention is the clinical diagnosis of compartment syndrome or impending vascular compromise due to a circumferential eschar. In the setting of severe burns, prophylactic escharotomies are often indicated for circumferential deep partial-thickness or full-thickness burns of the extremity to prevent the mechanical tourniquet effect. Fasciotomies are strictly indicated when intracompartmental pressures exceed 30 mmHg, or when the delta pressure (diastolic blood pressure minus compartment pressure) falls below 30 mmHg. In high-voltage electrical injuries, deep fasciotomy is almost universally indicated due to the high likelihood of hidden myonecrosis, regardless of initial surface pressure readings.
Contraindications in the acute phase are exceedingly rare, as failure to release a compartment syndrome inevitably leads to limb loss or permanent severe dysfunction (Volkmann's ischemic contracture). However, relative contraindications include profound systemic hemodynamic instability where the patient cannot tolerate anesthesia or operative blood loss, though this is often managed by performing rapid bedside escharotomies under local anesthesia or light sedation.
Chronic Phase Interventions
Indications for secondary contracture reconstruction include functionally limiting scar contractures that have failed conservative management (aggressive splinting, pressure garments, and serial casting). Surgery is typically deferred until the scar has matured—often 6 to 12 months post-injury—when the scar is soft, pliable, and no longer actively hyperemic. Early intervention may be indicated for severe contractures causing joint subluxation, progressive neurological deficits (e.g., severe carpal tunnel syndrome secondary to volar scarring), or exposure of vital structures.
Contraindications for secondary reconstruction include active, immature, and highly vascular scars, which are prone to massive intraoperative bleeding and immediate postoperative recurrence. Patients with severe, uncorrectable systemic comorbidities, ongoing psychiatric instability precluding postoperative rehabilitation compliance, or limbs with such profound neurological destruction that releasing the contracture will not yield a functional gain, are poor candidates for extensive reconstructive efforts.
| Parameter | Acute Escharotomy/Fasciotomy | Secondary Contracture Reconstruction |
|---|---|---|
| Absolute Indications | Compartment syndrome ($\Delta$P < 30 mmHg); Absent distal pulses (late sign); Circumferential full-thickness burn with vascular compromise. | Functionally limiting contracture; Joint subluxation/dislocation due to scar; Nerve compression secondary to scar. |
| Relative Indications | High-voltage electrical injury (>1000V) with any extremity involvement; Progressive pain on passive stretch. | Failure of conservative therapy (splinting/garments); Need to improve hygiene in severe flexion contractures. |
| Absolute Contraindications | None (life/limb-saving procedure). | Active local infection; Non-viable distal limb (requires amputation). |
| Relative Contraindications | Extreme hemodynamic instability (perform bedside); Irreversible ischemia (>12-24 hours) where release may cause fatal reperfusion injury. | Immature, hyperemic scar (<6 months post-injury); Patient inability to comply with rigorous postoperative therapy. |
Pre-Operative Planning, Templating, and Patient Positioning
Preoperative planning in the burned upper extremity is an exercise in anticipating disaster and preparing multiple reconstructive lifeboats. We leave absolutely nothing to chance, recognizing that the intraoperative findings, particularly in electrical burns, often dictate a radical departure from the initial surgical plan.
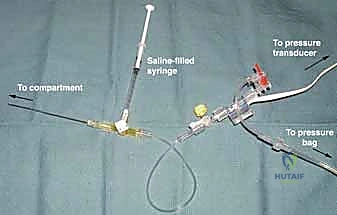
Patient Assessment and Systemic Resuscitation
For acute burn injuries, particularly those involving high voltage or a large total body surface area (TBSA), the orthopaedic surgeon must work in lockstep with the trauma and burn critical care teams. The Advanced Trauma Life Support (ATLS) and Advanced Burn Life Support (ABLS) protocols are paramount. Aggressive crystalloid fluid resuscitation, often guided by the Parkland formula or modern variations, is initiated. The surgeon must be acutely aware that this massive fluid influx will rapidly exacerbate third-spacing and soft-tissue edema, dramatically increasing the risk of compartment syndrome within the first 12 to 36 hours.
A meticulous history regarding the mechanism of injury is vital. High-voltage injuries, enclosed space fires (inhalation injury risk), and blast components all alter the surgical calculus. Because extensive burn debridements and fasciotomies can incur massive, rapid blood loss, preoperative optimization must include ensuring that adequate blood products (packed red blood cells, fresh frozen plasma) are cross-matched and physically present in the operating room prior to incision.
Clinical Examination: The Cornerstone of Diagnosis
In the acute setting, clinical judgment supersedes all other diagnostic modalities. The hallmark of acute compartment syndrome is pain out of proportion to the apparent injury, specifically progressive, unrelenting pain that is exacerbated by passive stretch of the involved muscle groups. This is the earliest and most sensitive indicator of muscle ischemia. For example, severe pain upon passive extension of the fingers strongly suggests volar forearm compartment ischemia.
Neurological deficits, such as diminished two-point discrimination or paresthesias, are later signs. Crucially, the absence of a distal pulse is an end-stage finding; waiting for pulselessness before intervening guarantees irreversible muscle death. The compartments will feel tense, non-compressible, and "wood-like" to palpation. If the clinical picture is equivocal, particularly in an intubated or obtunded patient, invasive compartment pressure monitoring using an arterial line transducer or a handheld manometer is mandatory.
Positioning and Preparation
For both acute and chronic procedures, the patient is positioned supine with the affected extremity extended on a radiolucent hand table. A pneumatic tourniquet is applied to the proximal arm. In the acute setting of impending compartment syndrome, the use of a tourniquet is highly controversial; if used, it should be inflated only for the briefest possible time to control massive hemorrhage, as it exacerbates ischemia. In chronic contracture reconstruction, the tourniquet is essential for maintaining a bloodless field to identify delicate neurovascular structures encased in dense scar tissue. The entire upper extremity, from the fingertips to the axilla, is prepped and draped to allow for extensive extensile exposures and the harvesting of local or regional flaps if necessary.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution must be methodical, anatomical, and uncompromising. We will divide this masterclass into the acute decompression phase and the chronic reconstructive phase.
Acute Phase: Escharotomy and Fasciotomy


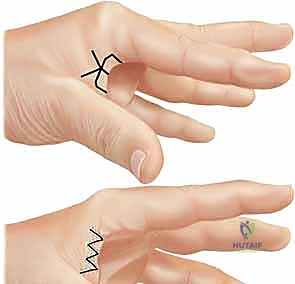
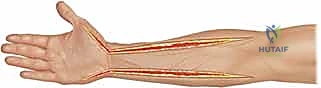
Escharotomies are performed by incising the burned skin down to, but not through, the subcutaneous fat. In the upper extremity, this is typically performed along the mid-medial and mid-lateral lines of the arm and forearm. If the underlying muscle remains tense, or in cases of electrical injury, a formal fasciotomy is required.
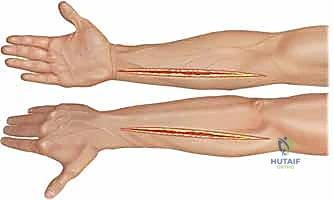
For the volar forearm, we utilize an extensile Henry approach or a modified ulnar approach. The incision begins proximal to the antecubital fossa, crossing the elbow crease obliquely to prevent subsequent flexion contracture, and proceeds distally down the forearm. At the wrist, the incision must cross the flexion creases obliquely and incorporate a formal carpal tunnel release to decompress the median nerve. The antebrachial fascia is incised longitudinally. We must systematically explore both the superficial and deep volar compartments. In electrical injuries, the flexor digitorum profundus and pronator quadratus must be directly inspected, as they lie adjacent to the radius and ulna and are highly susceptible to core heating. Any overtly necrotic, non-contractile, non-bleeding muscle must be aggressively debrided.
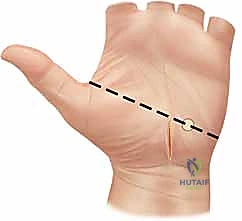
The dorsal forearm is decompressed via a longitudinal incision over the mobile wad, allowing access to both the dorsal and lateral compartments. In the hand, two to four longitudinal dorsal incisions are made over the metacarpals to release the dorsal and volar interosseous compartments. The thenar and hypothenar compartments are released via separate longitudinal incisions along their respective borders, taking care to avoid the recurrent motor branch of the median nerve and the ulnar neurovascular bundle.
Chronic Phase: Contracture Release and Reconstruction
Reconstruction of the burned hand requires a highly tailored approach. The fundamental principle is to incise or excise the restricting scar tissue until normal, unyielding tissue is reached, allowing the joint to be passively manipulated into a functional position.
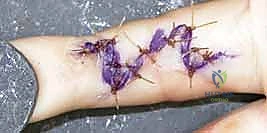
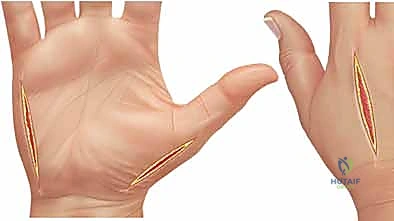
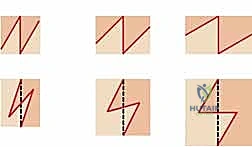
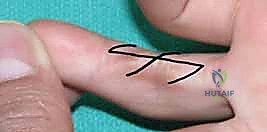
For linear contractures, multiple Z-plasties or W-plasties are employed to redirect the scar tension lines and lengthen the contracted tissue. The angles of the Z-plasty flaps are typically designed at 60 degrees to provide maximum theoretical lengthening (up to 73%) while maintaining adequate flap vascularity.
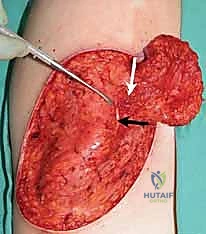
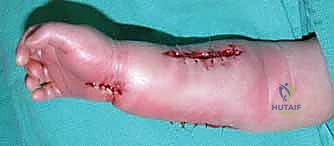
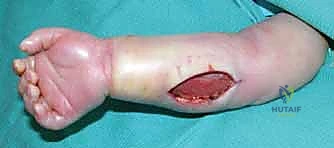
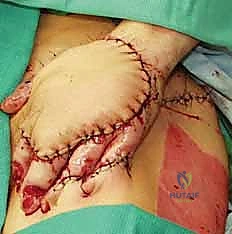
For broad, sheet-like contractures, particularly over the dorsum of the hand or the volar wrist, the scar must be completely incised (incisional release) or excised (excisional release). Once the contracture is released, a massive soft-tissue defect is created. This defect cannot be closed primarily. We rely heavily on full-thickness skin grafts (FTSG) or thick split-thickness skin grafts (STSG) to resurface these areas. FTSGs are preferred over joints and in the web spaces because they undergo significantly less secondary contraction compared to STSGs.
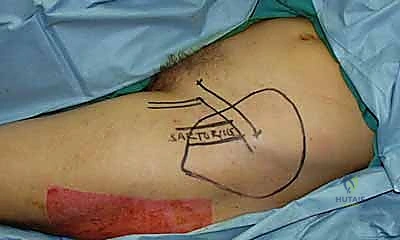
If the release exposes bare bone, tendon without paratenon, or vital neurovascular structures, skin grafting is contraindicated. In these scenarios, vascularized tissue transfer is mandatory. This may involve local random pattern flaps, regional pedicled flaps (e.g., radial forearm flap, reverse posterior interosseous flap), or, in severe cases, free tissue transfer (e.g., anterolateral thigh flap, lateral arm flap) utilizing microvascular anastomosis.
Following the soft-tissue release, the underlying joints often remain stiff due to capsular contractures. Sequential release of the collateral ligaments and volar plate may be necessary. To maintain the corrected position postoperatively while the grafts or flaps heal, the joints are frequently stabilized using percutaneous Kirschner wires (K-wires) driven across the PIP or DIP joints in full extension, or the first web space in maximum abduction.
Complications, Incidence Rates, and Salvage Management
The surgical management of upper extremity burns is fraught with potential complications, demanding constant vigilance and a readiness to employ salvage techniques. The physiological insult of the burn, combined with the extensive surgical trauma, creates an environment ripe for adverse outcomes.
The most devastating acute complication is the failure to adequately decompress a compartment syndrome, leading to Volkmann's ischemic contracture. This results in irreversible necrosis of the flexor musculature and profound neuropathy. The incidence of compartment syndrome in high-voltage electrical injuries approaches 40-80%, whereas in isolated thermal burns, it is significantly lower but highly dependent on the depth and circumferential nature of the burn. If myonecrosis is established, the necrotic tissue must be radically excised to prevent life-threatening sepsis and myoglobinuric renal failure. Salvage management for established Volkmann's contracture involves late reconstructive procedures, including free functioning muscle transfers (e.g., gracilis transfer) to restore prehension, or complex tendon transfer algorithms.
Infection is a omnipresent threat, given the loss of the cutaneous barrier and the presence of avascular eschar. Postoperative graft loss or flap necrosis due to infection, hematoma, or inadequate vascular bed preparation occurs in approximately 10-15% of complex reconstructive cases. Immediate salvage requires aggressive serial debridements, targeted antimicrobial therapy, and eventual re-grafting or the use of a higher-order flap once the wound bed is sterile and highly vascularized.
Recurrence of the contracture is the most common long-term complication, particularly in pediatric patients who outgrow their grafts or in patients who are non-compliant with rigorous postoperative splinting and
