Tibiotalocalcaneal Fusion with Femoral Head Allograft: A Masterclass in Complex Hindfoot Reconstruction
Key Takeaway
Join us in the OR for a masterclass on tibiotalocalcaneal fusion with femoral head allograft. We'll meticulously reconstruct severe talar defects, addressing avascular necrosis or failed total ankle arthroplasty. Learn precise surgical anatomy, intricate intraoperative steps, critical pearls, and comprehensive postoperative management to achieve stable, pain-free hindfoot function.
Comprehensive Introduction and Patho-Epidemiology
Tibiotalocalcaneal (TTC) fusion utilizing a bulk femoral head allograft represents a formidable, salvage-level intervention within the armamentarium of complex hindfoot reconstructive surgery. This masterclass procedure is specifically reserved for a uniquely challenging patient cohort presenting with catastrophic talar bone loss, where conventional arthrodesis techniques or revision arthroplasty are anatomically and biomechanically precluded. The fundamental objective of this extensive reconstruction is to restore a stable, plantigrade, and functional lower extremity, thereby preventing the ultimate morbid endpoint: major lower limb amputation. The evolution of this technique underscores a paradigm shift in orthopedic salvage, moving from simple, often shortening fusions toward anatomically restorative bulk allografting that preserves limb length and optimizes the biomechanical axis.
The patho-epidemiology necessitating such a profound structural reconstruction is multifaceted, predominantly driven by severe talar body avascular necrosis (AVN), post-traumatic osteonecrosis, Charcot neuroarthropathy, and the increasingly prevalent complication of failed total ankle arthroplasty (TAA). The talus, characterized by its extensive articular cartilage coverage (approximating 60% of its surface area) and a notoriously tenuous, retrograde blood supply, is inherently vulnerable to ischemic insult. Disruption of the artery of the tarsal canal, the artery of the sinus tarsi, or the deltoid branches—whether through high-energy trauma, systemic corticosteroid use, or idiopathic causes—precipitates osteocyte death, subchondral microfractures, and eventual macroscopic structural collapse. Once the talar body architecture disintegrates, the intricate kinematics of the ankle and subtalar joints are irrevocably destroyed, leading to debilitating pain, progressive deformity, and profound functional impairment.
Concurrently, the exponential rise in primary total ankle arthroplasties performed globally has generated a parallel increase in complex revision scenarios. Aseptic loosening, implant subsidence into the talus, periprosthetic osteolysis, and deep periprosthetic joint infections (PJI) frequently result in massive cavitary and segmental bone defects upon explantation. When the remaining talar bone stock is insufficient to support a revision implant or a standard fusion construct, the surgeon is faced with a massive void. The utilization of a structural femoral head allograft provides the necessary osteoconductive scaffold and immediate mechanical stability to bridge this critical-sized defect, effectively spanning the compromised talus to unite the distal tibia directly to the calcaneus.
Understanding the complex interplay between bone viability, mechanical alignment, and soft tissue envelope integrity is paramount. This procedure is not merely a carpentry exercise in filling a void; it is a meticulously orchestrated biological and mechanical reconstruction. The fresh-frozen femoral head allograft acts as a massive spacer that restores limb length, tension to the myofascial sleeve, and a stable platform for weight-bearing. However, it relies entirely on the slow, unpredictable process of creeping substitution for long-term biological incorporation. Therefore, the surgeon must execute flawless technique to maximize host-graft contact, achieve absolute rigid internal fixation, and optimize the local biological milieu to foster osteogenesis across massive, relatively avascular interfaces.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of the surgical anatomy of the distal leg, ankle, and hindfoot is absolutely critical for the safe execution of a transfibular tibiotalocalcaneal fusion. The standard lateral approach exploits the internervous and intermuscular planes, but it is fraught with potential hazards. The lateral malleolus and the distal fibula serve as the primary anatomic landmarks. The fibula not only provides dynamic stability to the ankle mortise but also dictates the lateral contour of the joint. In this procedure, a distal fibulectomy is routinely performed to gain unhindered access to the tibiotalar and subtalar joints, and the excised fibula serves as an invaluable source of autogenous bone graft.

The neurovascular structures in the lateral and posterolateral compartments demand meticulous attention. The sural nerve, formed by the union of the medial sural cutaneous nerve and the sural communicating branch, courses distally along the posterolateral aspect of the calf, intimately associated with the small saphenous vein. It passes posterior to the lateral malleolus and provides sensation to the lateral aspect of the foot. This nerve is highly susceptible to iatrogenic injury during the initial incision, superficial dissection, and aggressive posterior retraction. Similarly, the superficial peroneal nerve, which supplies the lateral compartment musculature before piercing the crural fascia to provide dorsal cutaneous foot sensation, can be compromised by errant proximal dissection or excessive anterior retraction.
Deep to the crural fascia, the peroneus longus and brevis tendons are encountered within the retromalleolar groove. These tendons, vital for foot eversion and dynamic lateral ankle stability, must be carefully mobilized and retracted posteriorly to expose the lateral capsule of the ankle and subtalar joints. The vascular anatomy is equally unforgiving. While the primary approach is lateral, the surgeon must remain acutely aware of the medial neurovascular bundle—comprising the posterior tibial artery, accompanying venae comitantes, and the tibial nerve—situated posterior to the medial malleolus. Aggressive medial joint preparation, over-penetration of reamers, or errant placement of transfixing hardware can result in catastrophic vascular compromise or debilitating neuropathic pain.
Biomechanically, the normal ankle and subtalar joints function as a complex universal joint, facilitating dorsiflexion, plantarflexion, inversion, and eversion to accommodate uneven terrain and absorb impact loads. A TTC fusion inherently obliterates this motion, converting the leg and foot into a rigid lever arm. The introduction of a massive femoral head allograft dramatically alters load transfer mechanics. The construct must withstand immense bending moments and torsional forces during the stance phase of gait. To mitigate the risk of hardware failure or allograft fracture, the fusion must be aligned in slight valgus (approximately 5 degrees), neutral dorsiflexion, and slight external rotation. This specific alignment optimizes the mechanical axis, minimizes varus thrust (which is poorly tolerated and leads to lateral column overload), and facilitates an energy-efficient, plantigrade gait despite the loss of hindfoot articulation.
Exhaustive Indications and Contraindications
The decision to proceed with a tibiotalocalcaneal fusion utilizing a femoral head allograft must be predicated on a rigorous, individualized patient assessment. This is a high-stakes, salvage procedure with a substantial complication profile; thus, patient selection is arguably the most critical determinant of success. The primary indication is massive, uncontained talar bone loss where the remaining viable bone is insufficient to achieve a primary arthrodesis or support a revision arthroplasty component.
Talar body avascular necrosis (AVN) with structural collapse remains a leading indication. When the talar dome flattens and fragments, the congruency of the ankle joint is lost, leading to rapid, painful osteoarthritis. If the necrosis extends deeply into the talar body, simple joint preparation will only expose dead, avascular bone incapable of supporting a fusion mass. Similarly, the dramatic increase in primary total ankle arthroplasty (TAA) has yielded a complex subset of patients presenting with catastrophic implant failure. Aseptic loosening with profound osteolysis, or explantation secondary to deep periprosthetic joint infection (after appropriate staging and eradication), often leaves massive cavitary defects spanning both the distal tibia and the remaining talus.
Other indications include severe, post-traumatic segmental bone loss following high-energy crush injuries or open fractures of the hindfoot, where the talus is either extruded, highly comminuted, or subsequently resected due to deep infection. End-stage Charcot neuroarthropathy with complete destruction of the talar body and subsequent hindfoot collapse may also necessitate this approach to restore a plantigrade, braceable foot and avert amputation. Furthermore, wide resection of primary or metastatic bone tumors of the talus can leave a void ideally suited for bulk allograft reconstruction.
| Category | Specific Conditions | Clinical Rationale |
|---|---|---|
| Absolute Indications | Talar Body AVN with massive collapse | Insufficient viable bone for standard fusion; need to restore height. |
| Failed TAA with profound osteolysis | Cavitary defects in tibia/talus preclude standard revision or fusion. | |
| Post-traumatic talar extrusion/loss | Missing structural intercalary segment requires bulk replacement. | |
| Resection of talar neoplasms | Oncologic void requires structural filling and fusion. | |
| Relative Indications | Severe Charcot neuroarthropathy | Restoration of plantigrade foot in the setting of massive bone loss. |
| Chronic osteomyelitis (post-eradication) | Reconstruction of segmental defect after aggressive debridement. | |
| Absolute Contraindications | Active, untreated deep infection | High risk of allograft seeding and catastrophic failure. |
| Severe peripheral vascular disease | Inadequate perfusion to heal the massive surgical insult. | |
| Uncorrectable severe malalignment | Extreme valgus/varus creates impossible soft tissue tension. | |
| Profound medical comorbidities | Patient unable to tolerate prolonged anesthesia or rehabilitation. | |
| Relative Contraindications | Active smoking / Nicotine use | Significantly increased risk of nonunion and wound breakdown. |
| Uncontrolled Diabetes Mellitus | Impaired healing, increased infection risk, poor bone quality. | |
| Severe osteoporosis | Insufficient host bone stock to secure massive intramedullary hardware. | |
| Patient non-compliance | Inability to adhere to strict, prolonged non-weight-bearing protocols. |
Contraindications must be strictly respected to avoid disastrous outcomes. Active, untreated deep infection is an absolute contraindication; placing a massive, avascular allograft into an infected bed guarantees failure. Severe peripheral vascular disease that precludes wound healing must be identified and optimized, or an alternative, less invasive strategy (including primary amputation) should be considered. Furthermore, severe, rigid hindfoot malalignment—particularly extreme valgus exceeding 25 degrees—presents a unique contraindication for the lateral transfibular approach. Attempting to correct profound valgus and insert a bulk allograft from the lateral side places immense, often insurmountable tension on the lateral soft tissue envelope, leading to inevitable skin necrosis, wound dehiscence, and secondary infection.
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the bedrock upon which the success of this complex reconstruction is built. Standard weight-bearing anteroposterior, lateral, and mortise radiographs of the ankle and foot are mandatory for initial assessment of deformity, bone loss, and overall alignment. However, advanced cross-sectional imaging is non-negotiable. A fine-cut computed tomography (CT) scan with 3D sagittal and coronal reconstructions provides a precise volumetric assessment of the talar defect, the quality of the remaining distal tibial plafond, and the integrity of the calcaneal body. This allows the surgeon to map the exact zones of viable, bleeding bone that will serve as the host bed for the allograft.
Templating is critical for both the allograft sizing and the hardware selection. Using digital templating software on the CT reconstructions, the surgeon can estimate the required diameter of the femoral head allograft. It is imperative to communicate with the tissue bank well in advance to secure a fresh-frozen, non-irradiated whole femoral head allograft of appropriate size. Irradiation, while decreasing disease transmission risk, significantly degrades the structural integrity and osteoinductive potential of the graft, rendering it unsuitable for this high-stress application. Furthermore, the surgeon must template for the fixation construct, most commonly a robust, retrograde intramedullary TTC nail. The length, diameter, and optimal trajectory of the nail must be determined to ensure adequate purchase in the calcaneus, passage through the center of the allograft, and secure fixation in the tibial isthmus.
Patient positioning and operating room setup must be executed flawlessly to facilitate surgical flow and optimal radiographic visualization. The patient is positioned supine on a radiolucent operating table. A substantial hip bump—often a specialized foam wedge or tightly rolled blankets—is placed under the ipsilateral hemipelvis. This bump forces the lower extremity into internal rotation, bringing the lateral malleolus and the entire lateral aspect of the hindfoot perpendicular to the floor, providing a direct, unhindered trajectory for the surgical approach. The bump must be securely taped to the table to prevent shifting during aggressive reaming and manipulation.
Anesthesia typically involves a combination of a regional block (spinal or popliteal/saphenous nerve blocks) for superior postoperative pain control, combined with general anesthesia to ensure complete muscle relaxation. A high thigh tourniquet is applied over generous padding. Following meticulous skin preparation and sterile draping—which must allow access from the mid-tibia to the toes—the leg is exsanguinated with an Esmarch bandage, and the tourniquet is inflated to 250-300 mm Hg. Fluoroscopy is positioned on the contralateral side of the table, ensuring that true AP and lateral views of the tibia, ankle, and foot can be obtained effortlessly without compromising the sterile field.
Step-by-Step Surgical Approach and Fixation Technique
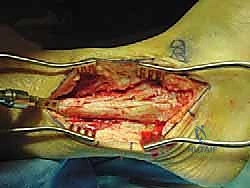
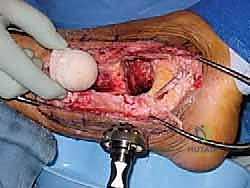
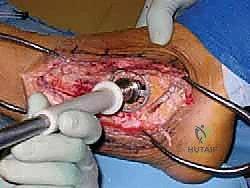
We resume the surgical execution from the point of defect sizing and preparation. "With the ankle and hindfoot held precisely in this neutral position, proceed with reaming the combined tibial and calcaneal surfaces to create a unified, hemispherical concavity." The objective here is to transform the irregular, necrotic void into a perfectly spherical socket that will intimately match the contour of our femoral head allograft. Utilizing a series of sequentially expanding male acetabular reamers on power, the surgeon meticulously decorticates the distal tibial plafond and the superior aspect of the calcaneus (the posterior facet).
This reaming process demands absolute precision and constant spatial awareness. The surgeon must apply even pressure, ensuring the reamer does not drift anteriorly (risking the anterior neurovascular bundle) or medially (threatening the posterior tibial artery and tibial nerve). Copious cold saline irrigation is mandatory during reaming to prevent thermal necrosis of the crucial host bone bed. The endpoint of reaming is reached when punctate bleeding is observed from healthy, cancellous host bone across the entire concavity, and the defect is deemed large enough to accept a structurally sound portion of the allograft. The final reamer size dictates the precise diameter to which the femoral head allograft must be shaped.
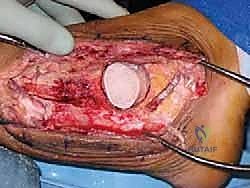
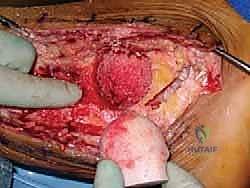
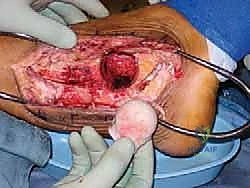
Simultaneous to the host bed preparation, the back-table team prepares the fresh-frozen femoral head allograft. Once completely thawed, all remaining articular cartilage and soft tissue attachments (such as the ligamentum teres) must be meticulously removed using a combination of a surgical scalpel, curettes, and a high-speed burr. Cartilage is an absolute barrier to osteointegration; leaving any remnants will guarantee a nonunion. Once denuded of cartilage, the allograft is shaped using female acetabular reamers that exactly match the final male reamer size used in the patient. This ensures a perfect, congruent, "press-fit" interface between the host bone and the allograft, which is the paramount biomechanical requirement for creeping substitution.
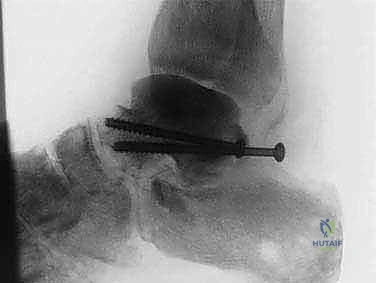
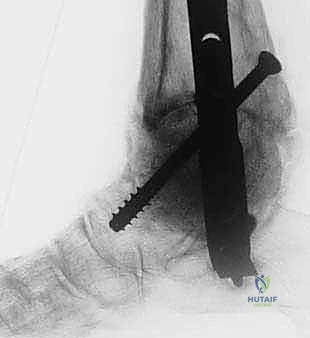
With the host bed prepared and the allograft perfectly shaped, the graft is impacted into the defect. The surgeon must assess the restoration of limb length and the tension of the soft tissue envelope. If the graft is too large, the lateral skin will not close without undue tension; if too small, the construct will lack structural stability. Once the optimal fit is achieved, the focus shifts to rigid internal fixation. A robust, retrograde intramedullary TTC nail is the gold standard for this reconstruction. A guidewire is introduced through the plantar aspect of the heel, centered on the calcaneal tuberosity, and driven superiorly through the calcaneus, directly through the center of the femoral head allograft, and up into the medullary canal of the tibia.
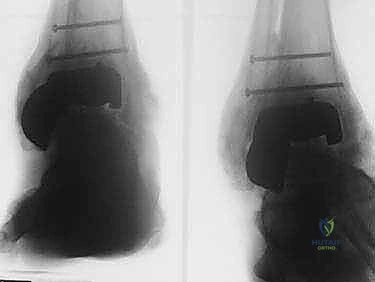
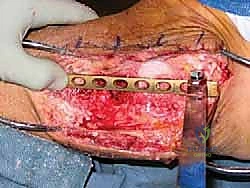
Fluoroscopy is utilized continuously to confirm perfect guidewire trajectory in both AP and lateral planes. The canal is then sequentially reamed over the guidewire. Reaming through the dense, sclerotic bone of the allograft can be highly challenging and requires sharp reamers and patience to avoid thermal necrosis or catastrophic fracturing of the graft. Once reamed to the appropriate diameter (typically 1-1.5 mm larger than the intended nail size), the selected intramedullary nail is inserted and driven across the calcaneus, allograft, and into the tibia.
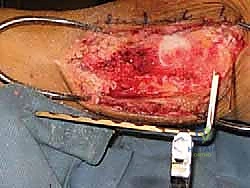
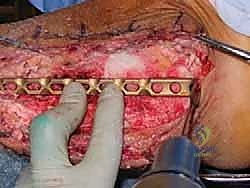
To maximize compression across the massive fusion interfaces, internal compression mechanisms built into modern TTC nails are utilized. Alternatively, or in addition, external compression can be applied manually before locking the nail. The nail is secured with multiple locking screws in the calcaneus, the allograft (if the nail design permits), and the proximal tibia. To augment the biological healing potential, the morcelized autograft generated from the initial fibulectomy, often combined with orthobiologics such as demineralized bone matrix (DBM) or bone marrow aspirate concentrate (BMAC), is densely packed into any remaining interstices around the allograft. Meticulous hemostasis is achieved, a deep suction drain is placed, and the lateral soft tissues are closed in layers, paying extreme care to achieve a tension-free skin closure to prevent catastrophic wound breakdown.
Complications, Incidence Rates, and Salvage Management
Tibiotalocalcaneal fusion with bulk femoral head allografting is a massive physiological insult and carries one of the highest complication profiles in foot and ankle surgery. Surgeons undertaking this procedure must be acutely aware of the potential pitfalls and possess the advanced reconstructive skills required to manage catastrophic failures. The utilization of a massive, avascular piece of donor bone introduces profound biological and mechanical vulnerabilities that persist for years postoperatively.
The most formidable and frequent complication is nonunion, which occurs at either the tibiotalar or subtalar interface, or both. Because the allograft relies entirely on creeping substitution—a slow process where host osteoclasts resorb the dead donor bone while osteoblasts lay down new host bone—the graft remains mechanically vulnerable for an extended period. If rigid fixation is lost, or if the biological milieu is inadequate, the graft will fail to incorporate. Nonunion rates reported in the literature range from 15% to over 30%, depending on patient comorbidities and the specific indication. Management of an aseptic nonunion typically requires revision surgery with removal of hardware, aggressive debridement of the fibrous nonunion site, massive autogenous bone grafting (often utilizing the Reamer-Irrigator-Aspirator (RIA) system to harvest from the femur), and revision internal fixation, potentially augmenting the IM nail with a robust lateral locking plate.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage Management Strategy |
|---|---|---|---|
| Nonunion / Delayed Union | 15% - 35% | Poor host bed, inadequate compression, smoking, massive avascular graft. | Revision fixation, massive autogenous bone grafting (RIA), orthobiologics. |
| Deep Infection / PJI | 5% - 15% | Prolonged OR time, massive foreign body, poor soft tissue envelope. | Aggressive I&D, hardware removal, antibiotic spacer, prolonged IV antibiotics. |
| Hardware Failure | 10% - 20% | Fatigue failure of nail/screws prior to solid bony union. | Hardware extraction, revision fixation with larger/stiffer construct. |
| Wound Dehiscence / Necrosis | 10% - 25% | Excessive lateral tension, poor vascularity, severe preoperative valgus. | Local wound care, negative pressure wound therapy, rotational or free flaps. |
| Allograft Resorption / Collapse | 5% - 15% | Immunologic reaction, mechanical overload, incomplete creeping substitution. | Revision with structural autograft, custom 3D printed cage, or amputation. |
| Nerve Injury (Sural/SPN) | 5% - 10% | Iatrogenic transection or traction during lateral approach. | Neuroma excision, targeted muscle reinnervation (TMR), neuropathic medications. |
Deep infection is a devastating complication that threatens not only the limb but potentially the patient's life. The massive allograft acts as a large, avascular foreign body, providing an ideal nidus for bacterial biofilm formation. If a deep infection occurs, aggressive surgical intervention is mandatory. This typically involves radical debridement, complete removal of all hardware, and excision of the entire infected allograft. The resulting massive void is managed with an antibiotic-impregnated polymethylmethacrylate (PMMA) spacer to maintain soft tissue tension while delivering high local doses of antibiotics. Systemic intravenous antibiotics, guided by intraoperative cultures, are administered for a minimum of 6 weeks. Once the infection is definitively eradicated, the surgeon faces a monumental reconstructive challenge, often requiring a custom 3D-printed titanium cage or a massive structural autograft to attempt a secondary fusion, though at this stage, major lower extremity amputation (typically a below-knee amputation) becomes a highly realistic and sometimes preferred salvage option.
Hardware failure, specifically fracture of the intramedullary nail or breakage of the locking screws, is a direct consequence of the race between biological healing and mechanical fatigue. If the fusion does not consolidate rapidly enough, the cyclic loading of weight-bearing will eventually cause the rigid metal construct to fail. Regular radiographic surveillance is critical to identify delayed union before catastrophic hardware failure occurs. Finally, wound complications, including marginal necrosis and frank dehiscence, are common due to the tenuous vascularity of the lateral ankle skin and the tension placed on the envelope by the bulky allograft. Prompt recognition and involvement of plastic surgery colleagues for local rotational flaps or free tissue transfer may be necessary to salvage the limb.

Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following a tibiotalocalcaneal fusion with a massive bulk allograft is a grueling, protracted process that demands extreme patience and absolute compliance from the patient. The biological reality of creeping substitution dictates that the construct remains mechanically fragile for months. Premature weight-bearing is the most common iatrogenic cause of catastrophic construct failure, allograft collapse, and nonunion. Therefore, the rehabilitation protocol must be rigidly phased and strictly guided by serial radiographic evidence of progressive bone consolidation.
Phase I: Absolute Protection and Soft Tissue Healing (Weeks 0-6)
Immediately postoperatively, the patient is placed in a bulky, well-padded Jones dressing with a posterior plaster splint to maintain neutral alignment and accommodate anticipated swelling. The patient is strictly non-weight-bearing (NWB) on the operative extremity. Deep venous thrombosis (DVT) prophylaxis is mandatory during this period of immobility. At the two-week mark, the surgical dressings are removed, the wounds are meticulously inspected, and sutures are removed if the incision is fully healed. The patient is then transitioned into a rigid, short-leg fiberglass cast. The primary goals of this phase are uninterrupted wound healing, strict edema control through continuous elevation, and the prevention of early mechanical overload.
Phase II: Continued Protection and Early Mobilization (Weeks 6-12)
At six weeks postoperatively, the cast is removed, and the first set of post-operative radiographs (AP, lateral, and mortise views) are obtained to assess alignment and hardware integrity; however, significant bony union is rarely visible at this early stage. If the incisions are pristine, the patient is transitioned into a removable Controlled Ankle Motion (CAM) boot. Strict non-weight-bearing must continue. The transition to a removable boot allows the patient to begin gentle, active range of motion exercises of the toes, knee, and hip to prevent proximal disuse atrophy and contractures. The patient may also remove the boot for hygiene but must sleep in the boot to prevent inadvertent trauma.
Phase III: Graduated Weight-Bearing and Functional Restoration (Weeks 12-24+)
At the 12-week mark, a critical clinical and radiographic assessment is performed. The surgeon looks for definitive signs of radiographic consolidation: bridging trabecular bone across the tibiotalar and subtalar interfaces, obliteration of the host-graft radiolucent lines, and the absence of hardware migration or lucency. A CT scan may be obtained if plain radiographs are equivocal. If, and only if, sufficient healing is evident, the patient may initiate a highly structured, progressive weight-bearing protocol. This typically begins with 25% partial weight-bearing in the CAM boot using crutches or a walker, advancing by 25% every 1-2 weeks as tolerated. Formal physical therapy is initiated to focus on gait retraining, proprioception, and strengthening of the proximal musculature. Transition to regular footwear, often requiring custom orthotics or a rigid ankle-foot orthosis (AFO) for prolonged support, typically occurs between 4 and 6 months postoperatively, though maximum medical improvement may take up to 18-24 months.

Summary of Landmark Literature and Clinical Guidelines
The literature surrounding tibiotalocalcaneal fusion with bulk femoral head allograft is characterized by retrospective case series and cohort studies, reflecting the salvage nature and relative rarity of the procedure. However, several landmark studies have shaped the current standard of care and established benchmark expectations for clinical outcomes and union rates. The evolution of this technique is a testament to the continuous refinement of internal fixation and biological augmentation strategies.
Early reports by Myerson and colleagues in the late 1990s and early 2000s demonstrated the feasibility of using fresh-frozen femoral head allografts to bridge massive defects, primarily in the setting of failed total ankle arthroplasty and severe post-traumatic AVN. These early series highlighted the immense technical difficulty of the procedure and reported union rates hovering around 70-75%, with a correspondingly high rate of complications, particularly nonunion and infection. These studies established the fundamental principle that achieving a congruent, press-fit interface between the host bone and the allograft is the most critical technical factor in promoting union.
More contemporary studies, such as those by Jeng et al. and Berlet et al., have evaluated the outcomes of this procedure utilizing modern, robust retrograde intramedullary nailing systems with internal compression capabilities. These studies have shown improved union rates, often approaching 80-85%, attributed to the superior biomechanical stability provided by modern locked intramedullary devices compared to older blade plate or crossed-screw constructs. Furthermore, recent literature emphasizes the critical role of biological augmentation. The routine use of local autogenous bone graft (harvested from the excised fibula), combined with cellular allografts, DBM, or BMAC, has become a standard clinical guideline to enhance the osteogenic potential at the avascular host-graft interfaces.
Current clinical guidelines strongly recommend meticulous preoperative optimization, including smoking cessation, strict glycemic control in diabetic patients, and exhaustive evaluation to rule out indolent periprosthetic joint infection prior to undertaking this massive reconstruction. While the complication rates remain undeniably high, the literature consistently demonstrates that when successful union is achieved, patients report significant reductions in pain, restoration of a plantigrade foot, and a profound improvement in their overall quality of life, successfully averting the devastating physical and psychological impact of a major lower extremity amputation.
