ECU Subsheath Reconstruction: An Intraoperative Masterclass for Ulnar Wrist Stability
Key Takeaway
Join us in the OR for an immersive masterclass on Extensor Carpi Ulnaris (ECU) subsheath reconstruction. We'll meticulously cover surgical anatomy, patient positioning, and granular intraoperative execution of retinacular sling and graft techniques. Learn to identify and manage critical pitfalls, ensuring optimal outcomes for patients suffering from painful ECU subluxation and associated DRUJ instability.
Welcome, fellows, to the operating theater. Today, we are addressing a common yet frequently mismanaged condition: Extensor Carpi Ulnaris (ECU) subluxation. This procedure demands meticulous attention to detail, a profound understanding of ulnar-sided wrist anatomy, and precise surgical technique to restore both dynamic stability and complex wrist kinematics. We are going to walk through this step-by-step, as if you are scrubbed in right next to me across the operating table.
Comprehensive Introduction and Patho-Epidemiology
The Extensor Carpi Ulnaris (ECU) tendon is an indispensable dynamic stabilizer of the distal radioulnar joint (DRUJ) and serves as a powerful ulnar deviator and wrist extensor. Its stability at the wrist is paramount for normal function, particularly in activities requiring forceful rotation and deviation. When the structural integrity of the ECU subsheath is compromised, the tendon is permitted to subluxate or frankly dislocate out of the ulnar osseous groove. Clinically, this manifests as a painful, mechanical click, often exacerbated by resisted supination, ulnar deviation, and palmar flexion. If left untreated, this seemingly simple mechanical snapping can rapidly progress to severe tendinopathy, partial intrasubstance tearing, and debilitating ulnar-sided wrist pain.
Epidemiologically, ECU subluxation is frequently observed in high-demand athletes involved in racket sports, baseball, golf, and ice hockey. The classic mechanism of injury involves a sudden, traumatic event characterized by active, forceful ECU contraction combined with forced supination, palmar flexion, and ulnar deviation. This specific vector of forces directs the ECU tendon volarly and ulnarly, placing immense stress on the radial attachment of the ECU subsheath. While acute traumatic ruptures are common in the athletic population, we also see chronic, attritional subluxation in patients with underlying inflammatory arthropathies, such as rheumatoid arthritis, where the supporting soft tissues become progressively attenuated.
The natural history of ECU subsheath injuries is highly variable and depends heavily on the morphology of the tear and the depth of the patient's native ulnar groove. In the acute phase, patients often present with profound swelling, localized tenderness, and apprehension during forearm rotation. As the acute inflammation subsides, the condition frequently transitions into a chronic, recurrent subluxation. The repetitive friction of the tendon translating over the sharp cortical edge of the ulnar groove leads to progressive mechanical attrition. This chronic state not only destroys the tendon substance but also effectively removes a critical dynamic stabilizer of the DRUJ, placing increased secondary stress on the triangular fibrocartilage complex (TFCC).
Differentiating true ECU subluxation from other causes of ulnar-sided wrist pain is the hallmark of a skilled wrist surgeon. The differential diagnosis is extensive and includes isolated ECU tenosynovitis, TFCC peripheral tears, lunotriquetral ligament instability, ulnocarpal impaction syndrome, and DRUJ arthrosis. While isolated tenosynovitis presents with fullness and pain along the tendon sheath, it lacks the palpable, dynamic translation of the tendon out of its groove. Careful clinical examination, combined with a high index of suspicion, is necessary to accurately identify the pathoanatomy and prevent the long-term sequelae of chronic instability.
Detailed Surgical Anatomy and Biomechanics
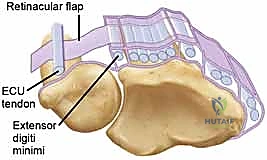
Before we make a surgical incision, a masterful understanding of the local anatomy is non-negotiable. The ECU tendon resides within the sixth dorsal compartment, a unique fibro-osseous tunnel situated along the dorsum of the distal ulna. Unlike the other five extensor compartments of the wrist, which are primarily defined by the overlying extensor retinaculum, the sixth compartment is structurally distinct. The critical stabilizing structure here is the ECU subsheath, a robust, separate fibro-osseous layer. It originates from the radial wall of the ulnar osseous groove, arcs tightly over the ECU tendon, and inserts securely onto the ulnar wall.
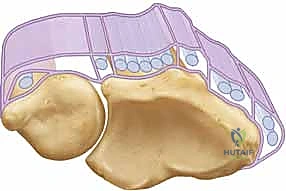
This subsheath ensheathes approximately 1.5 to 2.0 cm of the distal ulna, maintaining the tendon securely within its groove throughout the full arc of forearm rotation. To understand the biomechanics, we must examine the broader picture of the extensor retinaculum, which consists of two primary layers. The supratendinous retinaculum is the superficial layer originating proximal to the radiocarpal joint and inserting at the carpometacarpal joints. Critically, while this superficial layer provides a block to subluxation for compartments one through five, it simply drapes over the sixth compartment and does not prevent volar or ulnar subluxation of the ECU tendon.
The true restraint is the infratendinous retinaculum. This deeper layer runs from the radiocarpal to the carpometacarpal joints, passing deep to the fourth and fifth extensor compartments. The ECU's own separate fibro-osseous subsheath is anatomically considered a specialized duplication of this infratendinous retinaculum. During supination, the ECU tendon requires maximum restraint as the ulnar groove rotates away from the tendon's line of pull. The subsheath must withstand significant tension to keep the tendon seated. If the subsheath is torn, the tendon bowstrings volarly, losing its mechanical advantage as an extensor and acting solely as a deforming flexor force.
Furthermore, the ECU subsheath is not an isolated anatomical island; it contributes significantly to the dorsal structural integrity of the triangular fibrocartilage complex (TFCC). The deep fibers of the subsheath interdigitate with the dorsal radioulnar ligament. Therefore, a high-grade disruption of the ECU subsheath can lead to not only ECU instability but also static instability of the DRUJ. The dislocation of the ECU tendon effectively removes a dynamic stabilizer of the DRUJ, further compromising complex wrist mechanics and accelerating the progression toward degenerative joint disease.
Exhaustive Indications and Contraindications
| Parameter | Indications for Surgery | Contraindications for Surgery |
|---|---|---|
| Clinical Presentation | Painful, reproducible snapping; persistent ulnar wrist pain | Asymptomatic snapping; painless hypermobility |
| Duration of Symptoms | Chronic (>3-6 weeks) failing conservative care; Acute with massive tear | Acute (<3 weeks) amenable to cast immobilization |
| Pathoanatomy | Inoue Type A (non-repairable), B, or C lesions; shallow ulnar groove | Active local infection; severe overlying skin compromise |
| Associated Conditions | Concomitant repairable TFCC tear; DRUJ instability | End-stage DRUJ osteoarthritis; severe fixed deformity |
Surgical reconstruction of the ECU subsheath is primarily indicated in patients with clinically significant, persistent symptoms related to painful subluxation of the ECU tendon who have failed a dedicated course of nonoperative management. In the acute setting (less than 3 weeks post-injury), a trial of conservative treatment utilizing an above-elbow cast or a rigid Muenster splint in pronation and slight wrist extension is often attempted. However, if symptoms persist beyond 4 to 6 weeks, or if the patient experiences immediate recurrence of painful snapping upon removal of immobilization, surgical intervention is strongly warranted to restore function and prevent tendon attrition.
The guiding principles for surgical repair are heavily dictated by the specific morphology of the osteofibrous sheath lesion. We rely on the Inoue and Tamura classification to guide our intraoperative strategy. A Type A lesion involves disruption of the fibro-osseous sheath from the ulnar wall. If acute and tissue quality is excellent, direct repair is indicated; if chronic or attenuated, reconstruction is required. A Type B lesion represents disruption from the radial wall, often with the tendon resting on top of the inverted sheath, preventing any chance of anatomical healing. Type B lesions almost universally require subsheath reconstruction. Finally, a Type C lesion involves the sheath stripping from the periosteum while remaining in continuity, forming a redundant false pouch that requires imbrication.
Contraindications to soft tissue reconstruction of the ECU subsheath must be carefully respected. The most absolute contraindication is asymptomatic subluxation. Many individuals, particularly those with generalized ligamentous laxity, exhibit a painless snap of the ECU during rotation; operating on these patients introduces unnecessary risk without functional benefit. Additionally, the presence of active local or systemic infection precludes elective reconstructive surgery.
Furthermore, patients presenting with advanced, end-stage osteoarthritis of the distal radioulnar joint are poor candidates for isolated ECU soft-tissue reconstruction. In these scenarios, the altered joint kinematics and bony deformity will rapidly lead to failure of any soft tissue sling. Such patients are better served by salvage procedures directed at the DRUJ itself, such as a Darrach procedure, a Sauvé-Kapandji procedure, or total DRUJ arthroplasty, which inherently alters the ECU mechanics and addresses the primary source of the patient's pathology.
Pre-Operative Planning, Templating, and Patient Positioning
Before we even consider making an incision, a rigorous and exhaustive preoperative assessment is mandatory. The physical examination must isolate the ECU pathology while ruling out confounding conditions. Palpation will reveal tenderness localized strictly to the distal ulna and the sixth dorsal compartment. The hallmark of the examination is stability testing: we passively move the wrist through supination, palmar flexion, and ulnar deviation. If the ECU tendon frankly dislocates with passive motion, we are dealing with gross instability. If active ECU contraction against resistance is required to elicit the dislocation (the synergy test), some inherent stability remains, but it is functionally insufficient.
Imaging studies are critical for preoperative templating. Routine posteroanterior, lateral, and oblique radiographs are essential to evaluate ulnar variance, assess the depth of the ulnar groove, and rule out ulnar styloid nonunions or DRUJ arthrosis. However, Magnetic Resonance Imaging (MRI) is our most sensitive diagnostic tool.
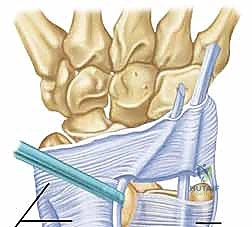
To maximize diagnostic yield, MRI should ideally be performed dynamically, or at least with the wrist positioned in full supination, to capture the tendon in its subluxated state. We look for the "perched" ECU tendon, increased signal within the tendon substance indicating tendinopathy, and the integrity of the subsheath itself.
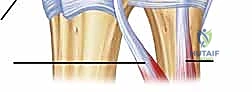
An MRI arthrogram provides unparalleled detail, depicting subsheath tears by demonstrating contrast extravasation into the sixth extensor compartment, while simultaneously evaluating the peripheral TFCC and intercarpal ligaments.
Patient positioning in the operating room must facilitate both excellent exposure and dynamic intraoperative testing. The patient is placed supine on the operating table with the affected extremity extended on a radiolucent hand table. A well-padded pneumatic tourniquet is applied to the proximal arm to ensure a bloodless surgical field. Initially, the arm is positioned in full pronation, which places the ECU tendon in its most reduced and anatomically accessible position on the dorsum of the wrist.
Preoperative templating also involves planning for graft harvest, should the local retinacular tissue prove insufficient for a robust reconstruction. While we primarily utilize a radially based slip of the extensor retinaculum, we must always be prepared with a backup plan. The ipsilateral palmaris longus tendon is our primary autograft choice, and its presence should be confirmed clinically prior to induction of anesthesia. If the palmaris longus is absent, a partial-thickness strip of the flexor carpi ulnaris (FCU) or a slip of the extensor carpi radialis brevis (ECRB) can be templated for harvest.
Step-by-Step Surgical Approach and Fixation Technique
We begin with a longitudinal incision over the dorsal ulnar aspect of the wrist, centered precisely over the sixth dorsal compartment. The incision typically extends 4 to 5 centimeters, allowing adequate proximal and distal exposure.
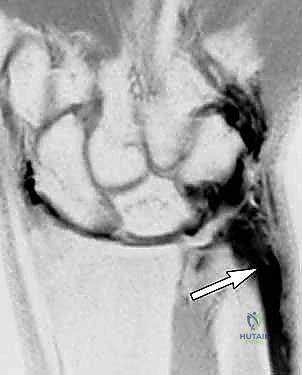
Meticulous subcutaneous dissection is critical here to identify and protect the dorsal sensory branches of the ulnar nerve (DSBUN). These nerve branches often cross directly over the operative field and are highly susceptible to iatrogenic injury or traction neuritis. We carefully mobilize the nerve branches and retract them gently using vessel loops.
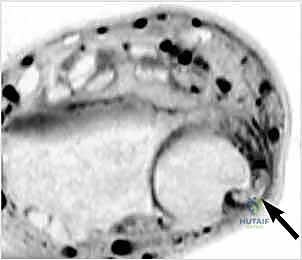
Once the superficial fascia is cleared, the extensor retinaculum is exposed. We identify the supratendinous layer and carefully define the borders of the sixth compartment.
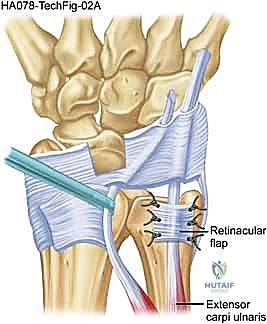
At this stage, we incise the superficial retinaculum longitudinally, slightly radial to the sixth compartment, to expose the underlying ECU subsheath and tendon. We systematically evaluate the subsheath to classify the lesion according to Inoue and Tamura.

Simultaneously, the ECU tendon itself is delivered from the wound and inspected circumferentially. Chronic subluxation frequently results in longitudinal split tears or severe attritional tendinopathy. If a split tear is present, it must be meticulously debrided and tubularized with a running locking 4-0 non-absorbable suture to restore a smooth gliding surface.
Next, we evaluate the morphology of the ulnar osseous groove. A congenitally shallow or flat groove is a significant predisposing factor for recurrent instability. If the groove is deemed insufficient to capture the tendon, a groove deepening procedure is mandatory. We use a high-speed burr or a fine rongeur to decorticate the floor of the groove, deepening it by 2 to 3 millimeters. It is imperative to preserve the cortical margins of the ulnar rim, as these edges will serve as the foundation for our soft tissue anchor placement.
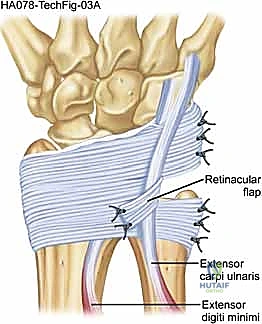
For the reconstruction itself, we elevate a radially based flap of the extensor retinaculum. This flap is typically 1.5 to 2.0 cm wide and is elevated from ulnar to radial, leaving its radial attachment intact near the fifth compartment.
The ECU tendon is reduced into the newly prepared ulnar groove. The retinacular flap is then passed over the ECU tendon and secured to the ulnar cortical rim.
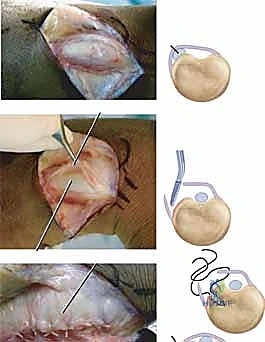
We utilize small 1.5 mm or 2.0 mm suture anchors placed precisely along the ulnar border of the groove.
The sutures are passed through the retinacular flap using a horizontal mattress configuration. Before tying the knots, we perform a dynamic intraoperative assessment. We take the wrist through a full arc of supination, pronation, flexion, and ulnar deviation. The reconstruction must hold the tendon securely in the groove without bowstringing, yet it must not be so tight as to cause stenosing tenosynovitis. Once perfect tension is achieved, the knots are tied, and the stability is re-confirmed.

The wound is then irrigated, and the skin is closed in a layered fashion.
Complications, Incidence Rates, and Salvage Management
| Complication | Estimated Incidence | Avoidance and Salvage Management |
|---|---|---|
| DSBUN Neuroma/Injury | 5 - 10% | Avoidance: Meticulous blunt dissection; use of loupe magnification. Salvage: Neuroma excision and burying into pronator quadratus. |
| Stenosing Tenosynovitis | 8 - 15% | Avoidance: Dynamic intraoperative testing before final knot tying; avoid over-constriction. Salvage: Surgical release of the tight retinacular sling. |
| Recurrent Subluxation | 5 - 12% | Avoidance: Adequate groove deepening; robust anchor fixation. Salvage: Revision reconstruction with free tendon autograft (Palmaris Longus). |
| Hardware Irritation | 2 - 5% | Avoidance: Use of knotless or all-suture anchors; burying knots deep to fascia. Salvage: Anchor removal after biological healing is complete. |
The most devastating, yet entirely preventable, complication during ECU subsheath reconstruction is iatrogenic injury to the dorsal sensory branch of the ulnar nerve (DSBUN). This nerve is highly variable in its arborization over the ulnar aspect of the wrist. Even vigorous retraction without direct transection can lead to a debilitating traction neuritis or a painful neuroma, which often overshadows the success of the tendon stabilization. Prevention requires loupe magnification, meticulous blunt dissection in the subcutaneous plane, and the use of soft vessel loops for gentle retraction. If a neuroma develops postoperatively and fails conservative management (gabapentinoids, targeted injections), salvage requires excision of the neuroma and burying the proximal nerve stump deep into the pronator quadratus muscle belly.
Over-constriction of the reconstructed subsheath is another frequent pitfall, leading to iatrogenic stenosing tenosynovitis. Surgeons, in their zeal to prevent recurrent instability, often tie the retinacular flap under excessive tension. This restricts the normal 1.5 to 2.0 cm longitudinal excursion of the ECU tendon during wrist flexion and extension, resulting in severe pain, restricted range of motion, and secondary tendinopathy. Avoidance relies entirely on dynamic intraoperative testing prior to final fixation. The tendon must glide smoothly without catching. If postoperative stenosis occurs and fails corticosteroid injections and therapy, a surgical release and slight lengthening of the reconstructed sheath are necessary.
Recurrent subluxation represents a catastrophic mechanical failure of the reconstruction. This typically occurs due to either premature return to heavy activity, failure to recognize and address a shallow ulnar groove, or pull-out of the suture anchors from osteopenic bone. Incidence rates vary but are generally reported between 5% and 12% in high-demand athletic populations. Salvage management for a failed primary reconstruction is highly complex. It almost universally requires revision surgery utilizing a free tendon autograft, such as the palmaris longus or a strip of the FCU, routed through transosseous tunnels to create a rigid, non-yielding anatomical sling, coupled with aggressive deepening of the ulnar osseous groove.
Finally, hardware irritation from suture anchors is a known complication, particularly in thin patients with minimal subcutaneous fat over the distal ulna. Prominent suture knots or backing-out of solid anchors can cause localized pain and skin breakdown. To mitigate this risk, modern techniques favor the use of all-suture anchors or knotless fixation devices, ensuring that no prominent hardware sits above the cortical line. If hardware irritation becomes symptomatic after biological healing of the subsheath is complete (typically after 6 months), simple removal of the offending anchor or suture knots is curative.
Phased Post-Operative Rehabilitation Protocols
Phase I: Maximum Protection (Weeks 0 to 4)
The immediate postoperative phase is dedicated to protecting the fragile soft tissue reconstruction. Upon completion of the surgery, the patient is placed in a bulky, non-removable sugar-tong splint or a Muenster cast. The forearm is strictly immobilized in neutral to slight pronation, and the wrist is placed in 15 to 20 degrees of extension with neutral radioulnar deviation. This specific position completely unloads the ECU tendon and removes all tension from the newly reconstructed subsheath. During this phase, absolutely no active or passive wrist motion or forearm rotation is permitted. Patients are instructed on digital range of motion exercises to prevent intrinsic hand stiffness and shoulder/elbow exercises to maintain proximal limb conditioning.
Phase II: Controlled Mobilization (Weeks 4 to 8)
At the four-week mark, biological healing of the retinacular flap to the ulnar bone is sufficient to begin controlled stress. The rigid cast is removed, and the patient is transitioned to a custom-molded, removable short-arm orthosis. Therapy is initiated under the strict guidance of a certified hand therapist. Active and active-assisted range of motion for wrist flexion and extension is commenced in a safe, sagittal plane. Forearm rotation is introduced gradually, starting from full pronation and moving toward neutral. Crucially, combined movements of supination, palmar flexion, and ulnar deviation—the exact mechanism of injury—are strictly prohibited during this phase to prevent stretching the healing graft.
Phase III: Strengthening and Proprioception (Weeks 8 to 12)
By week eight, the reconstruction should be biologically robust. The protective splint is gradually weaned, initially during the day and eventually at night. The focus of rehabilitation shifts toward restoring the dynamic stabilizing function of the ECU muscle-tendon unit. We begin with submaximal isometric exercises and progress to isotonic strengthening using light weights and resistance bands. Proprioceptive neuromuscular facilitation (PNF) techniques are highly beneficial in this phase to retrain the complex firing patterns of the wrist stabilizers. Eccentric loading of the ECU is carefully introduced to promote tendon remodeling and maximize tensile strength.
Phase IV: Return to Sport and Heavy Labor (Weeks 12+)
The final phase of rehabilitation bridges the gap between clinical recovery and functional performance. For our athletic patients, particularly those in racket sports or golf, sport-specific drills are incorporated. We focus on kinetic chain integration, ensuring that power is generated from the core and shoulder rather than placing isolated stress on the ulnar wrist. Criteria for unrestricted return to sport include painless, full active range of motion, symmetrical grip strength, and the ability to perform explosive, sport-specific maneuvers without apprehension or mechanical snapping. Full, unprotected activity is typically cleared between 3.5 to 4 months postoperatively, provided all clinical milestones have been met.
Summary of Landmark Literature and Clinical Guidelines
The evolution of our understanding of ECU subluxation is rooted in the landmark anatomical studies of Spinner and Kaplan in the 1970s. They were the first to definitively describe the sixth dorsal compartment not merely as a space beneath the extensor retinaculum, but as a distinct, specialized fibro-osseous tunnel. Their work established the critical concept that the superficial extensor retinaculum does not prevent ECU subluxation, highlighting the necessity of addressing the deep infratendinous layer—the subsheath—during surgical reconstruction. This paradigm shift moved the surgical community away from simple retinacular reefing toward true anatomical subsheath reconstruction.
Building upon this anatomical foundation, Inoue and Tamura published their seminal work in 1996, which remains the gold standard for classifying ECU subsheath injuries. By categorizing lesions into Types A, B, and C based on the specific location of the subsheath avulsion, they provided a rational, algorithm-driven approach to surgical management. Their clinical guidelines dictated that while clean, ulnar-sided avulsions (Type A) in the acute setting could be directly repaired, radial-sided avulsions (Type B) or chronic, attenuated lesions necessitated reconstruction using an ext
