Scapholunate Ligament Bone-Ligament-Bone Reconstruction: An Intraoperative Masterclass
Key Takeaway
This masterclass details bone-ligament-bone reconstruction for scapholunate ligament tears. We cover comprehensive anatomy, precise preoperative planning, and granular intraoperative steps from incision to graft fixation. Fellows will learn critical pearls, pitfalls, and post-operative management, ensuring optimal outcomes for carpal instability. This immersive guide emphasizes meticulous technique and rationale in the operating theater.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues, to the operating theater. Today, we are undertaking a technically demanding yet profoundly rewarding procedure: the Bone-Ligament-Bone (BLB) Reconstruction of the Scapholunate Ligament. This intervention transcends the simple repair of a torn structure; it represents a sophisticated biomechanical restoration of the wrist's intricate kinematic chain. Our primary objective is to arrest the relentless progression of carpal destabilization, thereby preventing the predictable and debilitating onset of degenerative arthritis, and ultimately restoring high-level function to our patients. The evolution of scapholunate (SL) reconstruction has moved from simple capsulodesis and soft-tissue tenodesis toward anatomic restorations. The BLB technique stands as the gold standard for chronic, reducible instability because it relies on the biologically superior principle of bone-to-bone healing, providing a robust construct capable of withstanding the substantial shear and tensile forces across the carpus.
Scapholunate ligament tears are, unequivocally, the most common form of carpal instability encountered in orthopedic practice. The epidemiology of this injury skews heavily toward young, active individuals and manual laborers, typically resulting from high-energy trauma or sports-related injuries. If left unrecognized or inadequately treated, the disruption of the SL kinematic linkage leads to a predictable and devastating cascade of degenerative changes. This progression culminates in Scapholunate Advanced Collapse (SLAC), a condition that fundamentally alters the load-bearing mechanics of the radiocarpal and midcarpal joints. The BLB reconstruction is our definitive surgical countermeasure to this pathophysiological cascade, offering a durable, anatomic restoration of the crucial dorsal scapholunate ligament complex.
Understanding the patho-epidemiology requires an appreciation for the subtle clinical presentations that often mask profound structural damage. Many patients initially dismiss their injury as a simple "wrist sprain," leading to delayed presentations. By the time these patients arrive in our clinics, the acute inflammatory phase has long passed, and the secondary stabilizers of the wrist—such as the dorsal intercarpal (DIC) and dorsal radiocarpal (DRC) ligaments—have begun to attenuate under the uncoupled loads of the scaphoid and lunate. It is this chronicity that mandates a structural graft; primary repair is no longer biologically viable, and soft-tissue tenodesis often stretches out over time. The BLB graft provides the immediate mechanical rigidity and the long-term biological integration necessary to restore carpal harmony.
Detailed Surgical Anatomy and Biomechanics
Before we make any incision, we must possess an absolute, uncompromising mastery of the regional anatomy and the complex biomechanics we are attempting to restore. The carpus is not merely a collection of bones; it is a finely tuned, intercalated segment that relies entirely on ligamentous integrity for its stability.
The Scapholunate Interosseous Ligament Complex
The scapholunate interosseous ligament (SLIL) is an intrinsic, intra-articular ligament composed of dense collagen fascicles, representing a true marvel of biological engineering. It is anatomically partitioned into three distinct regions, each possessing unique histological and biomechanical properties critical to carpal stability.
The Dorsal Portion is the primary mechanical stabilizer and the absolute focus of our reconstructive efforts. It is the thickest and strongest of the three components, typically measuring 2 to 3 mm in thickness and 3 to 5 mm in length. Its dense type I collagen bundles are oriented transversely, making it supremely effective at resisting distraction, torsion, and dorsal-palmar translation between the scaphoid and lunate. Crucially, its most dorsal aspect merges seamlessly with the overlying dorsal capsular ligaments. Biomechanical studies have demonstrated that this dorsal band can withstand over 300 Newtons (N) of tensile stress before ultimate failure.
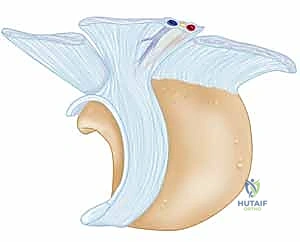
FIG 1 • The scapholunate ligament viewed from the proximal radial side with the scaphoid removed. Note the distinct dorsal, membranous, and volar components.
The Volar Portion is notably thinner, measuring approximately 1 mm thick and 4 to 5 mm long. Its collagen bundles are oriented obliquely. While less robust than the dorsal band, it functions synergistically with the proximal portion to limit dorsal-palmar rotation and acts as a secondary restraint to distraction. It is intersected by the loose, vascular radioscapholunate ligament (ligament of Testut), which functions more as a neurovascular conduit than a mechanical tether. The volar portion typically fails at approximately 150 N of applied force.
The Proximal (Membranous) Portion is the weakest and thinnest segment, composed predominantly of avascular fibrocartilage. It lacks significant neurovascular penetration, a physiological reality that severely limits its intrinsic healing potential following injury. Mechanically, it acts primarily as a synovial seal between the radiocarpal and midcarpal joints rather than a primary load-bearing structure. A mere 25 N of shear or tensile stress can precipitate failure in this membranous zone.
Kinematics and the Pathogenesis of Instability
To comprehend the necessity of BLB reconstruction, one must understand the inherent kinematic antagonism between the scaphoid and lunate. Under axial loading, the scaphoid naturally tends to flex and pronate, driven by the geometric slope of the scaphoid fossa and the pull of the STT (scaphotrapezio-trapezoidal) articulation. Conversely, the lunate naturally tends to extend, influenced by the triquetrum via the lunatotriquetral (LT) ligament. The intact SLIL acts as the critical mechanical linkage, coupling these opposing forces and ensuring the proximal carpal row moves synchronously.
The typical mechanism of injury is a high-energy fall on an outstretched hand (FOOSH). Mayfield, Johnson, and Kilcoyne elegantly described this pathogenesis as a sequence of perilunate instability driven by an axial load combined with extreme dorsiflexion, ulnar deviation, and midcarpal supination. This force vector drives the capitate proximally, acting as a wedge that violently forces the scaphoid radially and dorsally, while driving the lunate ulnarly and palmarly. The SLIL is the first structure to fail in this sequential cascade (Mayfield Stage I).
When the SLIL is completely ruptured, the vital kinematic connection is severed. The scaphoid, freed from the lunate's extending influence, collapses into severe flexion and pronation. The lunate, now unconstrained by the scaphoid but still tethered to the triquetrum, falls into extension, creating the classic Dorsal Intercalated Segment Instability (DISI) deformity. It is imperative to understand that for a fixed DISI deformity to manifest, the secondary stabilizers—specifically the dorsal extrinsic ligaments—must also be injured or chronically attenuated. The natural history of this uncoupled state is a predictable progression of altered contact stresses, leading directly to the SLAC wrist phenomenon.
Exhaustive Indications and Contraindications
Patient selection is the cornerstone of a successful BLB reconstruction. The procedure is technically demanding and relies on specific biological and mechanical prerequisites. Proper staging of the SL instability, often utilizing the Garcia-Elias classification, is critical for determining surgical candidacy.
| Category | Clinical and Radiographic Parameters |
|---|---|
| Primary Indications | Chronic SLIL tears (> 6 weeks old) where primary repair is impossible due to tissue retraction or poor quality. |
| Reducible SL diastasis and reducible DISI deformity (Garcia-Elias Stages 3 and 4). | |
| Persistent radial-sided wrist pain and subjective weakness failing conservative management. | |
| High-demand patients (e.g., manual laborers, athletes) requiring robust structural restoration. | |
| Absolute Contraindications | Established SLAC wrist arthrosis (Stage II or III) involving the capitolunate joint. |
| Fixed, irreducible DISI deformity (indicates severe secondary ligamentous contracture). | |
| Inadequate bone stock in the scaphoid or lunate (e.g., advanced cystic changes, avascular necrosis). | |
| Active local or systemic infection. | |
| Relative Contraindications | Advanced age or low-demand functional status (where palliative procedures may suffice). |
| SLAC Stage I (radial styloid arthrosis) without concurrent radial styloidectomy. | |
| Poor patient compliance, as the postoperative rehabilitation protocol is rigorous and prolonged. | |
| Heavy tobacco use (significantly impairs bone graft incorporation and soft tissue healing). |
The decision to proceed with a BLB reconstruction hinges on the reducibility of the carpus and the absolute absence of midcarpal arthritis. If the capitate has already migrated proximally and eroded the lunate fossa (SLAC II/III), a BLB graft will fail to alter the degenerative course and will likely exacerbate the patient's pain. In such advanced stages, salvage procedures such as proximal row carpectomy (PRC) or four-corner arthrodesis are the mandated interventions.
Pre-Operative Planning, Templating, and Patient Positioning
Thorough pre-operative planning is the foundation of flawless intraoperative execution. The diagnostic workup must be exhaustive, combining nuanced physical examination techniques with advanced, multi-modal imaging to precisely define the geometry of the instability and the quality of the host bone.
Clinical and Radiographic Evaluation
Patients typically present with chronic, activity-related radial-sided wrist pain, a subjective sense of giving way, and diminished grip strength. On physical examination, exquisite point tenderness is almost universally localized to the dorsal SL interval, situated just distal to Lister's tubercle. Range of motion may be preserved initially but often demonstrates terminal restriction in extension and radial deviation as the scaphoid begins to impinge on the radial styloid.
The cornerstone of the physical examination is the Watson Scaphoid Shift Test. The examiner stabilizes the distal radius and lunate with one hand while applying firm, dorsally directed pressure to the scaphoid tuberosity with the thumb of the other hand. The wrist is then passively moved from ulnar deviation (where the scaphoid is extended) to radial deviation (where the scaphoid normally flexes). In a positive test, the torn SL ligament fails to constrain the scaphoid, causing its proximal pole to dorsally subluxate over the dorsal rim of the radius. Release of thumb pressure results in a palpable, and often audible, "clunk" as the scaphoid reduces back into the elliptical fossa.
Imaging begins with a meticulously acquired plain radiographic series. We require a true posteroanterior (PA), true lateral, scaphoid view, and, crucially, dynamic stress views including clenched-fist PA and ulnar/radial deviation views.
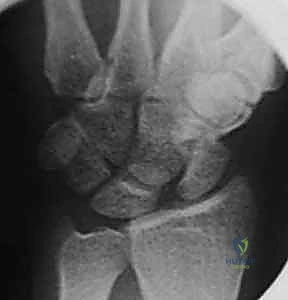
FIG 2 • A. PA radiograph demonstrating an increased scapholunate gap and a scaphoid ring sign. B. A fluoroscopic image of another example of an increased scapholunate gap. C. The contralateral wrist demonstrates a normal scapholunate interval.
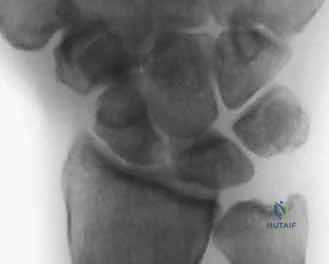
FIG 2 • A. PA radiograph demonstrating an increased scapholunate gap and a scaphoid ring sign. B. A fluoroscopic image of another example of an increased scapholunate gap. C. The contralateral wrist demonstrates a normal scapholunate interval.
On the PA view, we look for the classic static instability signs. An SL interval greater than 3 mm is diagnostic of diastasis, colloquially known as the Terry Thomas sign. Furthermore, as the uncoupled scaphoid collapses into severe flexion, it projects end-on in the coronal plane, creating the pathognomonic "Scaphoid Ring Sign."
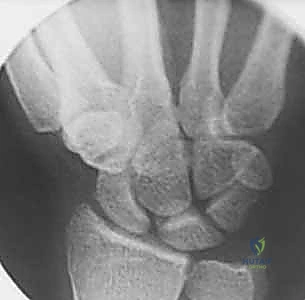
FIG 2 • (continued) D. A lateral radiograph demonstrating an increased scapholunate angle.
On the true lateral radiograph, we assess the intercarpal angles. The normal SL angle ranges from 30 to 60 degrees. An angle exceeding 60 degrees indicates pathological scaphoid flexion. Concurrently, a radiolunate angle greater than 15 degrees of extension confirms the presence of a DISI deformity.

FIG 2 • (continued) E,F. Scapholunate advanced collapse (SLAC) wrist. E. Early-stage arthrosis from a chronic scapholunate ligament injury affecting the radial styloid.
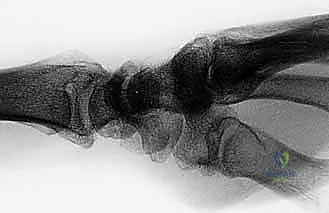
FIG 2 • (continued) F. This PA view on the right demonstrates a more advanced stage. Bone–ligament–bone reconstruction may not be the preferred choice of treatment here.
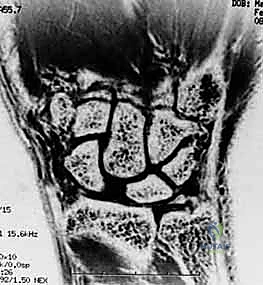
FIG 2 • (continued) F. This PA view on the right demonstrates a more advanced stage. Bone–ligament–bone reconstruction may not be the preferred choice of treatment here.
High-resolution Magnetic Resonance Imaging (MRI), preferably a 3-Tesla scan with dedicated wrist coils, is indispensable. MRI not only confirms the complete rupture of the dorsal SLIL but also allows us to meticulously evaluate the articular cartilage of the radiocarpal and midcarpal joints, ruling out occult SLAC changes that would contraindicate the BLB procedure. Furthermore, MRI assesses the vascularity of the proximal pole of the scaphoid, ensuring adequate biological capacity for graft incorporation.
FIG 2 • (continued) MRI demonstrating a complete rupture of the scapholunate ligament with associated fluid in the interval.
Surgical Setup and Anesthesia
The procedure is performed under regional anesthesia (supraclavicular or axillary brachial plexus block) combined with intravenous sedation, or general anesthesia, depending on patient factors and expected surgical duration. The patient is positioned supine with the operative extremity extended on a radiolucent hand table.
A well-padded pneumatic tourniquet is applied to the proximal arm and inflated to 250 mmHg following exsanguination with an Esmarch bandage. Prophylactic intravenous antibiotics (typically a first-generation cephalosporin) are administered within one hour prior to incision.
Intraoperative fluoroscopy is absolutely critical. The C-arm monitor is positioned directly across from the surgeon for ergonomic viewing. Prior to prepping and draping, a fluoroscopic "time-out" is performed to ensure adequate visualization of the entire carpus in both PA and lateral planes, unobstructed by table hardware. Meticulous skin preparation and sterile draping are performed, leaving the hand and forearm exposed up to the tourniquet.
Step-by-Step Surgical Approach and Fixation Technique
The execution of the BLB reconstruction demands microsurgical precision, rigorous handling of the osteochondral elements, and a deep understanding of carpal kinematics to achieve an anatomic reduction before graft fixation.
Dorsal Approach to the Wrist and Joint Preparation
We begin with a dorsal longitudinal incision, approximately 6 to 8 cm in length, centered over Lister's tubercle and extending distally toward the base of the third metacarpal. Careful subcutaneous dissection is performed to identify and protect the dorsal sensory branches of the radial nerve and the dorsal branch of the ulnar nerve.
The extensor retinaculum is exposed, and the third extensor compartment is opened. The extensor pollicis longus (EPL) tendon is transposed radially. We then elevate the fourth extensor compartment subperiosteally from the radius, retracting the extensor digitorum communis (EDC) tendons ulnarly. This exposes the dorsal wrist capsule.
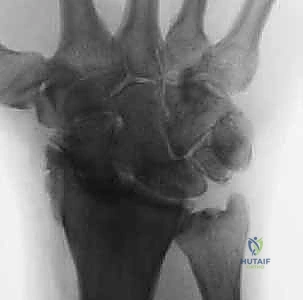
FIG 3 • Extensor retinaculum exposed and compartments identified prior to capsulotomy.
We utilize a ligament-sparing capsulotomy, specifically the Berger dorsal capsular flap. This involves incising the capsule along the axis of the dorsal intercarpal (DIC) ligament and the dorsal radiocarpal (DRC) ligament, creating a radially based, V-shaped flap. This preserves the critical secondary stabilizers and provides excellent exposure of the radiocarpal and midcarpal joints.
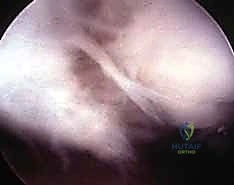
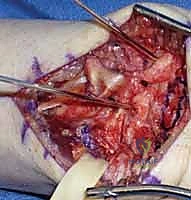
FIG 4 • The Berger dorsal capsular flap elevated, exposing the radiocarpal joint and the scapholunate interval.
Upon entering the joint, the pathology is immediately evident. The SL interval is typically widened and filled with reactive, fibrotic scar tissue. We meticulously debride this fibrous non-union using a combination of a surgical blade, fine rongeurs, and a motorized shaver. It is imperative to clear the interval down to bleeding bone to facilitate eventual fibro-osseous integration, while taking extreme care not to damage the underlying articular cartilage of the proximal scaphoid and lunate.
Carpal Reduction and Trough Preparation
Before any graft can be placed, the carpal kinematics must be perfectly restored. We insert two stout K-wires (0.045 or 0.062 inches) to act as joysticks: one into the dorsal pole of the scaphoid and one into the dorsal aspect of the lunate.
Using these joysticks, the surgeon corrects the DISI deformity. The scaphoid joystick is used to extend and supinate the scaphoid, while the lunate joystick is used to flex the lunate. Once anatomic reduction is confirmed visually and fluoroscopically (restoring the SL angle to approximately 45 degrees and closing the diastasis), the reduction is provisionally secured. We typically drive two 0.045-inch K-wires from the radial side of the scaphoid into the lunate, and a third wire from the scaphoid into the capitate to neutralize the midcarpal joint.
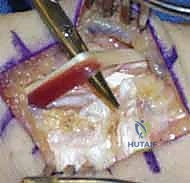
FIG 5 • Joysticks in place reducing the scaphoid and lunate, with provisional K-wire fixation securing the interval.
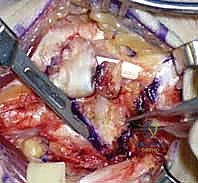
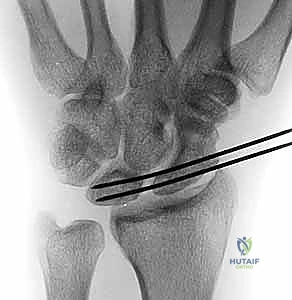
FIG 6 • Fluoroscopic confirmation of the reduced scapholunate interval prior to trough creation.
With the carpus rigidly stabilized, we prepare the recipient troughs in the dorsal aspects of the scaphoid and lunate. Using a high-speed burr with continuous saline irrigation (to prevent thermal necrosis), we create rectangular troughs spanning the SL interval. The dimensions must precisely match our intended graft, typically measuring 4 to 5 mm in width, 4 to 5 mm in depth, and extending 5 to 6 mm into both the scaphoid and lunate.
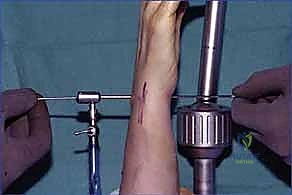
FIG 7 • Creation of the recipient troughs in the dorsal scaphoid and lunate using a high-speed burr.
FIG 8 • The completed recipient troughs, demonstrating bleeding cancellous bone beds ready for graft insertion.
Graft Harvest and Insertion
Several autograft options exist, including the third metacarpal-capitate graft, the iliac crest graft, or the Lister's tubercle-scaphoid graft. For this masterclass, we will describe the harvest of a bone-retinaculum-bone graft from the distal radius, which provides excellent structural integrity and local availability without requiring a secondary surgical site.
We identify a robust section of the extensor retinaculum overlying the distal radius. Using a fine oscillating saw and osteotomes, we harvest two small bone blocks (approximately 4x4x4 mm) connected by a 10 to 12 mm strip of robust retinacular tissue. The graft is carefully elevated, ensuring the soft tissue remains rigidly attached to the bone plugs.
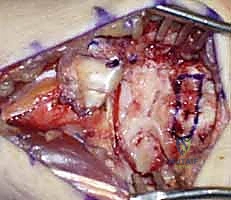
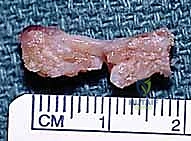
FIG 9 • Harvest of the bone-ligament-bone autograft.
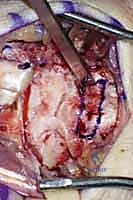
FIG 10 • The harvested BLB graft, demonstrating the two bone plugs connected by the robust ligamentous tissue.
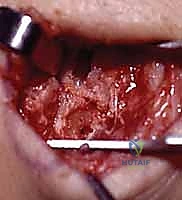
The graft is then transferred to the prepared carpal troughs. The bone plugs are press-fit into the scaphoid and lunate defects. The ligamentous portion must sit flush across the interval, perfectly recreating the dorsal SLIL under appropriate tension.
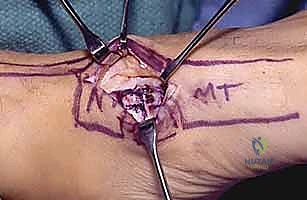
FIG 11 • Insertion of the BLB graft into the recipient troughs.
Fixation of the bone plugs is paramount to achieve primary stability and allow for creeping substitution. We utilize either micro-interference screws (e.g., 2.0 mm or 2.4 mm titanium or PEEK screws), intraosseous wire loops, or rigid suture anchors. The fixation must compress the cancellous bone of the graft against the bleeding bed of the recipient trough.