Mastering Flexor and Extensor Tenosynovectomy: An Intraoperative Masterclass for Rheumatoid Hand Surgery
Key Takeaway
Join us in the OR for a comprehensive masterclass on flexor and extensor tenosynovectomy. This guide provides fellows with detailed surgical anatomy, meticulous step-by-step intraoperative execution, critical pearls, and robust postoperative management strategies for inflammatory tenosynovitis, particularly in rheumatoid arthritis. Master precise dissection, hardware application, and complication avoidance for optimal patient outcomes.
Welcome, fellows, to the operating theater. Today, we are tackling a crucial procedure in the management of rheumatoid arthritis and other inflammatory arthropathies: flexor and extensor tenosynovectomy. This is not merely an exercise in excising inflamed tissue; it is a highly technical, organ-preserving operation designed to maintain tendon excursion, prevent catastrophic rupture, and restore the mechanical integrity of the hand and wrist. As orthopedic surgeons, you must approach this procedure with a profound understanding of the intricate regional anatomy, the meticulous surgical steps required for thorough clearance without devascularization, and the critical intraoperative decision-making necessary to achieve optimal functional outcomes. Let us scrub in and dissect the nuances of this masterclass procedure.
Comprehensive Introduction and Patho-Epidemiology
The synovial membrane is a highly specialized mesenchymal tissue that lines diarthrodial joint spaces and, critically for our surgical focus, the tendon sheaths of the hand and wrist. Its primary physiological role is the secretion of synovial fluid, a viscous, hyaluronic acid-rich ultrafiltrate of blood plasma that serves as an essential boundary lubricant. This lubrication is paramount for minimizing friction during the repetitive, high-excursion gliding of tendons within their respective fibro-osseous canals. When this delicate homeostatic environment is disrupted by systemic autoimmune processes, the resulting pathology fundamentally alters the biomechanics of the entire upper extremity.
In the context of rheumatoid arthritis (RA), the primary indication for tenosynovectomy, the pathophysiology is characterized by chronic, aggressive synovial inflammation. The normal, delicate synovial lining undergoes massive cellular hypertrophy and hyperplasia, transforming into a destructive, locally invasive tissue known as pannus. This pannus is densely populated with activated macrophages, T-lymphocytes, and synovial fibroblasts. These cells create a highly toxic microenvironment by releasing a cascade of pro-inflammatory cytokines, most notably Tumor Necrosis Factor-alpha (TNF-α), Interleukin-1 (IL-1), and Interleukin-6 (IL-6). Furthermore, the hypertrophic synovium secretes copious amounts of matrix metalloproteinases (MMPs) and collagenases, which directly and enzymatically degrade the structural Type I collagen fibers of the tendon substance.
Beyond enzymatic degradation, the sheer volume of the proliferating pannus introduces a severe mechanical detriment. As the hypertrophic synovium expands within the confined, rigid spaces of the extensor compartments or the digital flexor sheaths, it causes acute mechanical impingement. This leads to a dramatic increase in friction during tendon excursion, creating a vicious cycle of microtrauma, further inflammation, and progressive ischemic necrosis of the tendon due to vascular compromise of the mesotendon and vincula. The increased pressure within these closed spaces, particularly the carpal tunnel, also precipitates secondary compressive neuropathies, most commonly affecting the median nerve.
The natural history of untreated rheumatoid tenosynovitis is inexorable tendon attrition and eventual rupture. This process is clinically insidious; inflammatory tenosynovitis in RA can often be surprisingly painless in its early stages, masking the underlying structural degradation. Tendon rupture is a devastating complication that severely impairs hand function and requires complex salvage reconstructions. Specific anatomical zones are highly susceptible to this attrition. The Vaughan-Jackson syndrome describes the sequential rupture of the digital extensors, typically beginning with the extensor digiti minimi (EDQ) and progressing radially, driven by a combination of invasive synovitis and mechanical abrasion against a dorsally subluxated, eroded distal ulna. Similarly, the Mannerfelt lesion refers to the attrition rupture of the flexor pollicis longus (FPL) tendon as it continuously glides over a sharp, osteophytic spur projecting from the palmar aspect of the scaphoid or trapezium, secondary to scaphotrapeziotrapezoid (STT) joint destruction.
Detailed Surgical Anatomy and Biomechanics
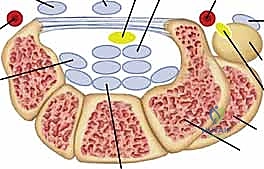
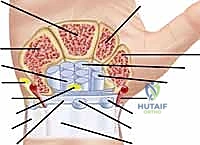
Mastery of flexor and extensor tenosynovectomy demands an encyclopedic knowledge of the wrist and hand's compartmental anatomy. The extensor tendons are organized into six distinct fibro-osseous compartments beneath the extensor retinaculum, a thickened band of deep fascia that prevents tendon bowstringing during active wrist extension. The first compartment houses the abductor pollicis longus (APL) and extensor pollicis brevis (EPB). Surgeons must be acutely aware of the superficial branch of the radial nerve (SBRN), which emerges from beneath the brachioradialis and arborizes directly over or adjacent to this compartment. Furthermore, the first compartment frequently contains multiple sub-slips of the APL and a separate sub-compartment for the EPB; failure to release and debride these anomalous septations is a primary cause of recurrent De Quervain's-like symptoms postoperatively.

Moving ulnarly, the second compartment contains the extensor carpi radialis longus (ECRL) and brevis (ECRB), the primary wrist extensors. The third compartment is highly specialized, containing only the extensor pollicis longus (EPL). The EPL takes an acute, 45-degree ulnar turn around Lister's tubercle on the dorsal radius. This fulcrum provides the biomechanical advantage necessary for the EPL to elevate the thumb out of the palmar plane, but it also creates a zone of high mechanical stress and relative hypovascularity, making the EPL highly vulnerable to attrition rupture from underlying radiocarpal synovitis or bony irregularity. The fourth compartment is the most capacious, housing the four slips of the extensor digitorum communis (EDC) and the extensor indicis proprius (EIP). The terminal branch of the posterior interosseous nerve (PIN) lies deep to the fourth compartment on the interosseous membrane, providing nociceptive innervation to the dorsal wrist capsule; while not directly in the superficial field, it is encountered during deep capsulotomies or synovectomies extending into the radiocarpal joint.

The fifth and sixth compartments possess unique biomechanical and structural characteristics. The fifth compartment contains the extensor digiti minimi (EDQ), which is often the sole independent extensor of the small finger metacarpophalangeal (MCP) joint. The sixth compartment houses the extensor carpi ulnaris (ECU). Unlike the other compartments, the ECU is stabilized within the ulnar groove by a distinct, robust fibrous subsheath that is separate from the overlying extensor retinaculum. This subsheath is critical for stabilizing the ECU tendon during forearm supination and wrist flexion. Furthermore, the ECU and its subsheath are integral dynamic stabilizers of the distal radioulnar joint (DRUJ). Aggressive surgical disruption of this subsheath during synovectomy, without meticulous reconstruction, will result in painful ECU subluxation and exacerbation of DRUJ instability.
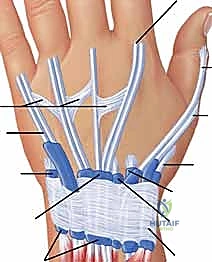
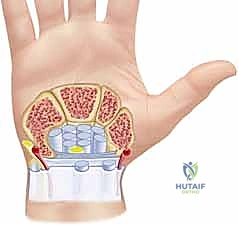
On the volar aspect, the digital flexor tendons (Flexor Digitorum Superficialis [FDS] and Flexor Digitorum Profundus [FDP]) transition from an extrasynovial environment in the distal forearm to an intrasynovial environment as they enter the carpal tunnel. Within the carpal tunnel, the nine flexor tendons (including the FPL) and the median nerve are tightly packed beneath the thick transverse carpal ligament. The hypertrophic synovium here not only degrades the tendons but also dramatically increases intracompartmental pressure, leading to ischemic compression of the median nerve. Distally, within the digits, the flexor tendons glide within a highly specialized fibro-osseous canal formed by a series of annular (A1-A5) and cruciate (C1-C3) pulleys. The A2 and A4 pulleys, located over the proximal and middle phalanges respectively, are the primary biomechanical anchors that prevent flexor tendon bowstringing. During digital flexor tenosynovectomy, the absolute preservation of the A2 and A4 pulleys is a non-negotiable surgical mandate to maintain the moment arm and mechanical efficiency of digital flexion.
Exhaustive Indications and Contraindications
The decision to proceed with operative tenosynovectomy requires a nuanced evaluation of the patient's clinical trajectory, responsiveness to medical management, and the imminent threat of structural tendon failure. The primary indication for tenosynovectomy is chronic, proliferative tenosynovitis that has remained refractory to an optimized regimen of disease-modifying antirheumatic drugs (DMARDs) and biologic therapies for a period of three to six months. While modern rheumatologic protocols have drastically reduced the incidence of massive rheumatoid deformities, a subset of patients will exhibit persistent, localized synovial hypertrophy that escapes systemic control. In these instances, surgical intervention is prophylactic, aimed at halting the mechanical and enzymatic destruction of the tendon before irreversible rupture occurs.
A more urgent indication arises when there is clinical evidence of impending tendon rupture or early attritional changes. This may manifest as a sudden change in the character of the swelling, the onset of localized pain over critical fulcrums (such as Lister's tubercle or the distal ulna), or a subtle loss of active range of motion despite full passive mobility. The presence of a "triggering" digit that fails corticosteroid injection, or the development of a palpable nodule within the tendon substance, strongly suggests invasive pannus that requires immediate surgical excision. Furthermore, the actual rupture of one or more tendons is an absolute indication for exploration, tenosynovectomy of the remaining intact tendons to prevent sequential failure, and concurrent tendon transfer or grafting to restore lost function.
Contraindications to tenosynovectomy are generally relative and center on patient optimization and the structural integrity of the surrounding tissues. A severe, active systemic flare of rheumatoid arthritis is a contraindication; surgery should be delayed until the systemic inflammatory burden is medically suppressed to minimize the risk of postoperative disease exacerbation and profound stiffness. Poor soft tissue envelopes, characterized by paper-thin, fragile rheumatoid skin with active ulceration or severe vasculitis, represent a significant risk for catastrophic wound breakdown and deep infection. In such cases, plastic surgery consultation for flap coverage may be necessary prior to or concurrent with tendon surgery.
| Category | Specific Factors | Clinical Rationale / Management Strategy |
|---|---|---|
| Indications | Refractory Synovitis | Persistent swelling/pain >3-6 months despite optimized DMARD/Biologic therapy. |
| Indications | Impending Rupture | Pain over Lister's tubercle, distal ulna, or STT joint; subtle lag in active extension/flexion. |
| Indications | Active Tendon Rupture | Loss of active motion (e.g., dropped finger, loss of thumb IP flexion). Requires concurrent tendon transfer/reconstruction. |
| Indications | Compressive Neuropathy | Severe carpal tunnel syndrome secondary to florid flexor tenosynovitis. |
| Contraindications (Absolute) | Active Local Infection | Cellulitis or septic arthritis in the operative field. Requires eradication prior to elective synovectomy. |
| Contraindications (Relative) | Severe Systemic Flare | High systemic inflammatory markers; risk of postoperative stiffness and poor wound healing. Optimize medically first. |
| Contraindications (Relative) | End-Stage Joint Destruction | If the underlying joint is completely destroyed and fixed, isolated tenosynovectomy will not restore function; requires arthrodesis/arthroplasty. |
| Contraindications (Relative) | Fragile/Compromised Skin | Severe rheumatoid vasculitis or paper-thin skin. High risk of wound necrosis. Consider modified incisions or flap coverage. |
Furthermore, the presence of fixed, end-stage joint deformities (e.g., a rigidly subluxated radiocarpal joint or a fixed swan-neck deformity) alters the surgical algorithm. Performing an isolated tenosynovectomy over a severely destroyed, unstable joint is a futile endeavor, as the underlying bony pathology will rapidly destroy the tendons again. In these scenarios, tenosynovectomy must be combined with definitive osseous stabilization, such as a radiocarpal arthrodesis, distal ulna resection (Darrach procedure), or total wrist arthroplasty, to provide a stable, smooth bed for the tendons to glide across.
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the foundation of a successful tenosynovectomy. The clinical examination remains paramount, but advanced imaging modalities are increasingly utilized to delineate the extent of the pathology. High-resolution ultrasonography is an invaluable, dynamic, and cost-effective tool. It allows the surgeon to visualize the volume of the synovial pannus, assess the presence of hypervascularity (via power Doppler), and dynamically evaluate tendon integrity and gliding during active motion. Magnetic Resonance Imaging (MRI) without and with intravenous contrast provides a comprehensive, static roadmap of the wrist and hand. It exquisitely details the extent of tendon substance invasion, fluid within the sheaths, and concurrent joint pathology such as erosions or ligamentous tears, aiding in the surgical decision to incorporate joint procedures.
Medical optimization requires close collaboration with the patient's rheumatologist. The perioperative management of biologic and non-biologic DMARDs is a critical component of surgical planning. According to current American College of Rheumatology (ACR) and American Association of Hip and Knee Surgeons (AAHKS) guidelines, traditional DMARDs like Methotrexate and Hydroxychloroquine are generally continued through the perioperative period, as the risk of a systemic flare outweighs the minimal risk of wound complications. However, biologic agents (e.g., TNF inhibitors like Adalimumab or Etanercept) should typically be withheld prior to surgery, timing the procedure for the end of the dosing cycle, and restarted only after complete wound healing (usually 14-21 days postoperatively) to mitigate the risk of deep surgical site infections.
Patient positioning for hand and wrist tenosynovectomy requires standard upper extremity protocols. The patient is placed supine with the operative arm extended on a radiolucent hand table. A well-padded pneumatic tourniquet is applied to the proximal arm. Exsanguination with an Esmarch bandage is performed prior to tourniquet inflation, which is typically set at 250 mmHg or 100 mmHg above the patient's systolic blood pressure. The use of a tourniquet is absolutely essential to provide the bloodless field required for the meticulous, microscopic dissection of pannus from the delicate tendon substance.
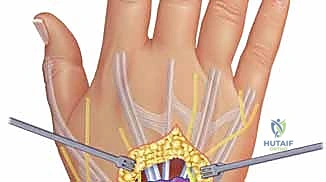
The surgical setup must include magnification loupes (minimum 2.5x to 3.5x) to safely identify and protect small cutaneous nerves and the intricate vascular supply of the tendons. Fine-tipped tenotomy scissors, delicate pickups (such as Adson or Bishop-Harmon forceps), and bipolar electrocautery are mandatory instruments. The surgeon must also be prepared with a variety of tendon repair and reconstruction materials, including non-absorbable braided sutures (e.g., 3-0 or 4-0 Ethibond or FiberWire) for tendon transfers, and monofilament sutures for skin closure, anticipating the need to address unexpected tendon ruptures discovered upon opening the compartments.
Step-by-Step Surgical Approach and Fixation Technique
Dorsal Extensor Tenosynovectomy and Retinacular Reconstruction
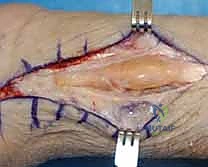
The dorsal approach begins with a longitudinal, slightly curvilinear incision centered over the dorsal wrist, typically extending from the distal third of the radius to the mid-metacarpal level. Straight incisions directly over the wrist crease should be avoided to prevent volar flexion contractures. Full-thickness fasciocutaneous flaps are meticulously elevated. The superficial sensory branches of the radial nerve (SBRN) radially, and the dorsal cutaneous branch of the ulnar nerve (DCBUN) ulnarly, must be identified, mobilized, and gently retracted with vessel loops. Injury to these nerves results in debilitating, painful neuromas that can overshadow a successful tenosynovectomy.
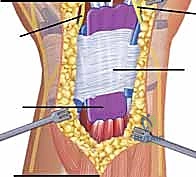
Exposure of the extensor compartments involves elevating the extensor retinaculum. A preferred technique is a step-cut or an ulnar-based flap. The retinaculum is incised longitudinally over the third compartment (EPL). The radial half is elevated to expose the first and second compartments, while the ulnar half is elevated to expose the fourth, fifth, and sixth compartments. Care is taken to preserve the deep septa attaching the retinaculum to the radius where possible, though these are often destroyed by the disease process. Once the compartments are unroofed, the true extent of the tenosynovitis is revealed. The hypertrophic, often grayish-purple pannus is systematically excised.
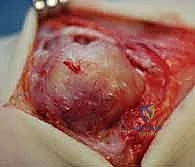
The tenosynovectomy must be thorough but respectful of the tendon's intrinsic blood supply. Using tenotomy scissors and blunt dissection, the pannus is stripped from the epitenon. If the pannus has invaded the tendon substance, it must be sharply excised, even if this results in thinning of the tendon. It is better to leave a thinned, healthy tendon than a thick tendon riddled with destructive inflammatory tissue. If a tendon is found to be ruptured (most commonly the EPL or EDQ), a reconstructive plan is immediately executed. For an EPL rupture, an Extensor Indicis Proprius (EIP) to EPL transfer is the gold standard. The EIP is harvested at the level of the index MCP joint, rerouted subcutaneously, and woven into the distal stump of the EPL using a Pulvertaft weave, tensioned with the wrist in neutral and the thumb in full extension. Following the synovectomy, the extensor retinaculum is reconstructed. To prevent recurrent attrition, the retinaculum is often placed deep to the extensor tendons, creating a smooth fascial floor over the joint capsule, leaving the tendons in a subcutaneous position.
Volar Flexor Tenosynovectomy and Carpal Tunnel Release
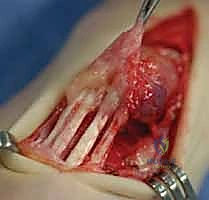
The volar approach requires an extended carpal tunnel incision. The incision begins in the palm, paralleling the thenar crease, and extends proximally across the wrist crease in a zig-zag or Brunner fashion to prevent scar contracture. The superficial palmar fascia is incised, and the transverse carpal ligament is completely divided under direct vision, ensuring the absolute protection of the median nerve and its recurrent motor branch. The median nerve is identified, neurolysed if necessary, and gently retracted radially with a Penrose drain.
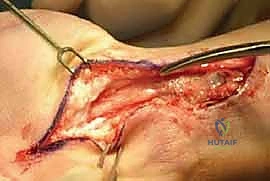
Within the carpal tunnel, the flexor tendons are often encased in a massive, boggy tenosynovium. The synovectomy begins by separating the individual FDS and FDP tendons. The visceral synovium enveloping each tendon is meticulously stripped using a combination of sharp dissection and wiping with a moist sponge. Particular attention must be paid to the FPL tendon, tracing it proximally and distally to ensure clearance of any Mannerfelt-type lesions near the STT joint.
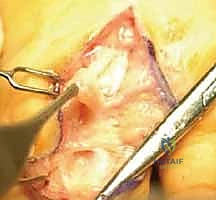
If a flexor tendon rupture is identified, primary repair is rarely possible due to tendon retraction and substance loss. For an FPL rupture, options include an FDS transfer (typically using the FDS of the ring or middle finger) or a free tendon graft (e.g., palmaris longus) if the proximal muscle belly remains functional. If the distal stump of the FPL is inadequate, an interphalangeal (IP) joint arthrodesis is a highly reliable salvage procedure that restores strong pinch mechanics. After thorough clearance of the carpal tunnel, the tourniquet is deflated, meticulous hemostasis is achieved using bipolar cautery, and the skin is closed loosely to accommodate postoperative swelling. The transverse carpal ligament is left open.
Digital Flexor Tenosynovectomy
Digital flexor tenosynovectomy is indicated for persistent triggering or loss of active flexion due to intrathecal pannus. The approach utilizes mid-axial or volar Brunner (zig-zag) incisions over the affected digit. The neurovascular bundles are identified and protected throughout the dissection. The flexor tendon sheath is exposed.
The critical step in this region is the strategic venting of the pulley system to allow for synovectomy while absolutely preserving the biomechanically essential A2 and A4 pulleys. The A1, A3, and A5 pulleys, along with the cruciate pulleys, can be safely incised or partially excised to gain access to the tendons. The inflamed synovium is stripped from the FDS and FDP tendons. The relationship between the FDS and FDP at Camper's chiasm must be carefully inspected and cleared of invasive tissue. The tendons must be pulled distally and proximally into the wound to ensure clearance extending into the palm and distal phalanx. If the FDS is severely infiltrated and non-functional, it may be excised (FDS tenectomy) to decompress the sheath and improve the excursion of the intact FDP tendon.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, flexor and extensor tenosynovectomy in the rheumatoid patient carries a distinct complication profile. The systemic nature of the disease, combined with the immunosuppressive effects of medical therapies and the inherent fragility of rheumatoid tissues, elevates the risk of adverse events compared to non-inflammatory cohorts. Recognizing these complications early and possessing a robust armamentarium of salvage strategies is essential for the operating surgeon.
Wound healing complications are among the most frequent postoperative issues, with delayed healing, marginal necrosis, or frank dehiscence occurring in approximately 5% to 15% of cases. The dorsal wrist skin, in particular, is often atrophic and heavily dependent on a fragile subdermal plexus. Overly aggressive retraction, excessive electrocautery, or closure under tension will inevitably lead to skin slough. Superficial infections can usually be managed with targeted oral antibiotics and local wound care. However, deep space infections, though rare (<2%), are catastrophic, threatening the integrity of the newly cleared tendons and requiring urgent operative debridement, copious irrigation, and prolonged intravenous antibiotic therapy.
Recurrent tenosynovitis is a frustrating but recognized complication, occurring in 10% to 20% of patients over long-term follow-up. This is often driven by inadequate initial synovectomy (leaving microscopic rests of pannus) or a failure of medical management to control the systemic disease flare. Recurrence presents as returning pain, swelling, and a decline in tendon excursion. Postoperative tendon rupture, occurring in 2% to 5% of cases, can happen despite an adequate synovectomy if the tendon substance was severely compromised prior to surgery and subsequently fails under the loads of early rehabilitation.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage Management Strategy |
|---|---|---|---|
| Wound Dehiscence / Necrosis | 5% - 15% | Fragile rheumatoid skin, chronic steroid use, excessive retraction, closure under tension. | Local wound care, secondary intent healing. Severe cases require split-thickness skin grafting or local rotational flaps. |
| Recurrent Tenosynovitis | 10% - 20% | Inadequate surgical clearance, uncontrolled systemic disease, failure to optimize DMARDs. | Medical optimization first. Repeat tenosynovectomy if mechanically symptomatic or threatening rupture. |
| Postoperative Tendon Rupture | 2% - 5% | Pre-existing severe attritional thinning, aggressive early passive stretching, recurrent synovitis. | Tendon transfer (e.g., EIP to EPL), free tendon grafting, or joint arthrodesis if tendon reconstruction is unfeasible. |
| Nerve Injury (SBRN, DCBUN) | 1% - 3% | Iatrogenic transection or aggressive traction during flap elevation. | Immediate microsurgical repair if recognized intraoperatively. Late presentation requires neuroma excision and burying the stump into muscle/bone. |
| Tendon Bowstringing | < 1% | Iatrogenic destruction of A2/A4 pulleys (digits) or failure to reconstruct extensor retinaculum. | Pulley reconstruction using free tendon grafts (e.g., palmaris longus) or extensor retinacular slings. |
Iatrogenic nerve injuries, particularly to the SBRN or DCBUN during dorsal approaches, result in painful neuromas that severely limit patient satisfaction. Prevention through meticulous dissection is paramount. If a nerve is inadvertently transected, immediate primary microsurgical epineural repair is required. Biomechanical complications, such as tendon bowstringing, occur if the critical restraining structures (A2/A4 pulleys in the digits, or the extensor retinaculum at the wrist) are destroyed and not adequately reconstructed. Bowstringing drastically reduces the mechanical advantage and excursion of the tendon, leading to weakness and incomplete range of motion, necessitating complex secondary reconstructive procedures using free tendon grafts to recreate the pulley systems.
Phased Post-Operative Rehabilitation Protocols
The surgical tenosynovectomy is only the first half of the therapeutic equation; the ultimate functional outcome is heavily dependent on a rigorous,
Clinical & Radiographic Imaging Archive