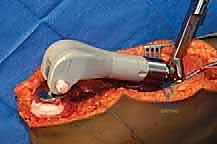
Comprehensive Introduction and Patho-Epidemiology
Welcome to the operating theater and the rigorous intellectual landscape of orthopaedic oncology. Today, we are not merely performing a routine arthroplasty; we are delving deep into the art and science of endoprosthetic reconstruction, which remains the absolute cornerstone of modern oncologic limb salvage. This discipline transcends the simple mechanical replacement of bone. It is fundamentally about the meticulous, margin-negative excision of aggressive disease, the restoration of complex biomechanical function, and the preservation of patient dignity and quality of life. The journey of limb salvage over the past century has been nothing short of revolutionary, transitioning from the grim, definitive certainty of amputation to a highly sophisticated realm of reconstructive possibilities driven by materials science, advanced imaging, and multidisciplinary oncology.

For decades, the standard, unquestioned surgical approach to malignant tumors of the extremities—particularly high-grade osteosarcomas and Ewing sarcomas—was immediate, radical amputation. This was the prevailing orthodoxy of the 1960s and 1970s, driven entirely by the imperative to achieve local disease control in an era before effective systemic therapies. Surgeons operated under the assumption that the widest possible anatomic barrier was the only defense against local recurrence. However, despite the diligent work of early orthopaedic oncologists in defining highly specific amputation levels and managing complex, massive soft-tissue wounds, these aggressive resections tragically failed to impact overall patient survival. Metastatic disease, primarily pulmonary, remained the relentless and primary cause of mortality, rendering the amputations largely palliative in the grand scheme of the disease process.
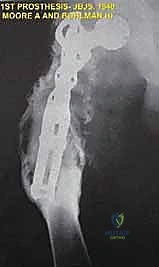
If we trace the lineage of our current techniques back to 1940, we find Austin Moore and Harold Bohlman publishing the first documented endoprosthetic reconstruction. They utilized a custom-cast Vitallium proximal femoral prosthesis for a patient with a massive giant cell tumor. This was a pioneering, audacious step that proved massive metallic implants could be tolerated by the human body. Yet, the true paradigm shift arrived later, in the early 1970s, ushered in by innovators like Kenneth Francis and Joseph Marcove. These pioneers developed the first generation of custom prostheses specifically designed for distal and total femoral replacement, primarily targeting patients with osteosarcomas who refused amputation.
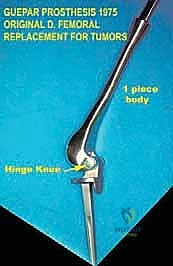
However, a significant, life-threatening hurdle quickly emerged in this early era of limb salvage: these were entirely custom-manufactured implants. The process required meticulous measurements from plain radiographs, followed by a 6- to 12-week manufacturing delay. During this prolonged interval, highly aggressive tumors inevitably progressed, often rendering the planned limb-salvage procedure impossible or resulting in devastating systemic metastasis. This specific logistical challenge directly catalyzed the birth of induction chemotherapy—what we now universally refer to as neoadjuvant chemotherapy. By employing newly proven, highly toxic but effective agents like high-dose methotrexate and doxorubicin during the implant manufacturing interval, oncologists could effectively halt tumor progression and treat micrometastatic disease. It was only with the introduction of these effective systemic protocols that alternatives to amputation truly became oncologically viable, expanding limb salvage from a highly selected few to the standard of care for 90-95% of extremity sarcomas today.
Detailed Surgical Anatomy and Biomechanics
To execute a flawless oncologic resection and subsequent endoprosthetic reconstruction, an absolute mastery of three-dimensional surgical anatomy and the biomechanical forces acting upon the reconstructed limb is mandatory. Let us consider the proximal femur, a frequent site for both primary bone sarcomas and destructive metastatic carcinomas. The proximal femur is enveloped by a complex, unforgiving array of musculature and neurovascular structures. The muscular envelope includes the gluteus medius and minimus (the primary abductors essential for a trendelenburg-free gait), the iliopsoas (the primary flexor), the adductor group medially, and the massive quadriceps anteriorly. Oncologic resections often require the sacrifice of one or more of these muscle groups to achieve negative margins, necessitating complex soft-tissue transfers or the acceptance of permanent functional deficits.
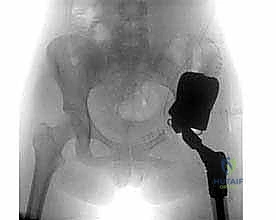
The neurovascular anatomy in this region demands meticulous dissection and profound respect. Anteriorly and medially lie the femoral nerve, artery, and vein. As we teach our residents, the relationship is critical: the femoral nerve lies lateral to the femoral artery, which in turn is lateral to the femoral vein (represented by the classic NAVEL mnemonic: Nerve, Artery, Vein, Empty space, Lymphatics, moving lateral to medial). Posteriorly, the sciatic nerve descends deep to the gluteus maximus and rests upon the short external rotators. Inadvertent traction, thermal injury from electrocautery, or direct transection of these structures during aggressive tumor mobilization will result in devastating, irreversible motor, sensory, and vascular compromise, effectively negating the functional benefits of limb salvage.
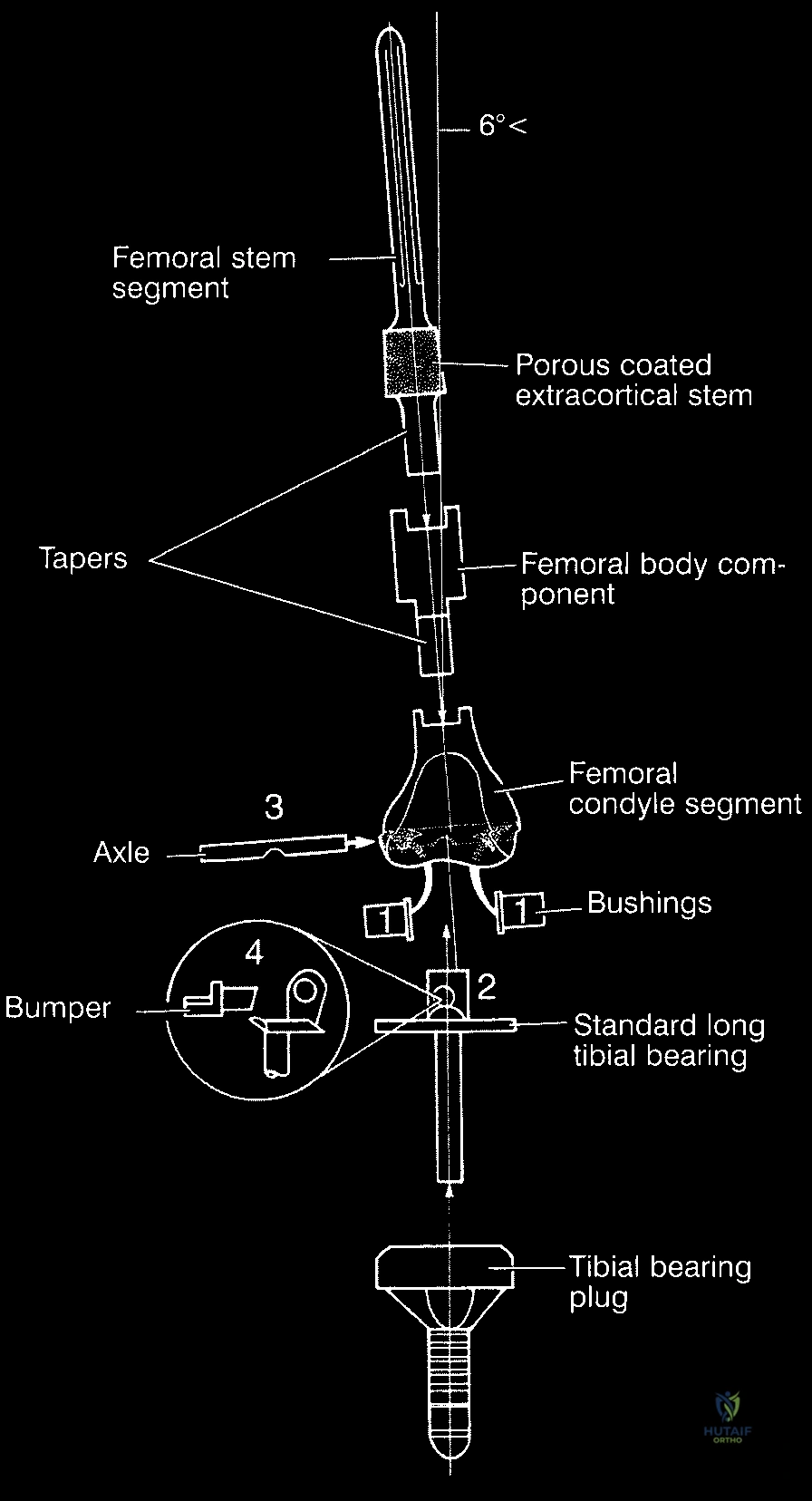
Biomechanically, the evolution of endoprosthetic design reflects a continuous battle against the immense forces transmitted across the major joints. Early custom implants, while revolutionary, suffered from catastrophic design flaws. The most common mode of mechanical failure in the 1980s was stem breakage or bending. This was typically due to inadequate stem diameters or the presence of severe stress risers caused by the sharp, un-radiused transition from the massive prosthetic body to the intramedullary stem. Advances in metallurgy, specifically the transition to forged titanium and cobalt-chromium alloys, along with the application of finite element analysis, allowed engineers to eliminate these stress risers and significantly improve the fatigue strength of "mega" prostheses.
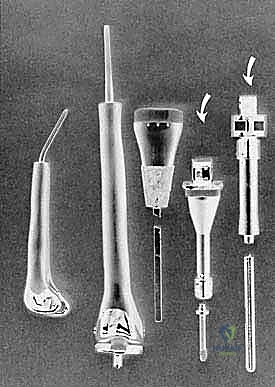
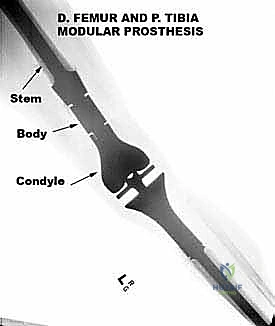
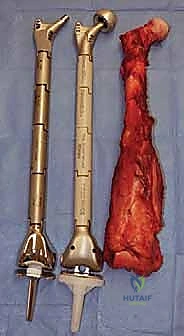
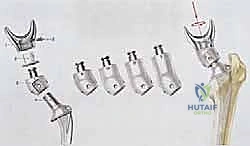
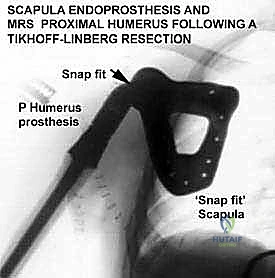
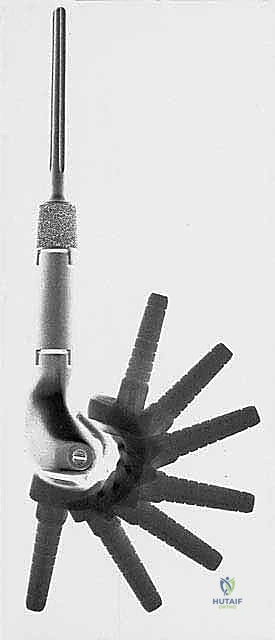
The most critical biomechanical leap, however, was the transition from custom to modular designs. Borrowing heavily from the booming total hip and knee arthroplasty markets, manufacturers developed interchangeable systems. While modularity introduced new potential failure points—specifically at the morse taper junctions between components—the benefits were undeniable. Systems like the Waldemar-Link Saddle Endoprosthesis for periacetabular resections, and the Howmedica Modular Replacement System (HMRS), allowed surgeons to intraoperatively adjust the length and rotation of the reconstruction. Today's third-generation systems, such as the Global Modular Replacement System (GMRS), utilize fluted, plasma-sprayed cementless stems that encourage extracortical bone bridging, fundamentally altering the biomechanical load transfer and reducing the incidence of stress shielding and aseptic loosening that plagued earlier cemented designs.
Exhaustive Indications and Contraindications
The decision to perform an endoprosthetic reconstruction is highly nuanced and requires a rigorous evaluation of oncologic, anatomic, and patient-specific factors. The primary indication remains the surgical management of primary malignant bone tumors of the appendicular skeleton, including osteosarcoma, Ewing sarcoma, and chondrosarcoma, where an en bloc resection of the affected bone segment is required to achieve wide margins. Furthermore, endoprostheses are increasingly utilized in the management of aggressive benign bone tumors (such as massive giant cell tumors with extensive cortical destruction) and in patients with solitary or oligometastatic bone disease presenting with impending or actual pathologic fractures that are not amenable to standard osteosynthesis techniques.
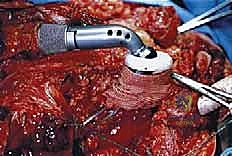
Beyond the realm of oncology, the indications for these massive implants have expanded significantly into complex revision arthroplasty and severe orthopaedic trauma. In cases of catastrophic aseptic loosening, periprosthetic joint infection with massive cavitary or segmental bone loss, or highly comminuted periarticular fractures in osteoporotic bone where standard fixation will inevitably fail, modular endoprostheses provide immediate structural stability and allow for rapid mobilization. This "off-label" use of oncologic implants has become a vital tool in the armamentarium of the complex joint reconstruction surgeon.
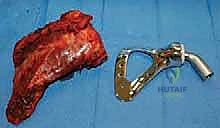
However, stringent contraindications must be respected to avoid catastrophic outcomes. Absolute contraindications include the inability to achieve negative oncologic margins without sacrificing the critical neurovascular bundle required for a functional and viable extremity. If the major motor nerves or the primary arterial inflow must be resected to clear the tumor, and reconstruction is impossible or will leave a flail, insensate limb, an amputation is functionally and oncologically superior. Additionally, active, uncontrollable local infection precludes the implantation of massive metallic hardware, as the biofilm burden will inevitably lead to systemic sepsis and implant failure.

Relative contraindications require careful multidisciplinary consideration. In skeletally immature patients, standard endoprostheses will result in significant limb length discrepancies as the contralateral physis continues to grow; here, expandable prostheses (either mechanically or electromagnetically lengthened) are indicated, though they carry higher complication rates. Severe patient comorbidities, profound malnutrition, or a severely compromised soft-tissue envelope (due to prior radiation or massive tumor fungation) also serve as relative contraindications, as the risk of catastrophic wound breakdown and subsequent deep periprosthetic infection may outweigh the benefits of limb salvage.
| Indication Category | Specific Conditions | Contraindication Category | Specific Conditions |
|---|---|---|---|
| Primary Malignancies | Osteosarcoma, Ewing Sarcoma, Chondrosarcoma requiring segmental resection. | Absolute Oncologic | Inability to achieve R0 margins without sacrificing critical neurovascular structures. |
| Metastatic Disease | Destructive lesions (renal cell, thyroid, breast) with actual/impending pathologic fracture. | Absolute Infectious | Active, uncontrolled local or systemic infection. |
| Aggressive Benign | Massive Giant Cell Tumor, recurrent Aneurysmal Bone Cyst with joint destruction. | Relative Anatomic | Severely compromised soft tissue envelope precluding implant coverage. |
| Non-Oncologic Salvage | Massive bone loss in revision arthroplasty, severe comminuted periarticular trauma. | Relative Patient Factors | Extreme medical comorbidities, skeletal immaturity (requires expandable implants). |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the absolute blueprint for success in oncologic limb salvage; battles are won and lost before the scalpel ever touches the skin. This phase begins with an exhaustive review of advanced imaging modalities. Magnetic Resonance Imaging (MRI) is the gold standard for defining the intraosseous marrow extent of the tumor, identifying "skip" metastases within the same bone, and precisely delineating the extraosseous soft-tissue mass and its relationship to critical neurovascular bundles. T1-weighted longitudinal images are essential for determining the exact level of the planned bone osteotomy, while axial STIR or T2-weighted images map the soft-tissue margins. Computed Tomography (CT) is utilized to assess cortical integrity, identify subtle pathologic fractures, and evaluate the precise geometry of the remaining bone stock for implant fixation.
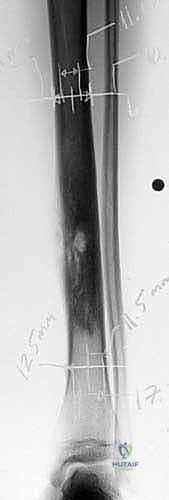
A critical, non-negotiable aspect of the preoperative plan is the management of the previous biopsy tract. The biopsy tract must be considered contaminated with microscopic tumor cells. Therefore, the surgical incision must be meticulously planned to encompass the entire biopsy tract, ensuring it is excised en bloc with the main tumor specimen. Failure to do so dramatically increases the risk of local recurrence. Incisions should be longitudinal and extensile, avoiding transverse components that compromise collateral blood flow and limit future reconstructive options if flap coverage becomes necessary.
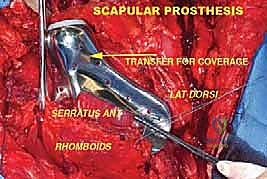
Digital templating using the chosen modular endoprosthetic system is mandatory. By overlaying digital templates onto appropriately scaled orthogonal radiographs and CT scanograms, the surgeon estimates the required resection length, the appropriate diameter and length of the intramedullary stems, and the necessary modular body segments to restore limb length, offset, and version. However, a master surgeon knows that templating provides a roadmap, not a contract. Modularity grants intraoperative flexibility, allowing the surgical team to adjust to unexpected tumor extension. Contingency planning is vital; backup implant sizes, alternate reconstructive systems, and even formal consent for amputation must be secured in the event that safe oncologic margins cannot be achieved with limb salvage.
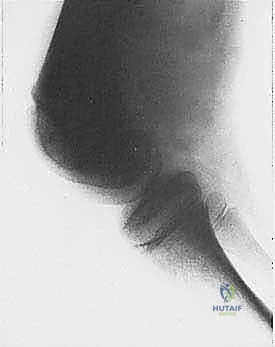
Patient positioning in the operating room must facilitate both the complex resection and the subsequent reconstruction, while allowing for intraoperative fluoroscopy. Patients are typically positioned supine or in the lateral decubitus position on a radiolucent table, depending on the anatomic location of the tumor. The entire limb is prepped and draped free to allow for dynamic assessment of joint stability, range of motion, and limb length comparison. A multidisciplinary approach is essential; if a massive soft-tissue defect is anticipated, plastic and reconstructive surgical colleagues must be integrated into the preoperative plan to ensure immediate availability for pedicled or free tissue transfer, such as a gastrocnemius or latissimus dorsi flap, to achieve robust coverage of the mega-prosthesis.
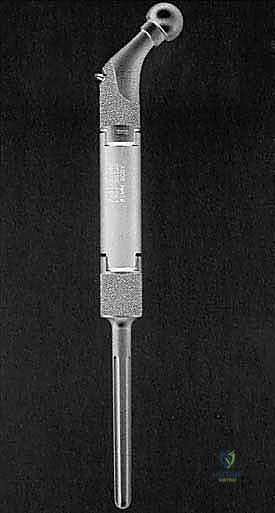
Step-by-Step Surgical Approach and Fixation Technique
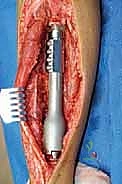
A successful limb-sparing procedure rests upon three interdependent stages, performed sequentially and with absolute precision: Tumor Resection with Appropriate Oncologic Margins, Reconstruction and Stabilization of the Involved Bone and Joints, and Restoration of the Soft Tissue Envelope. The first stage is non-negotiable. Our primary goal is always complete, en bloc tumor removal with clear, wide (R0) margins. The dissection proceeds from normal, healthy tissue toward the tumor pseudocapsule, taking a cuff of normal muscle and fascia. The neurovascular bundle is meticulously skeletonized and protected, often requiring the ligation of major venous and arterial branches feeding the tumor. The bone osteotomy is performed at the pre-determined level, typically 2 to 3 centimeters beyond the MRI-defined marrow extent, using a cool oscillating saw to prevent thermal necrosis of the host bone.

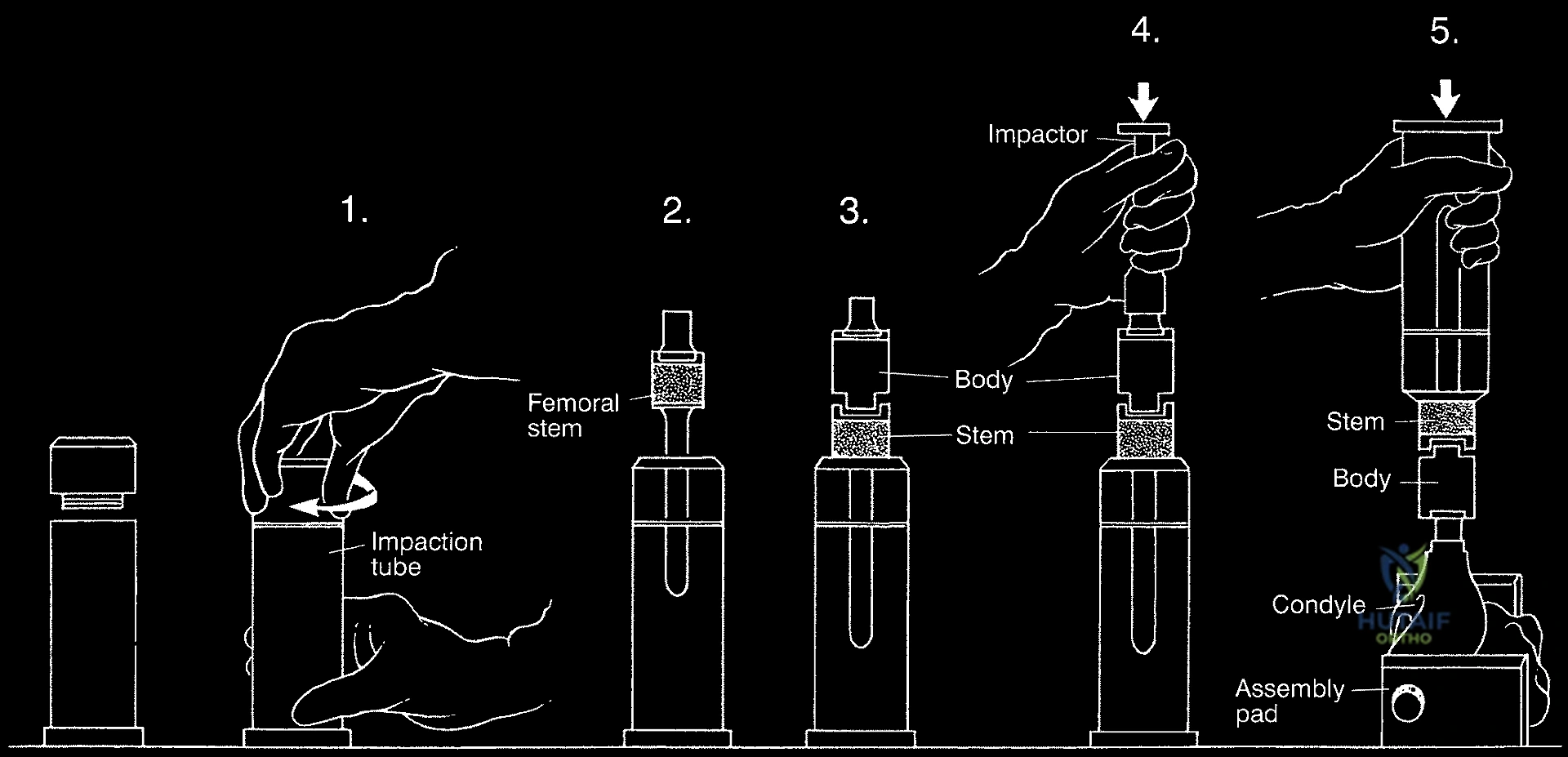
Once the specimen is removed and sent for pathological evaluation of the margins, the reconstructive phase begins. This is where modern modular endoprosthetic systems demonstrate their value, providing immediate structural integrity. The host bone canal is sequentially reamed and broached to accommodate the prosthetic stem. Fixation philosophy dictates the approach: cemented stems rely on a robust polymethylmethacrylate (PMMA) mantle for immediate, rigid fixation, ideal for older patients with poor bone stock or metastatic disease. Cementless, press-fit stems rely on initial mechanical stability achieved through diaphyseal scratch fit, often augmented by extracortical flanges and cross-locking screws, followed by long-term biologic osteointegration.
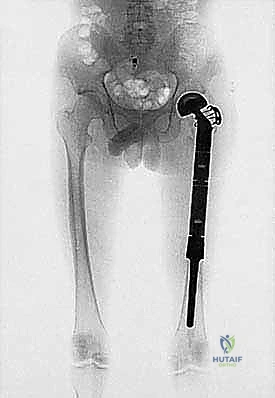
The assembly of the modular components is a critical step requiring meticulous attention to detail. The morse tapers must be completely clean and dry before impaction to ensure a cold-weld lock; any interposed fat, blood, or soft tissue will dramatically increase the risk of taper fretting, corrosion, and eventual mechanical dissociation. Trial components are utilized extensively to fine-tune limb length, myofascial tension, and joint stability. In the proximal femur, restoring the correct abductor lever arm (offset) and femoral anteversion is paramount to prevent postoperative dislocation. In the distal femur or proximal tibia, achieving a balanced flexion and extension gap with the rotating hinge knee mechanism is essential for optimal kinematics and implant longevity.
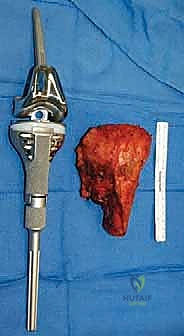
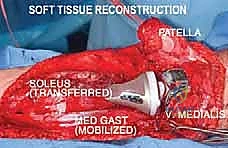
The final, and often most challenging, pillar is the restoration of the soft tissue envelope. A mega-prosthesis left exposed or covered only by thin, irradiated skin is destined for disastrous infection. In proximal femoral replacements, the abductor mechanism must be securely reattached to the prosthesis, often utilizing specialized trochanteric grip components, heavy non-absorbable sutures, or synthetic meshes like Trevira tubes to facilitate soft-tissue ingrowth. In proximal tibial replacements, the extensor mechanism (patellar tendon) must be robustly reattached to the implant or a composite allograft, often augmented with a medial gastrocnemius rotational flap to provide a highly vascularized envelope over the anterior tibia, promoting healing and serving as a barrier against infection.

Complications, Incidence Rates, and Salvage Management
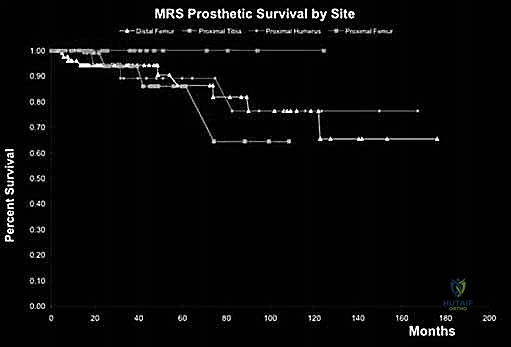
Despite profound advancements in surgical technique and implant metallurgy, endoprosthetic reconstruction remains fraught with a high incidence of complications. The survival of a megaprosthesis is significantly lower than that of standard primary arthroplasty, and surgeons must be intimately familiar with the Henderson Classification of Failure for limb salvage implants, which categorizes complications into five distinct types. Type 1 failures involve soft tissue failures, including wound dehiscence, flap necrosis, and joint instability or dislocation. Dislocation is particularly prevalent in proximal femoral replacements (up to 15% incidence) due to the extensive resection of the hip capsule and abductor musculature. Management relies on aggressive soft-tissue reconstruction, bracing, or revision to constrained acetabular liners.
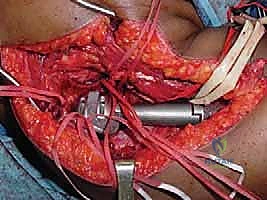
Type 2 failures represent aseptic loosening, historically the most common mode of failure in cemented stems, driven by the massive mechanical stresses transferred across the bone-cement interface. While modern cementless, hydroxyapatite-coated, fluted stems have reduced this incidence, stress shielding and subsequent osteolysis remain significant long-term concerns, particularly in young, active patients. Type 3 failures are structural failures of the implant itself, including stem fractures, morse taper dissociations, or severe polyethylene bushing wear within hinge knee mechanisms. These mechanical failures necessitate complex revision surgeries, often requiring the extraction of well-fixed stems using specialized trephines and ultrasonic tools.

Type 4 failures—periprosthetic joint infection (PJI)—are the most dreaded and devastating complications in orthopaedic oncology. The incidence of PJI in megaprostheses ranges from 5% to 15%, significantly higher than standard arthroplasty, due to extensive soft-tissue stripping, prolonged operative times, and the immunosuppressive effects of neoadjuvant chemotherapy and radiation. The massive surface area of the implant provides a fertile ground for bacterial biofilm formation. Management almost universally requires a grueling two-stage revision protocol: complete hardware explantation, placement of an antibiotic-impregnated cement spacer, prolonged intravenous antibiotics, and eventual reimplantation. To combat this, modern implants are increasingly utilizing silver, iodine, or titanium-nitride coatings to leverage antimicrobial properties.
Finally, Type 5 failures represent local tumor recurrence. While advancements in imaging and neoadjuvant therapies have reduced local recurrence rates to approximately 5-10%, when it occurs adjacent to a megaprosthesis, the consequences are dire. Recurrence often implies an aggressive, chemo-resistant tumor biology or microscopic positive margins. Salvage management for Type 5 failures frequently requires radical ablative surgery—amputation or disarticulation—as further limb-sparing attempts carry an unacceptably high risk of further recurrence and systemic metastasis. Rigorous oncologic surveillance with serial imaging is mandatory to detect these failures early.
| Henderson Classification | Failure Mechanism | Typical Incidence | Primary Management Strategy |
|---|---|---|---|
| Type 1 | Soft Tissue Failure (Instability, Dislocation, Wound) | 5 - 15% | Bracing, constrained liners, flap coverage, soft-tissue repair. |
| Type 2 | Aseptic Loosening (Bone-implant interface failure) | 10 - 20% (Long-term) | Revision to longer/thicker stem, impaction bone grafting. |
| Type 3 | Structural Failure (Implant fracture, bushing wear) | 5 - 10% | Component exchange, extraction of broken stems, revision. |
| Type 4 | Infection (Periprosthetic Joint Infection) | 5 - 15% | Two-stage revision, aggressive debridement, suppressive antibiotics. |
| Type 5 | Tumor Recurrence (Local oncologic failure) | 5 - 10% | Radical resection, often requiring amputation/disarticulation. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following massive endoprosthetic reconstruction is a highly specialized, phased process that must balance the competing demands of rapid mobilization to prevent systemic complications with the absolute necessity of protecting fragile soft-tissue repairs and allowing for biologic osteointegration. The immediate postoperative phase (0 to 2 weeks) is focused heavily on wound healing, pain management, and the prevention of deep vein thrombosis (DVT). Due to the extensive surgical trauma and prolonged immobilization, these patients are at an exceptionally high risk for venous thromboembolism; aggressive chemical and mechanical prophylaxis is mandatory. Weight-bearing status during this phase is strictly dictated by the method of implant fixation. Cemented constructs generally allow for immediate weight-bearing as tolerated, whereas cementless, press-fit stems typically require a period of protected, partial weight-bearing (e.g., 50% for 6 weeks) to prevent micromotion and facilitate bone ingrowth.
The early rehabilitation phase (2 to 6 weeks) introduces controlled, progressive range of motion while rigorously protecting the specific anatomic repairs. For proximal femoral replacements, strict hip precautions are instituted to prevent dislocation; active abduction is strictly prohibited to protect the reattached gluteus medius and minimus, and flexion past 90 degrees is avoided. For proximal tibial reconstructions, the primary concern is the extensor mechanism repair. Patients are typically placed in a hinged knee brace locked in full extension, allowing only passive flexion to a limited degree (e.g., 0-30 degrees initially) to prevent catastrophic rupture of the patellar tendon reattachment. Active quadriceps firing is delayed until early biologic healing is evident.
Entering the intermediate phase (6 to 12 weeks), the focus shifts toward restoring muscle strength, improving proprioception, and normalizing gait mechanics. As soft-tissue healing solidifies, active-assisted and eventually active range of motion exercises are initiated. Patients transition from walkers to crutches or canes, aiming to eliminate the use of assistive devices by the end of this phase. Aquatic therapy can be highly beneficial during this period, providing a buoyant environment that reduces joint loading while allowing for resistance training. However, the physical therapist must remain vigilant for signs of complication, such as sudden increases in pain, new-onset instability, or wound drainage, which require immediate surgical evaluation.
Long-term functional outcomes and expectations must be clearly communicated to the patient. While modern endoprostheses provide excellent limb salvage, the reconstructed limb will never equate to a normal, native extremity. Functional outcomes are rigorously tracked using standardized metrics such as the Musculoskeletal Tumor Society (MSTS) scoring system, which evaluates pain, function
Clinical & Radiographic Imaging Archive
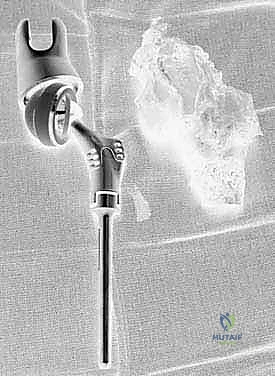
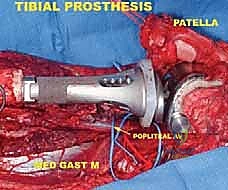

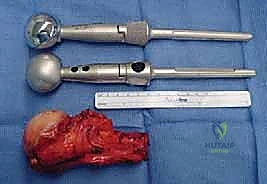
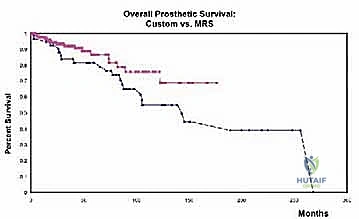


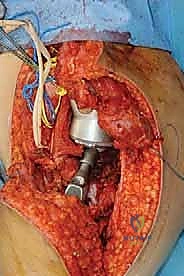

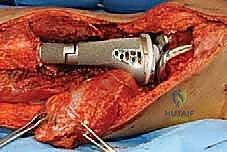

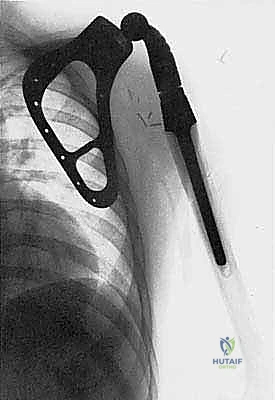

Detailed Chapters & Topics
Dive deeper into specialized chapters regarding overview-of-endoprosthetic-reconstruction
