Sartorial Canal Tumor Resection: An Intraoperative Masterclass in Limb Salvage
Key Takeaway
This masterclass guides fellows through the complex resection of sartorial canal tumors. We'll meticulously cover preoperative planning, patient positioning, and real-time surgical execution, emphasizing comprehensive anatomy, neurovascular protection, and oncologic principles. Learn precise dissection, vascular management, and soft tissue reconstruction techniques for optimal limb salvage and functional outcomes, including handling potential complications.
Comprehensive Introduction and Patho-Epidemiology
The management of soft tissue sarcomas and aggressive benign neoplasms within the extremities has evolved dramatically over the past four decades, shifting from a paradigm of routine amputation to sophisticated limb salvage surgery. Among the most technically demanding anatomical regions for limb salvage is the sartorial canal. Tumors residing deep within this confined anatomical space present a unique oncologic and surgical challenge. These are not merely standard soft tissue masses; they are extracompartmental space tumors whose intimate and unyielding relationship with the primary neurovascular axis of the lower extremity demands absolute surgical precision, profound anatomical knowledge, and a profound respect for tissue planes.
Sartorial canal tumors are relatively rare, but when they occur, they are predominantly soft tissue sarcomas, including undifferentiated pleomorphic sarcomas, synovial sarcomas, liposarcomas, and leiomyosarcomas. Leiomyosarcomas, in particular, hold a special significance in this region as they may arise directly from the tunica media of the superficial femoral artery or vein, radically altering the surgical approach. Benign but locally aggressive lesions, such as deep-seated hemangiomas, aggressive fibromatosis (desmoid tumors), and schwannomas, also frequently manifest in this conduit. The patho-epidemiology of these tumors dictates that early, definitive resection is paramount. Because the sartorial canal is a tightly constrained extracompartmental space, expanding neoplastic lesions rapidly exhaust the available volume, leading to early displacement, compression, and eventual direct invasion of the superficial femoral vessels.
The primary objective in this masterclass is to elucidate the principles of oncologic resection within this high-rent district. The surgeon must balance the absolute necessity of achieving negative histological margins (R0 resection) with the goal of preserving a functional, viable extremity. A compromised margin in the sartorial canal inevitably leads to catastrophic local recurrence, often necessitating morbid hemipelvectomy or high transfemoral amputation. Conversely, overly aggressive resection without a clear plan for vascular reconstruction can result in acute limb ischemia. Therefore, mastering the sartorial canal requires a synthesis of orthopedic oncology, vascular surgery, and microvascular reconstructive techniques.
Detailed Surgical Anatomy and Biomechanics
Before a scalpel ever touches skin, the orthopedic oncologist must possess an eidetic understanding of the regional anatomy. The sartorial canal, historically referred to as the subsartorial canal, Hunter's canal, or the adductor canal, serves as the primary neurovascular conduit of the medial thigh. It acts as an aponeurotic tunnel, extending from the apex of the femoral triangle proximally, and transitioning distally into the popliteal fossa via the adductor hiatus. This canal is approximately 15 centimeters in length in the average adult and dictates the surgical approach to the middle third of the thigh.
Boundaries of the Sartorial Canal
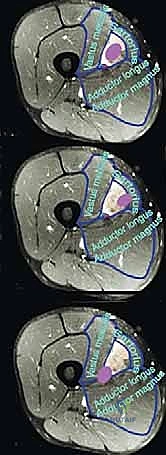
To conceptualize the canal intraoperatively, one must imagine an inverted, asymmetrical triangle in cross-section. The boundaries are formed by robust muscular and fascial structures that dictate the pathways of tumor extension. The roof of this canal is formed by the sartorius muscle, which courses obliquely from anterior to medial. The deep fascia underlying the sartorius, known as the vastoadductor membrane, spans from the adductor muscles to the vastus medialis, forming the true anatomical roof. This muscle and its underlying fascia serve as our initial surgical landmark and, depending on tumor extension, may require partial or complete en bloc resection.
The floor of the canal is muscular, composed primarily of the adductor longus muscle in its proximal extent, and the adductor magnus muscle more distally. These muscles form a dense bed upon which the neurovascular bundle rests. Tumors originating from the floor often push the vessels anteriorly, increasing their vulnerability during the initial surgical approach. The lateral border is defined by the thick, unyielding fascia overlying the vastus medialis muscle. The posterior border is primarily the adductor magnus muscle and its dense fascial covering. Understanding these boundaries is critical because sarcomas respect fascial boundaries until late in their course; thus, these muscular fascial envelopes often provide our definitive surgical margins.
Neurovascular Contents and the Fascial Barrier
Within this confined, aponeurotic space lie the vital structures that govern the viability and function of the distal extremity. The superficial femoral artery (SFA) is the primary arterial supply to the distal limb and the most critical structure within the canal. The superficial femoral vein (SFV) typically lies posterior and lateral to the artery, although its position is subject to significant anatomical variance, occasionally duplicating or forming a plexus around the artery.
Accompanying these vessels are two critical neurological structures. The saphenous nerve, the largest cutaneous branch of the femoral nerve, is purely sensory. It accompanies the SFA, initially lying lateral to it, then crossing anteriorly, and finally residing medial to the artery as it pierces the vastoadductor membrane to exit the canal. It supplies sensation to the medial aspect of the leg, ankle, and foot. Iatrogenic damage to the saphenous nerve is a common complication, resulting in debilitating neuropathic pain and sensory deficits. The nerve to the vastus medialis, a robust motor branch of the femoral nerve, typically courses within the proximal aspect of the canal before entering the vastus medialis muscle.
Crucially, from an oncologic perspective, the superficial femoral artery and vein are encased throughout their length by a highly dense, fibrous fascial sheath. This perivascular sheath serves as a vital oncologic barrier. In many instances, a tumor may abut this sheath, compressing the vessels without directly invading the adventitia. This sheath provides a potential, albeit microscopic, safe plane of subadventitial dissection, allowing for limb salvage in cases that might otherwise require vascular sacrifice. The vessels typically exit the canal distally through the adductor hiatus—a fibrous archway in the distal adductor magnus tendon—to become the popliteal artery and vein. Tumors located at the adductor hiatus are notoriously difficult to resect due to the transition from the anterior to the posterior compartment of the leg.
Exhaustive Indications and Contraindications
The decision to proceed with limb-salvage tumor resection in the sartorial canal is complex and requires a multidisciplinary tumor board consensus. The primary goal is always patient survival, followed by local tumor control, and finally, functional preservation. The indications and contraindications must be meticulously weighed.
| Category | Specific Criteria | Clinical Rationale |
|---|---|---|
| Absolute Indications | Biopsy-proven soft tissue sarcoma confined to the sartorial canal. | Standard of care dictates wide local excision for local control and survival. |
| Absolute Indications | Symptomatic, locally aggressive benign tumors (e.g., aggressive fibromatosis) causing neurovascular compromise. | Prevention of irreversible ischemic or neuropathic damage to the distal limb. |
| Relative Indications | Isolated, resectable metastatic lesions within the canal (e.g., renal cell carcinoma metastasis). | Palliative local control or curative intent in oligometastatic disease. |
| Relative Indications | Recurrent sarcoma following previous inadequate excision or radiation failure. | Salvage therapy; carries a significantly higher risk of complications and amputation. |
| Absolute Contraindications | Encasement of the sciatic nerve combined with femoral nerve involvement. | Resection would yield a flail, insensate limb that is functionally inferior to a prosthesis. |
| Absolute Contraindications | Inability to achieve negative margins without sacrificing essential structures that cannot be reconstructed. | R1 or R2 resections in sarcomas lead to guaranteed recurrence; amputation is preferred. |
| Absolute Contraindications | Severe, uncorrectable peripheral vascular disease prohibiting graft reconstruction. | Vascular reconstruction failure will result in acute limb ischemia and emergent amputation. |
| Relative Contraindications | Diffuse, progressive metastatic disease with a life expectancy of less than 3 months. | Surgical morbidity outweighs the oncologic benefit; palliative care or radiation is preferred. |
Pre-Operative Planning, Templating, and Patient Positioning
The battle against a sartorial canal tumor is frequently won or lost in the preoperative planning phase. Given the catastrophic consequences of an intraoperative vascular injury or an inadvertent positive margin, our planning must be exhaustive. We operate under the strict assumption that every mass in this region is a high-grade sarcoma until proven otherwise by definitive pathology.
Advanced Imaging and Staging Modalities
Our comprehensive imaging suite provides the critical roadmap for the surgical approach. Plain radiography is always the initial step. While soft tissue tumors are radiolucent, plain films are essential to rule out direct periosteal reaction or invasion of the underlying femoral diaphysis. Furthermore, the presence of specific soft tissue calcifications can be pathognomonic; for instance, phleboliths suggest a hemangioma, while amorphous calcifications may indicate a synovial sarcoma.
Computed Tomography (CT) with 3D reconstruction and timed arterial contrast is an invaluable asset. In the confined space of the sartorial canal, expanding tumors distort normal anatomy rapidly, displacing the native vessels. The 3D reconstructions allow the surgical team to visualize this distortion in multiple planes, mapping the exact trajectory of the displaced superficial femoral artery.
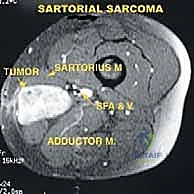
Magnetic Resonance Imaging (MRI) remains the undisputed gold standard for soft tissue evaluation in orthopedic oncology. An MRI, utilizing T1, T2, and STIR sequences with and without gadolinium contrast, precisely delineates the tumor's volume, its peritumoral edema (which often harbors microscopic satellite cells), and its invasion into neighboring muscular boundaries. MRI is critical for determining the proximal and distal extent of the tumor into the femoral triangle or the popliteal space, thereby dictating the length of our surgical incision.

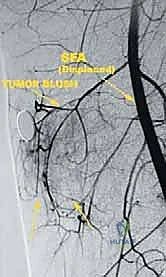
Angiography is frequently performed, not just for mapping, but to assess the intrinsic vascularity of the tumor. The presence of a "tumor blush" during the late arterial phase is highly indicative of a high-grade, hypervascular malignancy. Most critically, angiography helps determine whether the femoral artery is merely displaced by the mass or if the tumor has directly invaded the tunica adventitia, which necessitates en bloc vascular resection.
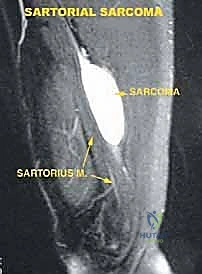
Venography is equally essential to rule out occult venous thrombosis, mural tumor thrombus extending into the lumen, or direct tumor involvement of the superficial femoral vein. This is crucial for planning potential venous sacrifice or reconstruction. Finally, systemic staging, including a high-resolution CT of the chest and a Technetium-99m bone scan, is mandatory to rule out distant metastatic disease prior to undertaking a massive local resection.
Topographic Tumor Classification System
Based on our advanced imaging, we classify sartorial canal tumors into three distinct topographic types. This classification is not merely academic; it directly dictates the surgical strategy, the required margins, and the necessity for a vascular surgery team on standby.
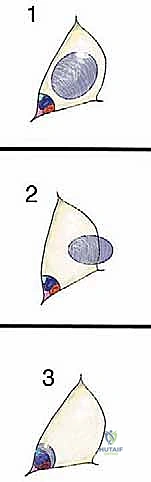
Type 1 (Luminal) Tumors arise from within the areolar tissue of the sartorial canal space itself, typically originating from fat, lymphatic, or fibrous tissue. They lie relatively "loose" within the space. While they may approximate the vessel walls or surrounding muscles, they are not intimately adherent. These are generally resected with a thin, continuous cuff of normal surrounding tissue.
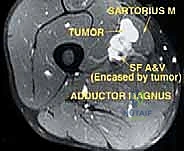
Type 2 (Wall) Tumors originate from one of the muscular boundaries bordering the canal—most commonly the sartorius, vastus medialis, adductor magnus, or adductor longus. They arise from within the muscle belly or its investing fascia and push into the canal space. Resection of Type 2 tumors requires an en bloc excision of the involved muscle from origin to insertion, or at least a wide margin of normal muscle tissue surrounding the lesion.
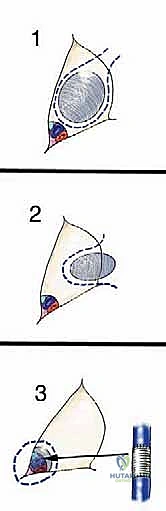
Type 3 (Vessel) Tumors represent the most formidable surgical challenge. These tumors directly involve the arteries, veins, or major nerves. They either originate from the vessel wall itself (e.g., a primary vascular leiomyosarcoma) or have aggressively invaded through the fascial sheath into the adventitia. These tumors cannot be peeled off the vessels; attempting to do so guarantees a positive margin. They must be resected en bloc with the involved neurovascular structures, necessitating immediate vascular reconstruction.

Biopsy Strategy and Patient Positioning
The biopsy strategy for the sartorial canal deviates from standard orthopedic oncology protocols. Typically, a core needle biopsy is the standard of care for soft tissue masses. However, in the sartorial canal, a core needle or open incisional biopsy carries unacceptable risks. A biopsy-induced hematoma can easily track along the neurovascular bundle, contaminating the entire length of the extremity and potentially converting a limb-sparing procedure into a mandatory amputation. Furthermore, if the tumor is a vascular sarcoma, a needle puncture can cause catastrophic, uncontrollable hemorrhage or a pseudoaneurysm.
Therefore, the preferred strategy is an excisional biopsy with immediate frozen section analysis at the time of definitive surgery, provided the lesion is small enough. For larger lesions where pre-operative neoadjuvant therapy is required, an extremely carefully planned, image-guided core biopsy is performed by an experienced musculoskeletal radiologist, utilizing a trajectory that will be completely excised during the definitive surgery.
Patient positioning is critical for optimal exposure. The patient is placed in the supine position on a radiolucent operating table. The entire affected lower extremity, from the umbilicus and iliac crest down to the toes, is meticulously prepped and draped free. This allows for intraoperative manipulation, full range of motion, and unhindered access to the entire length of the canal and the popliteal fossa. Crucially, the contralateral lower extremity must also be prepped and draped into the sterile field. This serves as our backup plan for harvesting a reverse greater saphenous vein graft should an unanticipated arterial or venous reconstruction become necessary.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution requires a seamless blend of blunt and sharp dissection, maintaining absolute hemostasis to ensure visualization of fascial planes. The surgeon must operate with a constant awareness of the proximity of the superficial femoral vessels.
Initial Incision and Flap Elevation
The procedure commences with a generous longitudinal skin incision. Inadequate exposure is the most common pitfall in sartorial canal surgery, leading to dangerous traction on the vessels and compromised oncologic margins. Using a #10 blade, a long, curvilinear incision is made directly over the palpable mass, following the anatomical course of the sartorius muscle. The incision is extended proximally into the femoral triangle and distally towards the medial epicondyle of the femur.
Subcutaneous flaps are elevated medially and laterally. It is imperative to raise full-thickness fasciocutaneous flaps, leaving the deep fascia intact over the musculature. Thin skin flaps are prone to postoperative necrosis, which can lead to disastrous wound breakdown and exposure of underlying vascular grafts. As the flaps are mobilized, the greater saphenous vein is identified superficially. If it is not involved by the tumor, it is carefully preserved and retracted, as it provides vital venous drainage, especially if the deep superficial femoral vein requires resection.
Proximal Vascular Control and Distal Dissection
The fundamental axiom of vascular and oncologic surgery in the extremities is to obtain proximal and distal control of the major vessels before ever manipulating the tumor itself. The deep fascia is incised longitudinally over the proximal sartorius muscle in the femoral triangle, well proximal to the superior pole of the tumor. The sartorius is mobilized and retracted laterally or medially, depending on tumor location, to expose the proximal extent of the sartorial canal.
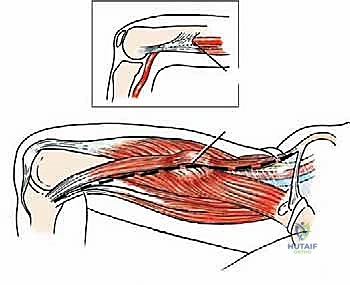
At this proximal location, the superficial femoral artery and vein are systematically identified. Vessel loops (typically red for artery, blue for vein) are passed around the structures to secure proximal control. The femoral nerve branches, particularly the saphenous nerve and the nerve to the vastus medialis, are identified and protected if they are free of disease.
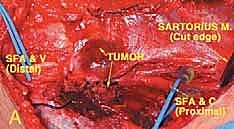
Once proximal control is secured, attention is directed distally. The incision is carried down to the adductor hiatus. The distal superficial femoral vessels (or proximal popliteal vessels) are identified as they dive through the hiatus. Distal vessel loops are applied. Only with secure proximal and distal control established can the surgeon safely begin the dissection of the tumor mass itself.
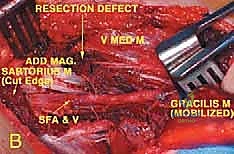
En Bloc Resection and Vascular Reconstruction
The dissection proceeds from normal, uninvolved tissue toward the tumor, utilizing the principle of "en bloc" resection. The surgeon must never visualize the tumor capsule directly; seeing the tumor implies a marginal or intralesional resection, which is an oncologic failure.
For Type 1 and Type 2 tumors, the dissection plane is established outside the reactive zone of the tumor. If the tumor abuts the vessels, the thick perivascular fascial sheath is meticulously incised longitudinally, well away from the tumor mass. Using sharp dissection and magnification loupes, the surgeon carefully dissects the sheath off the adventitia of the artery and vein. If the sheath strips away cleanly, the vessels can be preserved, and the tumor is resected with the sheath acting as the deep margin.
For Type 3 tumors, where the vessels are encased or directly invaded, no attempt is made to separate the tumor from the vasculature. The patient is systemically heparinized. The superficial femoral artery and vein are clamped proximally and distally. The vessels are transected en bloc with the tumor mass, achieving wide oncologic margins.
Following removal of the specimen, immediate vascular reconstruction is mandatory to prevent irreversible limb ischemia. For arterial reconstruction, an autologous reverse greater saphenous vein graft (harvested from the prepped contralateral leg) is the conduit of choice due to its superior patency rates and resistance to infection compared to synthetic grafts (e.g., PTFE or Dacron). The graft is anastomosed end-to-end using continuous non-absorbable monofilament sutures (e.g., 6-0 Prolene).
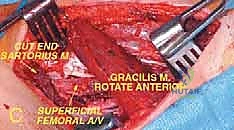
Venous reconstruction is more controversial. While some collateral venous drainage exists via the profunda femoris system, ligation of the superficial femoral vein can lead to severe, chronic postoperative edema and compartment syndrome. Therefore, reconstruction of the SFV with a vein graft or a ringed PTFE graft is highly recommended whenever feasible. Before closure, the tourniquet (if used) or vascular clamps are released, and meticulous hemostasis is achieved. Closed suction drains are placed deep within the canal, ensuring they do not rest directly on the vascular anastomoses.
Complications, Incidence Rates, and Salvage Management
Surgical intervention in the sartorial canal carries a high morbidity profile. The surgeon must be prepared to manage complex postoperative complications rapidly to prevent limb loss.
| Complication | Estimated Incidence | Pathophysiology & Clinical Presentation | Salvage Management & Intervention |
|---|---|---|---|
| Wound Dehiscence / Flap Necrosis | 10% - 15% | Ischemia of thin skin flaps, exacerbated by postoperative radiation. Presents as eschar, wound breakdown, and exposed deep structures. | Aggressive debridement. Coverage with local rotational flaps (e.g., medial gastrocnemius) or free tissue transfer. |
| Vascular Graft Thrombosis | 5% - 8% | Technical error at anastomosis, hypercoagulable state, or kinking of the graft. Presents as acute loss of distal pulses, pallor, and pain. | Emergent return to OR for thrombectomy, revision of anastomosis, or placement of a new bypass graft. |
| Postoperative Hematoma | 5% - 10% | Inadequate intraoperative hemostasis or coagulopathy. Can compress vascular grafts leading to secondary ischemia. | Immediate operative evacuation if expanding or causing neurovascular compromise to prevent graft failure. |
| Deep Vein Thrombosis (DVT) | 15% - 20% | Endothelial injury, venous stasis, and oncologic hypercoagulability. Presents as severe limb swelling and pain. | Therapeutic anticoagulation (Heparin bridge to DOAC/Warfarin). Close monitoring for pulmonary embolism. |
| Saphenous Nerve Neuropathy | 20% - 30% | Iatrogenic traction, transection, or entrapment in scar tissue. Presents as medial leg numbness and severe neuropathic pain. | Pharmacologic management (Gabapentin/Pregabalin). Desensitization therapy. Rarely requires surgical neurolysis. |
| Local Tumor Recurrence | 10% - 25% | Microscopic positive margins (R1/R2) or aggressive tumor biology. Presents as a new palpable mass or pain. | Restaging. Often necessitates radical salvage procedures, including high transfemoral amputation or hip disarticulation. |
Phased Post-Operative Rehabilitation Protocols
Rehabilitation following a major sartorial canal resection is a delicate balance between protecting the surgical reconstruction and preventing devastating joint contractures. The protocol must be individualized, particularly if a vascular graft has been placed.
Phase I: Maximum Protection (Weeks 0-2)
The primary goal is wound healing and protection of the vascular anastomosis. The patient is kept on strict bed rest for the first 24 to 48 hours. The limb is elevated to minimize edema. If a vascular graft crosses the knee joint (into the popliteal space), the knee is immobilized in a hinged brace locked in extension to prevent kinking of the graft. Weight-bearing is typically restricted to toe-touch or partial weight-bearing with assistive devices. Gentle, passive range of motion of the ankle is encouraged to promote venous return.
Phase II: Controlled Mobility (Weeks 2-6)
Once the incision has healed and the drains are removed, the focus shifts to restoring mobility. The hinged knee brace is gradually unlocked, allowing progressive flexion. However, extreme flexion (greater than 90 degrees) is strictly avoided if a vein graft is present, as this can cause graft occlusion. Active-assisted range of motion exercises for the hip and knee are initiated. Weight-bearing is advanced as tolerated based on the extent of muscular resection.
Phase III: Strengthening and Functional Restoration (Weeks 6-12+)
At this stage, the vascular reconstruction is considered stable. Intensive physical therapy focuses on strengthening the remaining musculature of the thigh. If the sartorius or vastus medialis was resected, compensatory strengthening of the rectus femoris and remaining adductors is crucial. Gait training is emphasized to eliminate compensatory limps. Patients are typically cleared for activities of daily living, though high-impact sports are generally permanently restricted to protect the limb and the vascular conduits.
Summary of Landmark Literature and Clinical Guidelines
The modern approach to sartorial canal tumors is built upon decades of foundational orthopedic oncology research. The principles of wide en bloc resection were definitively established by Enneking et al. in the 1980s, who demonstrated that functional limb salvage is oncologically equivalent to amputation provided that negative margins are achieved.
Contemporary guidelines from the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO) mandate a multidisciplinary approach for all extremity soft tissue sarcomas. For high-grade tumors larger than 5 centimeters in the sartorial canal, the standard of care now involves neoadjuvant external beam radiation therapy (EBRT). Radiation significantly sterilizes the reactive zone, forming a dense pseudocapsule that facilitates margin-negative surgical resection and significantly reduces the rate of local recurrence.
Furthermore, vascular surgical literature, notably studies by the Society for Vascular Surgery, strongly supports the use of autologous vein grafts over synthetic materials in the setting of oncologic resection, citing lower rates of catastrophic graft infection in the face of large dead spaces and irradiated tissue beds. The continuous evolution of microvascular techniques and targeted biological therapies promises to further enhance limb salvage rates and functional outcomes for patients afflicted with these formidable tumors.
