Mastering Shoulder Girdle Resections: An Intraoperative Guide to Limb-Sparing Oncologic Surgery
Key Takeaway
This masterclass provides an exhaustive, real-time guide to shoulder girdle resections for high-grade sarcomas. We delve into comprehensive surgical anatomy, meticulous preoperative planning, and granular intraoperative execution from the surgeon's perspective. Learn precise dissection techniques, neurovascular protection, and strategies for achieving wide oncologic margins. Critical pearls, pitfalls, and postoperative management are extensively covered, preparing fellows for complex limb-sparing procedures.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues, to the definitive exploration of one of the most mechanically and anatomically demanding procedures in orthopaedic oncology: the shoulder girdle resection for high-grade sarcomas. Historically, tumors of the proximal humerus and scapula that escaped the confines of the bone were managed almost exclusively with forequarter amputations. Today, the modern orthopaedic oncologist is tasked not merely with the eradication of the malignancy, but with the meticulous preservation of a functional limb, the maintenance of the patient's quality of life, and the strict adherence to the highest oncologic principles. We will be detailing the execution of a modified extra-articular resection, drawing heavily upon the foundational principles established by Tikhoff and Linberg, and subsequently refined by Malawer and contemporary reconstructive surgeons, to achieve a wide margin while seamlessly reconstructing the resulting massive defect.
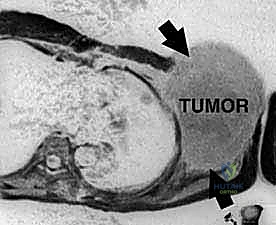
The patho-epidemiology of shoulder girdle sarcomas presents unique challenges that dictate our surgical approach. The proximal humerus is the third most common site for primary bone sarcomas, following the distal femur and proximal tibia. Osteosarcoma, chondrosarcoma, and Ewing sarcoma frequently present in this metaphyseal-epiphyseal region. Unlike the lower extremity, where thick fascial septa define distinct muscular compartments that naturally contain tumor growth, the shoulder girdle is characterized by a complex, interdependent network of muscles with comparatively weak fascial boundaries. This anatomic reality allows high-grade sarcomas to disseminate more readily through tissue planes, demanding a highly aggressive and meticulously planned resection strategy to achieve negative margins.
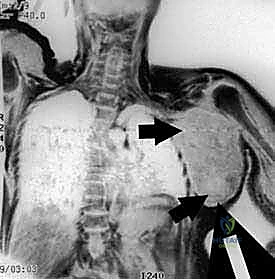
Furthermore, the proximity of the brachial plexus and the axillary vessels to the proximal humerus and scapula introduces a critical variable into the oncologic equation. High-grade bone sarcomas in this region exhibit a characteristic centripetal growth pattern, compressing surrounding muscles and reactive tissue into a pseudocapsular layer. This pseudocapsule is deceptive; while it appears to macroscopically contain the tumor, it invariably harbors microscopic satellite nodules and finger-like projections of malignant cells. Consequently, enucleation or marginal excision is absolutely contraindicated. A wide, compartmental—or often extra-articular—resection must be performed, removing the tumor en bloc with its entire pseudocapsular envelope and a cuff of normal, healthy tissue.
The evolution of neoadjuvant chemotherapy has dramatically altered the landscape of these resections. By inducing tumor necrosis and potentially shrinking the reactive pseudocapsular zone, systemic therapy often converts a previously unresectable tumor into a candidate for limb salvage. However, the surgeon must operate based on the pre-chemotherapy MRI extent of the tumor, as microscopic disease may persist in areas where the macroscopic tumor has seemingly retreated. This chapter will serve as your exhaustive intraoperative guide, bridging the gap between advanced cross-sectional imaging and the tactile reality of the operating theater, ensuring you are equipped to navigate the treacherous anatomy and complex reconstructive demands of the shoulder girdle.
Detailed Surgical Anatomy and Biomechanics
Before a scalpel is ever passed, an intimate, three-dimensional understanding of the functional anatomy of the shoulder girdle is absolutely paramount. This region represents a complex, highly mobile interplay of osseous structures, dynamic muscular stabilizers, and critical neurovascular conduits. Tumors here, particularly high-grade sarcomas, demand an encyclopedic knowledge of these relationships, as they invariably grow along paths of least resistance, displacing, compressing, and occasionally directly invading vital structures.
Osteology and Articulations of the Shoulder Girdle
The osseous foundation of the shoulder girdle comprises the proximal humerus, the scapula, and the clavicle. The proximal humerus includes the articular head, the anatomical neck, the surgical neck, the greater and lesser tuberosities, and the bicipital groove. Primary bone tumors frequently originate in the highly active metaphyseal bone of the proximal humerus. From this epicenter, they can extend proximally into the epiphysis and the glenohumeral joint, distally into the diaphysis, or radially through the cortex into the surrounding soft tissues. The inherently unconstrained nature of the glenohumeral joint, heavily reliant on soft tissue for stability, means that oncologic resection inherently destabilizes the entire upper extremity.
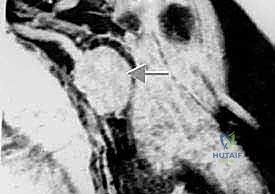
The scapula is a complex, flat, triangular bone featuring a body, spine, acromion, coracoid process, and the glenoid fossa. Its broad ventral and dorsal surfaces provide massive surface areas for the origin of the rotator cuff and periscapular musculature. Scapular sarcomas commonly arise from the neck or body, growing centrifugally and rapidly involving the massive muscle bellies of the infraspinatus and subscapularis. Because the scapula is suspended entirely by muscular attachments to the axial skeleton (with the exception of the acromioclavicular articulation), resections of the scapula dramatically alter the biomechanics of shoulder elevation and abduction.
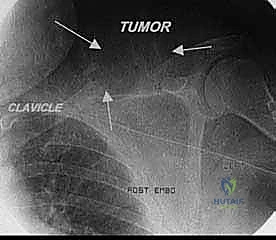
The clavicle, an S-shaped strut connecting the upper extremity to the axial skeleton, plays a pivotal role in both exposure and oncologic clearance. The distal third articulates with the acromion to form the AC joint. In extensive shoulder girdle resections, particularly the classic Tikhoff-Linberg procedure, osteotomy or resection of the distal clavicle is frequently required to facilitate exposure of the brachial plexus and to ensure adequate superior and medial oncologic margins.

Muscular Compartments and the Pseudocapsule
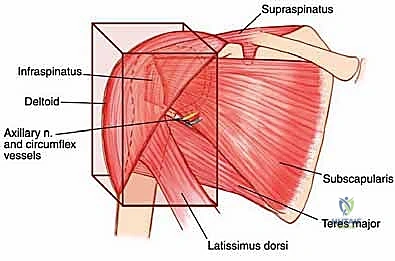
As previously noted, sarcomas compress surrounding muscles into a pseudocapsular layer. The functional compartment of the proximal humerus is defined by the deltoid laterally, the subscapularis anteriorly, the remaining rotator cuff muscles superiorly and posteriorly, the latissimus dorsi and teres major distally, and the brachialis and triceps in the arm. The glenoid and scapular neck are functionally contained within this compartment due to the enveloping rotator cuff and robust joint capsule. Tumors extending beyond the humeral cortices compress these muscles, often following the path of least resistance directed towards the glenoid and scapular neck.
Anteriorly, the tumor is frequently covered by the subscapularis, which bulges and displaces the neurovascular bundle medially. Rarely, a massive tumor may protrude anteriorly through the rotator interval. A wide, compartmental resection for a high-grade sarcoma in this location mandates the en bloc inclusion of the surrounding muscles forming the pseudocapsule. This typically involves resecting the deltoid (or its involved portions), the lateral insertions of the rotator cuff, the axillary nerve, the humeral circumflex vessels, and the glenoid, culminating in an extra-articular resection.
The scapular compartment presents its own unique challenges. Most high-grade scapular sarcomas arise from the neck region. The compartmental borders consist of the rotator cuff muscles, the teres major, and the latissimus dorsi. The subscapularis, infraspinatus, and teres muscles originate directly on the scapular surfaces and are intimately involved early in the disease process. The deltoid, while attaching to the scapular spine and acromion, is often protected by the intervening rotator cuff muscles unless the tumor is massive. Because the head of the proximal humerus is contained within the scapular compartment by the rotator cuff, wide resection of a high-grade scapular sarcoma frequently necessitates the inclusion of the rotator cuff and the humeral head to prevent capsular breach.
Critical Neurovascular Structures
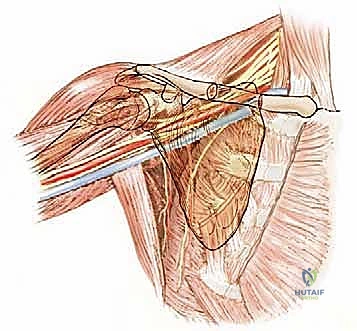
The relationship of the neurovascular bundle to the tumor is the single most significant anatomic factor determining the feasibility of limb salvage versus amputation. The brachial plexus, originating from C5-T1, courses through the axilla deep to the pectoralis minor. The axillary artery and vein, supplying and draining the entire upper limb, lie in intimate association with the cords and branches of the plexus. The main neurovascular bundle passes anterior to the subscapularis and latissimus dorsi muscles. Crucially, these muscles and their investing fascial layers often act as a robust protective barrier, displacing the bundle away from the expanding tumor and preventing direct vascular invasion in many cases.
However, the axillary nerve and the posterior humeral circumflex vessels are highly vulnerable. The axillary nerve, a terminal branch of the posterior cord, courses inferior to the capsule of the shoulder joint and wraps around the surgical neck of the humerus, passing through the quadrangular space to innervate the deltoid and teres minor. Because these structures physically enter the functional compartment of the proximal humerus, they are frequently enveloped by the reactive pseudocapsule.
In cases of extra-articular resection of the proximal humerus, the axillary nerve and circumflex vessels are often deliberately sacrificed to ensure a wide margin. While the main trunk of the axillary nerve is theoretically outside the true bone compartment, preserving it when the pseudocapsule abuts the quadrangular space carries an unacceptably high risk of local recurrence. Intraoperative judgment is paramount; if the tumor has not directly invaded the nerve or its surrounding fascial sleeve, and a clear margin can be visualized, meticulous neurolysis may allow for preservation, though this is the exception rather than the rule in high-grade lesions.
Tumor Spread Mechanisms Across the Shoulder Joint
High-grade bone sarcomas are particularly insidious in their propensity for intra-articular or pericapsular involvement. Understanding these distinct pathways of spread is crucial for achieving clear margins. The primary mechanism is direct capsular extension, where the tumor breaches the metaphyseal cortex and directly invades the joint capsule. Once the capsule is breached, the entire glenohumeral joint is considered contaminated, absolutely necessitating an extra-articular resection (resecting the humerus and glenoid en bloc without opening the joint).
The long head of the biceps tendon represents a secondary, highly specific conduit for tumor spread. Originating from the supraglenoid tubercle and superior labrum, this tendon passes intra-articularly before exiting through the bicipital groove. Tumor cells can track along the synovial sheath of this tendon, extending far distally into the arm or proximally into the joint space. The surgeon must routinely resect the biceps tendon distal to the tumor margin to prevent this avenue of recurrence.
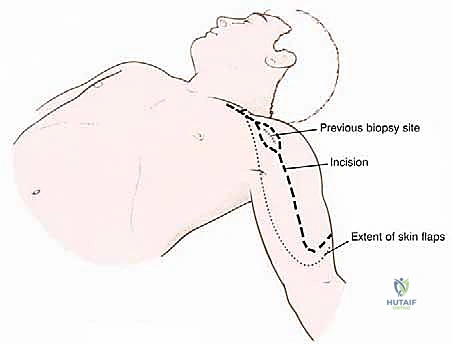
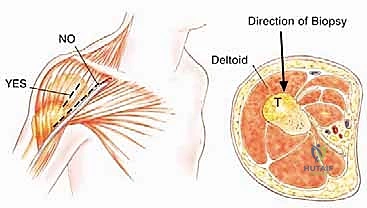
Pathologic fractures and poorly planned biopsies are iatrogenic or mechanical causes of widespread contamination. A pathologic fracture disrupts the hematoma, spreading malignant cells widely into the surrounding soft tissues and joint space, frequently converting a limb-salvage candidate into a forequarter amputation. Similarly, an improperly placed biopsy tract can contaminate adjacent, otherwise clean compartments. For proximal humerus tumors, a core needle biopsy through the anterior third of the deltoid is the gold standard. Great care must be taken to avoid the pectoralis major, the deltopectoral interval, and the axillary vessels. Because the deltoid is innervated posteriorly by the axillary nerve, the anterior biopsy tract can be excised en bloc with the tumor without compromising the remaining deltoid function.
Exhaustive Indications and Contraindications
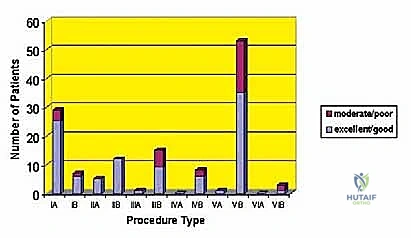
The decision to proceed with a limb-sparing shoulder girdle resection is highly complex and must be made within the context of a multidisciplinary tumor board. The goal is to achieve local control equivalent to that of an amputation while providing a limb that is functionally superior to a prosthesis. Patient selection is rigorously guided by Enneking staging, the patient's physiological status, and the anatomic extent of the disease.
The primary indication for a limb-sparing shoulder girdle resection is a primary, high-grade malignant bone or soft tissue sarcoma (Enneking Stage IIA or IIB) located in the proximal humerus, scapula, or periscapular soft tissues, provided that wide margins can be achieved without compromising the major neurovascular bundle. Common histologies include osteosarcoma, chondrosarcoma, Ewing sarcoma, and undifferentiated pleomorphic sarcoma. Additionally, aggressive benign lesions (Enneking Stage 3), such as massive giant cell tumors or aneurysmal bone cysts that have destroyed the articular surface and are not amenable to joint-preserving curettage, may also necessitate this approach. Isolated, solitary metastatic lesions (e.g., renal cell carcinoma, thyroid carcinoma) that are highly destructive and symptomatic may also be treated with resection and endoprosthetic reconstruction to palliate pain and restore function, provided the patient has a reasonable life expectancy.
Contraindications are strictly defined to prevent catastrophic oncologic or surgical failures. Absolute contraindications include major neurovascular encasement by the tumor that precludes reconstruction. If the brachial plexus is directly invaded by a high-grade sarcoma, limb salvage will likely result in a flail, painful, and useless extremity, making forequarter amputation the superior functional and oncologic choice. Similarly, if the axillary artery and vein are encased over a long segment and cannot be bypassed with clear margins, amputation is indicated. Uncontrolled systemic sepsis or severe medical comorbidities that preclude a massive, prolonged surgical intervention are also absolute contraindications.
Relative contraindications require nuanced clinical judgment. Extensive metastatic disease is a relative contraindication; however, if the primary tumor is fungating, severely painful, or impending fracture, palliative resection may still be warranted. Extensive soft tissue contamination from a poorly placed prior biopsy or a massive pathologic fracture hematoma significantly increases the risk of local recurrence and may tip the scales toward amputation. Finally, a poor soft tissue envelope, perhaps due to previous extensive irradiation, may preclude adequate coverage of a massive endoprosthesis, increasing the risk of catastrophic postoperative infection.
| Category | Specific Conditions | Management Implication |
|---|---|---|
| Primary Indications | High-grade bone sarcomas (Stage IIA/IIB) of proximal humerus/scapula. | Proceed with wide extra-articular resection and endoprosthetic reconstruction. |
| Primary Indications | Aggressive benign bone tumors (Stage 3) with massive articular destruction. | En bloc resection and reconstruction; joint preservation usually impossible. |
| Absolute Contraindications | Direct macroscopic invasion of the brachial plexus cords/trunks. | Forequarter amputation required for oncologic clearance and functional reality. |
| Absolute Contraindications | Circumferential encasement of axillary vessels precluding bypass. | Forequarter amputation required. |
| Relative Contraindications | Massive pathologic fracture with extensive soft tissue hematoma. | High risk of local recurrence; consider amputation vs. highly morbid extended resection. |
| Relative Contraindications | Poor soft tissue envelope (prior radiation, severe scarring). | High risk of implant exposure/infection; requires complex free tissue transfer planning. |
Pre-Operative Planning, Templating, and Patient Positioning
The success of a massive limb-sparing resection is dictated long before the patient enters the operating room. Exhaustive preoperative planning is the blueprint that translates two-dimensional imaging into a flawless three-dimensional surgical execution. This phase requires a synthesis of advanced imaging modalities, precise templating, and strategic patient positioning to ensure optimal exposure and safety.
High-resolution, multi-planar Magnetic Resonance Imaging (MRI) with and without intravenous contrast is the cornerstone of oncologic planning. T1-weighted images are essential for defining the intramedullary extent of the tumor and planning the osseous osteotomy level. T2-weighted and STIR sequences are critical for evaluating the extraosseous soft tissue mass, identifying the reactive edema zone, and delineating the pseudocapsule. The relationship of the tumor mass to the axillary neurovascular bundle must be scrutinized on axial and coronal sequences. If the fat plane between the tumor and the vessels is obliterated, vascular involvement must be assumed, and the surgical team must be prepared for vascular resection and grafting.
Computed Tomography (CT) provides unparalleled detail of the bony anatomy, cortical destruction, and matrix mineralization. Thin-slice CT is now routinely used to generate 3D models and patient-specific instrumentation (PSI). 3D printed cutting guides allow the surgeon to replicate the planned osteotomy perfectly in the operating room, ensuring negative bone margins while maximizing the preservation of healthy bone stock for endoprosthetic fixation. Furthermore, CT or MR angiography is highly recommended for massive tumors to map the vascular tree, identify feeding vessels for potential preoperative embolization (especially in highly vascular tumors like metastatic renal cell carcinoma or thyroid cancer), and plan for potential bypass.
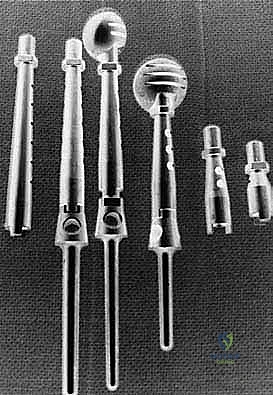
The templating process involves superimposing digital templates of various endoprosthetic systems over the patient's scaled radiographs and CT scans. The surgeon must determine the required resection length, the diameter of the remaining medullary canal, and the necessary offset and version of the reconstructive implant. Modular endoprosthetic systems are standard, allowing intraoperative flexibility to adjust length and soft tissue tension. The surgeon must also plan for soft tissue reconstruction, anticipating the need for local muscle transfers (e.g., latissimus dorsi or pectoralis major pedicled flaps) to cover the metallic implant and restore active motion.
Patient positioning is a critical, highly specific step. For extensive shoulder girdle resections, particularly those involving the scapula or requiring a posterior approach, the lateral decubitus position is preferred. The patient is positioned with the unaffected side down on a radiolucent table. A beanbag or specialized rigid lateral positioners are utilized to secure the torso perfectly perpendicular to the floor. An axillary roll is mandatory to protect the dependent brachial plexus. The entire affected upper extremity, shoulder girdle, and hemithorax (from the sternal notch to the spine, and from the mandible to the costal margin) are prepped and draped free. This allows the limb to be freely manipulated during the procedure, facilitating circumferential access to the tumor and permitting dynamic assessment of joint stability and soft tissue tension during reconstruction.
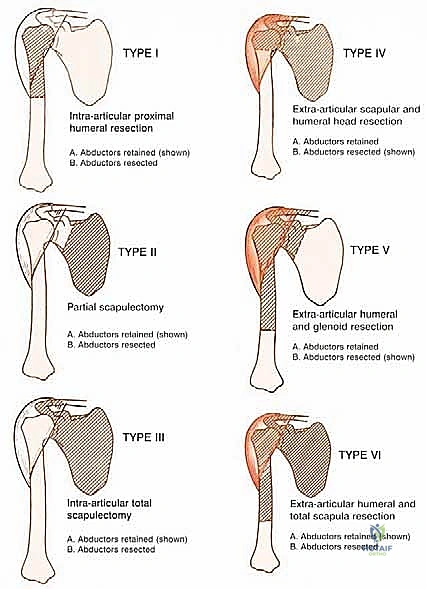
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution of a modified extra-articular shoulder girdle resection is a masterclass in anatomic dissection and oncologic discipline. The procedure is broadly divided into three distinct phases: the neurovascular exposure, the en bloc oncologic resection, and the complex endoprosthetic and soft tissue reconstruction.
The Utilitarian Exposure and Neurovascular Isolation
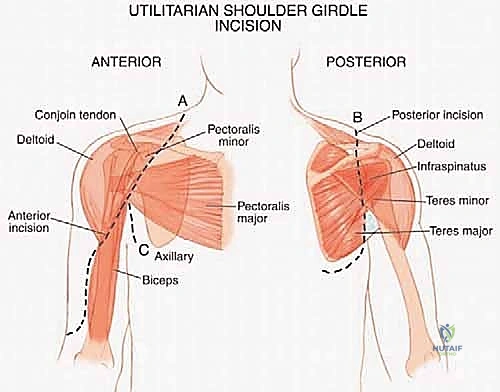
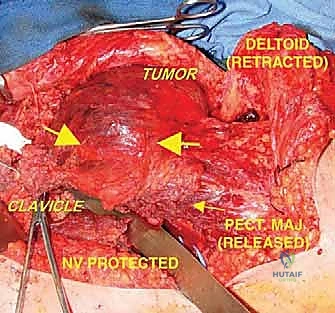
The procedure begins with a highly extensile utilitarian incision. The classic deltopectoral incision is utilized but extended significantly. Proximally, it tracks along the anterior border of the clavicle; distally, it extends down the anterolateral aspect of the arm following the lateral border of the biceps. Crucially, the prior biopsy tract must be incorporated into the incision design as an ellipse, ensuring it remains attached to the underlying tumor mass to be resected en bloc.
The initial and most critical phase of the deep dissection is the identification and isolation of the axillary neurovascular bundle. The deltopectoral interval is developed, taking care to retract the cephalic vein laterally (or ligate it if dictated by tumor proximity). The pectoralis major is identified and its insertion on the humerus is tagged and released. This exposes the clavipectoral fascia and the pectoralis minor. By retracting or releasing the pectoralis minor from the coracoid, the axillary sheath is exposed. The surgeon must meticulously dissect the axillary artery, axillary vein, and the cords of the brachial plexus, tracing them from proximal to distal. Vessel loops are placed around these critical structures to maintain absolute control and protect them throughout the remainder of the massive resection.
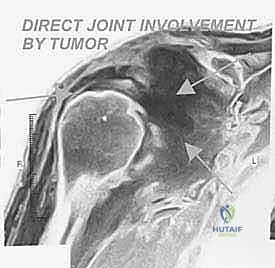
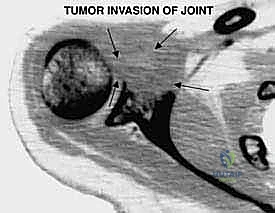
The En Bloc Oncologic Resection
Once the neurovascular bundle is secured and safely retracted medially, the compartmental resection begins. The insertions of the latissimus dorsi and teres major are identified on the medial aspect of the humerus, tagged, and released. The axillary nerve and posterior humeral circumflex vessels are identified as they enter the quadrangular space. In a true extra-articular resection for a high-grade proximal humerus sarcoma, these structures are typically ligated and transected to ensure the posterior pseudocapsule remains unbreached, sacrificing the deltoid muscle's innervation.
Attention is then turned to the osseous cuts. If an extra-articular resection is planned, the glenohumeral joint must not be opened. The coracoid process is osteotomized to release the short head of the biceps and coracobrachialis. The rotator cuff musculature (supraspinatus, infraspinatus, teres minor, subscapularis) is transected at its musculotendinous junction, far medial to the tumor pseudocapsule. A Gigli saw or oscillating saw is then used to perform the glenoid neck osteotomy, separating the glenoid from the body of the scapula.
Distally, the humeral diaphysis is exposed at the pre-planned resection level, ensuring a wide margin from the intramedullary extent of the tumor as determined by the preoperative MRI. The anterior compartment muscles (biceps, brachialis) are elevated off the humerus. The radial nerve must be identified and protected as it spirals around the posterior humerus in the spiral groove. The humeral osteotomy is performed, and the entire specimen—comprising the proximal humerus, the intact glenohumeral joint, the glenoid, the biopsy tract, and the surrounding muscular pseudocapsule—is removed en bloc and sent for immediate pathologic margin assessment.
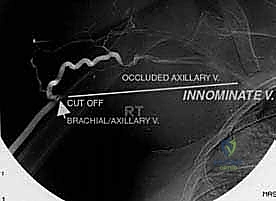
Endoprosthetic and Soft Tissue Reconstruction
With clear margins confirmed, the reconstructive phase commences. The goal is to restore skeletal stability and provide a durable soft tissue envelope. A modular proximal humeral endop
Clinical & Radiographic Imaging Archive