Mastering Intercalary Reconstruction: Allografts and Segmental Endoprostheses
Key Takeaway
Step into the operating room for a masterclass on intercalary reconstruction. This guide covers comprehensive preoperative planning, precise anatomical considerations, and granular intraoperative execution for allografts and segmental prostheses. Learn critical techniques for oncologic resection, fixation, and managing potential pitfalls. Gain insights into postoperative care for optimal patient outcomes in complex limb salvage procedures.
Comprehensive Introduction and Patho-Epidemiology
Welcome to the operating theater, colleagues. Today, we are undertaking a masterclass in one of the most technically demanding yet profoundly rewarding procedures in the realm of orthopaedic oncology: the intercalary reconstruction. This surgical endeavor represents a triumph of modern limb-salvage surgery, moving far beyond the mere replacement of a resected bone segment. It is a meticulously orchestrated effort to restore biomechanical function, preserve the native limb, and, above all, achieve definitive oncologic control. As you scrub in with me, understand that every preoperative calculation, every intraoperative osteotomy, and every choice of fixation hardware carries profound implications for the patient's survival and quality of life.
An intercalary reconstruction is precisely defined as the structural replacement of the diaphyseal portion of a long bone following a segmental skeletal resection, a procedure we formally term a diaphysectomy. The paramount objective of this approach is the preservation of the patient's native articular joints, both proximal and distal to the reconstruction zone. By maintaining the native joint mechanics, proprioception, and capsuloligamentous structures, this approach typically yields markedly superior functional outcomes when compared to more extensive osteoarticular resections or total joint arthroplasties. The paradigm shift from amputation to limb salvage over the past four decades has made intercalary reconstruction a cornerstone in the management of diaphyseal malignancies.
The patho-epidemiology of lesions necessitating this procedure is diverse. Primary malignant bone tumors, such as osteosarcoma and Ewing sarcoma, frequently arise in the metadiaphyseal or diaphyseal regions of long bones, particularly in the pediatric and young adult populations. Additionally, aggressive benign lesions like adamantinoma, fibrous dysplasia, and giant cell tumors, as well as isolated metastatic carcinomas and myeloma, frequently mandate segmental resection. The biological behavior of these tumors dictates the required oncologic margins, which in turn defines the magnitude of the resulting osseous defect.

To bridge these massive diaphyseal defects, we primarily rely on two distinct reconstructive avenues: bulk structural allografts or modular intercalary endoprostheses. We frequently favor bulk allografts in younger, higher-demand patients due to their potential for biological integration, restoration of bone stock, and long-term durability through osseous healing via creeping substitution. However, allograft reconstruction is highly dependent on favorable host biology. In scenarios characterized by poor host factors—such as compromised regional vascularity, extremely short residual host bone segments, or in patients undergoing aggressive adjuvant chemotherapy and radiation that significantly impede osteogenesis—an intercalary endoprosthetic reconstruction becomes the superior, and often necessary, alternative.
Detailed Surgical Anatomy and Biomechanics
Before a scalpel ever touches the skin, an encyclopedic understanding of regional cross-sectional anatomy and biomechanics is absolute. We are not merely carpenters working on inert structural supports; we are navigating a highly vascularized, innervated, and dynamically loaded biological landscape. The success of an intercalary reconstruction hinges on our ability to manipulate this environment without causing iatrogenic harm.
Femoral Considerations
When operating on the femur, we must meticulously account for its natural anterior bow and the physiological degree of femoral anteversion. These anatomical features are not merely cosmetic; they are the biomechanical foundation for proper load distribution and rotational control. Reconstructing a straight segment in a curved bone alters the mechanical axis, potentially leading to eccentric loading and premature failure of the implant or allograft. Most critically, we must maintain acute awareness of the superficial femoral artery (SFA). As the SFA courses distally through the adductor canal and passes through the adductor hiatus to become the popliteal artery, it lies in perilous proximity to the posteromedial aspect of the distal femoral diaphysis. Unplanned retraction or errant osteotomies in this zone can lead to catastrophic limb ischemia.
Tibial Considerations
The tibia presents a unique set of challenges, primarily due to its notoriously limited soft tissue envelope along the anteromedial border. This anatomical reality makes primary wound closure over bulky allografts or endoprostheses exceedingly difficult and drastically elevates the risk of wound dehiscence and deep periprosthetic infection. Posteriorly, the posterior tibial neurovascular bundle—comprising the posterior tibial artery, accompanying veins, and the tibial nerve—runs deep to the soleus, in close proximity to the posteromedial tibial cortex. Precise subperiosteal or extraperiosteal dissection, depending on the required oncologic margin, is vital to protect these structures from thermal necrosis during sawing or mechanical injury during tumor mobilization.
Humeral Considerations
In the humerus, the radial nerve is our most critical anatomical landmark and the primary structure at risk. It spirals around the posterior mid-humeral shaft within the spiral groove, accompanied by the profunda brachii artery, before piercing the lateral intermuscular septum. During any diaphyseal dissection, especially for tumor resection requiring wide margins, meticulous identification, neurolysis, and protection of the radial nerve are absolutely essential to prevent devastating iatrogenic wrist drop and loss of finger extension. Biomechanically, the humerus is subjected to significant torsional forces rather than the massive axial loads seen in the lower extremity. Therefore, our fixation constructs here must be exceptionally robust against rotational failure.
Forearm Considerations
Finally, for the forearm, particularly when reconstructing the radius, we must respect and restore the natural radial bow. This curvature is the mechanical fulcrum necessary for normal forearm pronation and supination. A perfectly straight reconstruction of the radius will invariably lead to a severe loss of rotational range of motion. Furthermore, the interosseous membrane must be carefully managed; its disruption can lead to longitudinal radioulnar dissociation (Essex-Lopresti type mechanics), compromising the stability of both the wrist and the elbow.
Exhaustive Indications and Contraindications
The decision algorithm for selecting the appropriate reconstructive modality is complex, multifactorial, and highly individualized. It requires a delicate balance between achieving immediate mechanical stability and pursuing long-term biological durability, all while factoring in the patient's oncologic prognosis and systemic health.
The primary indication for an intercalary reconstruction is a primary bone sarcoma, aggressive benign tumor, or solitary metastasis confined to the diaphysis, where an adequate oncologic margin can be achieved while preserving at least 4 to 5 centimeters of healthy, structurally sound epiphyseal/metaphyseal bone adjacent to the proximal and distal joints. This residual bone stock is the absolute minimum required to secure the stems of an endoprosthesis or to achieve stable osteosynthesis with an allograft. If this minimum bone stock cannot be preserved, the surgeon must abandon the intercalary approach and convert to an osteoarticular reconstruction or a total joint endoprosthesis.
When deciding between an allograft and a modular endoprosthesis, we must evaluate the patient's biological capacity for healing. Structural allografts are indicated in younger, active patients with a reasonable life expectancy who can tolerate a prolonged period of restricted weight-bearing and rehabilitation. Conversely, intercalary endoprostheses are indicated in older patients, those with metastatic disease and limited life expectancy, or patients undergoing aggressive adjuvant therapies that would reliably thwart allograft incorporation. Endoprostheses offer the distinct advantage of immediate structural stability, allowing for rapid mobilization and weight-bearing, which is crucial for preventing the systemic complications of prolonged immobility in oncologic patients.
Contraindications to intercalary reconstruction must be strictly respected. Absolute contraindications include intra-articular tumor extension, which mandates joint resection; massive soft tissue contamination or neurovascular encasement that precludes a limb-sparing approach entirely; and active, uncontrolled regional infection. Relative contraindications for allograft use include heavy prior radiation to the surgical bed, severe malnutrition, or systemic immunosuppression, all of which dramatically increase the risk of nonunion and deep infection.
| Factor | Bulk Structural Allograft | Modular Intercalary Endoprosthesis |
|---|---|---|
| Primary Indication | Young, active patients; high demand; long life expectancy; adequate soft tissue. | Older patients; metastatic disease; short life expectancy; need for immediate weight-bearing. |
| Biological Integration | Yes (creeping substitution, osteoconduction). | No (relies on mechanical fixation, though extracortical bridging can occur at collars). |
| Immediate Stability | Moderate (requires protected weight-bearing for 6-12 months). | Excellent (allows for immediate, full weight-bearing). |
| Impact of Chemotherapy | Significantly delays union; increases risk of nonunion and infection. | Minimal impact on immediate mechanical stability. |
| Late Complications | Fracture, nonunion, late deep infection. | Aseptic loosening, fatigue failure of stems/screws. |
| Revision Options | Can be revised to endoprosthesis if failed. | Modular design allows for relatively straightforward exchange or conversion to total joint. |
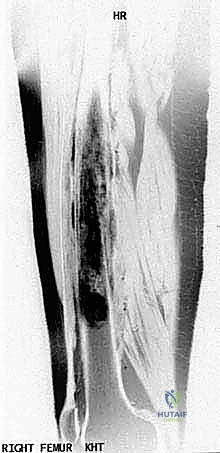
Pre-Operative Planning, Templating, and Patient Positioning
The bedrock of success in these high-stakes oncologic cases is obsessive, meticulous preoperative planning. This is not merely about reviewing images; it is about crafting a precise, millimeter-accurate blueprint for the entire surgical resection and subsequent reconstruction. We are dealing with life and limb, and intraoperative surprises are generally the result of preoperative oversights.
Comprehensive Imaging Review and Templating
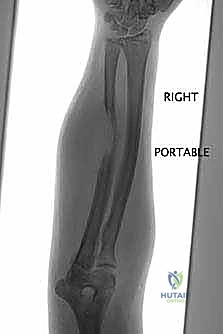
Our intelligence gathering begins with a multi-modality imaging approach. We start with high-quality, orthogonal plain radiographs. We assess the overall bone geometry, the lytic or blastic nature of the lesion, and critically, the remaining host bone stock. We must confirm that the residual metaphyseal segments can support our planned fixation. Radiographs also serve as the foundation for our initial digital templating, allowing us to estimate the required length of the allograft or the modular segments of the endoprosthesis.
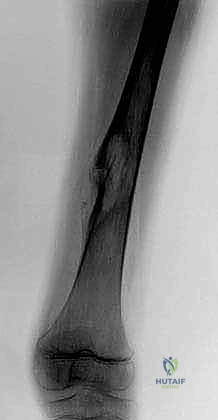
Next, Computed Tomography (CT) scanning is indispensable. CT provides exquisite, cross-sectional detail of cortical destruction and the exact bony footprint of the tumor. We utilize 3D CT reconstructions to plan our osteotomy levels with exacting precision, ensuring we achieve our planned oncologic margins while maximizing host bone preservation. Furthermore, CT allows us to measure the intramedullary canal diameter, which is vital for selecting the appropriate stem sizes for endoprostheses or intramedullary nails.
Magnetic Resonance Imaging (MRI) is our definitive window into the true extent of the disease. It accurately delineates the intraosseous marrow extent of the tumor, identifying any "skip" metastases within the same bone. Crucially, MRI defines the extraosseous soft tissue extension and its relationship to major neurovascular bundles. We rely heavily on T1-weighted coronal and sagittal images to measure the exact distance from stable, reproducible anatomical landmarks (such as the joint line) to our planned osteotomy levels.
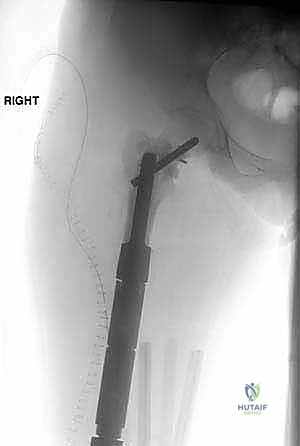

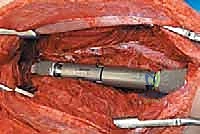
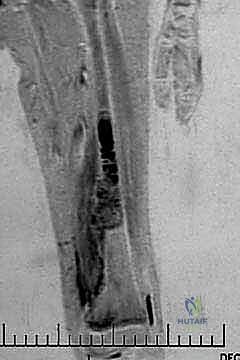
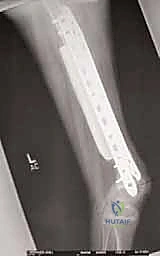
Surgical Strategy and Implant Selection
Armed with this imaging data, we finalize our reconstructive strategy. If an allograft is chosen, we coordinate with the tissue bank, providing them with our templated dimensions to secure a structurally matched graft. The allograft must match the host bone not only in length but also in cortical diameter and intramedullary canal size to ensure a flush osteosynthesis. For modular endoprostheses, we must ensure the vendor has the appropriate inventory of stems, intercalary segments, and collars available. If a custom, 3D-printed implant is required, we must account for the 3-to-4-week manufacturing lead time.
Patient Positioning: The Foundation of Exposure
Proper patient positioning dictates our surgical exposure, our ability to utilize intraoperative fluoroscopy, and our capacity to manage potential vascular complications. For femoral resections, the patient is typically positioned supine on a radiolucent table. A bump under the ipsilateral hemipelvis can facilitate a lateral approach, but the entire limb must be prepped and draped free to allow for dynamic assessment of rotation and length. Tibial cases are performed supine, again with the leg draped free. Humeral reconstructions may utilize the supine or beach-chair position, depending on the proximal extent of the tumor. In all cases, an examination under anesthesia to assess the resting rotational profile of the contralateral, healthy limb provides a vital reference point for restoring appropriate version during the reconstruction.
Step-by-Step Surgical Approach and Fixation Technique
The surgical approach is dictated primarily by the location of the tumor and the necessity of excising the prior biopsy tract en bloc with the specimen. We favor extensile, longitudinal incisions that can be extended proximally or distally if the intraoperative findings necessitate a wider resection or conversion to a joint-replacing procedure.
Tumor Resection and Osteotomy
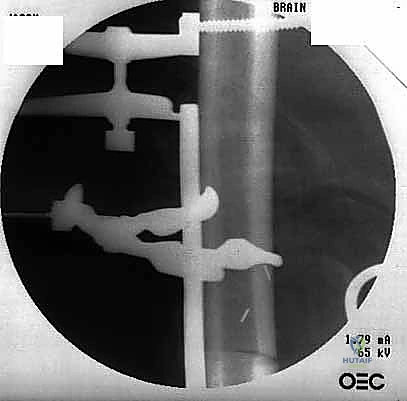
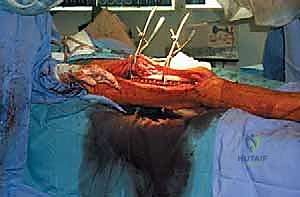
The dissection proceeds through healthy, uncontaminated tissue planes. We meticulously isolate and protect the major neurovascular structures early in the procedure. The tumor is mobilized with a cuff of normal muscle tissue, adhering strictly to the principles of wide oncologic margins. Once the diaphysis is exposed, we translate our preoperative MRI measurements to the bone using a sterile ruler and intraoperative fluoroscopy. We place K-wires at the planned osteotomy sites to mark our cuts. The osteotomies are performed using an oscillating saw under continuous saline irrigation to prevent thermal necrosis of the host bone, which would compromise subsequent healing or implant fixation. After the specimen is removed en bloc, marrow margins are sent for immediate frozen section analysis to confirm we are free of disease before proceeding with reconstruction.
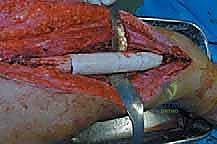
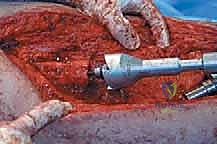

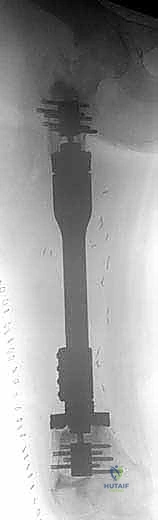
Allograft Preparation and Fixation
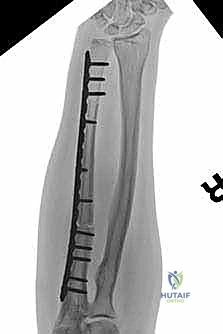
If utilizing a bulk allograft, it is thawed in warm antibiotic saline. We cut the allograft to precisely match the length of the resected defect. Step-cut osteotomies provide superior rotational stability and increase the surface area for osseous healing compared to simple transverse cuts, though they are technically more demanding. Fixation is achieved through rigid osteosynthesis. We frequently employ dual-plate constructs or a combination of an intramedullary nail and a supplementary plate. Compression across the host-graft junctions is absolutely critical to stimulate primary bone healing. In cases of massive defects or heavily irradiated beds, we may utilize the Capanna technique, placing a vascularized free fibula graft within the medullary canal of the massive allograft to provide a living, osteogenic core that drastically reduces the rate of nonunion and late fracture.
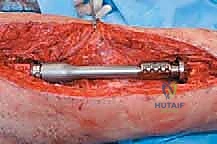


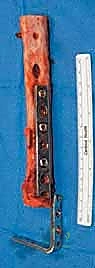
Endoprosthetic Reconstruction
For endoprosthetic reconstructions, the medullary canals of the proximal and distal host segments are sequentially reamed to accommodate the implant stems. We must decide between cemented or cementless fixation. Cementless, press-fit stems with hydroxyapatite-coated collars are generally preferred in younger patients with good bone quality, as they allow for extracortical bone bridging and biological integration at the collar-bone interface, reducing the risk of long-term aseptic loosening. Cemented stems are reserved for older patients with osteoporotic bone or those with short life expectancies. The modular intercalary segments are then assembled to the exact length required to restore limb length and soft tissue tension. Rotational alignment is meticulously checked against our preoperative clinical assessment and the linea aspera before final seating or cementing of the implants. Finally, meticulous soft tissue closure is performed, utilizing local or free muscle flaps (such as a medial gastrocnemius flap for proximal tibial defects) if primary closure over the implant is under tension.
Complications, Incidence Rates, and Salvage Management
Despite our most meticulous planning and execution, intercalary reconstructions carry a formidable complication profile. The nature of these massive structural replacements, combined with the immunosuppressive effects of adjuvant oncology treatments, creates a perfect storm for potential failures. Recognizing, preventing, and managing these complications is a hallmark of the experienced orthopaedic oncologist.
Nonunion and Structural Failure
Nonunion at the host-allograft junction is a frequent complication, occurring in 15% to 25% of allograft reconstructions. Chemotherapy and radiation are significant independent risk factors, as they severely depress osteoblastic activity. If a nonunion is identified and remains symptomatic or shows hardware failure, intervention is required. This typically involves aggressive decortication of the nonunion site, application of copious autogenous cancellous bone graft (often harvested from the iliac crest or utilizing reamer-irrigator-aspirator (RIA) techniques), and revision of the osteosynthesis to provide absolute rigid stability. Furthermore, allografts undergo a phase of creeping substitution where they become temporarily osteopenic and mechanically weaker before revascularizing. During this vulnerable period (typically 2 to 3 years post-operatively), late allograft fractures can occur, necessitating revision plating or conversion to an endoprosthesis.
Periprosthetic Infection
Deep periprosthetic infection is the absolute nemesis of limb salvage surgery. The incidence ranges from 5% to 15%, significantly higher than in standard arthroplasty, due to extensive soft tissue dissection, prolonged operative times, and immunocompromised hosts. Bacteria, notably Staphylococcus aureus and Staphylococcus epidermidis, rapidly form impenetrable biofilms on the avascular surfaces of massive allografts and metallic endoprostheses. Management of a deep infection rarely succeeds with antibiotics alone. It almost universally requires aggressive surgical debridement. For infected endoprostheses, a two-stage revision utilizing an antibiotic-loaded cement spacer is the gold standard. For infected massive allografts, the graft must typically be entirely excised, followed by spacer placement and eventual reconstruction with an endoprosthesis once the infection is eradicated. In recalcitrant cases, amputation may be the only life-saving salvage option.
Aseptic Loosening and Implant Failure
While less common in intercalary endoprostheses compared to joint-replacing mega-prostheses (due to the absence of a bearing surface generating wear debris), aseptic loosening can still occur at the stem-bone interface. This is typically driven by stress shielding or insufficient initial fixation. Furthermore, the immense bending and torsional moments placed on these implants can lead to fatigue fracture of the stems or the modular junction screws. Revision involves extraction of the failed components and
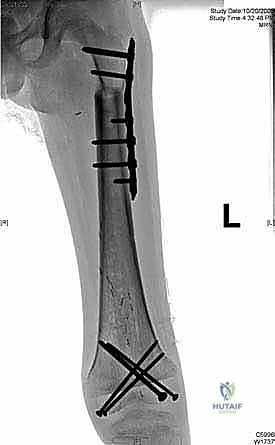
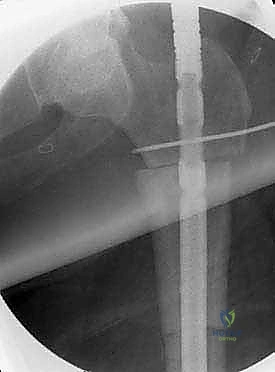
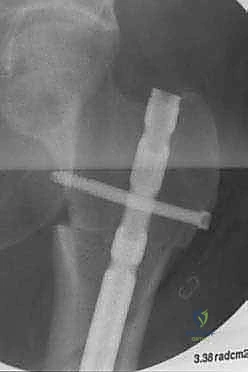
Clinical & Radiographic Imaging Archive