Distal Femoral Endoprosthetic Reconstruction: A Masterclass in Oncologic Limb Salvage
Key Takeaway
This masterclass provides an exhaustive, real-time guide to distal femoral endoprosthetic reconstruction for limb salvage. Fellows will learn comprehensive surgical anatomy, meticulous preoperative planning, and granular intraoperative execution from incision to implant. We cover precise dissection, oncologic margins, hardware application, and soft tissue management, alongside critical pearls, pitfalls, and postoperative care, ensuring a functional, pain-free outcome for complex cases.
Comprehensive Introduction and Patho-Epidemiology
The evolution of orthopedic oncology over the past five decades represents one of the most profound paradigm shifts in modern surgical science. Historically, the diagnosis of a malignant primary bone tumor of the distal femur—most notably osteosarcoma—was synonymous with high-level amputation, typically a hip disarticulation or high transfemoral amputation, carrying a dismal five-year survival rate of less than twenty percent. Today, we stand in a vastly different era. The advent of efficacious multi-agent neoadjuvant and adjuvant chemotherapy protocols, pioneered in the 1970s and 1980s, fundamentally altered the systemic management of these aggressive sarcomas. Concurrently, surgical pioneers such as Ralph Marcove and Kenneth Francis relentlessly pushed the boundaries of limb salvage, establishing the conceptual framework for wide en bloc resection coupled with durable endoprosthetic reconstruction. This chapter serves as an exhaustive masterclass on the distal femoral endoprosthetic reconstruction, a procedure that epitomizes the delicate balance between aggressive oncologic extirpation and sophisticated biomechanical restoration.
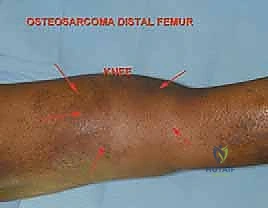
Patho-epidemiologically, the distal femur is the single most common anatomic site for primary malignant bone tumors, harboring approximately forty percent of all osteosarcomas and a significant proportion of chondrosarcomas, Ewing sarcomas, and undifferentiated pleomorphic sarcomas of bone. The unique vascularity and rapid cellular proliferation at the distal femoral physis during the adolescent growth spurt create a microenvironment highly susceptible to malignant transformation. Furthermore, the distal femur is a frequent repository for metastatic carcinoma (particularly from breast, prostate, lung, thyroid, and renal cell primaries) and multiple myeloma. The fundamental goal of surgical intervention in this region is tripartite: achieve a wide, margin-negative oncologic resection to minimize local recurrence; provide an immediately stable, robust skeletal reconstruction that permits early mobilization; and ensure durable, well-vascularized soft tissue coverage to facilitate wound healing and optimize functional outcomes.
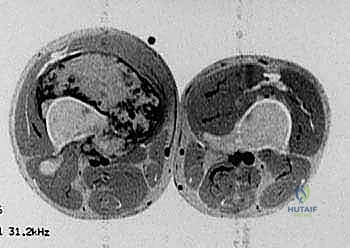
Understanding the biological behavior of these tumors is an absolute prerequisite for surgical planning. Osteosarcomas typically arise in the intramedullary metaphysis and rapidly penetrate the cortex, elevating the periosteum (producing the classic Codman's triangle) and extending into the surrounding soft tissues. This extraosseous extension dictates the required margin of resection, necessitating a meticulous dissection through virgin tissue planes to avoid tumor capsular violation. Conversely, metastatic lesions often present with extensive lytic destruction, predisposing the patient to impending or completed pathologic fractures. In such scenarios, the surgical field is frequently contaminated by hematoma, mandating a more radical resection to encompass the entire zone of hemorrhage. The interplay between the tumor's biological aggressiveness, its response to neoadjuvant therapy (assessed via histologic necrosis mapping), and its precise anatomical footprint ultimately dictates the feasibility and extent of the distal femoral endoprosthetic reconstruction.
Detailed Surgical Anatomy and Biomechanics
Before the scalpel ever meets the skin, the operating surgeon must possess an intimate, three-dimensional understanding of the distal thigh's complex anatomy. The distal femur serves as a critical anatomical crossroads, acting as the primary load-bearing strut of the lower extremity while simultaneously serving as the origin and insertion for the massive musculature of the thigh and leg. The osteology of the distal femur is characterized by a transition from the dense, cylindrical cortical bone of the diaphysis to the flared, cancellous bone of the metaphysis and epiphysis. This metaphyseal flare is biomechanically crucial, as it dissipates the immense compressive and torsional forces transmitted across the knee joint. When planning an osteotomy, the surgeon must carefully evaluate the bone quality at the intended resection margin via high-resolution computed tomography (CT), ensuring adequate cortical thickness to support the stem of the endoprosthesis, whether it is destined for cemented or press-fit fixation.
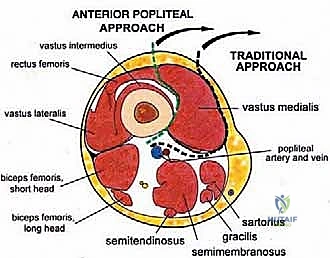
Muscular Intervals and Compartmental Anatomy
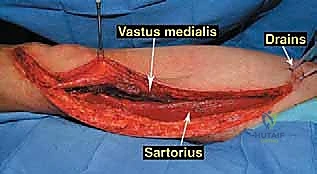
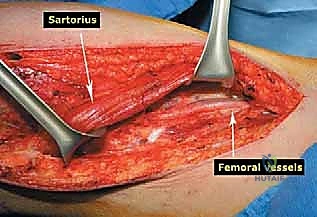
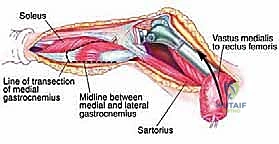
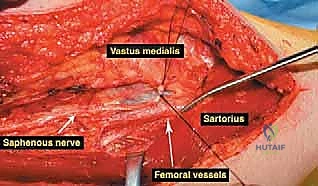
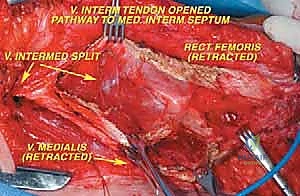
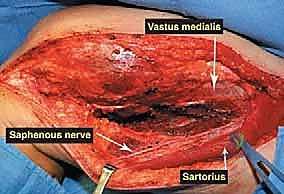
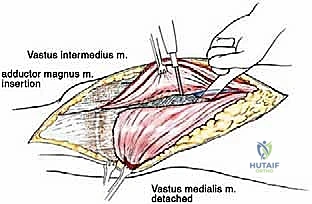
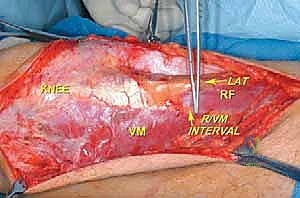
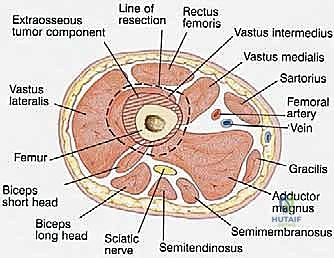
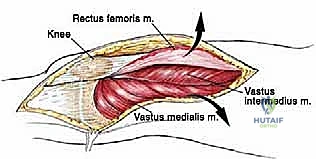
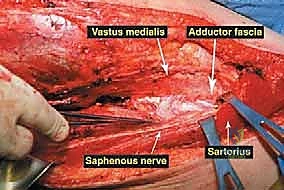
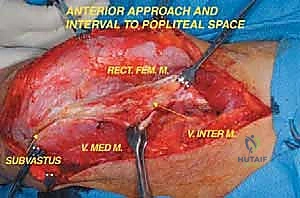
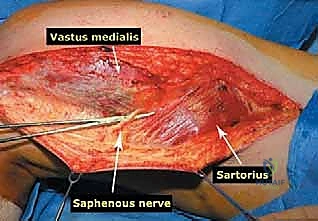
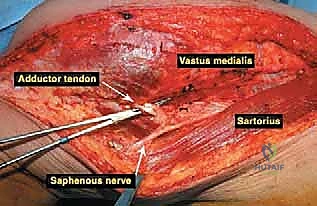
The musculature surrounding the distal femur is divided into three distinct compartments, each defined by robust fascial septa. The anterior compartment, dominated by the quadriceps femoris group, is our primary surgical corridor. The rectus femoris, vastus lateralis, vastus medialis, and vastus intermedius envelop the anterior and lateral aspects of the femur. The vastus intermedius, lying deep to the rectus femoris, is frequently sacrificed en bloc with the tumor to ensure an adequate anterior margin. The vastus medialis is of particular strategic importance; its distal muscular fibers (the vastus medialis obliquus) are essential for dynamic patellar tracking, while its deep fascial layer forms the anterior and lateral boundaries of the adductor (Hunter's) canal. Preserving the innervation to the vastus medialis, derived from the femoral nerve, is a critical objective during the approach.
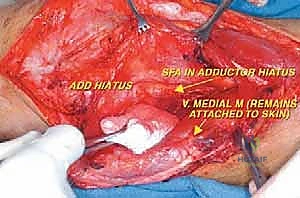
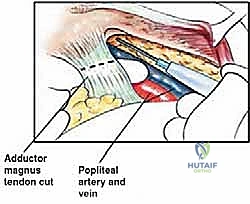
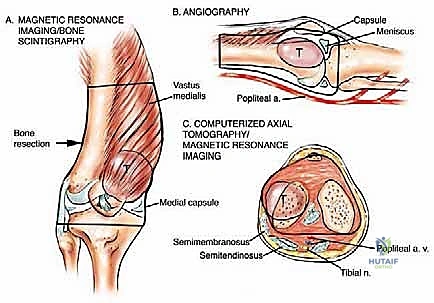
The medial compartment houses the sartorius muscle, the adductor longus, adductor brevis, and the massive adductor magnus. The sartorius, running obliquely across the thigh, forms the roof of the adductor canal. The adductor magnus is a critical landmark; its broad insertion along the linea aspera and the medial supracondylar ridge forms the posterior boundary of the adductor canal. Distally, the tendinous insertion of the adductor magnus onto the adductor tubercle creates the adductor hiatus, the anatomical gateway through which the superficial femoral vessels transition into the popliteal space. The posterior compartment, comprising the hamstring muscles (semitendinosus, semimembranosus, and biceps femoris), defines the superior boundaries of the popliteal fossa. These muscles are rarely directly invaded by distal femoral tumors unless the lesion is massive or arises from the posterior cortex, but they must be carefully retracted during posterior dissection.
Neurovascular Topography and Joint Considerations
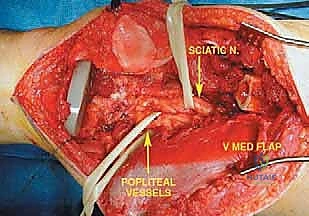
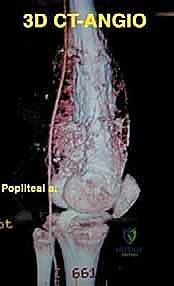
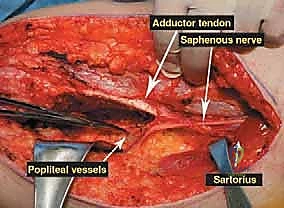
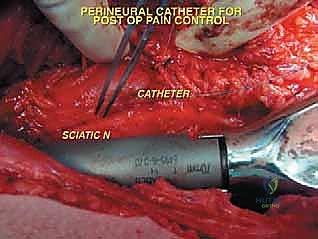
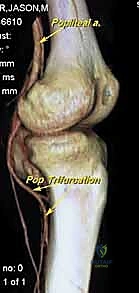
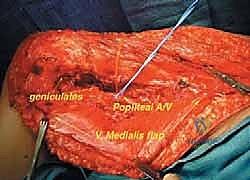
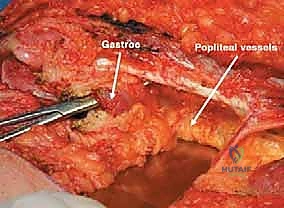
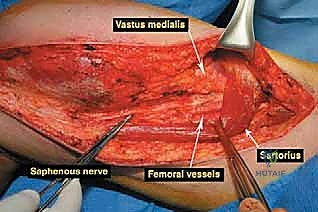
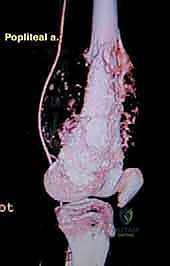
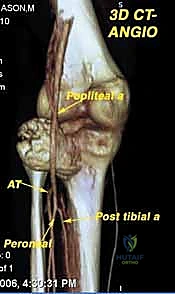
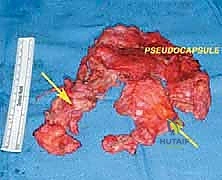
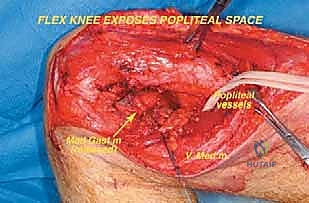
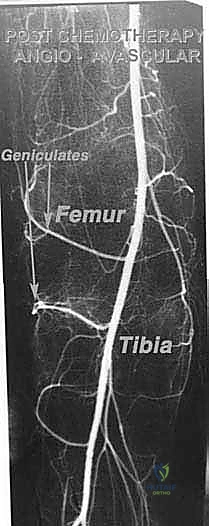
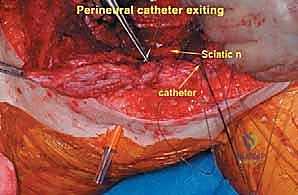
Navigating the neurovascular structures is arguably the most perilous aspect of this procedure. The adductor canal, located in the middle third of the thigh, houses the superficial femoral artery, femoral vein, and the saphenous nerve. In large, bulky tumors, this canal can be severely displaced medially or anteriorly. Crucially, the vessels are typically protected by a tough fascial sheath and the deep fascia of the vastus medialis; direct tumor penetration through this fascial barrier is rare but mandates immediate vascular surgical consultation if suspected. As the vessels pass through the adductor hiatus, they become the popliteal artery and vein, situated deep within the popliteal fossa. The popliteal vessels are intimately related to the posterior capsule of the knee joint and are frequently displaced posteriorly by expanding metaphyseal tumors. Dissection in this region must be meticulous, utilizing blunt techniques to separate the neurovascular bundle from the posterior tumor pseudocapsule.
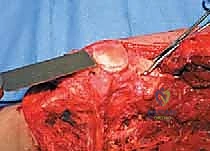
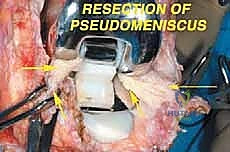
The knee joint itself presents unique oncologic challenges. While primary bone sarcomas rarely exhibit direct, trans-articular invasion into the joint space, contamination can easily occur via an inappropriately placed biopsy tract, microscopic extension along the intra-articular cruciate ligaments, or a pathologic intra-articular fracture. The cruciate ligaments, particularly at their bone-tendinous junctions within the intercondylar notch, lack a protective cartilaginous barrier and serve as a potential conduit for tumor spread. If preoperative magnetic resonance imaging (MRI) suggests cruciate ligament involvement, a true extra-articular resection—encompassing the entire knee joint capsule, the patella, and the proximal tibial articular surface en bloc—may be indicated. However, in the majority of cases, an intra-articular resection with en bloc removal of the cruciate ligaments and the distal femur is oncologically sufficient, preserving the proximal tibia and simplifying the subsequent reconstruction.
Exhaustive Indications and Contraindications
The indications for distal femoral endoprosthetic reconstruction have expanded dramatically over the past three decades. Initially conceived as a salvage procedure exclusively for primary malignant bone tumors, the unparalleled durability and modularity of modern rotating-hinge endoprostheses have broadened their application to a wide spectrum of complex orthopedic pathologies. The primary indication remains the surgical management of high-grade sarcomas (osteosarcoma, Ewing sarcoma, chondrosarcoma) of the distal femur, where wide en bloc resection creates a massive segmental osseous defect that cannot be managed with conventional osteosynthesis or allografting alone. Furthermore, locally aggressive benign conditions, such as Campanacci Stage III giant cell tumors of bone that have destroyed the articular surface or resulted in extensive cortical breakout, are excellent candidates for this reconstructive modality.
In the realm of metastatic disease, endoprosthetic reconstruction is indicated for highly destructive lesions that are not amenable to intralesional curettage and prophylactic internal fixation. Approximately ten percent of patients with metastatic bone disease to the distal femur present with massive bone loss, complete destruction of the metaphyseal-diaphyseal junction, or failed prior osteosynthesis. In these scenarios, a segmental resection and endoprosthetic replacement provide immediate, rigid stability, allowing for rapid mobilization and pain relief in a patient population with a limited life expectancy. Beyond oncology, this technology has been increasingly adopted by arthroplasty and trauma surgeons. It is utilized for catastrophic failure of total knee arthroplasty (TKA) with massive periprosthetic bone loss, highly comminuted supracondylar femur fractures in severely osteoporotic elderly patients, and primary knee replacements in patients with severe, rigid flexion contractures or profound ligamentous instability where conventional constrained TKA systems would invariably fail.
Despite its versatility, distal femoral endoprosthetic reconstruction is not without strict contraindications. The decision to proceed with limb salvage versus amputation requires a multidisciplinary approach, weighing the oncologic imperatives against the patient's physiological reserve and anticipated functional outcome. Absolute contraindications include major neurovascular encasement by the tumor that precludes margin-negative resection without sacrificing the sciatic nerve or popliteal artery (unless a vascular bypass is planned and feasible). Furthermore, active, uncontrolled periprosthetic or systemic infection is an absolute contraindication to the implantation of massive metallic hardware.
| Category | Indications | Contraindications |
|---|---|---|
| Oncologic (Primary) | High-grade osteosarcoma, Ewing sarcoma, Chondrosarcoma requiring wide resection. | Encasement of the sciatic nerve or major popliteal vessels precluding negative margins. |
| Oncologic (Benign/Metastatic) | Stage III Giant Cell Tumor with joint destruction; Massive metastatic osteolysis with impending/actual fracture. | Inability to achieve a wide margin without rendering the limb completely functionless. |
| Revision/Trauma | Catastrophic TKA failure with massive bone loss; Osteoporotic supracondylar non-unions. | Active, uncontrolled local periprosthetic joint infection (PJI) or systemic sepsis. |
| Patient Factors | Motivated patient capable of undergoing extensive rehabilitation. | Severe medical comorbidities precluding prolonged anesthesia; lack of soft tissue coverage. |
Pre-Operative Planning, Templating, and Patient Positioning
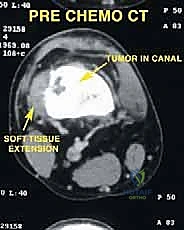
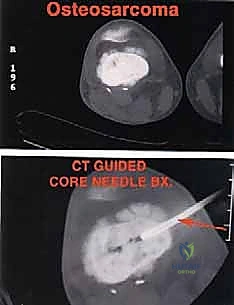
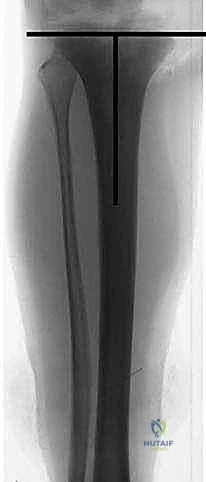
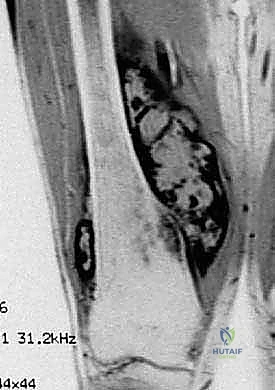
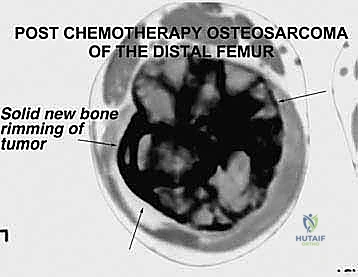
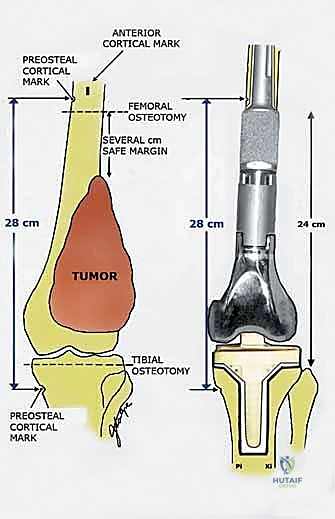
The success of a distal femoral endoprosthetic reconstruction is inextricably linked to the meticulousness of the preoperative planning phase. In the modern era, this process begins with a comprehensive review of multi-modal advanced imaging. High-resolution, contrast-enhanced MRI of the entire femur is the gold standard for delineating the intramedullary extent of the tumor (the "skip" lesions), the precise extraosseous soft tissue mass, and the relationship of the tumor pseudocapsule to critical neurovascular structures. The MRI dictates the exact level of the femoral osteotomy, which must be planned a minimum of 2 to 3 centimeters proximal to the most proximal extent of the intramedullary marrow abnormality. Concurrently, a fine-cut CT scan of the femur is essential for evaluating cortical integrity, measuring the endosteal canal diameter for stem sizing, and assessing the rotational profile of the limb.
Digital Templating and Biopsy Considerations
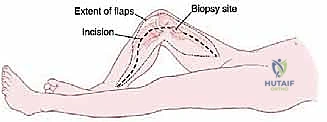
Digital templating is a non-negotiable step in the preoperative workflow. Using specialized orthopedic software, the surgeon overlays the modular endoprosthetic components onto the calibrated digital radiographs and CT multi-planar reconstructions. The surgeon must determine the precise resection length to restore leg length equality, select the appropriate stem diameter and length (aiming for intimate endosteal contact while avoiding cortical perforation in the anteriorly bowed femur), and choose the optimal bushing and axle sizes for the rotating hinge mechanism. Furthermore, the previous biopsy tract must be meticulously evaluated. Oncologic principles mandate that the biopsy tract—including the skin incision, subcutaneous tissues, and muscle trajectory—must be excised en bloc with the primary tumor specimen. Therefore, the definitive surgical incision must be planned to incorporate the biopsy site in an elliptical fashion.
Patient Positioning and Surgical Preparation
Patient positioning in the operating theater is critical for optimal exposure and intraoperative flexibility. The patient is placed strictly supine on a radiolucent operating table. A small bump or sandbag is placed under the ipsilateral hemipelvis to prevent external rotation of the limb, ensuring the patella points directly anteriorly. This neutral alignment is crucial for accurately assessing the rotational alignment of the femoral component during implantation. A sterile pneumatic tourniquet is applied as proximal as possible on the thigh; however, in cases of very proximal tumor extension, the tourniquet may be omitted entirely, and the surgeon must be prepared for rapid control of the femoral vessels. The entire limb is prepped and draped freely, from the iliac crest down to the toes, allowing for circumferential access to the limb and the ability to manipulate the knee through a full range of motion during the trialing phase. Intravenous prophylactic antibiotics, carefully selected based on institutional antibiograms, are administered 30 minutes prior to the skin incision.

Step-by-Step Surgical Approach and Fixation Technique
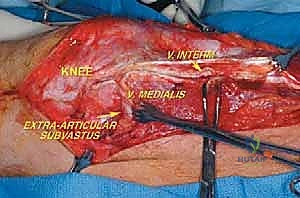
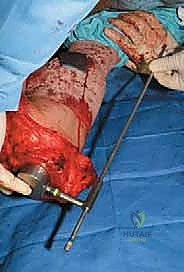
The surgical approach for a distal femoral resection is typically an extensile anteromedial or anterolateral incision, dictated primarily by the location of the tumor's extraosseous extension and the placement of the prior biopsy tract. The incision begins in the middle to proximal third of the thigh, centered over the femoral diaphysis, and extends distally over the patella, terminating at the tibial tubercle. If an anteromedial approach is utilized, the dissection proceeds through the subcutaneous tissues to identify the interval between the rectus femoris and the vastus medialis. The rectus femoris is mobilized and retracted laterally, exposing the vastus intermedius, which is often intimately involved with the anterior cortex of the distal femur and is routinely sacrificed en bloc with the specimen.
Vascular Mobilization and Posterior Dissection
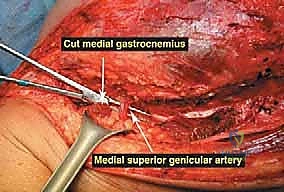
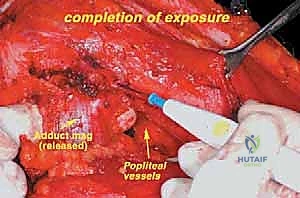
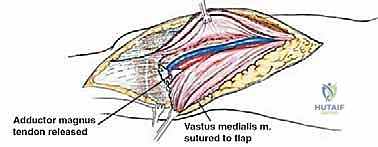
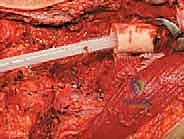
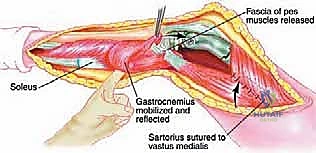
Once the anterior compartment is addressed, attention turns to the medial aspect of the distal femur. The deep fascia of the vastus medialis is incised to enter the adductor canal. The superficial femoral artery and vein are systematically identified, mobilized, and protected with vessel loops. As the dissection proceeds distally toward the adductor hiatus, the tendinous insertion of the adductor magnus is identified and sharply transected, allowing the vessels to be retracted posteriorly and laterally away from the tumor pseudocapsule. The posterior dissection is the most treacherous phase. Working from both medial and lateral sides, the surgeon uses blunt finger dissection and careful electrocautery to separate the popliteal vessels and the sciatic nerve branches from the posterior aspect of the tumor. The medial and lateral superior genicular arteries are frequently encountered and must be selectively ligated. The origins of the medial and lateral heads of the gastrocnemius muscle are detached from the posterior femoral condyles, further exposing the posterior joint capsule.
The Osteotomy and En Bloc Resection
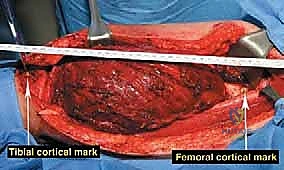
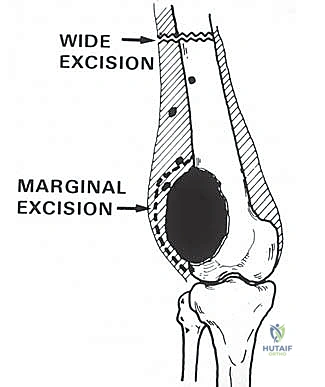
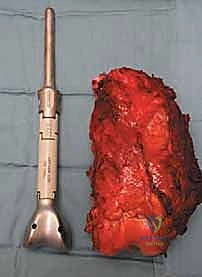
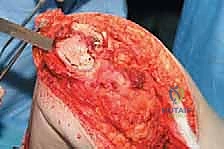
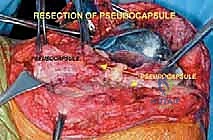
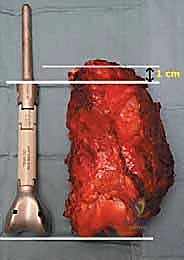
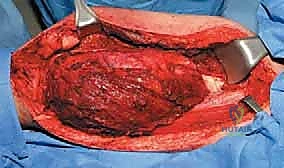
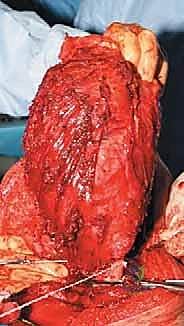
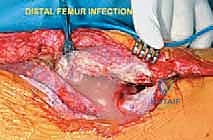
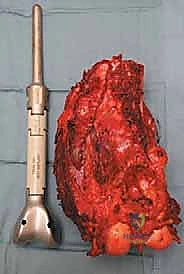
With the tumor circumferentially mobilized and the neurovascular structures protected, the definitive femoral osteotomy is performed. The exact level, predetermined during preoperative MRI templating, is measured meticulously from the joint line or the medial epicondyle. A sterile ruler and intraoperative fluoroscopy are utilized to confirm the level. The osteotomy is executed using a heavy-duty oscillating saw, ensuring a perfectly transverse cut to maximize contact with the collar of the endoprosthesis. Once the bone is transected, the distal segment is elevated, and the remaining posterior soft tissue attachments are divided. The knee joint capsule is incised circumferentially, and the cruciate ligaments are transected at their tibial insertions. The entire specimen—comprising the distal femur, the tumor, the pseudocapsule, the sacrificed vastus intermedius, and the biopsy tract—is removed en bloc and immediately handed to the pathology team for margin analysis.
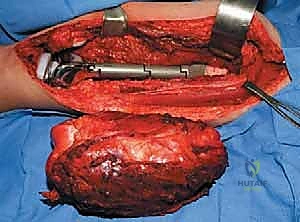
Endoprosthetic Reconstruction and Soft Tissue Closure
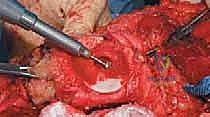
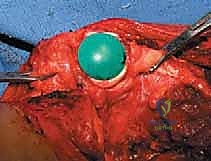
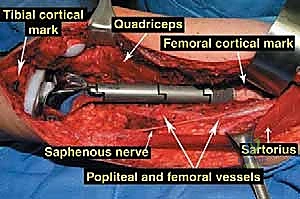
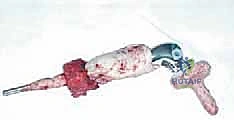
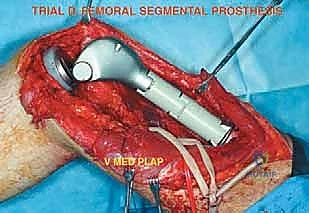
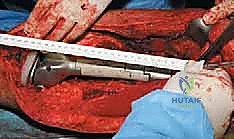
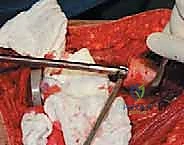
The reconstruction phase begins with the preparation of the proximal tibial plateau. A standard TKA tibial cut is performed, ensuring appropriate posterior slope and varus/valgus alignment. The tibial canal is reamed, and the tibial baseplate is implanted, typically using polymethylmethacrylate (PMMA) bone cement. Attention then returns to the femoral diaphysis. The medullary canal is sequentially reamed to accommodate the selected femoral stem. If cemented fixation is chosen (the gold standard for oncology patients with limited life expectancy or poor bone quality), the canal is thoroughly irrigated with pulsatile lavage, dried, and a restrictive cement plug is inserted. PMMA is injected in a retrograde fashion using a cement gun, and the femoral stem is pressurized into the canal. Rotational alignment is critical; the linea aspera serves as a reliable posterior landmark to ensure the femoral component is inserted with appropriate anteversion.
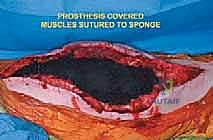
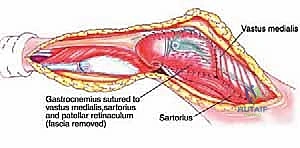
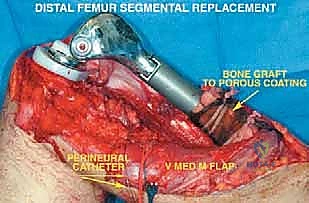
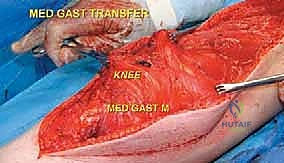
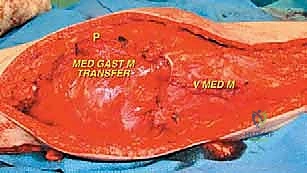
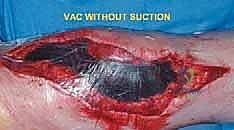
Once the cement has polymerized, the modular intercalary segments are connected via Morse taper junctions to restore the exact length of the resected bone. The rotating hinge mechanism is then assembled, connecting the femoral and tibial components with a high-molecular-weight polyethylene bushing and a metallic axle. The hinge allows for flexion and extension while permitting a controlled degree of internal and external rotation, significantly reducing the torsional stresses transmitted to the stem-bone interface. Finally, meticulous soft tissue reconstruction is paramount. The extensor mechanism must be centralized and securely reattached. If the vastus medialis was sacrificed, a medial gastrocnemius rotational flap may be required to provide robust, vascularized coverage over the metallic endoprosthesis, preventing hardware exposure and facilitating wound healing.
Complications, Incidence Rates, and Salvage Management
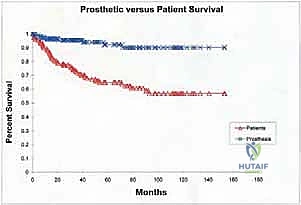
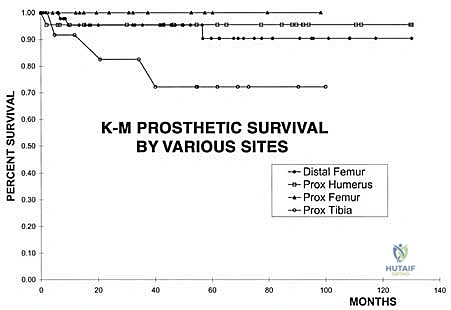
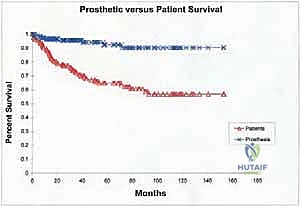
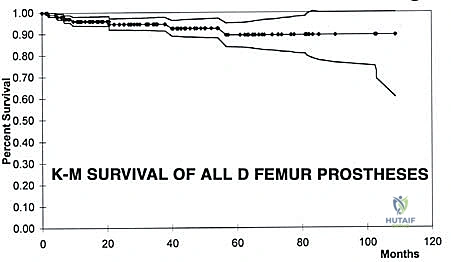
Despite remarkable advancements in implant metallurgy and surgical technique, distal femoral endoprosthetic reconstruction remains a massive undertaking fraught with potential complications. The sheer volume of foreign material implanted, combined with the immunocompromised state of the oncologic patient (often undergoing concurrent chemotherapy), creates a perfect storm for adverse events. The landmark classification system proposed by Henderson et al. categorizes endoprosthetic failures into five distinct modes: Type 1 (Soft Tissue Failure), Type 2 (Aseptic Loosening), Type 3 (Structural Failure), Type 4 (Infection), and Type 5 (Tumor Progression). Understanding the incidence and management of these complications is essential for the orthopedic oncologist.
Periprosthetic joint infection (PJI) (Henderson Type 4) is arguably the most devastating complication, occurring in 5% to 15% of cases. The extensive soft tissue dissection, prolonged operative time, and massive dead space contribute to this high risk. Management of PJI in the setting of a megaprosthesis typically requires a grueling two-stage revision protocol: complete hardware explantation, aggressive surgical debridement, placement of an antibiotic-impregnated PMMA spacer, and a prolonged course of intravenous antibiotics tailored to the offending organism. Only after eradication of the infection is confirmed can a new endoprosthesis be reimplanted. In refractory cases, or when soft tissue coverage is impossible, an above-knee amputation or hip disarticulation may be the only life-saving salvage option.
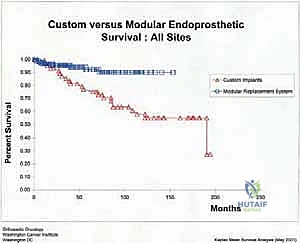
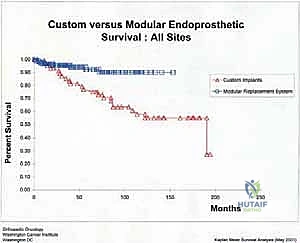
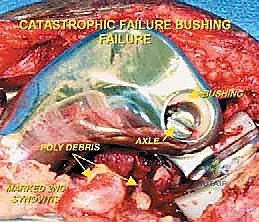
Aseptic loosening (Henderson Type 2) is the most common mode of long-term failure, particularly in young, active survivors of pediatric osteosarcoma. The immense stress-shielding at the metaphyseal-diaphyseal junction and the torsional forces acting on the rigid stem inevitably lead to osteolysis and implant micromotion. Revision surgery for aseptic loosening often requires the use of longer, larger-diameter stems, potentially utilizing porous-coated collars or extracortical bone grafting techniques to enhance biological fixation. Structural failures (Henderson Type 3), such as fracture of the femoral stem or catastrophic wear of the polyethylene hinge bushing, are less common with modern titanium and cobalt-chrome alloys but still occur, necessitating modular component exchange.
| Henderson Failure Mode | Description | Estimated Incidence | Salvage Management Strategy |
|---|---|---|---|
| Type 1 | Soft Tissue Failure (e.g., extensor lag, wound necrosis) | 10 - 15% | Gastrocnemius flap coverage; Extensor mechanism allograft reconstruction. |
| Type 2 | Aseptic Loosening (Stem subsidence or radiolucency) | 15 - 25% (Long-term) | Revision to longer/thicker stem; Impaction bone grafting; Cementless fixation. |
| Type 3 | Structural Failure (Stem fracture, bushing wear) | 5 - 10% | Modular component exchange; Bushing replacement via minor arthrotomy. |
| Type 4 | Periprosthetic Joint Infection (PJI) | 5 - 15% | Two-stage revision with antibiotic spacer; Suppressive antibiotics; Amputation. |
| Type 5 | Local Tumor Recurrence | 5 - 10% | Radical re-resection; Above-knee amputation or hip disarticulation. |