Distal Humeral Tumor Resection & Prosthetic Elbow Reconstruction: An Operative Masterclass
Key Takeaway
Join us in the OR for a masterclass on distal humeral tumor resection and prosthetic reconstruction. We'll meticulously cover intricate surgical anatomy, essential preoperative planning, and a granular, real-time breakdown of every intraoperative step. Learn critical techniques for neurovascular protection, precise osteotomy, modular prosthesis implantation, and soft tissue reconstruction, alongside crucial pearls and pitfalls for optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
The management of neoplastic lesions involving the distal humerus represents one of the most formidable challenges in orthopedic oncology. Historically, primary bone sarcomas and destructive metastatic lesions in this anatomical region were managed with radical amputation, given the profound difficulty of achieving wide oncologic margins while simultaneously preserving a functional limb. However, the advent of neoadjuvant chemotherapy, sophisticated advanced imaging modalities, and highly modular endoprosthetic reconstruction systems has catalyzed a paradigm shift toward limb-salvage surgery. Today, distal humeral tumor resection with prosthetic elbow reconstruction is the gold standard, offering patients not only local tumor control but also a durable, functional upper extremity.
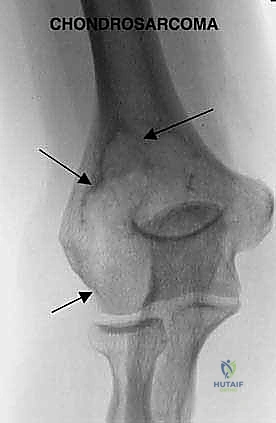
Primary bone sarcomas of the distal humerus, such as osteosarcoma, Ewing sarcoma, and chondrosarcoma, are relatively rare compared to their predilection for the distal femur or proximal tibia. Conversely, the distal humerus is a frequent site for metastatic dissemination, particularly from carcinomas of the breast, lung, thyroid, kidney, and prostate, as well as hematologic malignancies like multiple myeloma. Pathologic fractures in this region are notoriously difficult to manage with conventional osteosynthesis due to profound bone loss, poor bone quality, and the deleterious effects of adjuvant radiotherapy. In these scenarios, en bloc resection and endoprosthetic reconstruction provide immediate mechanical stability, rapid pain relief, and expedited restoration of function, which is paramount for patients with limited life expectancies.
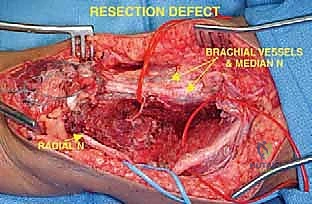
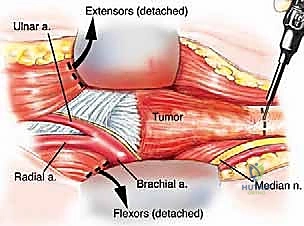
Executing this procedure requires an operative masterclass in regional anatomy, meticulous preoperative planning, and precise surgical technique. The distal humerus is intimately surrounded by a complex network of critical neurovascular structures—namely the brachial artery, and the median, ulnar, and radial nerves. The tumor often distorts these structures, displacing them from their native anatomical beds. The surgical objective is twofold: first, to perform an aggressive, wide en bloc resection that minimizes the risk of local recurrence; and second, to reconstruct the skeletal defect with a segmental hinged prosthesis that restores the highly constrained biomechanics of the elbow joint. This dual mandate demands that the orthopedic surgeon be equally adept at radical tumor extirpation and complex joint arthroplasty.
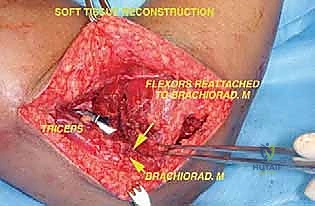
Furthermore, the soft tissue envelope surrounding the elbow is notoriously thin, making the reconstruction highly susceptible to wound breakdown and deep periprosthetic joint infection. The extensor mechanism, specifically the triceps brachii, must often be mobilized, partially resected, or detached to achieve adequate exposure and oncologic margins. The subsequent reattachment of the triceps to the metallic prosthesis is a critical determinant of postoperative functional outcome. Thus, the operation does not end with the implantation of the hardware; meticulous soft tissue handling, dead space management, and robust myofascial coverage are absolutely essential to prevent catastrophic postoperative complications.
Detailed Surgical Anatomy and Biomechanics
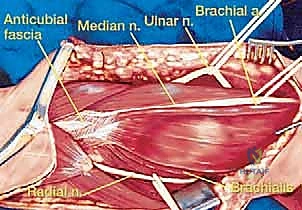
A profound, three-dimensional understanding of the neurovascular anatomy of the middle and distal arm is the bedrock of safe oncologic resection. The critical neurovascular structures in the middle one-third of the arm lie within a fibrous sheath in the groove between the biceps and triceps muscles, medial to the brachialis muscle. The brachial artery, accompanied by its venae comitantes, courses distally along the medial aspect of the arm. In the antecubital fossa, it rests on the anterior surface of the brachialis, immediately adjacent and lateral to the median nerve. Direct neoplastic invasion of the brachial artery and veins typically precludes limb-sparing surgery, necessitating amputation, whereas mere displacement allows for meticulous neurolysis and preservation.
The major peripheral nerves must be systematically identified in normal, virgin tissue proximal to the tumor bulk and traced distally. The median nerve travels distally in the arm, closely applied to the anterior aspect of the brachial artery, crossing medially as it approaches the antecubital fossa. The ulnar nerve occupies a position slightly more medial and posterior to the brachial artery in the mid-arm; it pierces the medial intermuscular septum to enter the posterior compartment, ultimately traversing the cubital tunnel posterior to the medial epicondyle. The radial nerve, perhaps the most vulnerable structure during distal humeral resections, courses through the spiral groove between the medial and lateral heads of the triceps before piercing the lateral intermuscular septum to enter the anterior compartment between the brachioradialis and brachialis muscles.
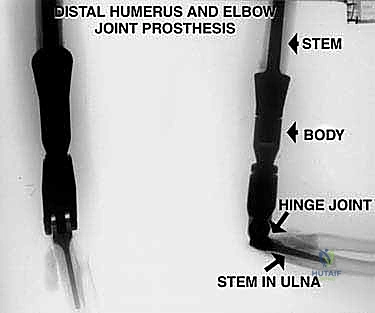
Biomechanically, the native elbow is a highly constrained, complex hinge joint that relies on the congruency of the ulnohumeral and radiocapitellar articulations, augmented by the medial and lateral collateral ligament complexes. When a distal humeral tumor is resected, these stabilizing structures are entirely sacrificed. Consequently, the reconstructive prosthesis must inherently provide the stability that was lost. Modern tumor endoprostheses utilize a linked, semi-constrained hinge mechanism. While this design prevents dislocation, it inherently transfers massive torsional and bending moments directly to the bone-cement interface at the humeral stem.
Because the humerus acts as a long lever arm during activities of daily living, the stresses concentrated at the prosthetic hinge and the intramedullary stem are immense. This biomechanical reality explains why aseptic loosening and bushing wear are the primary modes of long-term mechanical failure in these reconstructions. To mitigate these forces, the surgeon must ensure optimal stem sizing, meticulous cementing technique, and precise restoration of the anatomic joint line to preserve the resting tension of the surrounding musculature. Failure to restore the correct humeral length alters the moment arms of the biceps and triceps, leading to profound weakness and accelerated prosthetic wear.
Exhaustive Indications and Contraindications
The decision to proceed with a distal humeral tumor resection and prosthetic reconstruction requires a multidisciplinary approach, integrating the expertise of orthopedic oncology, medical oncology, radiation oncology, and musculoskeletal radiology. The primary indication is the presence of a high-grade or aggressive low-grade primary bone sarcoma (e.g., osteosarcoma, chondrosarcoma, Ewing sarcoma) localized to the distal humerus, provided that wide oncologic margins can be achieved without sacrificing the critical neurovascular bundle. Additionally, soft tissue sarcomas that secondarily invade the distal humerus or the elbow joint capsule are prime candidates for this en bloc resection technique.
For metastatic disease, the indications are nuanced. Patients with solitary metastatic lesions to the distal humerus, particularly from favorable primary histologies (e.g., renal cell carcinoma, thyroid carcinoma), may be treated with curative intent via wide resection. More commonly, patients present with massive, destructive metastatic lesions causing intractable pain or pathologic fractures that are not amenable to standard open reduction and internal fixation (ORIF). In these palliative scenarios, endoprosthetic reconstruction is indicated to provide immediate structural stability, allowing the patient to utilize the extremity for activities of daily living and personal hygiene during their remaining lifespan.
Contraindications must be strictly respected to avoid catastrophic oncologic or surgical failures. The absolute contraindication to limb salvage is the direct tumor encasement of the major neurovascular bundle—specifically, the brachial artery and vein combined with two or more major nerves (median, ulnar, radial). In such cases, a functional limb cannot be preserved, and amputation (e.g., above-elbow amputation or forequarter amputation) is mandated. Relative contraindications include extensive tumor contamination from a poorly placed prior biopsy, active deep infection, or a massive fungating tumor with overlying skin necrosis that precludes adequate soft tissue coverage.
| Clinical Scenario | Indication Status | Rationale / Surgical Consideration |
|---|---|---|
| Primary High-Grade Sarcoma (Intraosseous) | Absolute Indication | Requires wide en bloc resection. Neoadjuvant chemotherapy dictates timing. Limb salvage is standard if NV bundle is free. |
| Destructive Metastatic Carcinoma with Pathologic Fracture | Strong Indication | ORIF will fail due to poor bone stock. Prosthesis provides immediate stability and pain relief for palliative care. |
| Soft Tissue Sarcoma invading Distal Humerus | Strong Indication | Requires en bloc resection of the soft tissue mass along with the involved underlying bone. |
| Tumor Encasement of Brachial Artery & 2+ Major Nerves | Absolute Contraindication | Limb salvage impossible without leaving gross tumor or creating a flail, ischemic limb. Amputation required. |
| Active Deep Periprosthetic or Osseous Infection | Relative Contraindication | Implantation of a massive megaprosthesis in an infected bed guarantees failure. Requires staged management or amputation. |
| Poorly Placed Biopsy Tract (Contaminating NV Bundle) | Relative Contraindication | May convert a limb-salvage candidate into an amputation candidate if the tract cannot be excised en bloc. |
Pre-Operative Planning, Templating, and Patient Positioning
In orthopedic oncology, inadequate preoperative planning is a recipe for surgical disaster. The blueprint for success begins with an exhaustive review of all staging and diagnostic imaging. Plain radiographs serve as the initial scouts, providing a preliminary estimate of tumor extent and assessing the response to neoadjuvant chemotherapy in osteosarcomas (evidenced by extensive calcification and healing of pathologic fractures). Computed Tomography (CT) is indispensable for evaluating cortical destruction and detecting subtle mineralization. For metastatic lesions, CT helps delineate whether the remaining bone stock is sufficient for curettage and cementation or if segmental resection is mandatory.
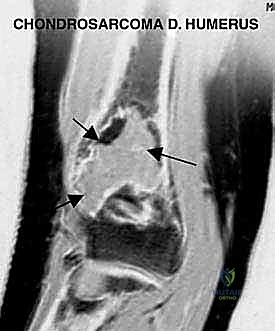
Magnetic Resonance Imaging (MRI) remains the gold standard for determining intra- and extraosseous tumor extent. T1-weighted coronal and sagittal sequences are critical for defining the proximal intramedullary extent of the tumor, which dictates the exact level of the humeral osteotomy. The transection level is typically planned 2 to 3 cm proximal to the most proximal MRI signal abnormality to ensure a negative marrow margin. Furthermore, axial T2-weighted and gadolinium-enhanced MRI sequences are meticulously scrutinized to assess the proximity of the extraosseous tumor component to the brachial vessels, median nerve, ulnar nerve, and radial nerve. Advanced modalities, such as Thallium scintigraphy or angiography, may be utilized to assess tumor viability and neovascularization, particularly following induction chemotherapy.
The biopsy is the first, and arguably most critical, surgical step. A poorly planned biopsy can hopelessly contaminate critical structures, converting a limb-salvage case into an amputation. For distal humeral tumors, the biopsy tract must be placed through the brachialis muscle, never through the biceps brachii, as the biceps must be preserved for postoperative elbow flexion. The tract must be perfectly in line with the proposed definitive surgical incision so it can be excised en bloc with the tumor specimen. An anterior approach, just lateral to the biceps tendon near the antecubital crease, is generally preferred.
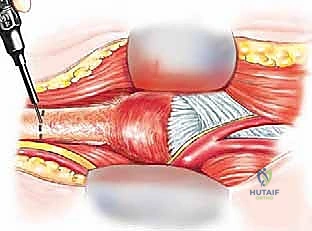
Patient positioning in the operating room is paramount for adequate exposure and ergonomic access. The patient is positioned supine with the affected arm abducted to 90 degrees and placed on a radiolucent arm board. A small bump is placed under the ipsilateral scapula to elevate the shoulder girdle, bringing the humerus into a more anterior and accessible plane. A sterile pneumatic tourniquet is applied high on the arm; it is typically left uninflated during the meticulous neurovascular dissection to aid in identifying vessels, and only inflated during the osteotomy and cementing phases to minimize blood loss. The entire upper extremity is prepped and draped free to allow full manipulation, flexion, and extension of the elbow during the reconstructive phase.
Step-by-Step Surgical Approach and Fixation Technique
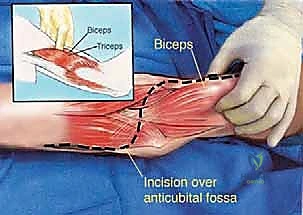
The operative execution is divided into three distinct phases: oncologic resection, skeletal reconstruction, and soft tissue coverage. The surgical approach is typically an extensile posterior or global approach, incorporating the previous biopsy tract. The skin incision is made, and full-thickness fasciocutaneous flaps are elevated. The immediate priority is the identification and protection of the major peripheral nerves. The ulnar nerve is identified proximally in the normal tissue of the posterior compartment, traced distally into the cubital tunnel, and meticulously mobilized. It is often transposed anteriorly to keep it out of the resection bed and away from the metallic hinge of the prosthesis.

Next, the radial nerve must be identified as it courses through the spiral groove. This is the most perilous portion of the dissection. The nerve is found proximal to the tumor, mobilized from the lateral intermuscular septum, and protected with a vessel loop. The median nerve and brachial vessels are similarly identified medially and retracted away from the tumor pseudocapsule. The dissection must remain strictly in the normal, healthy tissue planes surrounding the reactive zone of the tumor. The triceps mechanism is managed based on tumor extent; a triceps-sparing approach, a Bryan-Morrey reflection, or a TRAP (triceps-reflecting anconeus pedicle) approach may be utilized, provided the tumor does not directly invade the extensor mechanism. If the triceps is invaded, the involved portion must be resected en bloc.
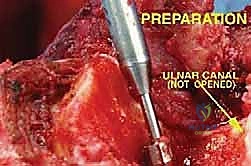
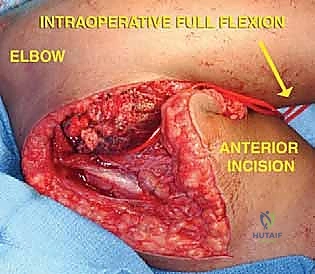
Once the neurovascular structures are safeguarded and the soft tissue envelope is mobilized, the humeral osteotomy is performed. Using the preoperative MRI measurements, the exact distance from the lateral epicondyle to the planned osteotomy site is marked on the humeral shaft. A sterile ruler and fluoroscopy are used to confirm the level. The osteotomy is performed with an oscillating saw. The medullary canal of the remaining proximal humerus is immediately sampled and sent for frozen section histological analysis to definitively confirm a negative marrow margin. The distal humerus, elbow joint capsule, and proximal radioulnar joint are then sharply dissected, and the entire specimen is delivered off the operative field en bloc.
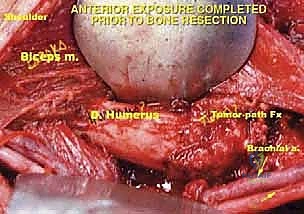
The reconstructive phase begins with the preparation of the proximal humeral canal and the proximal ulna. Sequential reaming is performed to accommodate the intramedullary stems of the modular endoprosthesis. Trial components are inserted to assess joint stability, soft tissue tension, and restoration of anatomical limb length. Once the trials are satisfactory, the canals are thoroughly irrigated with pulsatile lavage and dried. Third-generation cementing techniques are employed. A restrictive cement plug is placed, and polymethylmethacrylate (PMMA) bone cement is introduced in a retrograde fashion using a cement gun. The definitive humeral and ulnar stems are inserted, ensuring precise rotational alignment.
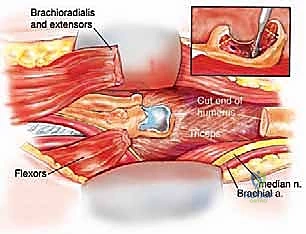
After the cement has fully polymerized, the ulnohumeral hinge mechanism is assembled and secured with the locking pin or bushing system specific to the implant design. The radial head may be resected, replaced, or left native depending on the specific prosthetic system utilized and the extent of the oncologic resection. The final, and crucial, step is soft tissue reconstruction. The triceps mechanism must be securely reattached to the proximal ulna or the prosthetic triceps-attachment flange using heavy, non-absorbable sutures through drill holes. If the soft tissue envelope is severely compromised, a pedicled latissimus dorsi flap or a free tissue transfer may be required to ensure robust coverage over the metallic hardware.

Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, distal humeral tumor resections with endoprosthetic reconstructions are fraught with a high complication profile. The patient population is often systemically compromised by malnutrition, neoadjuvant chemotherapy, and radiation therapy, dramatically altering the host's healing capacity. Complications can be broadly categorized into mechanical failures, biological failures (infection), and oncologic failures (local recurrence). Henderson et al. famously classified failure modes of tumor endoprostheses into five types: soft tissue failure (Type 1), aseptic loosening (Type 2), structural failure (Type 3), infection (Type 4), and tumor progression (Type 5).
Deep periprosthetic joint infection (PJI) is arguably the most devastating complication, with incidence rates ranging from 5% to 15% in the oncologic elbow population. The thin soft tissue envelope, extensive surgical dissection, and immunocompromised state create a perfect storm for bacterial colonization. Management of PJI in a megaprosthesis typically requires a two-stage revision protocol: removal of the prosthesis, insertion of an antibiotic-impregnated PMMA spacer, administration of targeted intravenous antibiotics for 6-8 weeks, and subsequent reimplantation. In cases of recalcitrant infection with massive soft tissue loss, amputation may be the only definitive salvage option.
Aseptic loosening and mechanical bushing wear are the most common long-term complications, inherent to the highly constrained biomechanics of the hinged elbow prosthesis. The long lever arm of the humerus transmits massive torque to the bone-cement interface. Over time, this leads to osteolysis, stem subsidence, and eventual loosening, presenting clinically as progressive pain and mechanical instability. Revision arthroplasty with longer stems, allograft-prosthetic composites (APCs), or total humeral replacement are the standard salvage strategies. Additionally, postoperative nerve palsies—particularly of the ulnar nerve—are common due to the extensive neurolysis and traction required during resection. Most are neuropraxias that resolve with time, but permanent deficits can occur.
| Complication Type | Estimated Incidence | Etiology / Risk Factors | Salvage Management Strategy |
|---|---|---|---|
| Deep Infection (PJI) | 5% - 15% | Immunosuppression, thin soft tissue envelope, prolonged OR time, radiation. | Two-stage revision with antibiotic spacer. Flap coverage. Suppressive antibiotics. Amputation if recalcitrant. |
| Aseptic Loosening | 10% - 25% (at 10 yrs) | High stress at bone-cement interface, constrained hinge mechanics, osteolysis. | Single-stage revision arthroplasty. Use of longer stems or Allograft-Prosthetic Composites (APC). |
| Ulnar Nerve Palsy | 10% - 20% | Traction injury during mobilization, thermal injury from cement, compression. | Observation (often neuropraxia). Gabapentinoids. Tendon transfers if permanent motor deficit persists. |
| Structural Failure (Bushing Wear / Fracture) | 5% - 10% | Material fatigue, high demand activity, excessive patient weight. | Modular component exchange (bushing replacement) or revision of fractured stems. |
| Local Tumor Recurrence | 5% - 10% | Inadequate surgical margins, highly aggressive tumor biology, skip metastases. | Repeat wide excision if feasible. Often requires amputation (e.g., forequarter) for definitive control. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following a distal humeral tumor resection and prosthetic reconstruction is a delicate balancing act. The therapist and surgeon must navigate the competing demands of protecting the fragile soft tissue reconstruction—specifically the reattached triceps mechanism—while simultaneously initiating early motion to prevent devastating elbow stiffness. The rehabilitation protocol is strictly phased and must be customized based on the intraoperative stability of the prosthesis and the integrity of the extensor mechanism repair.
Phase I (Weeks 0-4) is focused on wound healing, edema control, and protection of the reconstruction. The arm is typically immobilized in a well-padded posterior splint or a hinged elbow orthosis locked at 60 to 90 degrees of flexion to minimize tension on the triceps repair and the anterior skin incision. Active hand and wrist range of motion (ROM) is initiated immediately to prevent distal edema and stiffness. Shoulder pendulum exercises are permitted, but active elbow extension is strictly prohibited to protect the triceps reattachment. Passive elbow flexion may be gently introduced under the direct supervision of the physical therapist, provided the wound is healing optimally.
Phase II (Weeks 4-8) marks the transition to active-assisted range of motion (AAROM). The hinged brace is unlocked to allow a progressive arc of motion. Active elbow flexion and gravity-assisted elbow extension are initiated. The patient is instructed to avoid forceful active extension or pushing movements (e.g., pushing up from a chair). Isometric strengthening of the biceps and brachialis begins. Aquatic therapy can be highly beneficial during this phase, utilizing the buoyancy of water to assist with motion while minimizing joint loading.
Phase III (Weeks 8-12 and beyond) focuses on progressive strengthening and functional restoration. Active resistive exercises for the triceps are cautiously introduced, progressing from light bands to light weights. The goal is to achieve a functional arc of motion, typically defined as 30 to 130 degrees of flexion, which is sufficient for most activities of daily living. Patients must be counseled that the reconstructed elbow will never possess the strength or durability of a native joint. A permanent lifetime lifting restriction of 10 to 15 pounds is universally implemented to mitigate the risk of premature bushing wear and aseptic loosening of the intramedullary stems.
Summary of Landmark Literature and Clinical Guidelines
The evolution of distal humeral tumor resection and reconstruction is deeply rooted in landmark orthopedic oncology literature. The conceptual framework for staging and resecting musculoskeletal neoplasms was pioneered by William Enneking in the 1980s. Enneking's principles of achieving wide margins—resecting the tumor en bloc with a cuff of normal, reactive tissue—remain the absolute gold standard for primary bone sarcomas. His work demonstrated that marginal or intralesional resections of high-grade sarcomas inevitably lead to local recurrence, which drastically reduces overall survival.
The biomechanical understanding of the elbow joint and its implications for prosthetic design relies heavily on the seminal work of Bernard Morrey. Morrey's extensive research on elbow kinematics highlighted the highly constrained nature of the ulnohumeral joint. This constraint is the primary reason why early, rigid, fully constrained elbow prostheses failed catastrophically due to aseptic loosening. The transition to the modern "sloppy hinge" or semi-constrained modular endoprostheses, which allow a few degrees of varus/valgus and rotational toggle, significantly reduced the stresses transmitted to the bone-cement interface, thereby improving long-term survivorship.
Finally, the classification and management of complications in oncologic endoprostheses are universally guided by the Henderson classification system. Henderson et al. provided a standardized lexicon for reporting failure modes, which has allowed the global orthopedic oncology community to pool data and improve implant designs. Their research underscores that while mechanical failures (aseptic loosening, structural fracture) increase linearly with time, biological failures (infection) and oncologic failures (recurrence) typically occur within the first two to three years postoperatively. Familiarity with this literature is not merely academic; it is essential for anticipating complications, counseling patients, and executing a successful limb-salvage strategy.
