Upper Extremity Amputation: A Masterclass in Oncologic Reconstruction
Key Takeaway
Fellows, welcome to this masterclass on upper extremity amputations. We'll meticulously cover above-elbow and below-elbow techniques for advanced sarcomas, focusing on achieving wide oncologic margins while preserving maximal function. This session details preoperative planning, precise intraoperative dissection, crucial neurovascular management, robust myodesis, and comprehensive postoperative rehabilitation protocols to optimize patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
Welcome, esteemed colleagues and fellows, to the operating theater and to this definitive academic discourse. Today, we delve into a critical, albeit less common, aspect of musculoskeletal oncology: above-elbow and below-elbow amputations. While the modern paradigm of orthopedic oncology heavily champions limb-sparing surgery, there remain distinct, unavoidable clinical scenarios where the sheer extent of disease—particularly in the context of aggressive, high-grade sarcomas—necessitates amputation. This intervention is required to achieve definitive local tumor control and ensure the patient's long-term oncologic survival. It is paramount to understand that an amputation in this setting is not a failure of limb salvage; rather, it is a highly calculated, life-saving decision designed to prioritize oncologic safety and optimize functional outcomes when complex reconstruction is either anatomically impossible or would result in a painful, non-functional, and burdensome extremity.
Historically, the mere presence of local recurrence or a large primary tumor was considered a primary indication for radical amputation. However, the landscape of musculoskeletal oncology has undergone a seismic shift. With the advent of sophisticated neoadjuvant chemotherapy protocols, targeted radiation therapy, isolated limb perfusion, and highly advanced microvascular surgical techniques, the presence of a massive or recurrent sarcoma does not automatically mandate the loss of the limb. The ultimate deciding factor, rigorously debated in every multidisciplinary tumor board, is whether the surgical team can resect the tumor with wide, histologically clear margins without critically compromising the remaining neurovascular and biomechanical function of the extremity.

The epidemiology of upper extremity sarcomas dictates that these tumors are significantly less common than their lower extremity counterparts, accounting for roughly 15% to 20% of all extremity soft tissue and bone sarcomas. Despite their relative rarity, the compact, unforgiving anatomy of the upper limb—where critical nerves and vessels are densely packed within narrow compartments—makes achieving negative margins exceedingly difficult. When tumors breach compartmental boundaries, encase the brachial plexus or its terminal branches, or aggressively destroy the humeral or forearm diaphysis, the oncologic principles of margin status must supersede the desire for limb preservation.
Furthermore, the psychological impact of upper extremity amputation cannot be overstated. The upper limb is intimately tied to a patient’s sense of identity, independence, and interaction with the world. As oncologic surgeons, our responsibility extends far beyond the physical resection of the tumor. We must meticulously manage patient expectations, coordinate with psychiatric and occupational therapy teams pre-operatively, and integrate advanced prosthetic concepts—such as Targeted Muscle Reinnervation (TMR) and osseointegration—into our initial surgical planning. This comprehensive approach ensures that we are not merely removing a diseased limb, but actively reconstructing the patient's future functional capacity.
Detailed Surgical Anatomy and Biomechanics
Above-Elbow Amputation Anatomy
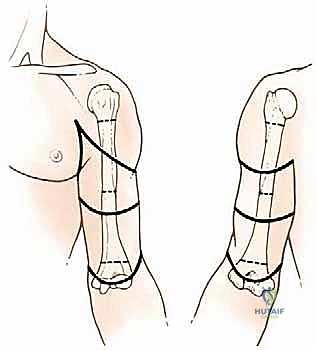
Above-elbow amputations are anatomically classified based on their specific level of bone resection: metaphyseal (high), diaphyseal, and supracondylar. High metaphyseal amputations occur proximal to the deltoid tuberosity, often involving the surgical neck or the proximal humerus itself. Diaphyseal amputations transect the mid-shaft of the humerus, while supracondylar amputations are performed just proximal to the olecranon fossa. The muscular anatomy of the proximal humerus is complex, serving as the insertion site for the deltoid (deltoid tuberosity), pectoralis major (lateral lip of the bicipital groove), latissimus dorsi (floor of the bicipital groove), and teres major (medial lip). Preserving the insertions of the deltoid and pectoralis major is a critical biomechanical goal, as these muscles are essential for stabilizing the shoulder girdle and powering a shoulder-driven prosthesis.
The neurovascular bundle of the arm is the next critical anatomical consideration. Coursing medially, it comprises the brachial artery, brachial veins, and the median, ulnar, and radial nerves. The brachial artery, a direct continuation of the axillary artery, runs in intimate association with the median nerve, providing the primary arterial inflow to the forearm and hand. The median nerve lies anterior to the brachial artery in the distal arm, while the ulnar nerve courses medially and posterior to the medial epicondyle. The radial nerve presents a unique surgical hazard; it winds around the posterior aspect of the humerus within the spiral groove before piercing the lateral intermuscular septum to enter the anterior compartment. Precise identification, proximal traction, and sharp transection of these nerves are vital to prevent symptomatic neuroma formation.
Biomechanically, the length of the preserved humeral stump directly correlates with the functional success of the prosthesis. A longer lever arm provides superior mechanical advantage, allowing the patient to control the prosthetic elbow and terminal device with less energy expenditure. Amputations that leave less than 30% of the humeral length drastically reduce the surface area available for prosthetic socket suspension, often necessitating a cumbersome shoulder disarticulation-style socket. Therefore, while oncologic margins dictate the ultimate level of resection, every safe millimeter of bone must be preserved, and the soft tissue envelope must be meticulously reconstructed to provide a robust, well-padded residuum.

Below-Elbow Amputation Anatomy
The overarching surgical objective for below-elbow amputations is the preservation of maximal length of both the radius and ulna, which is paramount for optimizing subsequent prosthetic function. The forearm is a highly specialized anatomical unit designed for pronation and supination, motions that are critical for spatial positioning of the hand. The muscular anatomy is divided into the volar flexor-pronator group (originating largely from the medial epicondyle) and the dorsal extensor-supinator group (originating from the lateral epicondyle). During a below-elbow amputation, these muscle groups must be carefully balanced and secured over the distal bone ends via myodesis to prevent a mobile, redundant soft tissue envelope that would chafe within a prosthetic socket.
The neurovascular anatomy of the forearm bifurcates significantly distal to the antecubital fossa. The brachial artery divides into the radial and ulnar arteries. The radial artery courses along the lateral aspect of the forearm deep to the brachioradialis, while the ulnar artery travels medially, deep to the flexor carpi ulnaris. The median nerve courses centrally, deep to the flexor digitorum superficialis, and the ulnar nerve runs alongside the ulnar artery. The radial nerve bifurcates into a superficial sensory branch and a deep motor branch (the posterior interosseous nerve), which dives through the supinator muscle. Each of these structures must be systematically identified and managed during the amputation.
A critical clinical pearl regarding below-elbow biomechanics is the minimum required stump length. A minimum of 2.5 to 3.0 centimeters of bony stump, measured from the distal aspect of the radial tuberosity, is absolutely required to preserve any useful function, primarily for the transmission of pronation and supination forces to a prosthesis. If tumor extent dictates a very short stump, releasing the distal biceps tendon from the radial tuberosity can sometimes provide just enough clearance to fit a specialized socket, relying on the brachialis muscle to provide elbow flexion. If this minimum length cannot be achieved, the functional outcome is often equivalent to, or worse than, an elbow disarticulation.
Exhaustive Indications and Contraindications
The decision to proceed with a major upper extremity amputation is an irreversible, life-altering intervention that is never taken lightly. It is the culmination of exhaustive multidisciplinary tumor board discussions involving orthopedic oncologists, medical oncologists, radiation oncologists, radiologists, and pathologists. The primary indication is always the inability to achieve wide, clear surgical margins while preserving a functional limb. This often occurs when a high-grade sarcoma exhibits extensive soft tissue and bone extension, infiltrating widely across multiple compartments. For instance, a massive pleomorphic sarcoma of the anterior arm that engulfs the biceps, brachialis, and intimately wraps around the humeral diaphysis often leaves no reconstructive options that would yield a functional extremity.
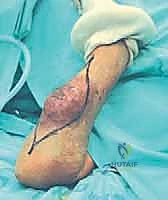
Major neurovascular involvement is another critical determinant. Tumors that extensively encase or frankly invade the main neurovascular bundle—particularly the brachial artery—frequently render limb salvage impossible or highly morbid. While venous reconstruction or reliance on collateral flow via the cephalic vein is sometimes feasible, extensive arterial resection and grafting in the setting of post-radiation tissue beds carry an unacceptably high risk of thrombosis, infection, and subsequent catastrophic limb loss. Similarly, the functional impact of major nerve sacrifice is paramount. While the loss of a single major nerve (e.g., the ulnar nerve) can be managed with tendon transfers, the simultaneous oncologic sacrifice of the median, ulnar, and radial nerves results in a flail, insensate, and entirely functionless extremity. In such scenarios, amputation followed by modern prosthetic rehabilitation offers a vastly superior quality of life.
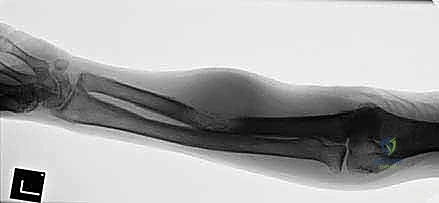
Recurrent, diffuse disease, particularly in previously irradiated fields, presents a uniquely challenging indication. When a sarcoma recurs locally after prior wide excision and maximum-dose radiotherapy, the surrounding tissue is often fibrotic, poorly vascularized, and highly susceptible to wound breakdown. Attempting a secondary limb salvage procedure in this hostile environment frequently necessitates massive soft tissue and bone resection, leading to an extensive defect that cannot be reliably covered even with free tissue transfer. Furthermore, extensive carcinomatosis, such as neglected or highly aggressive squamous cell carcinoma of the forearm that has invaded deep fascial planes and bone, mandates amputation to prevent fungation, intractable pain, and systemic sepsis.
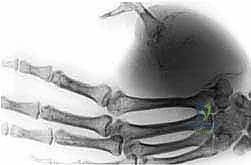
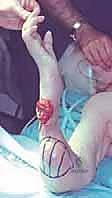
| Category | Specific Clinical Scenarios | Rationale for Decision |
|---|---|---|
| Absolute Indications | 1. Inability to achieve negative margins with limb salvage. 2. Encasement of the entire brachial neurovascular bundle. 3. Sacrifice of 3 major nerves (Median, Ulnar, Radial). 4. Intractable, fungating tumor with life-threatening hemorrhage or sepsis. |
Oncologic safety and overall patient survival mandate complete tumor extirpation. A functionless, insensate limb is a liability, not an asset. |
| Relative Indications | 1. Massive local recurrence in a previously heavily irradiated bed. 2. Severe baseline functional impairment of the affected limb. 3. Patient preference for faster return to activity over prolonged, multi-stage reconstruction. |
Limb salvage may technically be possible but carries an unacceptably high complication rate (infection, flap failure) or will result in a limb inferior to a prosthesis. |
| Absolute Contraindications | 1. Refusal of consent by a competent patient. 2. Terminal systemic disease where the patient is too unstable to survive anesthesia (unless for strictly palliative, life-saving hemorrhage control). |
Ethical and medical principles dictate that surgery cannot proceed without consent or if the immediate perioperative mortality risk outweighs the oncologic benefit. |
| Relative Contraindications | 1. Presence of distant metastases (unless amputation is required for palliation of severe pain or fungation). 2. Severe psychiatric illness precluding post-operative rehabilitation. |
In metastatic disease, systemic therapy takes precedence. Amputation is reserved for local symptom control. |
Pre-Operative Planning, Templating, and Patient Positioning
Advanced Imaging and Biopsy Principles
Before the scalpel ever touches the skin, exhaustive and meticulous staging is absolutely non-negotiable. This process is not merely about identifying the histological subtype of the tumor; it is about creating a high-fidelity, three-dimensional map of its anatomical extent to determine the precise level of amputation and the required soft tissue resection margins. Plain radiography provides the initial assessment of bone destruction, periosteal reaction, and matrix mineralization. Computed Tomography (CT) is crucial for evaluating subtle cortical breaches, intraosseous medullary extension, and the presence of calcified tumor matrix within the soft tissue components.
However, Magnetic Resonance Imaging (MRI) remains the gold standard for soft tissue evaluation and ultimate surgical planning. We strictly utilize whole-compartment T1-weighted images for anatomical detail, T2-weighted images with fat suppression (STIR or fat-sat) to highlight peritumoral edema and skip lesions, and post-contrast T1 images to delineate viable, enhancing tumor tissue from reactive, non-enhancing edema. The proximal extent of the tumor on MRI dictates the bone cut. A critical oncologic principle is that the more proximal of the two levels of involvement (bone versus soft tissue) dictates the final level of amputation. We must achieve a wide margin, typically 2 to 3 centimeters of healthy, uninvolved tissue proximal to the most proximal microscopic tumor extent.
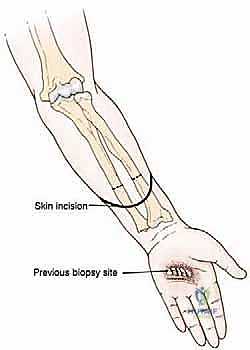
The biopsy is the final, critical step in pre-operative planning. A core needle biopsy, performed strictly by the operating musculoskeletal oncologist or a specialized interventional radiologist under direct guidance, is mandatory to establish a definitive diagnosis and tumor grade. The placement of the biopsy tract is of paramount importance; it must be meticulously planned so that the entire tract, including the skin puncture site, can be completely excised en bloc with the amputation specimen. An poorly placed biopsy tract that contaminates proximal, otherwise healthy tissue compartments can catastrophically force a higher level of amputation—converting a functional below-elbow amputation into an above-elbow amputation.
Anesthesia, Positioning, and Tourniquet Considerations
The pre-operative setup requires seamless coordination between the surgical and anesthesia teams. A regional block, such as an interscalene or supraclavicular brachial plexus block, combined with general anesthesia is the preferred modality. The regional block provides profound intraoperative muscle relaxation and excellent post-operative pain control, which significantly minimizes the need for high-dose systemic narcotics and reduces the incidence of phantom limb pain by preemptively blocking nociceptive pathways. The patient is placed strictly supine on the operating table, with the ipsilateral shoulder slightly elevated using a rolled blanket or gel pad to allow unhindered 360-degree access to the axilla and the entire circumference of the arm.
The affected arm is prepped circumferentially from the fingertips to the base of the neck, including the axilla and the ipsilateral chest wall, and draped free. This allows for full manipulation and rotation of the extremity during the procedure. A sterile pneumatic tourniquet is applied as proximally as possible on the arm. However, a critical oncologic caveat must be observed: exsanguination of the limb using an Esmarch bandage must NEVER be performed by tightly wrapping over the tumor mass, as this can force tumor emboli into the systemic circulation. Instead, the limb is simply elevated for 3 to 5 minutes to allow venous drainage via gravity before tourniquet inflation.
Finally, meticulous pre-operative templating is performed in the operating room. Using the pre-operative MRI as a direct reference, the planned incision lines and the proposed level of the bone osteotomy are clearly marked on the skin with a sterile surgical marker. This visual confirmation ensures that the surgical team remains precisely oriented to the required oncologic margins once the skin is incised and the normal anatomical landmarks become distorted by the dissection. Fluoroscopy (C-arm) is brought into the field to correlate the skin markings with the underlying bony anatomy, ensuring the osteotomy will be performed at the exact planned millimeter.
Step-by-Step Surgical Approach and Fixation Technique
Flap Design and Initial Incisions
With the patient prepped, draped, and the tourniquet inflated, the surgical execution begins. The standard approach for most upper extremity amputations involves the creation of anterior and posterior "fishmouth" flaps. These flaps are designed to be equal in length, allowing the final suture line to rest terminally over the bone end. However, in the context of oncologic resections, flap design is frequently atypical and entirely dictated by the location and proximity of the tumor. If the tumor is located anteriorly, a much longer posterior flap must be designed to ensure adequate skin coverage while maintaining a wide margin around the anterior tumor mass.
The skin incisions are made boldly through the dermis and subcutaneous fat down to the deep investing fascia. It is crucial to maintain full-thickness fasciocutaneous flaps whenever possible. By leaving the deep fascia attached to the subcutaneous tissue, the delicate subdermal vascular plexus is preserved, significantly reducing the risk of post-operative marginal necrosis and wound breakdown. The flaps are then elevated proximally to the level of the planned bone transection. Throughout this dissection, meticulous hemostasis is maintained using electrocautery, and the tissue planes are handled with atraumatic technique to preserve the viability of the soft tissue envelope that will ultimately cushion the prosthetic socket.
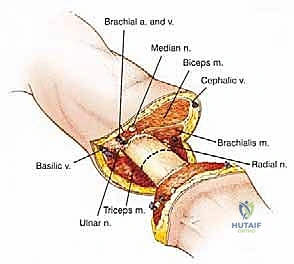
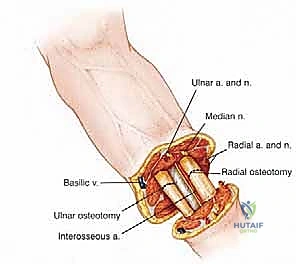
Neurovascular Handling and Targeted Muscle Reinnervation
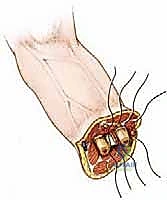
As the dissection deepens into the muscular compartments, the major neurovascular bundles are systematically identified, isolated, and managed. The vascular structures must be handled with absolute precision. The brachial artery (or radial/ulnar arteries in a below-elbow amputation) and their accompanying veins are carefully dissected free from the surrounding connective tissue. Crucially, the artery and the vein must be doubly ligated and divided separately. Ligation of the artery and vein together in a single mass tie creates a high risk for the development of an arteriovenous fistula, a complication that can cause severe localized pain and systemic hemodynamic issues. We utilize heavy non-absorbable sutures (e.g., 0-Silk) for the proximal ties and a transfixing suture ligature for absolute security.
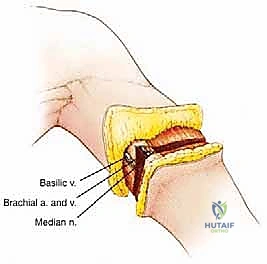
The management of the peripheral nerves is arguably the most critical step in preventing chronic post-amputation pain. The median, ulnar, and radial nerves are identified and isolated. Historically, nerves were simply placed under strong distal traction, transected sharply with a fresh scalpel blade, and allowed to retract deep into the proximal muscle bellies to avoid terminal neuroma formation near the skin closure. While this remains a fundamental principle, modern oncologic reconstruction now heavily incorporates Targeted Muscle Reinnervation (TMR).
In TMR, the transected ends of these major mixed motor-sensory nerves are meticulously coapted (using microsurgical techniques and 8-0 or 9-0 nylon sutures) to the redundant motor nerve branches of nearby expendable muscles that have been denervated by the amputation. For example, the median nerve can be transferred to the motor branch of the short head of the biceps. This provides the regenerating axons of the amputated nerve with a new target muscle to reinnervate, effectively preventing the disorganized axonal sprouting that leads to painful neuromas. Furthermore, the reinnervated muscle acts as a biological amplifier for the nerve signals, which can later be detected by surface electromyography (EMG) electrodes to provide highly intuitive, simultaneous control of advanced bionic prostheses.
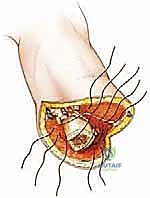
Osteotomy and Myodesis
Once the neurovascular structures are secured and the tumor-bearing soft tissues are mobilized distally, attention is turned to the bone. The periosteum is sharply incised circumferentially at the planned level of the osteotomy and elevated distally. It is critical not to strip the periosteum proximally, as this devascularizes the terminal bone end and promotes the formation of painful terminal ring sequestra. The osteotomy is performed using a high-speed oscillating saw. Continuous cold saline irrigation is directed onto the saw blade throughout the cut to dissipate heat and prevent thermal necrosis of the osteocytes, which can lead to delayed healing or bone resorption.
After the limb is removed from the field, the sharp cortical edges of the remaining bone stump are meticulously smoothed and contoured using a rasp or a rongeur. The anterior cortex is typically beveled to prevent a sharp bony prominence from eroding through the anterior skin flap during prosthetic wear. The medullary canal is inspected; if there is any suspicion of proximal intramedullary tumor extension, a medullary reaming is sent for immediate frozen section analysis.
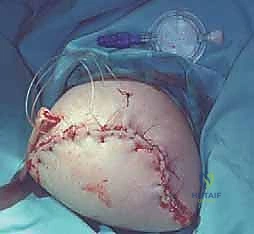
The final reconstructive step before closure is the myodesis. Unlike a simple myoplasty (where antagonist muscles are sutured to each other over the bone end), a myodesis involves directly anchoring the transected muscle bellies to the bone itself. Drill holes are placed through the distal cortex of the bone stump. Heavy, absorbable sutures (e.g., #1 or #2 Vicryl) are woven through the epimysium and deep fascia of the muscle bellies (e.g., the triceps and biceps in an above-elbow amputation) and passed through the drill holes. The muscles are secured to the bone under physiological tension. This technique provides a robust, stable soft tissue envelope, prevents muscle retraction, and preserves the length-tension relationship of the muscles, which is vital for maximizing the strength of the residual limb for prosthetic control. The tourniquet is deflated, meticulous hemostasis is achieved, a closed suction drain is placed deep to the fascia, and the skin is closed in layers without tension.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, upper extremity amputations carry a distinct profile of post-operative complications. The most immediate concerns are hematoma formation and surgical site infection, particularly in patients who have received neoadjuvant radiation or chemotherapy, which severely compromises local tissue healing and systemic immune response. Wound dehiscence can expose the underlying bone or neurovascular structures, necessitating urgent surgical debridement and potentially complex flap coverage.
Neuroma formation and Phantom Limb Pain (PLP) are the most notoriously difficult long-term complications to manage. While all transected nerves form neuromas as part of the natural healing process, symptomatic terminal neuromas occur when the nerve end becomes tethered in scar tissue near the skin surface or bone end, causing exquisite pain with prosthetic socket pressure. PLP, a complex neuropathic phenomenon where the patient experiences painful sensations in the missing limb, affects up to 80% of amputees to varying degrees. Proactive management with TMR during the index procedure has significantly reduced the incidence and severity of both symptomatic neuromas and PLP.
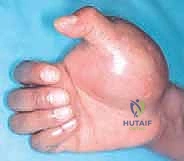
Bone overgrowth is a unique and challenging complication primarily seen in the pediatric oncologic population. Because the appositional growth of bone outpaces the growth of the surrounding soft tissue envelope, the bone end can literally spear through the skin. This phenomenon is most common in the humerus and fibula. Management often requires surgical revision
Clinical & Radiographic Imaging Archive