Sarcoma Resection Masterclass: Biology, Staging, & Limb-Sparing Principles
Key Takeaway
Welcome, fellows, to an immersive masterclass on musculoskeletal sarcomas. We'll delve into the fundamental biology, epidemiology, and staging systems that dictate our surgical approach. This session emphasizes meticulous preoperative planning, precise intraoperative execution for limb-sparing resections, and comprehensive postoperative care. Understanding tumor behavior, anatomic barriers, and potential pitfalls is paramount to achieving optimal oncologic and functional outcomes for our patients.
Comprehensive Introduction and Patho-Epidemiology
The surgical management of musculoskeletal malignancies demands an uncompromising mastery of oncologic principles, spatial anatomy, and advanced reconstructive biomechanics. As orthopedic oncologists, we operate at the intersection of cellular biology and macro-structural engineering. Understanding the basic biology and pathology of bone and soft tissue tumors is not merely an academic exercise; it is the absolute bedrock upon which every successful limb-sparing surgery is built. Without this foundational knowledge, the surgeon is operating blind, risking catastrophic local recurrence and compromising patient survival. Our approach is dictated by the intrinsic behavior of these tumors, which originate primarily from elements of the mesodermal embryonic layer. This common mesenchymal origin imparts unique patterns of biologic behavior, driving our staging systems, dictating our surgical margins, and forming the rationale for modern multimodality treatment strategies.
Soft tissue and bone sarcomas are rare, profoundly heterogeneous neoplasms, representing less than 1% of all adult malignancies and approximately 15% of pediatric malignancies. Despite their rarity, their clinical impact is devastating if mismanaged. Historically, mortality rates hovered around 30% for soft tissue sarcomas and a grim 45% for bone sarcomas. However, survival rates have dramatically improved over the past three decades. This paradigm shift is largely attributable to the evolution of limb-sparing surgery performed in concert with highly targeted neoadjuvant and adjuvant chemotherapy, as well as advanced radiation protocols. Today, we achieve oncologic cure in the majority of our patients, with limb-salvage resection performed in lieu of amputation in more than 90% of cases at specialized tertiary referral centers.
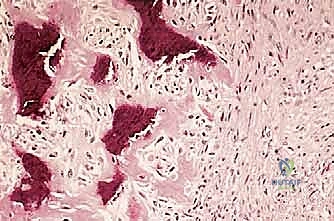
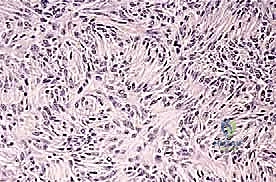
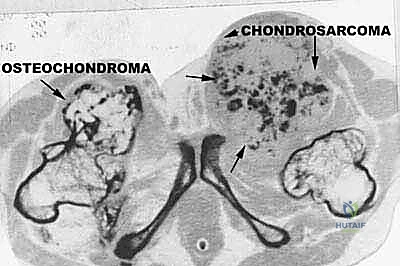
We classify soft tissue sarcomas according to the adult tissue they morphologically resemble, whereas bone sarcomas are typically classified by the type of extracellular matrix they produce—osteoid-producing for osteosarcomas, and chondroid-producing for chondrosarcomas. The most common soft tissue sarcomas encountered in adult practice include undifferentiated pleomorphic sarcoma (UPS, formerly malignant fibrous histiocytoma or MFH), liposarcoma, and leiomyosarcoma. In the pediatric and adolescent populations, rhabdomyosarcoma predominates in soft tissue, while osteosarcoma and Ewing sarcoma are the most prevalent primary bone tumors. Chondrosarcoma remains a distinct entity, typically presenting in older adults and characterized by its notorious resistance to standard cytotoxic chemotherapy and radiation, making surgical extirpation the sole curative modality.
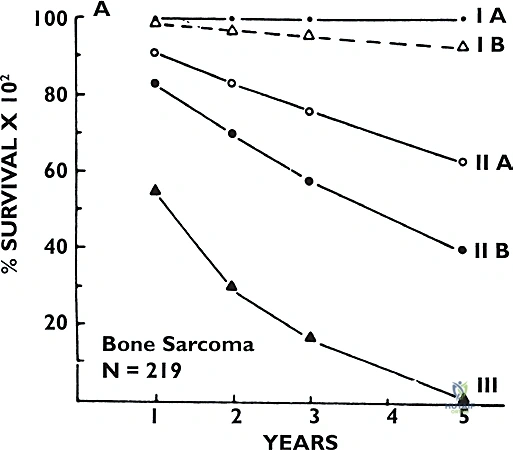
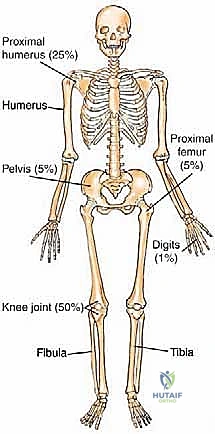
Prognostication in sarcoma management relies on a complex matrix of clinical and histopathologic factors. For bone sarcomas, the most critical prognostic indicators include histologic grade, maximum tumor dimension, extension beyond the bone cortex into the soft tissue envelope, the presence of regional or distant metastatic disease, and crucially, the histologic response to neoadjuvant chemotherapy (quantified as the necrosis rate, with >90% necrosis indicating a favorable response). Conversely, for soft tissue sarcomas, the primary prognostic factors encompass histologic grade, tumor size (with 5 cm serving as a critical threshold), depth relative to the investing fascia, patient age, surgical margin status, anatomic location (proximal lesions generally portend a worse prognosis than distal ones), specific histologic subtype, and the presence of metastatic disease at presentation.
The Pseudocapsule and Tumor Microenvironment
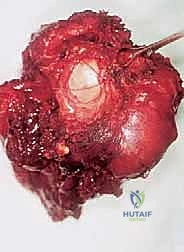
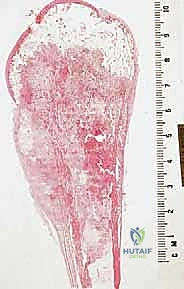
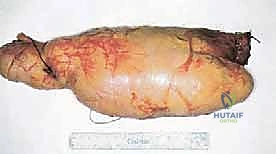
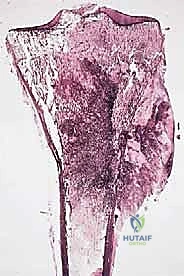
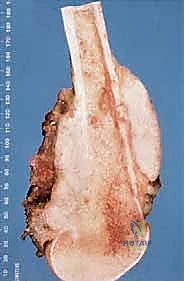
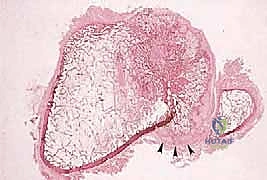
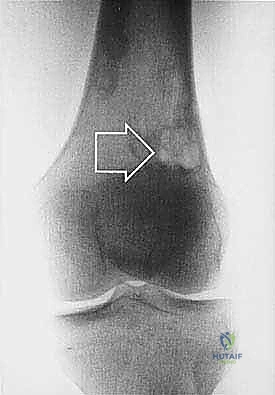
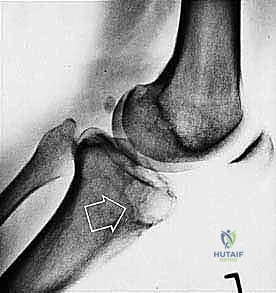
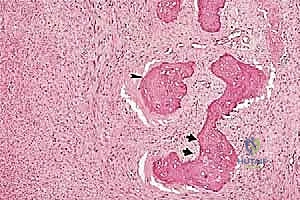
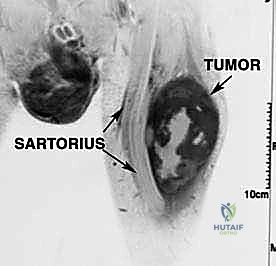
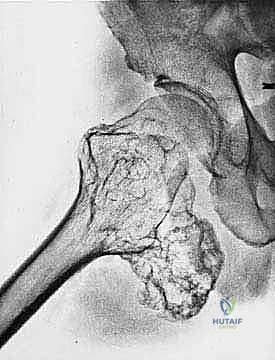
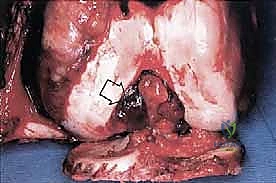
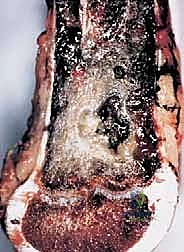
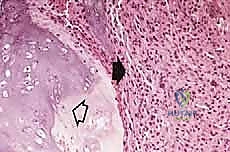
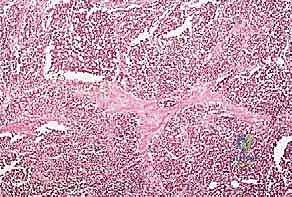
Sarcomas typically present as a solid, expanding mass that grows centrifugally. Crucially, the periphery of the lesion—the advancing front—is often the least mature and the most biologically aggressive. This centrifugal growth pattern has profound implications for our surgical margin planning. Unlike benign neoplasms (such as lipomas or fibromas), which are surrounded by a true capsule of compressed normal cells that allows for simple enucleation, sarcomas are typically enclosed by a deceptive anatomic entity known as the reactive zone, or pseudocapsule.
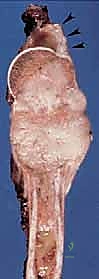
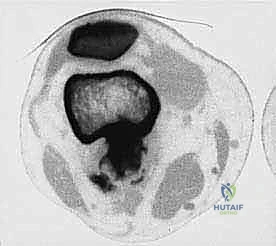
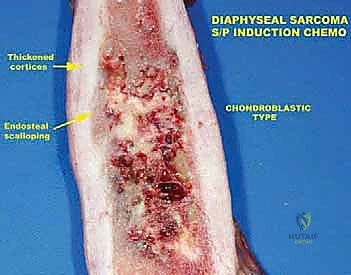
This pseudocapsule is an illusion of containment. It consists of compressed, anaplastic tumor cells intermixed with a fibrovascular zone of reactive normal tissue, heavily infiltrated by a variable inflammatory component. It interacts intimately with the surrounding normal host tissues, driven by tumor-derived angiogenic factors and matrix metalloproteinases. Treating this pseudocapsule as a true barrier and attempting marginal excision or enucleation is a catastrophic surgical error that guarantees leaving microscopic, high-grade disease behind in the tumor bed. The thickness and integrity of this reactive zone vary significantly with the histogenic type and the intrinsic grade of the sarcoma.
High-grade sarcomas often possess a poorly defined, highly permeable reactive zone that is aggressively invaded by the primary tumor mass. This micro-invasion is the primary mechanism responsible for local recurrences despite apparently "negative" margins reported on gross pathologic inspection. Even low-grade sarcomas, which may appear more circumscribed, regularly interdigitate into the reactive zone, sending microscopic tendrils of tumor cells into the surrounding fibrovascular tissue. Therefore, the fundamental principle of oncologic resection dictates that the surgical plane must be established well beyond this reactive zone, within entirely normal, un-reactive host tissue.
Satellite and Skip Lesions: Locoregional Dissemination
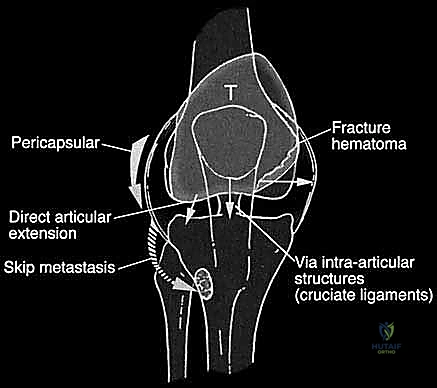
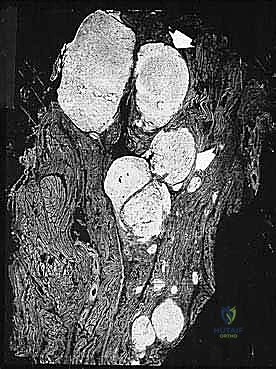
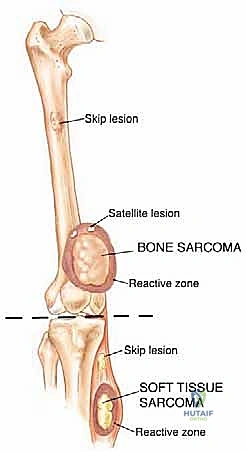
Within the reactive zone surrounding the primary tumor mass, we frequently encounter isolated tumor foci known as satellite lesions. These are essentially locoregional micrometastases that have separated from the main tumor body but remain confined within the immediate inflammatory pseudocapsule. They have not disseminated through the systemic circulation but represent localized, aggressive micro-invasion. The presence of satellite lesions underscores the absolute necessity of achieving wide surgical margins, as a marginal resection that merely skirts the pseudocapsule will inevitably transect and leave these satellite nodules in the patient.
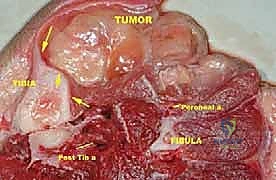
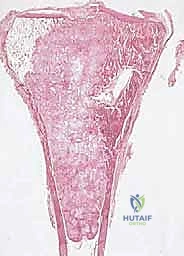
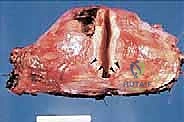
More ominously, high-grade sarcomas (and occasionally aggressive low-grade variants) possess the biological capacity to break entirely through the pseudocapsule and form skip metastases. Skip metastases are defined as distinct, discontinuous tumor nodules located within the same anatomic compartment as the primary tumor, but separated from the reactive zone by a region of completely normal intervening tissue. These are believed to arise from embolic spread along the microvasculature or lymphatic channels within the compartment's longitudinal axis.
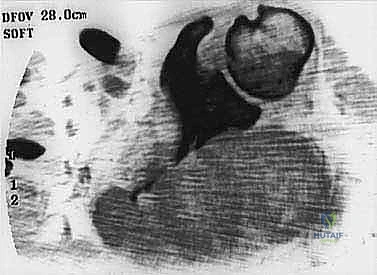
Skip metastases are a silent threat. They are typically not documented on standard preoperative imaging in more than 5% of patients, yet they represent a massive risk factor for local recurrence and systemic failure. When planning a resection, particularly for osteosarcoma or Ewing sarcoma of the long bones, the entire anatomic compartment (e.g., the entire femur from hip to knee) must be imaged with high-resolution MRI to meticulously rule out these discontinuous lesions. If skip metastases are identified, the surgical plan must be radically altered, often necessitating a total compartmental resection or amputation, as leaving a skip lesion behind equates to an incomplete oncologic resection.
Detailed Surgical Anatomy and Biomechanics
To execute a successful limb-sparing resection, the orthopedic oncologist must view human anatomy not merely as a collection of muscles, nerves, and vessels, but as a series of distinct, interconnected biological compartments. Sarcomas, in their initial stages of growth, respect these anatomic borders to a significant degree. Local anatomy heavily influences tumor growth by setting natural, biomechanical barriers to extension. Tumors initially take the path of least resistance, expanding longitudinally within the compliance of their native compartment. Only at later, more advanced stages are these compartment walls violated, allowing the tumor to break into surrounding extracompartmental spaces.
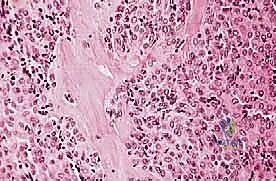
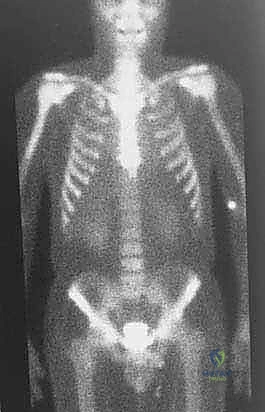
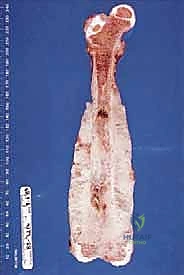
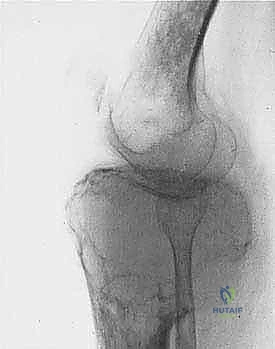
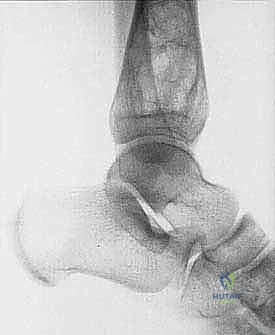
Understanding these typical anatomic barriers is our greatest ally in preoperative planning. Articular cartilage, for instance, is highly resistant to direct tumor invasion. The avascular nature of hyaline cartilage, combined with the presence of anti-angiogenic factors such as chondromodulin, makes it an exceptional natural barrier. This biologic reality allows us to perform precise, intra-articular resections for many distal femur or proximal tibia sarcomas, preserving the joint capsule and adjacent bone when the tumor abuts, but does not penetrate, the subchondral plate.
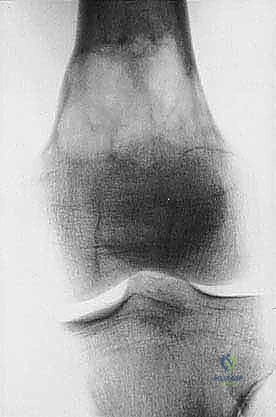
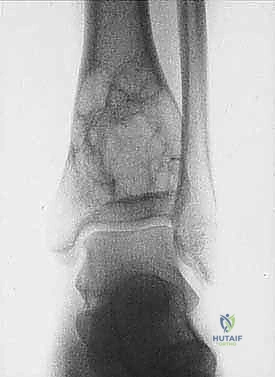
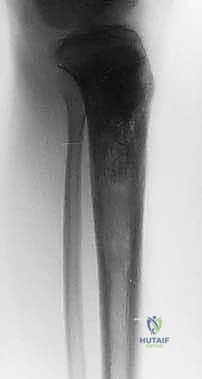
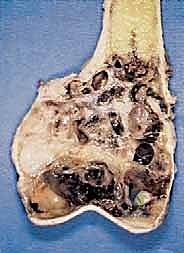
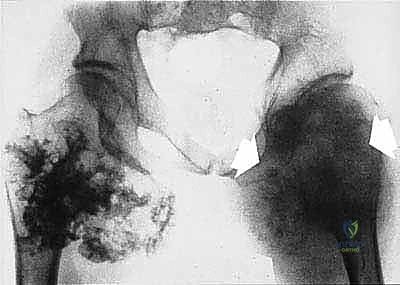
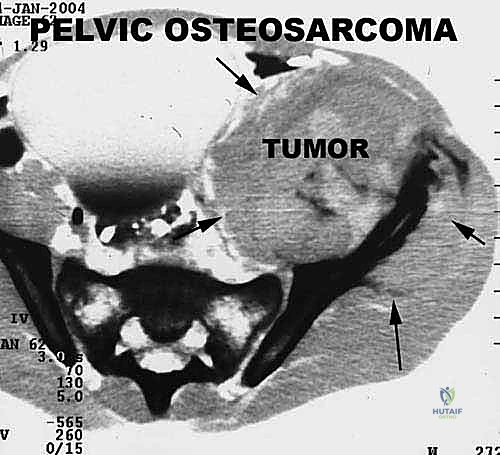
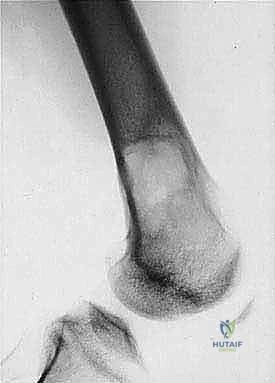
Cortical bone serves as another significant barrier. While high-grade primary bone sarcomas (like osteosarcoma) utilize osteoclast-mediated bone resorption to eventually destroy the cortex and extend into the adjacent soft tissues, thick cortical diaphyseal bone slows this progression. Similarly, thick fascial planes, such as the fascia lata, the intermuscular septa of the thigh, and the aponeuroses of major muscle groups, are robust barriers to soft tissue tumor extension. A tumor may grow to massive proportions within the anterior compartment of the thigh, yet remain entirely contained by the lateral and medial intermuscular septa.
Barrier Integrity and Compartmental Violation
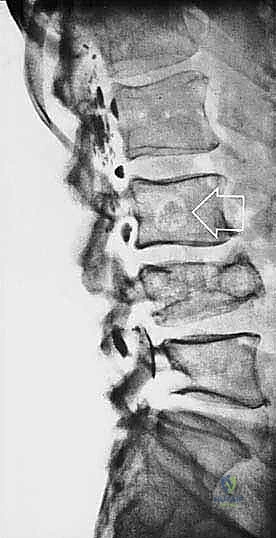
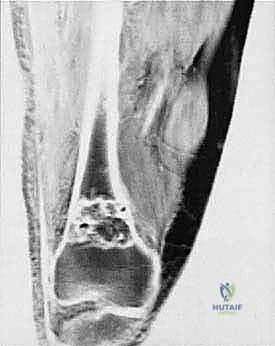
It is a common and dangerous misconception among trainees that the epiphyseal growth plate acts as an anatomic barrier to tumor spread. Crucially, the growth plate is NOT a reliable barrier. While it may temporarily slow tumor progression in very young children, the physis contains numerous transphyseal vascular channels that run directly from the metaphysis to the epiphysis. These vessels serve as a superhighway for microscopic tumor embolization. Therefore, when a metaphyseal sarcoma approaches the physis, the surgeon must assume microscopic epiphyseal involvement unless proven otherwise by high-resolution MRI, and the resection must be planned accordingly.

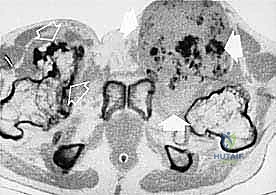
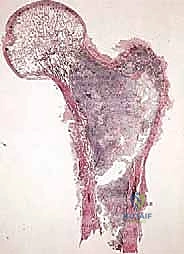
We categorize tumors based on their relationship to these anatomic boundaries. Intracompartmental tumors are completely encased within a defined anatomic compartment (e.g., entirely within the medullary canal of a bone, or entirely within the anterior muscle compartment of the thigh). Extracompartmental tumors, conversely, have either grown out through the compartment barrier (such as an osteosarcoma breaking through the cortex into the muscle) or originated in spaces that lack defined fascial boundaries, such as the popliteal fossa, the femoral triangle, or the axilla. Most high-grade bone sarcomas present as extracompartmental (Enneking Stage IIB) lesions, having already violated the cortex at the time of diagnosis.
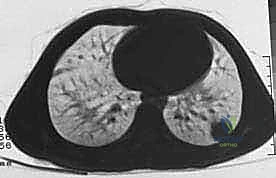
Unlike carcinomas, which frequently disseminate via the lymphatic system, bone and soft tissue sarcomas disseminate almost exclusively hematogenously. This spread is dictated by the venous drainage of the affected compartment. Hematogenous spread of extremity sarcomas typically manifests first as pulmonary metastases, as the lungs represent the first major capillary bed encountered by venous blood returning from the extremities. Bony metastases typically occur later in the disease process. Regional lymph node metastasis is exceptionally rare, observed in only 1-3% of soft tissue sarcomas (with notable exceptions like synovial sarcoma, epithelioid sarcoma, and clear cell sarcoma) and approximately 7% of bone sarcomas.
Exhaustive Indications and Contraindications
The decision to proceed with limb-sparing surgery versus amputation is one of the most critical junctures in orthopedic oncology. Historically, amputation was the standard of care for nearly all high-grade extremity sarcomas. However, landmark studies in the 1980s definitively proved that, when combined with appropriate adjuvant therapies, limb-salvage surgery offers equivalent overall survival rates to amputation, provided that negative surgical margins are achieved. The primary indication for limb salvage is the ability to completely resect the tumor with negative oncologic margins while preserving a limb that is ultimately more functional, or at least cosmetically superior, to a prosthetic replacement.

However, limb salvage is not universally applicable, and the surgeon must resist the temptation to perform heroic resections that leave the patient with a painful, insensate, or biomechanically useless extremity. A salvaged limb that lacks adequate motor control, suffers from chronic osteomyelitis, or requires continuous bracing to prevent collapse is fundamentally inferior to a well-fitted modern amputation prosthesis. The functional demands of the patient, their physiological age, and their psychological readiness for a potentially protracted reconstructive journey must be rigorously evaluated.
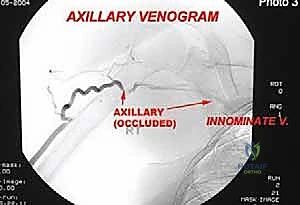
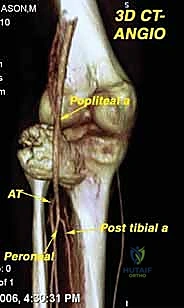
Contraindications to limb salvage are generally absolute when oncologic safety is compromised. Major neurovascular involvement was historically an absolute contraindication; however, with modern vascular bypass techniques, segmental resection and reconstruction of major vessels (e.g., the superficial femoral artery) is now routine. True absolute contraindications include major encasement of the primary motor nerve (e.g., the sciatic nerve in the proximal thigh) where resection would yield a flail limb, massive soft tissue extension precluding adequate soft tissue coverage, and the presence of severe, uncontrolled local infection in the tumor bed.
| Category | Indications for Limb-Sparing Surgery | Contraindications for Limb-Sparing Surgery |
|---|---|---|
| Absolute | Ability to achieve wide/negative surgical margins. | Inability to achieve negative margins without sacrificing the entire limb. |
| Absolute | Intact, functional neurovascular bundle (or reconstructable vessels). | Encasement of essential major motor nerves (e.g., high sciatic nerve) resulting in a useless, insensate limb. |
| Absolute | Adequate remaining soft tissue envelope for coverage or flap reconstruction. | Massive extra-compartmental extension precluding any form of soft tissue coverage. |
| Relative | Favorable response to neoadjuvant chemotherapy (high necrosis rate). | Poor response to neoadjuvant therapy with explosive tumor progression during treatment. |
| Relative | Patient preference and psychological readiness for complex rehab. | Severe patient comorbidities precluding prolonged anesthesia or complex reconstruction. |
| Relative | Anticipated functional outcome superior to amputation prosthesis. | Pathologic fracture with massive hematoma contaminating multiple compartments (historically absolute, now relative). |
Patient Selection and Multidisciplinary Tumor Board Review
No sarcoma resection should ever be planned in isolation. The standard of care mandates that every patient be presented at a formal Multidisciplinary Tumor Board (MDT) comprising orthopedic oncologists, medical oncologists, radiation oncologists, musculoskeletal radiologists, and specialized soft-tissue/bone pathologists. This collaborative approach ensures that the timing of surgery is perfectly synchronized with neoadjuvant chemotherapy or radiation, optimizing the biologic environment for resection.
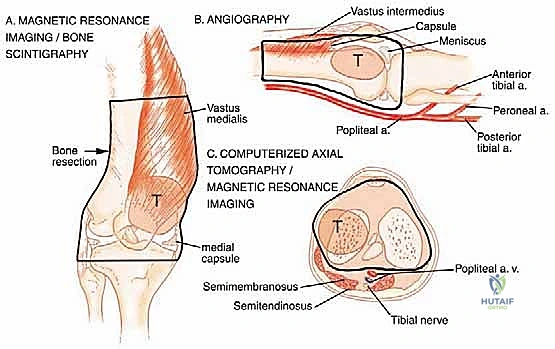
In the context of osteosarcoma and Ewing sarcoma, the tumor board evaluates the post-neoadjuvant MRI to assess for tumor shrinkage, ossification, and the development of a distinct fibrous rim, which facilitates a safer resection. For soft tissue sarcomas, the board must debate the merits of preoperative versus postoperative radiation therapy. Preoperative radiation offers the advantage of a lower total dose, a smaller radiation field, and a thickened pseudocapsule that aids surgical dissection; however, it carries a significantly higher risk of major postoperative wound complications (up to 35%).
Patient selection also demands a rigorous psychosocial evaluation. Limb salvage is not a single event; it is the beginning of a lifelong commitment to oncologic surveillance, intensive physical therapy, and the very real possibility of future reconstructive revisions (e.g., megaprosthesis aseptic loosening or periprosthetic joint infection). The surgeon must have a frank, documented discussion with the patient and family regarding these realities, ensuring their expectations align with the anticipated functional and oncologic outcomes.
Pre-Operative Planning, Templating, and Patient Positioning
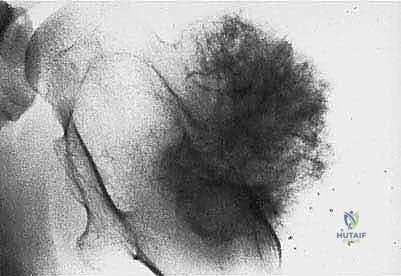
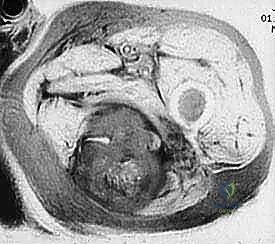
Meticulous preoperative planning is the hallmark of the master orthopedic oncologist. The margin of error in sarcoma surgery is measured in millimeters. Planning begins with a comprehensive review of advanced imaging modalities. A contrast-enhanced MRI of the entire involved bone or compartment is mandatory to define the intraosseous and extraosseous extent of the tumor, assess the relationship to the neurovascular bundle, and definitively rule out skip metastases. A high-resolution CT scan of the chest is required to stage for pulmonary metastases, while a PET-CT or whole-body bone scan evaluates for distant skeletal disease.
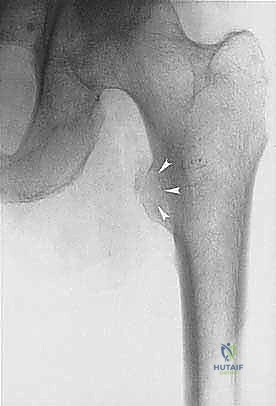
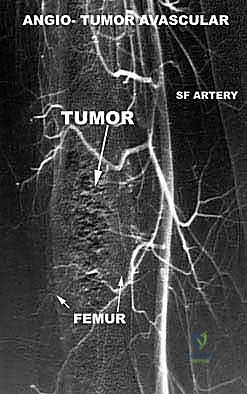

The biopsy is the first, and arguably most critical, step in the surgical intervention. A poorly planned biopsy can contaminate previously uninvolved compartments, converting a straightforward limb salvage into an unavoidable amputation. The biopsy incision must be strictly longitudinal, placed exactly within the planned definitive surgical resection tract, and meticulously hemostatic to prevent tracking of tumor cells via hematoma. The biopsy tract is considered contaminated tissue and must be excised en bloc with the primary tumor during the definitive resection.
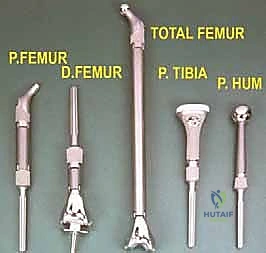
Modern preoperative planning increasingly relies on 3D templating and patient-specific instrumentation (PSI). Utilizing CT and MRI fusion software, the surgeon can virtually map the tumor volume and plan the exact osteotomy planes required to achieve wide margins. Custom 3D-printed cutting guides can be manufactured to ensure these precise osteotomies are replicated in the operating room, minimizing the resection of healthy bone while guaranteeing oncologic safety. Patient positioning must facilitate wide exposure, allow for potential extensile measures (such as vascular control proximal to the tumor), and permit the harvesting of local or free flaps if soft tissue reconstruction is anticipated.
Margin Assessment and Surgical Strategy
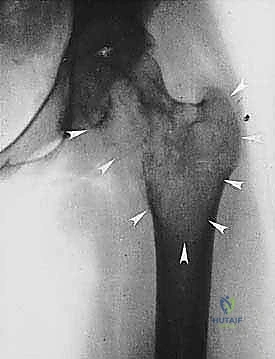
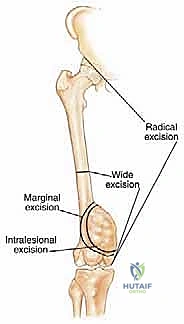
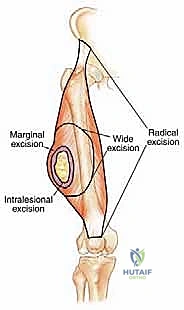
The surgical strategy is fundamentally dictated by the Enneking classification of surgical margins: Intralesional (macroscopic tumor left behind), Marginal (dissection through the reactive zone/pseudocapsule, leaving microscopic disease), Wide (dissection entirely through normal tissue, removing the tumor, pseudocapsule, and a cuff of normal tissue en bloc), and Radical (removal of the entire anatomic compartment). For curative intent in sarcoma surgery, a Wide margin is the absolute minimum acceptable standard.
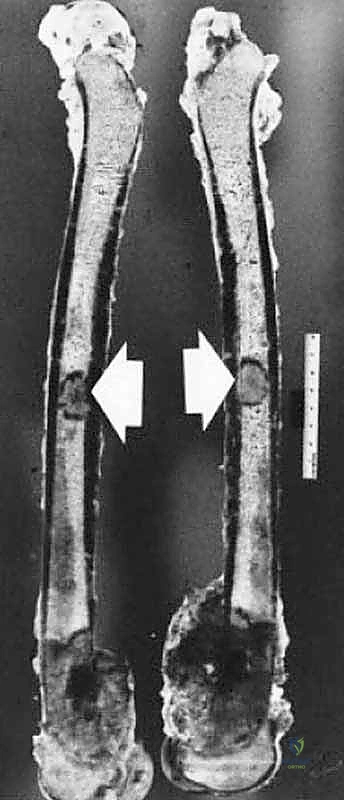
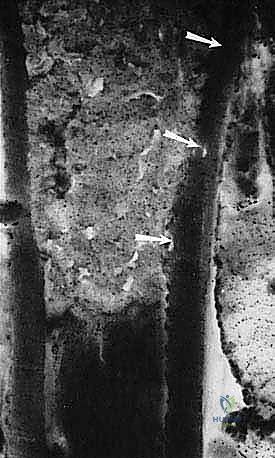
Defining a "Wide" margin in modern practice has evolved. Historically, arbitrary measurements (e.g., 3 cm or 5 cm) were advocated. However, contemporary oncologic principles recognize that the type of tissue comprising the margin is far more critical than the absolute distance in millimeters. A 2-millimeter margin consisting of intact, dense fascial tissue or articular cartilage provides a more robust oncologic barrier than a 3-centimeter margin of loose areolar fat or muscle. The surgeon must meticulously plan the resection to incorporate these natural anatomic barriers into the margin whenever possible.
Planning the osteotomy level requires precise correlation between the preoperative MRI and intraoperative landmarks. Typically, an osteotomy is planned 2 to 3 centimeters beyond the most proximal or distal extent of the marrow edema visible on T1-weighted MRI sequences. Intraoperatively, marrow margins are often sent for frozen section analysis to confirm the absence of microscopic tumor extension before proceeding with the reconstructive phase. Concurrently, the soft tissue envelope must be planned not just for tumor extirpation, but with a clear strategy for dead-space management and robust soft tissue coverage over the reconstruction.
Step-by-Step Surgical Approach and Fixation Technique
The operative execution of a sarcoma resection is an exercise in controlled, deliberate dissection. The procedure commences with an elliptical incision that incorporates the previous biopsy tract, ensuring a minimum of 1 to 2 centimeters of normal skin surrounding the scar. This skin island remains attached to the underlying tumor mass throughout the procedure. Dissection must always proceed from normal, uninvolved tissue toward the abnormal tumor bed. The surgeon must establish planes in healthy tissue and work circumferentially, avoiding any direct visualization of the tumor capsule.