Total Scapular Resection & Endoprosthetic Reconstruction: An Intraoperative Masterclass
Key Takeaway
This masterclass details total scapular resection with endoprosthetic reconstruction. We cover critical anatomy, meticulous preoperative planning, and a granular, real-time intraoperative guide. Learn precise dissection, neurovascular protection, hardware application, and comprehensive postoperative management, emphasizing pearls and pitfalls for optimal patient outcomes.
Welcome, colleagues, to the operating theater. Today, we are undertaking a highly complex and demanding oncologic procedure: a total scapular resection with endoprosthetic reconstruction for a high-grade periscapular sarcoma. This operation requires not only meticulous surgical precision but also an intimate, three-dimensional understanding of the intricate anatomy of the shoulder girdle. Our overarching objective is limb salvage that achieves definitive oncologic margins while yielding the best possible functional outcome—a profound paradigm shift from the forequarter amputations that dominated historical practice.
Comprehensive Introduction and Patho-Epidemiology
Tumors arising from the scapula, such as chondrosarcomas in the adult population or Ewing sarcomas and osteosarcomas in pediatric and adolescent cohorts, often reach a substantial volume prior to clinical detection. Initially, the robust periscapular musculature—comprising the trapezius, latissimus dorsi, rhomboids, and the rotator cuff—acts as an anatomic barrier, containing these lesions and protecting vital anterior neurovascular structures. Consequently, patients typically present late in the disease course with insidious shoulder pain, a palpable, firm mass, or a combination of both, often misdiagnosed initially as a sports-related injury or degenerative condition.
The critical decision to pursue limb-sparing surgery rather than amputation hinges entirely on exhaustive preoperative staging and a precise, millimeter-accurate understanding of the tumor's extent. The surgeon must evaluate the neoplasm's relationship to the key neurovascular bundles of the axilla, the brachial plexus, and the underlying thoracic rib cage. The primary oncologic mandate is the achievement of wide, negative margins; functional reconstruction is strictly secondary to patient survival.
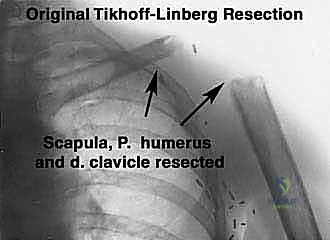
Historically, high-grade scapular sarcomas almost uniformly necessitated a forequarter amputation to achieve local control. However, pioneering surgical advancements, notably the work by Marcove et al. in 1977, built upon the foundation of the classical Tikhoff-Linberg resection. These early innovators demonstrated that aggressive limb-sparing techniques, involving en bloc extra-articular resection of the shoulder girdle, could achieve local tumor control and overall survival rates statistically comparable to amputation, all while preserving a highly functional hand, wrist, and elbow. Today, with the advent of neoadjuvant chemotherapy, advanced cross-sectional imaging, and modular or custom 3D-printed endoprosthetics, the vast majority of scapular malignancies are amenable to limb-sparing surgery.
Forequarter amputation, while increasingly rare, remains a crucial tool in the orthopedic oncologist's armamentarium. It is now strictly reserved for specific, dire clinical scenarios: massive, fungating, or actively infected tumors; local recurrences following failed limb-sparing attempts; or definitive, circumferential invasion of the major neurovascular structures (axillary artery/vein, cords of the brachial plexus) or deep chest wall that categorically precludes the attainment of clear surgical margins with a limb-salvage approach.
Detailed Surgical Anatomy and Biomechanics
Before the first incision is made, a rigorous mental rehearsal of the regional anatomy is mandatory. The shoulder girdle is a highly complex, dynamic web of muscular, vascular, and neural structures. Understanding their precise geometric relationships to the scapula is the foundation for a safe, oncologically sound resection and a stable reconstruction.
Osteology of the Scapula
The scapula is a broad, flat, triangular bone that forms the posterior foundation of the shoulder girdle, serving primarily as a highly mobile muscular attachment site rather than a weight-bearing pillar. Key osteologic features dictate the surgical approach:
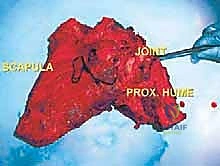
The glenoid fossa articulates with the humeral head. Tumors involving the glenoid, the scapular neck, or those breaching the articular cartilage necessitate an extra-articular resection. This requires transecting the proximal humerus and taking the entire glenohumeral joint capsule en bloc to prevent tumor seeding within the joint space. The coracoid process, a hook-like anterior projection, serves as a critical surgical landmark and an attachment point for the pectoralis minor, coracobrachialis, and the short head of the biceps.
The acromion represents the lateral extension of the scapular spine, forming the highest point of the shoulder and articulating with the distal clavicle. The scapular body itself is remarkably thin, making subtle cortical breakthrough or marrow involvement difficult to assess on standard plain radiographs. Finally, the scapular notch, located on the superior border deep to the transverse scapular ligament, is a critical danger zone through which the suprascapular nerve and vessels pass, requiring careful isolation during superior dissection.

Muscular Intervals and Attachments
The scapula is enveloped by a multitude of muscles that not only frequently contain the extraosseous extension of the tumor but also present significant dissection and reconstructive challenges. These muscles must be systematically detached, and decisions regarding their resection versus preservation dictate the functional outcome.
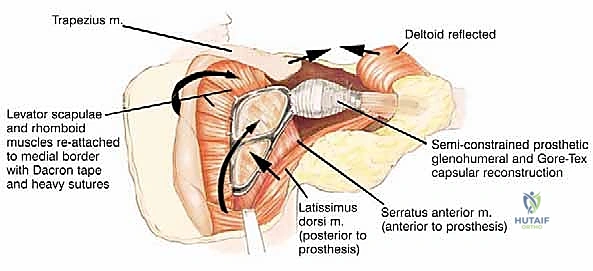
The posterior and medial musculature includes the trapezius, rhomboids (major and minor), latissimus dorsi, supraspinatus, infraspinatus, teres major, and teres minor. The anterior surface is dominated by the subscapularis and the serratus anterior, which glide over the thoracic cage. The deltoid overlies the entire shoulder joint, attaching broadly to the scapular spine and acromion.
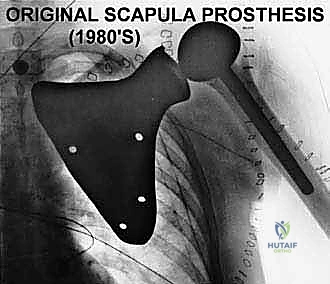
For a successful endoprosthetic reconstruction, the surgeon must strive to preserve the trapezius, deltoid, rhomboids, serratus anterior, and latissimus dorsi, provided they are free of tumor involvement. These specific muscles are absolutely crucial for providing adequate soft tissue coverage over massive metallic implants, providing dynamic suspension of the reconstructed shoulder, and allowing for potential functional restoration. If the trapezius and deltoid are unsalvageable due to tumor proximity, the proximal humerus or the prosthesis must be statically suspended directly from the remaining clavicle using heavy non-absorbable tapes or mesh, resulting in a flail but stable shoulder.
Neurovascular Bundle and Associated Structures
The axilla and the anterior shoulder house the most critical and unforgiving anatomy in this procedure. The axillary vessels and the brachial plexus form a tightly packed neurovascular bundle that is extremely vulnerable to both direct tumor extension and iatrogenic traction injury during scapular mobilization.
The axillary artery and vein represent the continuation of the subclavian vessels, passing beneath the middle third of the clavicle. Their course is medial and inferior to the coracoid process, intimately associated with the cords and branches of the brachial plexus. The branches of the axillary artery must be systematically identified and ligated. The supreme thoracic, thoracoacromial, and lateral thoracic arteries arise proximally. The subscapular artery is a massive branch that divides into the thoracodorsal artery and the circumflex scapular artery. The circumflex scapular artery wraps around the lateral border of the scapula, critically tethering the axillary vessels to the bone. Ligation and division of this specific vessel is the key maneuver that allows the neurovascular bundle to be swept anteriorly and safely away from the resected scapula. Furthermore, the anterior and posterior humeral circumflex arteries arise at the inferior border of the subscapularis. Early ligation of these circumflex vessels is an essential step to mobilize the neurovascular structures away from the tumor mass.
The brachial plexus surrounds the axillary vessels throughout their course. The musculocutaneous nerve arises from the lateral cord and passes through the coracobrachialis. Its path can vary significantly, and large anterior tumors often displace it anteriorly, making it superficial and highly susceptible to injury during the anterior approach. Preservation of this nerve is vital for postoperative elbow flexion. The axillary nerve courses with the posterior humeral circumflex vessels; while scapular tumors often stretch it, the subscapularis muscle usually offers a protective barrier. The radial nerve, arising from the posterior cord, passes anterior to the latissimus dorsi-teres major insertion and must be isolated and protected early in the inferior dissection. The upper and lower subscapular nerves are routinely ligated during a total scapulectomy, whereas the thoracodorsal nerve can usually be spared.
Exhaustive Indications and Contraindications
The decision algorithm for performing a total scapular resection with endoprosthetic reconstruction is complex and requires a multidisciplinary tumor board consensus. The primary indication is a primary malignant bone or soft tissue sarcoma (e.g., chondrosarcoma, osteosarcoma, Ewing sarcoma, undifferentiated pleomorphic sarcoma) that involves the majority of the scapula or the glenohumeral joint, where wide margins cannot be achieved with a partial scapulectomy.
Additionally, massive, aggressive benign lesions, such as advanced giant cell tumors of bone or aneurysmal bone cysts that have destroyed the osseous architecture of the scapula, may necessitate total resection and reconstruction. Metastatic disease to the scapula is a relative indication; it is typically managed non-operatively or with palliative radiation, but isolated, solitary metastases (e.g., from renal cell carcinoma or thyroid cancer) that are highly destructive and painful may warrant resection to improve quality of life.
Contraindications are primarily dictated by oncologic safety. If the tumor has directly invaded the major neurovascular bundle (axillary artery/vein or the cords of the brachial plexus) such that resection would leave a non-viable or completely insensate and paralyzed distal limb, a forequarter amputation is indicated. Similarly, massive invasion into the deep thoracic wall (ribs and pleura) that precludes clear margins is a contraindication to limb salvage. Active, uncontrolled local infection or fungating tumor masses that compromise the skin envelope also preclude the use of massive metallic endoprostheses.
Table of Indications and Contraindications
| Category | Specific Clinical Scenarios | Rationale / Considerations |
|---|---|---|
| Absolute Indications | High-grade primary bone sarcomas (Osteosarcoma, Ewing) involving >50% of scapula. | Requires wide en bloc resection; partial scapulectomy would yield positive margins. |
| Absolute Indications | High-grade soft tissue sarcomas with secondary massive scapular invasion. | Limb salvage is prioritized if neurovascular bundle is free of disease. |
| Relative Indications | Massive, recurrent benign aggressive tumors (e.g., Giant Cell Tumor, Chondroblastoma). | When structural integrity of the scapula is completely compromised. |
| Relative Indications | Solitary, radioresistant metastatic lesions (e.g., RCC, Thyroid) with intractable pain. | Palliative limb salvage to restore mechanical stability and relieve pain. |
| Absolute Contraindications | Circumferential encasement of the axillary artery/vein or brachial plexus cords. | Resection would result in an ischemic or completely flail, insensate distal extremity. Forequarter amputation required. |
| Absolute Contraindications | Deep, extensive invasion into the thoracic cavity (pleura/lung parenchyma). | Inability to achieve negative margins safely without unacceptable morbidity. |
| Relative Contraindications | Severe, uncontrolled local infection or fungating tumor ulcerating through the skin. | High risk of catastrophic periprosthetic joint infection (PJI) with massive hardware. |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the absolute cornerstone of a successful oncologic resection. The surgeon must transition from a two-dimensional understanding of the imaging to a three-dimensional operative blueprint, defining the tumor's exact intraosseous and extraosseous extent, its relationship to critical structures, and formulating a definitive plan for both margins and the reconstruction strategy.
Imaging Modalities and Staging Studies
Plain radiography is often the initial imaging modality. It reveals macroscopic bony involvement and patterns of mineralization, which are highly useful for differentiating an osteoid-producing osteosarcoma from a chondroid-producing chondrosarcoma. However, the thin body of the scapula is frequently obscured by the overlying rib cage, limiting the sensitivity of plain films.

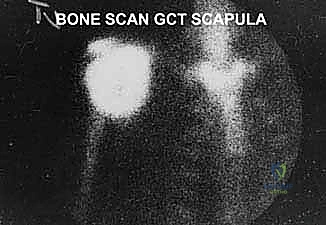
A Technetium-99m whole-body bone scan is valuable for assessing the entire osseous skeleton for synchronous metastatic disease or polyostotic conditions. It also aids in evaluating local biologic activity and extension into the proximal humerus or adjacent ribs. However, the surgeon must be aware that the thin scapular body may not demonstrate the true microscopic extent of the tumor as accurately as long bones; therefore, bone scans must always be correlated with advanced cross-sectional imaging.
Computed Tomography (CT) of the chest with intravenous contrast is mandatory. It is extremely important for evaluating the rib cage for subtle cortical erosion, which is far better visualized on CT than on MRI. CT is also excellent for detecting subtle matrix mineralization within the tumor and for pulmonary staging to rule out lung metastases. Contrast-enhanced CT is crucial for determining the proximity of the medial tumor mass to the axillary vessels.

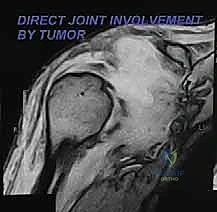
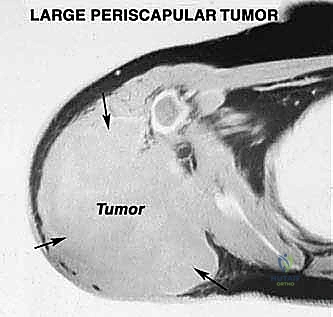
Magnetic Resonance Imaging (MRI) with and without gadolinium contrast is the gold standard and the most accurate modality for determining the precise intraosseous marrow extent and the extraosseous soft-tissue boundaries of the tumor. T1-weighted images are optimal for assessing marrow replacement, while fluid-sensitive T2-weighted or STIR sequences define the peritumoral edema and the true soft-tissue mass. MRI provides the necessary soft-tissue contrast to evaluate the tumor's relationship to the glenohumeral joint capsule, the rotator cuff musculature, and the neurovascular bundle.

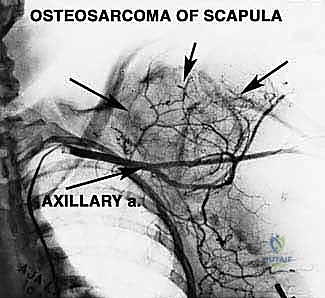
Angiography or CT Angiography (CTA) is frequently utilized to determine precise vascular involvement, reveal anatomic anomalies, and demonstrate the displacement of the axillary vessels. A displaced axillary artery is highly indicative of anterior tumor extension deep into the axilla. Preoperative angiograms can highlight this vascular displacement, allowing the surgeon to anticipate the distorted anatomy during the anterior dissection. In select cases, preoperative embolization of highly vascular tumors (e.g., renal cell metastases or aneurysmal bone cysts) can significantly reduce intraoperative blood loss.
Biopsy Principles and Techniques
The biopsy is a critical operative step that must be planned with the same rigor as the definitive resection. Fine-needle aspiration or core needle biopsies are routinely performed under CT or fluoroscopic guidance to ensure accurate sampling while protecting the adjacent neurovascular bundle and pleura.
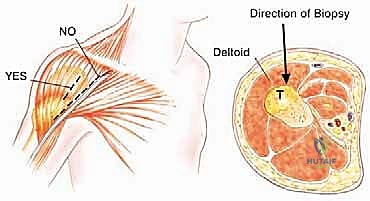
The technique demands strict adherence to oncologic principles. A single puncture site should be used, with the core needle introduced through that exact site at various angles to obtain multiple diagnostic cores. Crucially, the biopsy tract must be placed directly along the intended longitudinal or transverse incision line of the future definitive resection. This ensures that the entire biopsy tract, which is considered contaminated with tumor cells, can be excised en bloc with the specimen. For tumors arising within the body of the scapula, a direct posterior needle approach is highly recommended to avoid contaminating the virgin anterior axillary planes.
Patient Positioning and Anesthesia
The procedure is performed under general anesthesia, often supplemented with a regional interscalene block for postoperative pain control, provided the block does not interfere with intraoperative neuromonitoring (if utilized). The patient is typically positioned in a full lateral decubitus or a "floppy lateral" position, secured with a vacuum beanbag.
This positioning allows the arm to be draped free, granting the surgeon 360-degree access to the shoulder girdle. The entire forequarter, from the base of the neck to the costal margin, and across the midline anteriorly and posteriorly, must be prepped and draped. A sterile tourniquet is not feasible for this central location, making meticulous hemostasis and the availability of rapid blood transfusion protocols imperative.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution of a total scapular resection is a marathon of anatomic dissection, requiring constant vigilance to maintain the oncologic plane while preserving critical functional structures.
Incision and Superficial Dissection
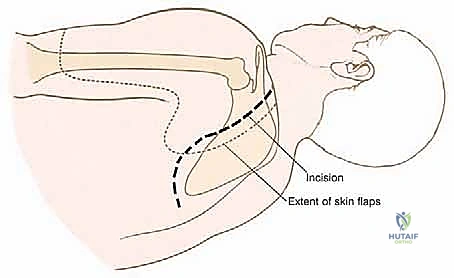
The standard approach utilizes an expansile, utilitarian incision. Often, a modified question-mark or a large elliptical incision is designed, incorporating the prior biopsy tract. The incision typically begins at the medial aspect of the clavicle, extends laterally over the acromioclavicular joint, curves posteriorly along the spine of the scapula, and descends along the medial border of the scapula toward the inferior angle.
Thick fasciocutaneous flaps are elevated anteriorly and posteriorly. It is vital to maintain the viability of these flaps by preserving the subdermal vascular plexus, as massive endoprostheses require robust soft-tissue coverage to prevent catastrophic postoperative wound breakdown. During the medial flap elevation, the spinal accessory nerve (Cranial Nerve XI) must be identified and carefully preserved as it courses on the deep surface of the trapezius, unless the trapezius itself is infiltrated by the tumor and requires en bloc resection.
Deep Dissection and Neurovascular Isolation
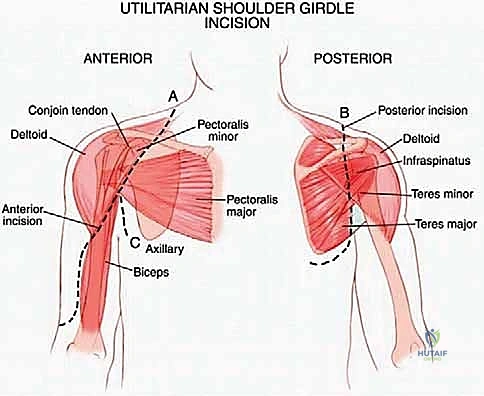
The anterior dissection is usually performed first to gain control of the major blood supply. The deltopectoral interval is developed. The pectoralis major is retracted or partially released, exposing the pectoralis minor, which is detached from the coracoid process. The coracobrachialis and short head of the biceps are similarly tagged and released from the coracoid.
This anterior exposure reveals the axillary sheath. The axillary artery, vein, and the cords of the brachial plexus are systematically skeletonized and looped with vessel loops. The surgeon must meticulously trace the axillary vessels distally, identifying, ligating, and dividing the arterial branches supplying the scapula. As emphasized in the anatomical review, the ligation of the circumflex scapular artery and the anterior/posterior humeral circumflex vessels is the critical maneuver that untethers the neurovascular bundle, allowing it to be safely retracted anteriorly away from the tumor pseudocapsule.
Scapular Mobilization and Resection
With the anterior neurovascular structures protected, attention is turned posteriorly and medially. The trapezius is detached from the scapular spine. The levator scapulae and the rhomboid major and minor are transected near their insertions on the medial border of the scapula. The latissimus dorsi is mobilized, and the teres major and minor are divided.
The deep dissection involves releasing the serratus anterior from the anterior medial border of the scapula, taking extreme care not to breach the parietal pleura of the underlying rib cage. The suprascapular nerve and vessels are identified at the scapular notch, ligated, and divided.
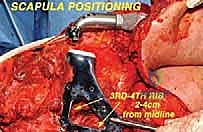
To free the shoulder girdle completely, the clavicle is either osteotomized at its distal third or the acromioclavicular ligaments are sharply divided. If the tumor involves the glenohumeral joint, an extra-articular resection is performed by osteotomizing the proximal humerus at a pre-planned, templated level. The entire scapula (and proximal humerus, if indicated) is then delivered en bloc from the surgical field and sent to pathology for immediate margin assessment.
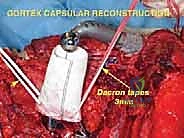
Endoprosthetic Reconstruction and Soft Tissue Tenodesis
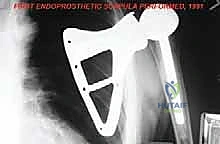
Following rigorous irrigation and confirmation of negative margins, the reconstructive phase commences. Modern total scapular endoprostheses are highly modular or custom 3D-printed titanium implants designed to replicate the geometry of the native scapula while providing numerous fenestrations and suture holes for soft-tissue attachment.
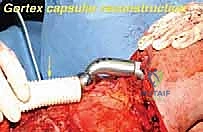
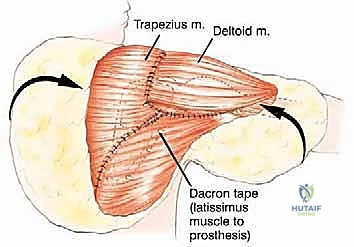
If the proximal humerus was resected, a modular proximal humeral component is cemented or press-fit into the remaining humeral shaft and coupled to the glenoid component of the scapular prosthesis. The entire construct must then be suspended to prevent inferior subluxation under the weight of the arm. This is achieved by securing the superior aspect of the scapular prosthesis to the remaining clavicle or the upper ribs using heavy, non-absorbable synthetic tapes (e.g., Trevira tube) or thick braided sutures.
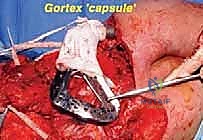
The soft tissue reconstruction is arguably as important as the bony fixation. The prosthesis is often wrapped in a synthetic mesh (such as Marlex or Trevira) to facilitate soft-tissue ingrowth. The remaining native musculature—specifically the trapezius, rhomboids, serratus anterior, and deltoid—must be meticulously tenodesed to the prosthesis and the mesh under appropriate tension. This dynamic muscle envelope provides essential soft-tissue coverage, reduces dead space, and offers the potential for limited but meaningful active shoulder motion. Closed suction drains are placed, and the thick fasciocutaneous flaps are closed in multiple layers to ensure a watertight seal.
Complications, Incidence Rates, and Salvage Management
Total scapular resection is a massive surgical insult, and complications are unfortunately common, requiring vigilant postoperative monitoring and aggressive management. The creation of a massive dead space, combined with extensive soft-tissue dissection and the introduction of a large volume of foreign material, sets the stage for several distinct challenges.
Intraoperative and Early Postoperative Complications
Intraoperative neurovascular injury is a catastrophic risk. Traction injuries to the brachial plexus, particularly the musculocutaneous and radial nerves, can occur during the retraction required to mobilize the massive tumor. Direct vascular injury to the axillary artery requires immediate vascular surgery consultation for primary repair or interposition grafting.
Infection is the most dreaded postoperative complication, with incidence rates for massive endoprostheses in the upper extremity ranging from 10% to 15%. The poor soft-tissue envelope, combined with neoadjuvant chemotherapy or radiation, impairs wound healing. Superficial flap necrosis can rapidly progress to deep periprosthetic joint infection (PJI). Aggressive, early surgical debridement, rotational flap coverage (e.g., a pedicled latissimus dorsi flap), and targeted intravenous antibiotics are required.
Mechanical complications, including prosthetic instability, glenohumeral dislocation, and aseptic loosening, also occur over time. The reconstructed shoulder is inherently unstable due to the loss of the dynamic rotator cuff stabilizers. Dislocation often requires closed reduction and prolonged immobilization, though recurrent instability may necessitate revision surgery to augment the capsular reconstruction or upsize the glenosphere in reverse-geometry setups.
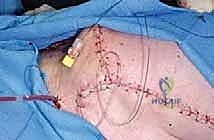
Table of Complications and Management Strategies
| Complication | Estimated Incidence | Prevention / Intraoperative Strategy | Salvage / Management Strategy |
|---|---|---|---|
| Wound Necrosis / Dehiscence | 15 - 25% | Preserve subdermal plexus; avoid excessive flap tension; meticulous hemostasis. | Early aggressive debridement; negative pressure wound therapy; pedicled latissimus or free tissue transfer. |
| Periprosthetic Infection | 10 - 15 |
