Surgical Management of Metastatic Bone Disease: Pelvic Lesions
Comprehensive Introduction and Patho-Epidemiology
Metastatic tumors of the pelvis represent a profound challenge in orthopedic oncology, frequently causing debilitating mechanical pain, catastrophic loss of function, and a complete inability to bear weight. The pelvis is a frequent site for skeletal metastases, secondary only to the spine, with primary carcinomas of the breast, prostate, lung, kidney, and thyroid accounting for the vast majority of these lesions. Due to the relatively large volumetric capacity of the pelvic cavity, the highly elastic nature of the visceral organs it contains, and the massive envelope of surrounding musculature, tumors in this anatomical region usually reach considerable, sometimes staggering, dimensions before becoming clinically symptomatic. Consequently, patients often present with advanced local disease characterized by extensive cortical destruction and significant soft tissue extension, necessitating highly complex, multidisciplinary surgical planning.
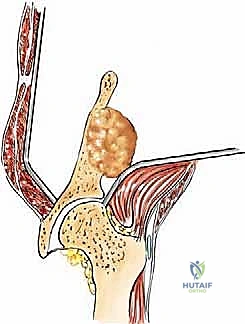
The precise anatomical location of the metastatic deposit within the pelvic ring dictates both the clinical presentation and the biomechanical implications for the patient. While some locations of metastases within the pelvis have virtually no immediate impact on pelvic ring stability and overall ambulatory function (such as isolated lesions of the iliac wing or the anterior pubic rami), tumors located in the critical load-bearing zones present a severe threat to the patient's mobility. Tumors of the posterior ilium and sacroiliac junction pose a direct threat to lumbosacral integrity and spinopelvic stability. More critically, tumors of the periacetabular region profoundly impair hip joint kinematics and the weight-bearing capacity of the entire lower extremity, often leading to rapid functional decline, bed rest, and the cascade of complications associated with immobility.
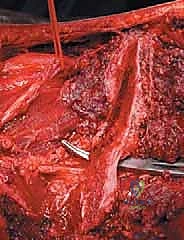
The pathophysiology of these lesions is driven by the complex interaction between tumor cells and the bone microenvironment, often described as the "vicious cycle" of bone metastasis. Tumor cells secrete factors such as parathyroid hormone-related protein (PTHrP) and interleukins, which stimulate osteoblast expression of RANKL. This, in turn, drives rampant osteoclastogenesis and subsequent osteolysis. In hypervascular lesions, such as metastatic renal cell carcinoma or follicular thyroid carcinoma, this osteolysis is accompanied by a massive proliferation of fragile, aberrant tumor neo-vasculature. These hypervascular metastases can bleed profusely, causing life-threatening exsanguination within minutes upon surgical exposure and intralesional curettage if not properly recognized and managed preoperatively.
The management of metastatic pelvic disease has evolved from a purely conservative approach to an aggressive, surgically driven paradigm aimed at immediate mechanical stabilization and pain relief. Both primary bone sarcomas and metastatic carcinomas usually present with considerable extension into the extraosseous soft tissues. However, due to the inherent sensitivity of most metastatic carcinomas to fractionated radiation therapy, the surgical management of metastatic lesions rarely requires the morbid, oncologically radical en bloc resection of overlying muscles and neurovascular structures that is standard for primary sarcomas. Instead, the modern surgical philosophy favors meticulous intralesional tumor excision, robust mechanical reconstruction with cemented hardware, and the treatment of microscopic residual disease with adjuvant external beam radiation therapy.
Detailed Surgical Anatomy and Biomechanics
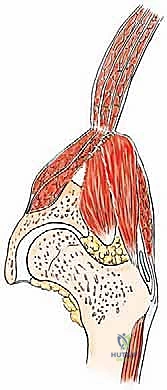
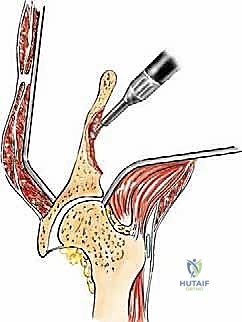
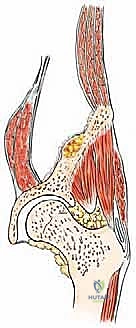
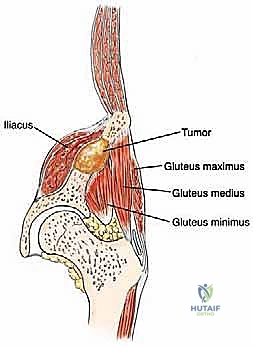
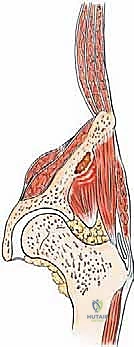
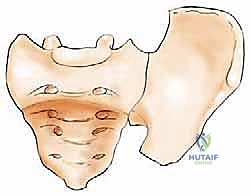
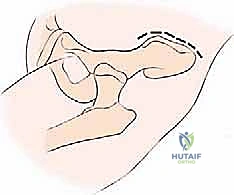
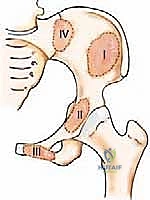
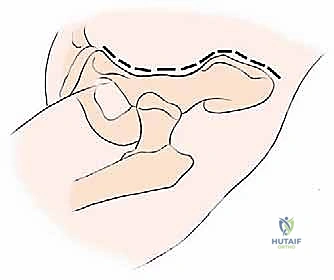
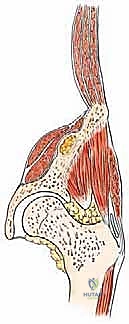
A profound understanding of pelvic osteology, muscular anatomy, and neurovascular topography is the absolute prerequisite for the safe and effective surgical management of pelvic metastases. The classification of pelvic resections, originally attributed to Enneking and Dunham, is based on the specific region of the innominate bone resected. Type I resections involve the ilium; Type II resections involve the periacetabular region; Type III resections involve the pubis and ischium; and Type IV resections (or extended Type I) involve the en bloc resection of the posterior ilium along with the sacral ala. Each of these zones presents unique biomechanical challenges. The acetabulum (Zone II) provides the critical upper-medial mechanical support of the hip joint, transferring the entire weight of the upper body from the axial skeleton, through the sacroiliac joint, down the sciatic buttress, and into the femoral head.
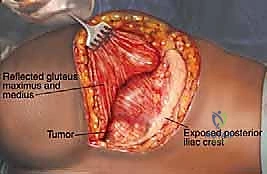
The muscular anatomy of the pelvis is complex and dictates the surgical exposures required for tumor resection. The iliac crest serves as the primary attachment site for the abdominal wall musculature superiorly and the quadratus lumborum posteriorly. The massive gluteal muscles (gluteus maximus, medius, and minimus) originate from and overlie the outer iliac table, providing essential hip extension and abduction forces. Conversely, the iliacus muscle overlies the inner iliac table, converging with the psoas major to form the primary hip flexor complex. In the anterior pelvis, the robust hip adductor complex takes its origin from the inferior aspect of the pubis and ischium. Notably, the acetabulum itself has no direct muscle attachments, which facilitates its exposure once the overlying muscular envelopes are reflected.
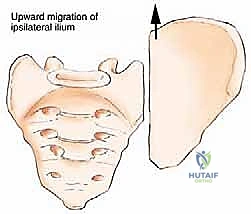
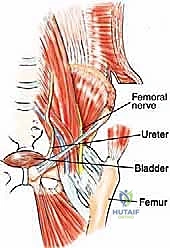
Neurovascular considerations in pelvic oncology are paramount, as iatrogenic injury can result in catastrophic hemorrhage or profound functional deficits. The femoral nerve lies medial to the inner iliac table, situated deep in the groove between the iliacus and the psoas muscles, and must be meticulously protected during medial exposures of the ilium and anterior acetabulum. The obturator neurovascular bundle runs along the anterior aspect of the pubis and must be isolated and mobilized during Type III resections. Posteriorly, the sciatic nerve exits the greater sciatic notch, lying in close proximity to the posterior column of the acetabulum and the ischial spine, making it highly vulnerable during posterior periacetabular reconstructions and Type IV sacropelvic resections.
Visceral anatomy further complicates anterior and medial pelvic approaches. The urinary bladder attaches in close proximity to the posterior wall of the pubis. Accessing the superior pubic ramus and symphysis requires careful entry into the space of Retzius (the retropubic space), necessitating the gentle mobilization of the bladder and protection of the delicate perivesical venous plexus. Furthermore, the external iliac vessels course along the pelvic brim, directly overlying the superior pubic ramus and anterior column of the acetabulum. Meticulous subperiosteal dissection is mandatory to avoid catastrophic vascular injury when addressing lesions in the medial periacetabular region or the superior pubic root.
Exhaustive Indications and Contraindications
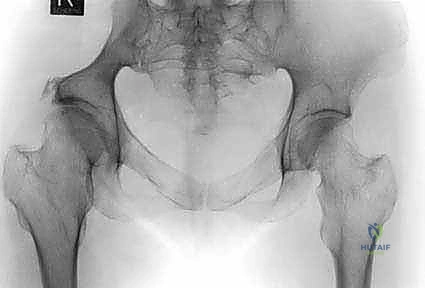
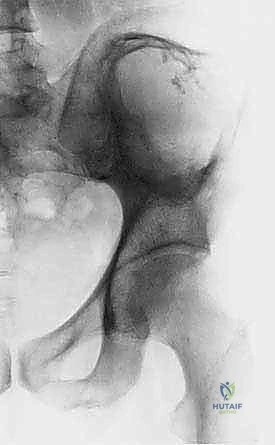
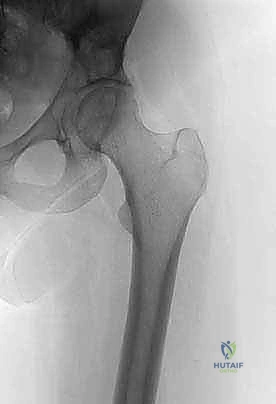
The decision to proceed with major surgical intervention for metastatic pelvic disease requires a delicate balance between the patient's overall oncologic prognosis, their current degree of mechanical instability, and the anticipated morbidity of the procedure. The primary indication for surgery is a pathological fracture of the acetabulum. Once the structural integrity of the acetabular dome or medial wall is breached, conservative management inevitably fails. The patient is rendered non-ambulatory, suffering from excruciating mechanical pain with any attempted movement of the lower extremity. In these scenarios, surgical reconstruction is not merely an option; it is an absolute necessity to prevent the rapid physical and psychological decline associated with terminal bed confinement.
Impending pathological fractures of the acetabulum represent another critical indication for operative intervention. These are defined as osteolytic lesions that extend directly to the subchondral bone of the acetabular roof, are associated with significant cortical destruction of the anterior or posterior columns, and present with considerable, escalating pain upon weight-bearing. Prophylactic stabilization of an impending fracture is technically less demanding, carries a lower complication profile, and yields superior functional outcomes compared to the reconstruction of a completely collapsed and fragmented acetabulum. The Harrington criteria are frequently utilized to assess the risk of periacetabular collapse, guiding the surgeon toward early, proactive cementoplasty or complex arthroplasty.
Intractable pain associated with locally progressive disease that has shown an inadequate response to high-dose narcotic analgesia and preoperative radiation therapy is a strong indication for palliative tumor debulking and stabilization. Even in the absence of a frank or impending fracture, massive tumor expansion stretches the sensitive periosteum and compresses adjacent lumbosacral nerve roots, causing severe neuropathic and somatic pain. Additionally, in highly selected patients presenting with a solitary bone metastasis—particularly from favorable primary histologies such as renal cell carcinoma or breast cancer—wide en bloc resection (rather than intralesional curettage) may be indicated to achieve local disease control and potentially extend overall survival.
Contraindications to major pelvic reconstruction must be rigorously respected to avoid inflicting unnecessary surgical trauma on a patient nearing the end of life. Absolute contraindications include a critically poor functional status (ECOG performance status 3 or 4), an anticipated life expectancy of less than 6 to 12 weeks, uncorrectable coagulopathies, and the presence of overwhelming, rapidly progressive visceral metastases (e.g., massive hepatic replacement or severe malignant pleural effusions leading to respiratory failure). Relative contraindications include extensive, bilateral pelvic destruction that precludes the secure fixation of reconstruction hardware, and active, untreated systemic infections.
| Clinical Parameter | Indications for Surgery | Contraindications to Surgery |
|---|---|---|
| Acetabular Status | Pathological fracture, Impending fracture (roof involvement) | Minimal asymptomatic cortical defect |
| Pain Level | Intractable mechanical pain, unresponsive to XRT/narcotics | Mild, easily controlled with oral analgesics |
| Oncologic Status | Solitary metastasis, chemo/radio-resistant histology | Widespread, rapidly fatal visceral metastasis |
| Patient Physiology | Life expectancy > 3 months, medically optimized | Life expectancy < 3 months, severe coagulopathy |
| Tumor Vascularity | Embolized hypervascular tumor, standard vascularity | Un-embolized hypervascular tumor (e.g., RCC) |
Pre-Operative Planning, Templating, and Patient Positioning
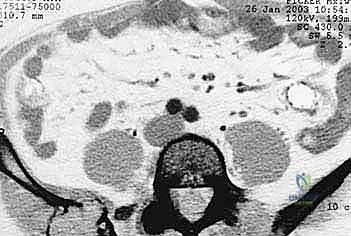
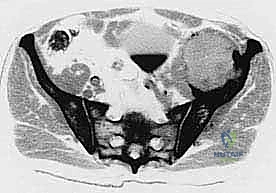
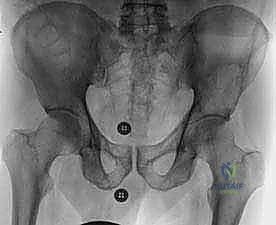
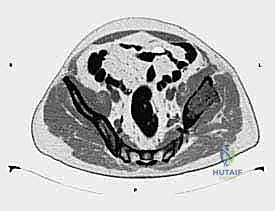
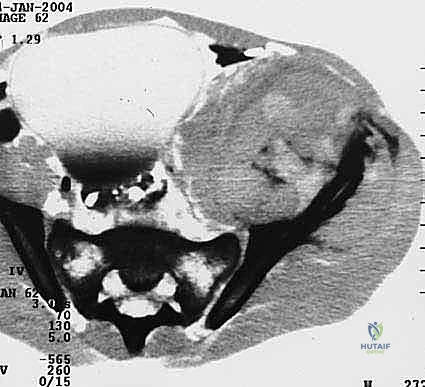
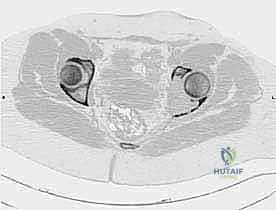
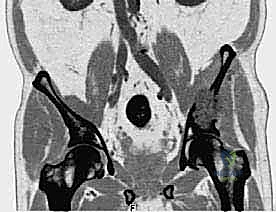
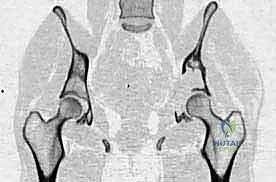
The complex, three-dimensional anatomy of the pelvic girdle mandates meticulously detailed preoperative imaging, exhaustive planning of the surgical exposure, and the precise selection of reconstruction techniques. Plain radiographs (anteroposterior, Judet obturator, and iliac oblique views) and high-resolution computed tomography (CT) of the pelvis and hip joints are absolutely mandatory. CT scans, particularly with multi-planar coronal and sagittal reconstructions, are essential to evaluate the full extent of trabecular bone destruction, the integrity of the remaining cortical struts, and the exact volumetric defects that will require cement or allograft filling. The surgeon must ascertain whether the subchondral bone of the acetabulum is intact, as this dictates whether a joint-preserving curettage or a total hip arthroplasty is required.
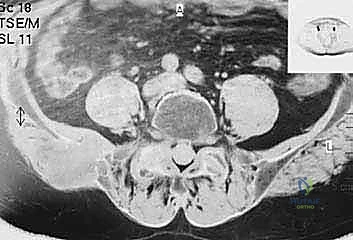
Magnetic resonance imaging (MRI) is generally utilized to assess extraosseous soft tissue extension and the proximity of the tumor mass to critical neurovascular structures. While MRI rarely adds additional information regarding cortical bone integrity compared to CT, it is highly indicated in lesions that have diffuse intramedullary extension. Pathologies such as multiple myeloma or lymphoma frequently exhibit extensive intramedullary marrow replacement that is commonly underestimated by CT imaging. Furthermore, total body bone scintigraphy (Tc-99m bone scan) or Positron Emission Tomography (PET-CT) is routinely performed to detect synchronous skeletal metastases elsewhere, which might alter the surgical plan or dictate a non-operative approach if the overall disease burden is overwhelming.
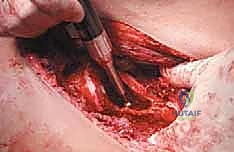
Preoperative tumor embolization is a critical adjunct in the management of pelvic metastases. Hypervascular lesions, classically metastatic renal cell carcinoma, thyroid carcinoma, and certain neuroendocrine tumors, can bleed profusely, causing life-threatening exsanguination within minutes upon tumor exposure and intralesional curettage. Preoperative selective arterial embolization, performed by an interventional radiologist 24 to 48 hours prior to the surgical procedure, is strongly advised to reduce intraoperative blood loss, improve visualization of the tumor cavity, and facilitate a more thorough intralesional excision.
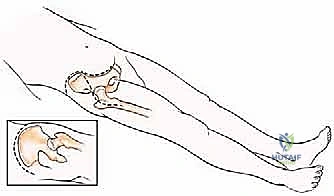
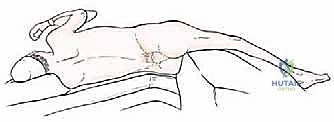
Patient positioning is dictated entirely by the Enneking zone of the planned resection. For Types I through III resections (involving the ilium, periacetabular region, and pubis), the patient is placed supine on a radiolucent operating table with the ipsilateral hip slightly elevated using a bump. This positioning allows unhindered access to the anterior iliac crest, the entire acetabular region, and the symphysis pubis, while permitting intraoperative fluoroscopy. Conversely, for Type IV resections involving the posterior ilium and sacrum, the patient is placed in a true lateral decubitus position with the affected side of the pelvic girdle uppermost. The operating table is bent (broken) with the breakage point just below the contralateral hip; this maneuver widens the space between the iliac crest and the lower aspect of the chest wall, allowing a comfortable approach and easier surgical maneuvering at the flank and posterior elements.
Step-by-Step Surgical Approach and Fixation Technique
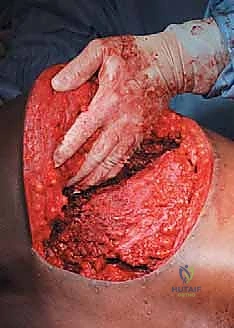
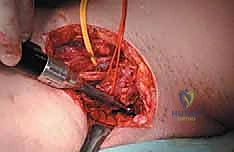
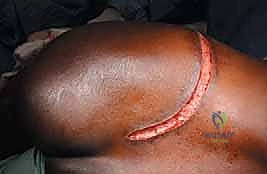
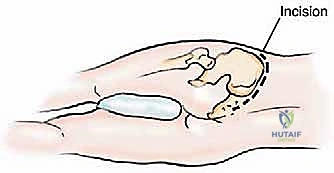
The most versatile and universally applicable approach to pelvic tumor resection is the utilitarian pelvic incision. This expansile exposure can be tailored; all or part of the incision can be utilized for adequate exploration and resection of pelvic girdle metastases depending on the specific Enneking zone involved. The classic incision begins at the posterior inferior iliac spine (PIIS) and extends anteriorly along the iliac crest to the anterior superior iliac spine (ASIS). From the ASIS, it separates into two distinct arms: one extends medially along the inguinal ligament towards the symphysis pubis, while the other turns distally over the anterior thigh, curving laterally just posterior to the shaft of the femur below the greater trochanter, following the insertion of the gluteus maximus muscle.
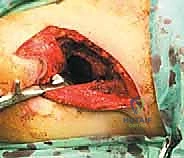
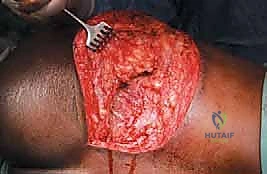
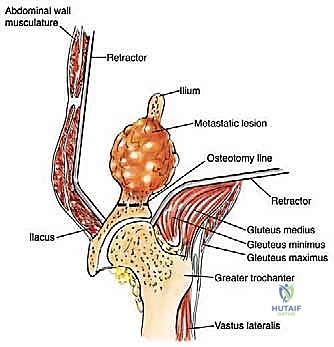
For Type I resections involving the iliac wing, the approach depends on the primary site of cortical destruction. For lesions with medial cortical destruction, the middle component of the utilitarian incision is used, extending to the ASIS with a 5-cm extension along the inguinal arm. Electrocautery is applied to detach and deflect the iliacus muscle from the inner iliac table, exposing the medial wall. For lesions with lateral destruction, the glutei are detached from the outer table. Type I resections typically involve a wide osteotomy of the ilium around the lesion with margins of 1 to 2 cm. Intralesional tumor curettage is neither feasible nor justified at the iliac wing, because a complete resection of the ilium that does not impair acetabular or sacroiliac joint integrity rarely has any long-term impact on patient function or gait.
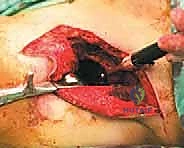
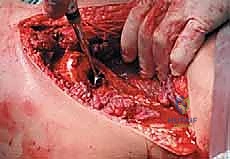
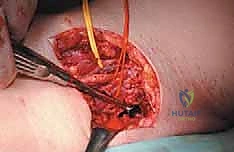
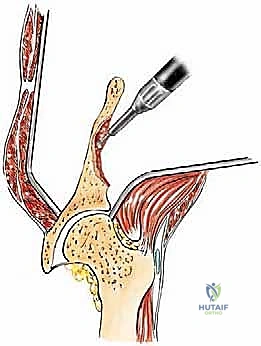
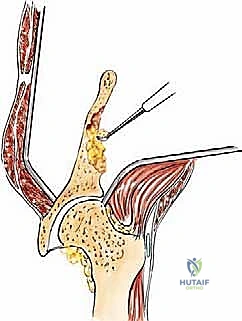
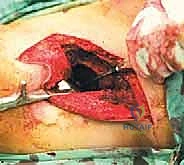
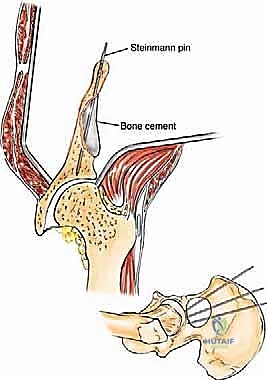
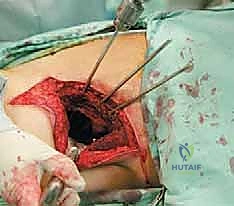
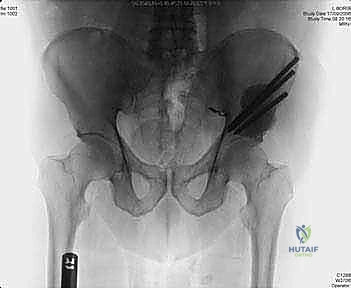
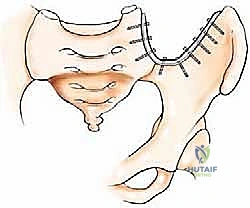
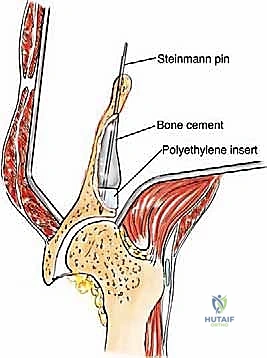
Type II (periacetabular) resections demand meticulous technique. For lesions with lateral cortical destruction, the glutei are reflected to expose the lateral acetabular wall. A wide cortical window is created directly over the lesion. Gross tumor is aggressively removed with large hand curettes. Curettage must be exhaustive, aiming to leave only microscopic disease within the cavitary defect. Adjuvant local treatments, such as high-speed burring, argon beam coagulation, or the application of phenol/alcohol, are frequently employed to extend the intralesional margin. Following tumor extirpation, the massive osseous defect is reconstructed using reinforced polymethylmethacrylate (PMMA) bone cement, often augmented with threaded Harrington pins directed up the intact sciatic buttress and into the sacroiliac joint. If the articular cartilage is compromised, this construct is paired with a total hip arthroplasty using a protrusio cage or custom triflange acetabular component.
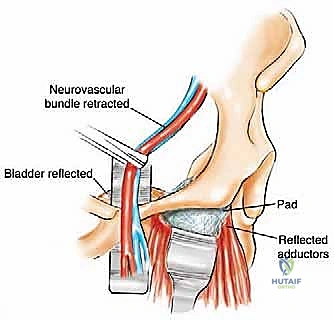
Type III and Type IV resections require highly specialized exposures. For Type III (pubic) resections, the inguinal component of the utilitarian incision is utilized. The external iliac neurovascular bundle is carefully isolated, marked with vessel loops, and mobilized laterally. The retropubic space is exposed, and a protective laparotomy pad is inserted between the urinary bladder and the posterior pubis. The adductor muscle attachments are then sharply detached from the inferior pubic ramus prior to osteotomy. For Type IV (posterior ilium/
Clinical & Radiographic Imaging Archive