Surgical Masterclass: Advanced Management of Metastatic Bone Disease
Key Takeaway
This masterclass guides fellows through the comprehensive surgical management of metastatic bone disease. We cover critical preoperative planning, precise intraoperative techniques for pathological fractures and impending lesions, including meticulous tumor debulking and robust fixation with cement. Emphasis is placed on neurovascular preservation, optimal implant selection, and managing complex reconstructions, ensuring immediate stability and restoring function for cancer patients.
Comprehensive Introduction and Patho-Epidemiology
The surgical management of metastatic bone disease (MBD) represents one of the most challenging, yet profoundly impactful, disciplines within orthopaedic oncology. This masterclass delves into the intricate decision-making paradigms and advanced surgical techniques required to navigate this complex pathology. Unlike the straightforward mechanical failure seen in traumatic fractures, MBD represents a systemic oncologic failure manifesting as localized skeletal destruction. The primary objective is rarely curative; rather, it is to restore immediate mechanical integrity, alleviate intractable pain, and optimize the patient’s functional independence and quality of life in the setting of advanced malignancy. The consequences of inadequate preoperative planning or suboptimal surgical execution can be devastating, frequently robbing an already vulnerable patient of their remaining mobility and dignity.
The skeleton is remarkably hospitable to metastatic seeding, representing the third most common site for metastatic disease, trailing only the pulmonary and hepatic systems. The microenvironment of the bone marrow, rich in growth factors and cytokines, provides a fertile "soil" for the "seed" of circulating tumor cells. We frequently encounter skeletal metastases originating from carcinomas of the breast, prostate, lung, kidney, and thyroid. Collectively, these five primary malignancies account for a staggering 80% of all skeletal involvement. The pathophysiology is primarily driven by the dysregulation of the normal bone remodeling cycle. Tumor cells secrete factors such as parathyroid hormone-related protein (PTHrP), which stimulates osteoblasts to upregulate Receptor Activator of Nuclear Factor Kappa-B Ligand (RANKL). This, in turn, drives massive osteoclastogenesis and subsequent osteolysis, releasing further growth factors (like TGF-beta) from the bone matrix that perpetually fuel tumor growth in a vicious cycle.
With the advent of advanced targeted systemic therapies, immunotherapies, and refined radiation protocols, oncology patients are achieving unprecedented survival rates. Consequently, the orthopaedic surgeon is encountering an ever-increasing volume of patients presenting with MBD who require durable skeletal reconstructions that will outlast their extended life expectancies. While the exact incidence remains difficult to pinpoint due to underreporting in asymptomatic patients, epidemiological estimates suggest that hundreds of thousands of individuals in the United States alone die annually with bone metastases. This underscores the immense prevalence and the crushing clinical burden of this condition on healthcare systems worldwide.
A fundamental paradigm shift is required when transitioning from trauma surgery to orthopaedic oncology. Traditional orthopaedic approaches for traumatic fractures rely heavily on intact biological bone healing and osteointegration. Autologous bone grafts, allografts, or allograft-prosthetic composites are generally contraindicated in the setting of MBD. The local healing environment is severely compromised by the tumor burden, systemic chemotherapy, malnutrition, and adjuvant radiation therapy. Therefore, our focus must pivot entirely away from biological union and toward achieving immediate, rigid, and durable mechanical stability. The construct must withstand immediate full weight-bearing and accommodate the inevitable delays in soft tissue healing associated with adjuvant therapies.
Detailed Surgical Anatomy and Biomechanics
Understanding the biomechanical alterations induced by metastatic lesions is paramount for selecting the appropriate reconstructive strategy. The skeletal system is subjected to complex, multi-planar forces including axial compression, bending, and torsion. A metastatic deposit disrupts the homogenous distribution of these forces, creating a significant stress riser. In the diaphysis of long bones, such as the femur or humerus, the cortex bears the brunt of the load. A lytic lesion acting as a cortical defect exponentially decreases the bone's load-bearing capacity. According to the principles of biomechanics, the torsional rigidity of a tubular bone is proportional to the fourth power of its radius. Therefore, even a relatively small open section defect in the cortex can reduce the bone's resistance to torsional forces by over 70%, predisposing the patient to a catastrophic spiral fracture during routine activities of daily living.
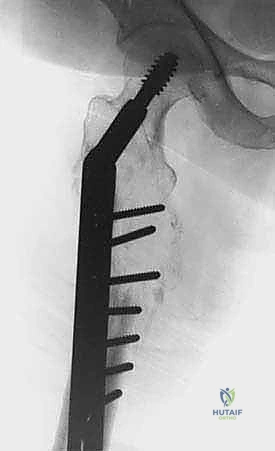
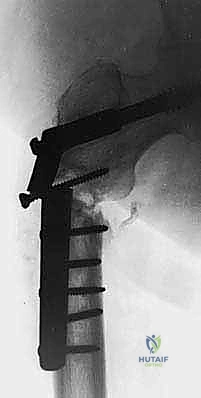
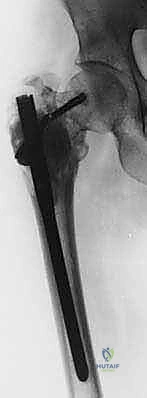
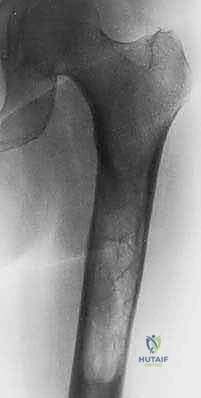
The proximal femur is the most common site for impending and completed pathological fractures requiring surgical intervention, given its role in transmitting the entire weight of the upper body to the lower extremities. The biomechanics here are uniquely unforgiving. The femoral neck and subtrochanteric regions are subjected to massive compressive forces medially and high tensile forces laterally. A lytic lesion in the subtrochanteric region, particularly on the medial calcar, destroys the primary compressive buttress of the proximal femur. Standard osteosynthesis with plates and screws is virtually guaranteed to fail in this scenario due to the massive bending moments and lack of medial support. Intramedullary devices or endoprosthetic reconstructions that bypass the defect and transfer loads directly to the intact distal diaphysis are absolutely required.

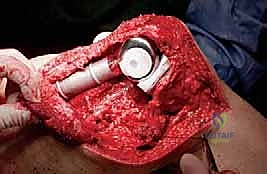
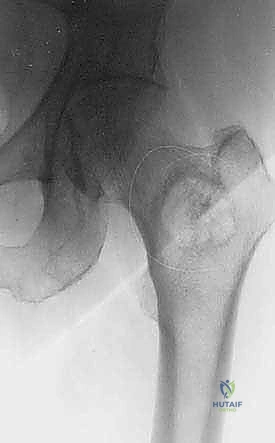
Conversely, the acetabulum presents a different set of anatomical and biomechanical challenges. Metastatic destruction of the periacetabular bone compromises the transmission of forces from the lower extremity to the axial skeleton. The superior dome, medial wall, and posterior column are critical for hip joint stability. When tumor osteolysis destroys these pillars, the femoral head can migrate centrally or superiorly, leading to severe pain and limb shortening. Reconstruction in this region cannot simply rely on standard total hip arthroplasty components, as there is often insufficient host bone for cup osteointegration. Techniques utilizing structural allografts are prone to failure due to non-union and infection. Instead, we must rely on complex constructs involving polymethylmethacrylate (PMMA) bone cement reinforced with Harrington pins, anti-protrusio cages, or custom triflange components to bridge the defect to intact iliac, ischial, or pubic bone.
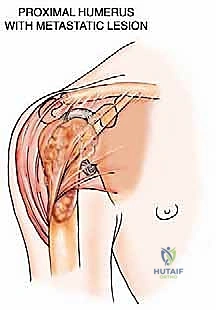
The upper extremity, while not subjected to the same repetitive axial loads as the lower extremity, is subjected to significant torsional and bending forces during lifting and pushing. The proximal humerus is frequently involved. Pathological fractures here severely compromise the shoulder girdle's function, rendering the arm useless for positioning the hand in space. Given the high rates of non-union in pathological humerus fractures treated non-operatively, surgical stabilization is aggressively pursued. The anatomical proximity of the radial nerve in the spiral groove must be meticulously respected during any diaphyseal intervention, and the axillary nerve must be protected during proximal reconstructions.
Exhaustive Indications and Contraindications
The decision to proceed with surgical intervention in the setting of MBD is highly nuanced and requires a delicate balance between the patient's overall prognosis, their current functional status, and the mechanical severity of the skeletal lesion. These indications are not arbitrary; they are driven by the imperative to alleviate suffering and prevent catastrophic skeletal-related events (SREs).

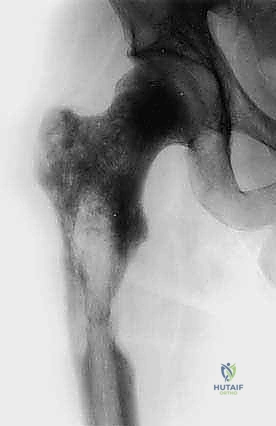
The most straightforward indication is an existing pathological fracture. A bone that has failed through an area weakened by metastatic disease generates acute, debilitating pain and immediate loss of function. Unlike traumatic fractures, these will not heal with simple immobilization. Surgical stabilization is emergent to control pain, facilitate nursing care, and mobilize the patient.
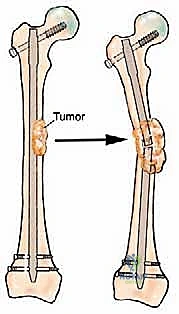
Prophylactic intervention for an impending pathological fracture is where the orthopaedic oncologist can significantly alter the disease trajectory. Identifying and stabilizing these lesions before catastrophic failure prevents an acute crisis, minimizes surgical morbidity, and reduces intraoperative blood loss. The classic criteria for an impending fracture include a lytic bone lesion measuring greater than 2.5 cm, circumferential destruction of 50% or more of the adjacent cortical bone, or an avulsion fracture of the lesser trochanter. The Mirels' scoring system is frequently utilized, assigning points based on the site of the lesion, the nature of the pain, the radiographic appearance (lytic vs. blastic), and the size of the defect. A Mirels' score of 9 or greater strongly dictates prophylactic fixation.
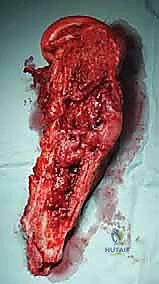
Intractable pain is another critical indication. When localized, progressive disease causes unremitting pain that is refractory to narcotic analgesia and preoperative radiation therapy, surgical intervention for local tumor debulking and mechanical stabilization becomes a vital palliative measure. Furthermore, in highly selected cases—such as a solitary metastasis from renal cell carcinoma or thyroid carcinoma presenting years after the primary excision—wide en bloc resection of the metastasis may confer a distinct survival advantage. However, this is the exception rather than the rule and requires rigorous multidisciplinary tumor board consensus.
| Indication / Contraindication Category | Specific Criteria | Clinical Rationale |
|---|---|---|
| Primary Surgical Indications | Existing Pathological Fracture | Immediate pain control, restoration of mobility, facilitation of nursing care. |
| Impending Pathological Fracture (Mirels >8) | Prevention of catastrophic failure, lower morbidity than treating a completed fracture. | |
| Intractable Pain | Palliation of pain unresponsive to XRT and high-dose narcotics. | |
| Solitary Metastasis (Selected Tumors) | Potential for extended disease-free survival or cure (e.g., late solitary RCC met). | |
| Absolute Contraindications | Life Expectancy < 4-6 Weeks | Surgical stress outweighs palliative benefit; patient unlikely to rehabilitate. |
| Medically Unfit for Anesthesia | Severe cardiopulmonary compromise, uncorrectable coagulopathy. | |
| Coma or Bedbound Status (Prior to Fx) | Surgery will not improve functional status or meaningful quality of life. | |
| Relative Contraindications | Active systemic infection | High risk of implant seeding; requires clearance prior to arthroplasty/fixation. |
| Highly radio-responsive tumors (e.g., Myeloma) | If no mechanical instability exists, XRT alone may suffice for pain and local control. |
Patient selection is heavily reliant on accurate prognostication. We generally consider surgical intervention appropriate for patients expected to survive for more than 3 months. Those with a life expectancy of less than 3 months are less likely to benefit from the physiological insult of an extensive operation. They frequently lack the physiological reserve for postoperative rehabilitation. For these patients, compassionate nonoperative approaches, such as splinting, bracing, and optimized pharmacological pain management, are more humane.
Pre-Operative Planning, Templating, and Patient Positioning
The preoperative planning phase is the most critical component of the surgical management of MBD. A meticulously executed surgery can still result in catastrophic failure if the preoperative assessment was inadequate. The surgeon must understand the lesion's precise morphology, its relationship to critical neurovascular structures, the overall skeletal staging, and the presence of any concurrent metastases that dictate simultaneous or staged interventions.
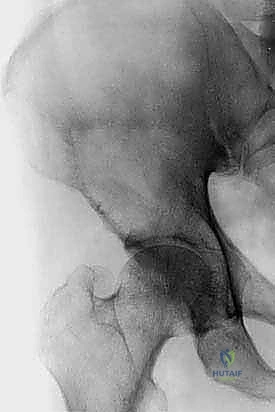
High-quality orthogonal plain radiography of the affected site is the initial step, defining the extent of gross bone destruction. However, plain films drastically underestimate the true extent of intramedullary tumor involvement. Therefore, advanced imaging is mandatory. Computed Tomography (CT) scans are invaluable for assessing cortical integrity, identifying subtle fractures, and evaluating the 3D geometry of periacetabular defects.
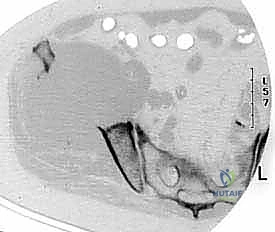
Magnetic Resonance Imaging (MRI) of the entire affected bone is essential to delineate the true longitudinal extent of marrow replacement and to identify skip lesions. Furthermore, MRI is critical for evaluating soft tissue extension, which may dictate the surgical approach or the need for a more extensive resection.
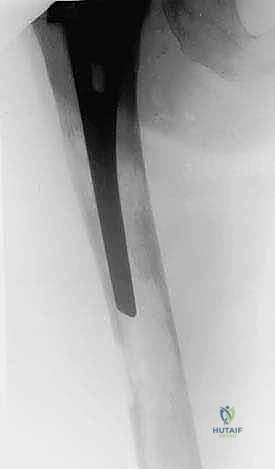
A paramount, non-negotiable rule in orthopaedic oncology is obtaining whole-bone imaging prior to any fixation. If a metastasis is identified in the proximal femur, the entire femur must be imaged down to the knee. Failure to identify a distal diaphyseal metastasis before placing a short cephalomedullary nail will inevitably lead to a stress riser and a subsequent pathological fracture at the tip of the implant, necessitating a massive revision surgery.
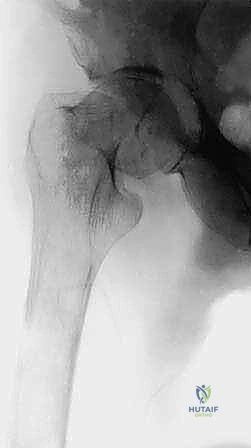
Systemic staging is equally critical. A total body technetium-99m bone scan provides a skeletal roadmap, identifying clinically silent metastases. However, surgeons must be acutely aware that highly aggressive, purely lytic tumors—such as multiple myeloma, renal cell carcinoma, and thyroid carcinoma—may not elicit an osteoblastic response and can present as "cold" or false-negative on a bone scan. In these cases, a PET-CT or whole-body MRI is a more reliable staging modality.
Laboratory evaluation must include a complete blood count, comprehensive metabolic panel, and coagulation profile. Hypercalcemia of malignancy is a frequent and potentially life-threatening complication that must be aggressively managed with hydration and bisphosphonates prior to anesthesia. Furthermore, tumors such as renal cell carcinoma and thyroid carcinoma are notoriously hypervascular. Preoperative angiographic embolization of these lesions 24 to 48 hours prior to surgery is strongly recommended to prevent massive, life-threatening intraoperative hemorrhage.
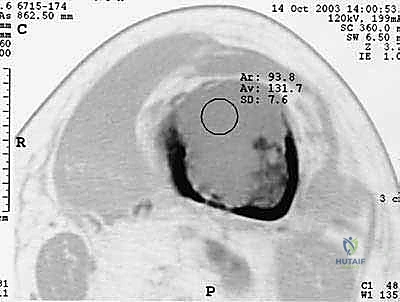
Patient positioning is dictated by the planned approach but must accommodate the fragility of the patient's skeleton. Careful padding and avoidance of excessive torque during positioning are vital to prevent iatrogenic fractures of uninvolved but osteopenic limbs. When utilizing a fracture table for femoral nailing, traction must be applied judiciously, as the weakened bone can easily fail under standard traction loads.
Step-by-Step Surgical Approach and Fixation Technique
The fundamental tenet of surgical intervention for MBD is to achieve immediate, rigid fixation that spans the entire bone, bypassing the area of weakness and protecting the remainder of the bone from future metastatic failure. The choice between osteosynthesis (intramedullary nailing or plating) and endoprosthetic reconstruction depends on the anatomical location, the extent of joint destruction, and the tumor's biology.
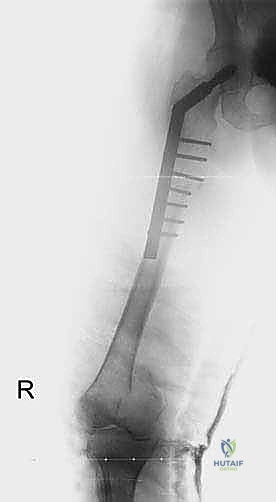
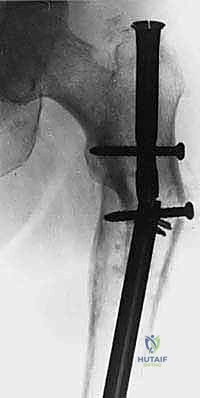
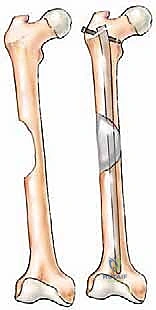
For diaphyseal lesions of the femur, humerus, and tibia, locked intramedullary (IM) nailing is the workhorse technique. IM nails offer superior biomechanical advantages over plates by resting closer to the neutral axis of the bone, thereby experiencing lower bending moments. The technique involves reaming the canal to bypass the tumor burden, which unfortunately risks systemic tumor dissemination, though this rarely alters the overall prognosis in advanced MBD.
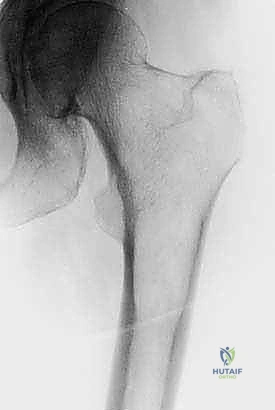
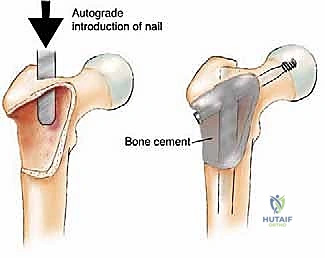
When performing IM nailing for a lytic defect, the nail alone provides torsional and bending stability but does not restore the compressive strength of the missing cortex. Therefore, for large defects (>3 cm or >50% cortical circumference), open intralesional curettage of the tumor and augmentation of the defect with polymethylmethacrylate (PMMA) bone cement is mandatory. The cement acts as a structural grout, filling the void, supporting the cortex, and significantly increasing the construct's fatigue life.
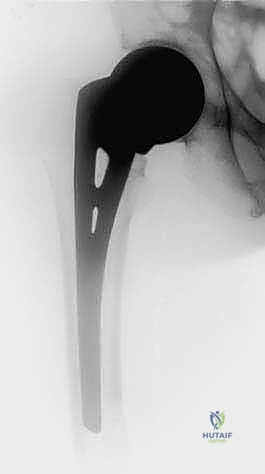
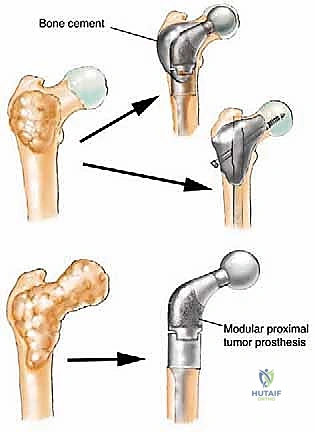
In the proximal femur, the approach varies based on the lesion's exact location. For intertrochanteric or subtrochanteric lesions with an intact femoral head and neck, a long cephalomedullary nail is preferred. The nail must span the entire femur and lock into the distal condyles. If the lesion involves the femoral neck or causes massive destruction of the trochanteric region, osteosynthesis will fail. In these scenarios, resection of the proximal femur and reconstruction with a cemented long-stem bipolar hemiarthroplasty or a modular proximal femoral replacement (megaprosthesis) is required.
Acetabular reconstruction is technically demanding. The approach typically involves a posterior or extensile iliofemoral exposure to visualize the entire column. The tumor is aggressively curetted until bleeding host bone is encountered. The resulting cavitary defect is then filled with PMMA. To transmit forces from the hip joint to the intact pelvis, threaded Steinmann pins (Harrington pins) are driven from the remaining acetabular roof into the dense bone of the sacroiliac joint and sciatic buttress. The PMMA is then molded around these pins, and an anti-protrusio cage and cemented polyethylene cup are impacted into the construct before the cement polymerizes.
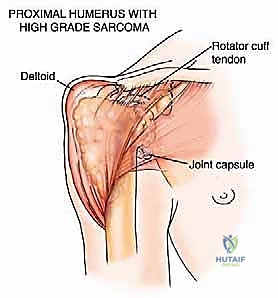
For upper extremity lesions, particularly the proximal humerus, cemented hemiarthroplasty or reverse total shoulder arthroplasty utilizing long-stem components is highly effective. If the deltoid and rotator cuff are compromised by tumor, a reverse geometry prosthesis provides superior functional outcomes by relying on the deltoid for elevation. Diaphyseal humeral lesions are excellently managed with locked antegrade or retrograde intramedullary nails, again utilizing PMMA augmentation for large structural voids.
Meticulous hemostasis is a critical surgical step. The use of tranexamic acid (TXA), bipolar electrocautery, and topical hemostatic agents is routine. Following curettage, the cavity can be packed with hydrogen peroxide-soaked sponges to induce chemical hemostasis and cause thermal necrosis of residual microscopic tumor cells before cementation.
Complications, Incidence Rates, and Salvage Management
Despite meticulous planning and execution, surgical intervention for MBD carries a high complication profile due to the patients' systemic frailty, immunocompromised state, and the aggressive nature of the underlying disease. The surgeon must be prepared to anticipate, recognize, and aggressively manage these complications.
Intraoperative complications can be acute and life-threatening. Massive hemorrhage is a primary concern, particularly during intralesional curettage of vascular tumors. If preoperative embolization was not performed, the surgeon must be prepared for rapid blood loss requiring massive transfusion protocols. Another severe intraoperative risk is Bone Cement Implantation Syndrome (BCIS) or fat embolism. Pressurizing PMMA or reaming the intramedullary canal forces marrow contents, fat, and tumor cells into the venous circulation, potentially causing acute right heart failure, hypoxia, and cardiovascular collapse. To mitigate this, the canal must be thoroughly lavaged and vented prior to cementation or nail insertion.
Postoperative complications include venous thromboembolism (VTE), surgical site infection, and implant failure. Cancer patients represent a hypercoagulable population; thus, aggressive mechanical and pharmacological DVT prophylaxis is mandatory unless absolutely contraindicated by bleeding risks. Infection rates in MBD surgery are significantly higher than in primary arthroplasty, ranging from 5% to 15%, driven by malnutrition, prior radiation, and chemotherapy-induced neutropenia.
Implant failure or disease progression leading to secondary fractures is the ultimate mechanical complication. If a construct fails, it is usually because the implant was too short, allowing disease progression at the tip, or because biological healing failed to occur, leading to eventual fatigue failure of the metal. Salvage management in these scenarios is exceptionally complex, often requiring removal of the broken hardware, massive endoprosthetic reconstruction, or, in extreme cases, amputation for local control and pain relief.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage / Management Strategy |
|---|---|---|---|
| Intraoperative Hemorrhage | 5 - 10% | Renal/Thyroid primary, lack of pre-op embolization. | Massive transfusion, rapid packing, PMMA packing for hemostasis. |
| Fat/Tumor Embolism (BCIS) | 1 - 3% | IM reaming, pressurizing cement in a closed canal. | Lavage/venting canal. 100% O2, vasopressors, supportive critical care. |
| Surgical Site Infection (SSI) | 5 - 15% | Radiation, malnutrition, neutropenia, prolonged OR time. | Aggressive I&D, suppressive antibiotics. Rarely, implant removal/amputation. |
| Implant Fatigue Failure | 3 - 8% | Patient outlives implant, lack of bone healing, short construct. | Revision to longer IM nail, structural PMMA augmentation, or Megaprosthesis. |
| VTE (DVT/PE) | 10 - 20% | Hypercoagulable state of malignancy, immobility. | LMWH or DOACs post-op, IVC filter if anticoagulation contraindicated. |
Phased Post-Operative Rehabilitation Protocols
The entire philosophy of MBD surgery is predicated on the ability to mobilize the patient immediately. Therefore, the post-operative rehabilitation protocol must be aggressive and initiated on post-operative day zero or one. The construct must be designed to withstand immediate, full weight-bearing as tolerated (WBAT). Restricting weight-bearing in a patient with a limited life expectancy defeats the primary purpose of the palliative intervention.
Phase I (Inpatient, Days 0-3) focuses on pain control, DVT prophylaxis, and immediate
Clinical & Radiographic Imaging Archive