Surgical Excision of Hand Vascular Tumors: An Intraoperative Masterclass
Key Takeaway
This masterclass provides a comprehensive, immersive guide to the surgical management of hand vascular tumors. We'll meticulously cover preoperative planning, precise patient positioning, and a granular, real-time breakdown of intraoperative execution. Fellows will learn critical surgical anatomy, instrument techniques, and how to navigate complex dissections. Extensive pearls, pitfalls, and postoperative protocols ensure thorough understanding for managing these diverse and challenging lesions.
Comprehensive Introduction and Patho-Epidemiology
Vascular lesions of the hand and upper extremity represent a profoundly diverse and often treacherous spectrum of pathology, demanding a rigorous understanding of both their embryological origins and their unpredictable natural histories. Comprising roughly 2% to 6% of all primary tumors, a significant proportion—approximately 26%—localize to the upper extremity, with a predilection for the hand and forearm. The fundamental paradigm shift in understanding these lesions originated with the biological classification introduced by Mulliken and Glowacki, later adopted and refined by the International Society for the Study of Vascular Anomalies (ISSVA). This framework strictly dichotomizes these lesions into two distinct pathophysiological entities: vascular tumors and vascular malformations. Conflating the two is a classic pitfall that leads to disastrous surgical decision-making.
Vascular tumors are defined by robust cellular proliferation, characterized by a high endothelial cell turnover rate. Hemangiomas remain the archetypal example. These lesions typically exhibit a dramatic, rapid proliferative phase during early infancy, followed by a plateau commensurate with the child's somatic growth, and ultimately, a prolonged phase of spontaneous involution. Histologically, they are densely packed with plump, proliferating endothelial cells and reliably stain positive for GLUT-1. Because approximately 50% of hemangiomas involute by age five, and 70% by age seven, the primary treatment modality is often disciplined observation. However, in the hand and digits, ulceration occurs in nearly 30% of cases, potentially precipitating chronic paronychia or deep space infections, particularly in pediatric patients prone to digit sucking. Hemangiomas are further subclassified into capillary (57%), cavernous (23%), and sclerosing (10%) variants. A critical, life-threatening permutation is the Kasabach-Merritt phenomenon, wherein a rapidly enlarging vascular tumor traps platelets, inducing a profound consumptive coagulopathy and thrombocytopenia that demands immediate, multidisciplinary medical intervention prior to any surgical consideration.
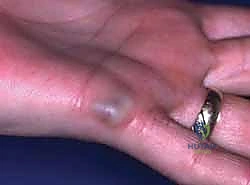
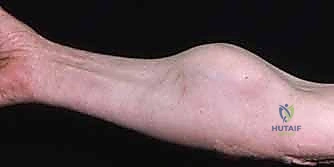
Conversely, vascular malformations are innate structural anomalies of capillary, venous, lymphatic, or arterial morphogenesis. They are present at birth—though they may not manifest clinically until hormonal triggers or trauma unmask them later in life—and they grow proportionally with the patient. Crucially, they exhibit a normal endothelial turnover rate and never spontaneously involute. We categorize them hemodynamically into low-flow and high-flow lesions. Low-flow malformations, encompassing capillary (port-wine stains), venous, and lymphatic anomalies, are the most prevalent. Venous malformations, the most common overall (40%), present as compressible, bluish masses that swell with dependency; the presence of phleboliths on plain radiography is pathognomonic. Lymphangiomas, though rarer, can infiltrate deep fascial planes, causing massive fluid accumulation, recurrent cellulitis, and secondary skeletal hypertrophy.
High-flow malformations are driven by arterial components, forming arteriovenous fistulas (AVFs) or complex arteriovenous malformations (AVMs). These are non-compressible, pulsatile masses that pose severe systemic risks, including distal ischemia (vascular steal syndrome) and high-output congestive heart failure. They are staged clinically (e.g., Schobinger staging) and anatomically. Type A lesions involve single AVFs; Type B lesions feature localized micro- or macrofistulas; and Type C lesions are diffuse, devastating anomalies infiltrating all tissue planes of the limb, carrying a high risk of compartment syndrome, ulceration, and ultimate limb loss. Congenital AVFs are often syndromic, associated with Parkes-Weber or Klippel-Trenaunay syndromes. Acquired lesions, such as true aneurysms (e.g., hypothenar hammer syndrome affecting the ulnar artery) and false aneurysms (pseudoaneurysms, comprising 83% of hand aneurysms), typically arise from blunt trauma or penetrating injuries. Glomus tumors, highly specialized neuromyoarterial mechanoreceptors regulating thermoregulation, account for 8% of vascular hand tumors. Finally, malignant vascular tumors—including angiosarcoma, hemangioendothelioma, and glomangiosarcoma—comprise less than 1% of upper extremity vascular lesions but require aggressive, wide-margin oncologic resection due to their high metastatic potential.
Detailed Surgical Anatomy and Biomechanics
Before initiating any surgical dissection, the operative surgeon must possess an encyclopedic mastery of the hand's intricate vascular architecture. The hand is a marvel of redundant but highly specific neurovascular networks, and the resection of vascular tumors often requires navigating distorted, hypertrophic, or aberrant anatomy. The primary arterial inflow is governed by the radial and ulnar arteries, which anastomose to form the superficial and deep palmar arches. The ulnar artery enters the hand volar to the flexor retinaculum via Guyon's canal, supplying the superficial palmar arch, which is typically the dominant and more complete arch. This arch gives rise to the common palmar digital arteries. The radial artery courses through the anatomical snuffbox, piercing the first dorsal interosseous muscle to form the deep palmar arch, which lies directly upon the metacarpal bases and provides critical collaterals to the digital circulation.
The digital vasculature is highly unforgiving. The common digital arteries bifurcate at the web spaces into the proper digital arteries, which travel along the midlateral aspect of each digit, slightly volar to the true midline. These vessels are intimately bound to the proper digital nerves within the neurovascular bundle. This bundle is tethered by Cleland's ligaments dorsally and Grayson's ligaments volarly. When excising an infiltrating venous malformation or a deep hemangioma, these fascial septa must be meticulously released to mobilize the neurovascular bundle without inducing traction neurapraxia or devascularizing the digit. Anatomical variations in the palmar arches are the rule rather than the exception; a pre-operative Allen test and intraoperative Doppler assessment are mandatory to confirm adequate collateral flow before ligating any feeding vessel.
The venous and lymphatic drainage systems, while less rigidly defined, are equally critical, particularly when addressing low-flow malformations. The superficial venous system, culminating in the dorsal venous arch, is frequently the epicenter of superficial venous malformations. The deep venous system consists of venae comitantes that parallel the arterial arches. Lymphatic channels densely populate the volar aspect of the digits and palm, draining dorsally. Disruption of these networks during wide excision can lead to intractable postoperative lymphedema, severely compromising digital flexion and overall hand biomechanics.
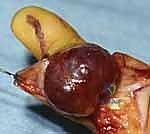
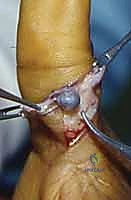
A specialized anatomical focus is required for the glomus body. This normal physiological structure is an arteriovenous shunt located primarily in the stratum reticulare of the dermis, highly concentrated in the subungual region of the distal phalanx. It consists of an afferent arteriole, the Sucquet-Hoyer canal (lined by cuboidal glomus cells), and an efferent venule. When these cells undergo hamartomatous proliferation, they form a glomus tumor. Because they are encased within the rigid confines of the distal phalangeal tuft and the nail plate, even microscopic tumors (<3 mm) generate excruciating, disproportionate pain due to localized pressure on the rich somatic nerve endings. Surgical access requires a flawless understanding of the nail bed (sterile and germinal matrix) to prevent permanent nail dystrophy during extirpation.
Exhaustive Indications and Contraindications
The decision to operate on a vascular lesion of the hand requires a nuanced calculus balancing the natural history of the specific lesion against the functional risks of surgical morbidity. Observation is the gold standard for infantile hemangiomas, as the vast majority will spontaneously involute. However, surgical intervention becomes an absolute indication in the presence of life-threatening complications, such as the Kasabach-Merritt phenomenon, where the lesion induces a profound consumptive coagulopathy. Similarly, any lesion exhibiting rapid, destructive growth, intractable ulceration with secondary deep-space infection, or biopsy-proven malignancy (e.g., angiosarcoma, epithelioid hemangioendothelioma) mandates immediate and aggressive surgical extirpation.
Relative indications for surgery are more common and revolve around the preservation of hand function and the alleviation of debilitating symptoms. Glomus tumors, characterized by the classic triad of severe pain, pinpoint point tenderness, and cold hypersensitivity, are a primary indication for excision, as the pain severely limits hand utilization. For vascular malformations (venous or lymphatic), surgery is indicated when the mass effect causes mechanical restriction of joint motion, compression neuropathies (e.g., carpal tunnel syndrome secondary to a palmar arch malformation), or recurrent thrombophlebitis. High-flow AVMs present a unique challenge; surgery is indicated for localized (Type A or B) lesions causing ischemic steal syndrome or high-output cardiac states, but only after precise angiographic mapping and, ideally, preoperative embolization.
Contraindications to surgical excision are primarily dictated by the anatomical extent of the lesion and the patient's systemic health. Diffuse, infiltrative high-flow AVMs (Type C) that permeate multiple tissue planes, including muscle and bone, represent an absolute contraindication to simple excision. Attempting partial resection in these cases inevitably leads to massive hemorrhage, rapid recurrence, and exacerbation of the collateral blood supply, often culminating in the need for limb amputation. In such scenarios, palliative embolization or systemic pharmacotherapy (e.g., mTOR inhibitors like sirolimus) are the preferred primary modalities. Furthermore, surgery is contraindicated in patients with severe, uncorrected coagulopathies or those who cannot tolerate the prolonged anesthesia required for complex microvascular reconstruction.
Indications and Contraindications Table
| Category | Indications for Surgical Intervention | Contraindications to Surgical Intervention |
|---|---|---|
| Vascular Tumors | Kasabach-Merritt phenomenon; intractable ulceration/infection; malignant transformation; failure to involute with functional deficit. | Uncomplicated infantile hemangiomas in the proliferative/early involution phase; medically unstable pediatric patients. |
| Low-Flow Malformations | Severe pain/recurrent thrombosis; mechanical restriction of joint ROM; compression neuropathy; significant cosmetic deformity affecting psychosocial development. | Diffuse intramuscular venous malformations where complete excision would destroy motor function; asymptomatic lesions. |
| High-Flow Malformations | Localized Type A/B lesions; ischemic steal syndrome with impending tissue loss; high-output cardiac failure; localized aneurysms. | Diffuse Type C AVMs involving the entire limb; lack of preoperative angiographic mapping; inability to secure proximal vascular control. |
| Acquired/Special | Glomus tumors (classic pain triad); symptomatic true/false aneurysms (e.g., Hypothenar Hammer Syndrome) causing distal ischemia. | Asymptomatic, stable pseudoaneurysms in non-critical areas (rare in the hand); active untreated local infection. |
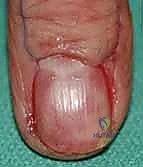
Pre-Operative Planning, Templating, and Patient Positioning
Every successful vascular tumor excision is won or lost in the preoperative planning phase. The clinical assessment must be exhaustive. A detailed history should elucidate the timeline of growth, exacerbating factors (e.g., dependency, hormonal changes, cold exposure), and any history of antecedent trauma. Physical examination requires meticulous inspection for color changes, palpable thrills, or audible bruits indicative of high-flow fistulas. The Allen test is mandatory to assess the patency of the palmar arches. For suspected glomus tumors, Love’s pin test (pinpoint tenderness localized with a paperclip) and Hildreth’s test (relief of pain with proximal tourniquet inflation) are highly specific clinical diagnostic tools.

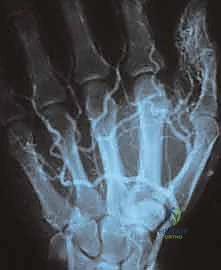
Imaging is the cornerstone of surgical templating. High-resolution Duplex Ultrasound is the ideal initial modality, differentiating cystic from solid structures and providing real-time hemodynamic data (arterial vs. venous waveforms). Magnetic Resonance Imaging (MRI) with and without intravenous gadolinium contrast is the gold standard for defining the anatomical extent of the lesion. Vascular tumors typically demonstrate intermediate signal on T1, hyperintensity on T2, and robust, homogeneous enhancement. Venous malformations show high T2 signal with characteristic signal voids representing phleboliths. For high-flow AVMs or aneurysms, Magnetic Resonance Angiography (MRA) or conventional catheter-directed Digital Subtraction Angiography (DSA) is essential to map the feeding arteries and draining veins. DSA also offers a therapeutic window; preoperative embolization of feeding vessels 24 to 48 hours prior to surgery can dramatically reduce intraoperative blood loss and shrink the nidus, making excision safer.
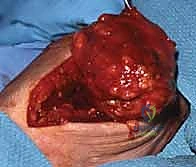
Patient positioning and tourniquet management require specific, non-standard protocols for vascular lesions. The patient is positioned supine with the operative arm extended on a radiolucent hand table. Crucially, the standard technique of exsanguinating the limb with an Esmarch bandage prior to tourniquet inflation is absolutely contraindicated for venous malformations and hemangiomas. Exsanguination empties the abnormal vascular channels, rendering the lesion flat, colorless, and visually indistinguishable from surrounding normal tissue, virtually guaranteeing an incomplete resection. Instead, the arm should be elevated for 60 seconds to allow gravity drainage of normal venous return, followed by rapid inflation of the pneumatic tourniquet to 250 mmHg. This traps blood within the malformation, keeping it engorged and clearly demarcated during dissection.
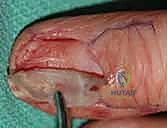
For glomus tumors, we plan a transungual or periungual approach depending on the exact subungual location mapped on MRI. The surgical field must be prepared for potential microsurgical reconstruction. Loupe magnification (minimum 3.5x) is mandatory for all cases, and an operating microscope should be on standby for digital artery repair or nerve neurolysis. The surgical template should utilize extensile incisions (Bruner zig-zag or mid-lateral) to allow broad exposure of the neurovascular bundles proximal and distal to the pathology, ensuring proximal control before entering the tumor bed.
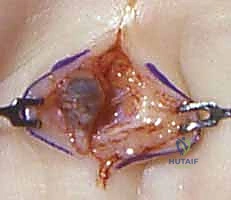
Step-by-Step Surgical Approach and Fixation Technique
The surgical approach to a vascular tumor is a masterclass in meticulous, bloodless micro-dissection. Once the tourniquet is inflated (using the gravity-elevation technique described above), the skin incision is made. For palmar or digital lesions, Bruner incisions are preferred to prevent flexion contractures. The skin flaps are elevated full-thickness, preserving the subdermal vascular plexus. The fundamental principle of this dissection is to identify normal, healthy anatomy proximal and distal to the lesion before attempting to dissect the tumor itself. The proper digital nerves and arteries must be identified in virgin tissue and traced into the zone of pathology.
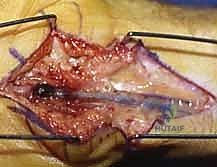
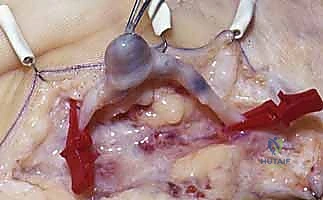
When excising a venous malformation or cavernous hemangioma, the lesion will appear as a spongy, purplish mass intimately intertwined with the surrounding fascia, tendon sheaths, or neurovascular bundles. Bipolar electrocautery is the instrument of choice; monopolar cautery risks thermal injury to adjacent nerves. The dissection proceeds circumferentially, utilizing a "peanut" sponge or fine tenotomy scissors to gently sweep normal tissue away from the pseudocapsule of the malformation. Feeding vessels are meticulously skeletonized, ligated with fine silk or metallic clips, and divided. If the malformation infiltrates a digital nerve (a common scenario), an operating microscope is brought into the field, and an epineurotomy is performed to dissect the vascular channels away from the nerve fascicles, accepting a subtotal resection if total excision would result in permanent anesthesia.
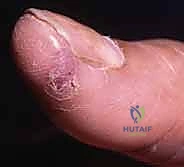
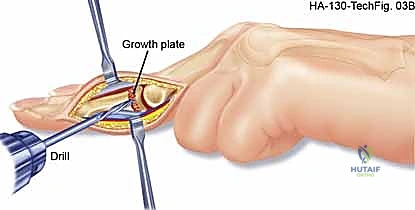
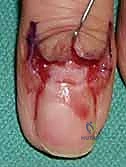
For subungual glomus tumors, the approach requires precision to prevent nail deformity. A digital block is performed, and a finger tourniquet is applied. The nail plate is carefully elevated and removed (or partially removed) using a Freer elevator. The sterile matrix is incised longitudinally directly over the bluish hue of the tumor. The tumor, typically a 2-5 mm encapsulated, pearl-like structure, is gently shelled out using a curette or fine iris scissors. It is imperative to inspect the bony floor of the distal phalanx for erosions and curette any residual cells. The nail bed is then meticulously repaired using 6-0 or 7-0 absorbable sutures (e.g., chromic gut) under magnification. The native nail plate, if intact, is fenestrated and replaced as a biological splint, tucked under the eponychial fold to prevent synechiae.
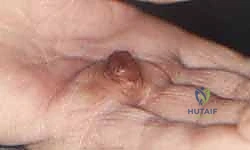
The management of acquired aneurysms (e.g., ulnar artery aneurysm in hypothenar hammer syndrome) or localized AVMs demands vascular reconstructive capabilities. Proximal and distal vascular control is obtained using vessel loops. The aneurysm is mobilized, and the ulnar nerve is carefully protected. If the Allen test confirms excellent radial collateral flow, simple ligation and excision of the aneurysm may be performed. However, to prevent cold intolerance or claudication, vascular reconstruction is preferred. The diseased arterial segment is resected until healthy intima is visualized. If a primary end-to-end anastomosis cannot be achieved without tension, a reversed interposition vein graft (typically harvested from the distal forearm or dorsal foot) is utilized. The anastomosis is performed under the microscope using 8-0 or 9-0 nylon sutures, and patency is confirmed intraoperatively with a micro-Doppler probe and the Acland strip test prior to closure.

Complications, Incidence Rates, and Salvage Management
Surgical intervention for hand vascular tumors carries a unique and formidable complication profile, primarily due to the dense concentration of critical structures within a confined space. Intraoperative hemorrhage is the most immediate threat, particularly when addressing high-flow AVMs without adequate proximal control or prior embolization. Inadvertent transection of a proper digital nerve or artery can occur if the lesion’s infiltrative nature distorts the normal anatomical planes. If an artery is compromised and the digit becomes ischemic upon tourniquet deflation, immediate microvascular repair or vein grafting is mandatory. Nerve injuries must be recognized and repaired primarily with epineurial sutures to prevent painful neuroma formation.
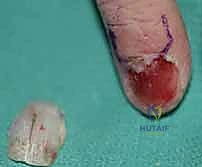
Early postoperative complications include hematoma formation, which can act as a space-occupying lesion, compromising flap viability or inducing a localized compartment syndrome. Meticulous hemostasis and the judicious use of closed suction drains in larger resections mitigate this risk. Wound dehiscence and flap necrosis may occur if the subdermal plexus is devascularized during superficial dissections. Late complications are often the most challenging. Recurrence is notoriously high for diffuse venous malformations and AVMs, sometimes exceeding 30-50% depending on the completeness of the initial excision. Recurrent glomus tumors (up to 15% incidence) are usually due to incomplete primary excision or the presence of synchronous, unrecognized satellite lesions.

Complex Regional Pain Syndrome (CRPS) and profound cold intolerance are devastating sequelae, particularly following extensive dissection around the palmar arches or digital nerves. Salvage management for recurrent or unresectable lesions increasingly relies on interventional radiology. Targeted sclerotherapy (using agents like sodium tetradecyl sulfate or bleomycin) is highly effective for recurrent venous malformations. For recurrent high-flow AVMs, serial transcatheter embolization is the mainstay of salvage therapy. In cases of intractable CRPS, aggressive occupational therapy, stellate ganglion blocks, or surgical sympathectomy may be required.
Complications, Incidence, and Salvage Management Table
| Complication | Estimated Incidence | Prevention & Salvage Management Strategy |
|---|---|---|
| Incomplete Excision / Recurrence | 10-50% (Highest in diffuse venous malformations and AVMs) | Prevention: MRI mapping, loupe magnification, non-exsanguinating tourniquet. Salvage: Re-excision, targeted sclerotherapy, or serial embolization. |
| Digital Nerve Injury / Neuroma | 2-5% | Prevention: Identify normal nerve proximal/distal to lesion first. Salvage: Microsurgical primary repair, nerve grafting, or neuroma excision with burying into muscle/bone. |
| Nail Dystrophy (Glomus Excision) | 5-15% | Prevention: Meticulous repair of sterile matrix with 7-0 absorbable suture. Salvage: Secondary nail bed reconstruction, split-thickness sterile matrix grafting. |
| Ischemic Compromise / Steal | 1-3% (Higher in AVM resections) | Prevention: Pre-op Allen test, intraoperative Doppler, microvascular reconstruction capability. Salvage: Urgent re-exploration, thrombectomy, interposition vein grafting. |
| Complex Regional Pain Syndrome | 2-10% | Prevention: Gentle tissue handling, adequate postoperative pain control, early ROM. Salvage: Aggressive hand therapy, gabapentinoids, sympathetic nerve blocks. |
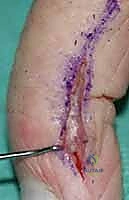
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following vascular tumor excision is heavily dictated by the extent of the soft tissue dissection and whether vascular or neural reconstruction was performed. The overarching goal is to balance the protection of healing tissues with the prevention of debilitating joint contractures and tendon adhesions. Phase I (0-2 weeks) focuses on wound healing and edema control. The hand is immobilized in a bulky, non-compressive soft dressing, typically reinforced with a volar plaster splint placing the wrist in slight extension and the metacarpophalangeal joints in 70 degrees of flexion (intrinsic-plus position). Strict elevation above the level of the heart is mandatory to minimize venous congestion and hematoma formation. Active range of motion (ROM) of the uninvolved digits, elbow, and shoulder is encouraged immediately.
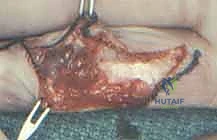
Phase II (2-6 weeks) commences with suture removal and the initiation of targeted hand therapy. If no microvascular reconstruction was performed, the splint is discontinued, and progressive active and active-assisted ROM exercises are instituted. Tendon gliding exercises are critical, particularly if the tumor was excised from the flexor tendon sheath, to prevent restrictive tenosynovitis. If a nerve neurolysis or repair was performed, gentle nerve gliding exercises are introduced to prevent tethering within the scar bed. Scar management begins once the wound is fully epithelialized, utilizing silicone gel sheeting and deep friction massage to soften the surgical bed and reduce hypersensitivity.
Phase III (6-12 weeks and beyond) focuses on functional restoration, strengthening, and desensitization. Patients recovering from glomus tumor excisions often require intensive desensitization protocols (using varied textures and fluidotherapy) due to the hyperalgesia associated with the distal tuft. Work conditioning and grip strengthening are progressively advanced. Long-term surveillance is an integral component of the rehabilitation phase. Patients with a history of venous malformations or AVMs require annual clinical evaluations and potentially follow-up MRI or ultrasound to monitor for asymptomatic recurrence, as early detection of a recurring nidus allows for less invasive salvage interventions, such as localized sclerotherapy.
Summary of Landmark Literature and Clinical Guidelines
The modern surgical management of hand vascular tumors is built upon a foundation of critical literature that has continuously refined our diagnostic and therapeutic paradigms. The most consequential paradigm shift was introduced by Mulliken and Glowacki in 1982, whose biological classification system fundamentally separated vascular tumors (characterized by endothelial hyperplasia) from vascular malformations (characterized by normal endothelium but dysplastic architecture). This work was subsequently codified and expanded by the ISSVA
Clinical & Radiographic Imaging Archive