Total Elbow Arthroplasty for Rheumatoid Arthritis: An Intraoperative Masterclass
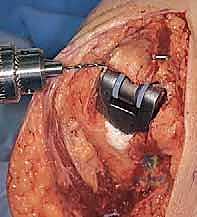
Key Takeaway
Master Total Elbow Arthroplasty for Rheumatoid Arthritis. This immersive guide covers detailed anatomy, precise surgical execution, critical decision-making, and comprehensive post-operative management. Learn from an expert surgeon's perspective, navigating every step from incision to implant, ensuring optimal patient outcomes in complex RA elbow cases.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues, to this definitive intraoperative masterclass on Total Elbow Arthroplasty (TEA) for end-stage Rheumatoid Arthritis (RA). The surgical management of the rheumatoid elbow represents one of the most challenging yet profoundly rewarding interventions in the orthopaedic reconstructive armamentarium. This procedure demands a meticulous understanding of altered biomechanics, compromised soft tissue envelopes, and often severe cavitary bone loss. Our primary objective is the restoration of a pain-free, functionally stable articulation that permits the patient to reclaim independence in their activities of daily living, particularly given the profound disability associated with upper extremity polyarticular disease.
Rheumatoid arthritis is a chronic, systemic, autoimmune inflammatory condition of complex and multifactorial etiology, affecting approximately 1% to 2% of the global population. It exhibits a strong predilection for females, who are affected two to three times more frequently than males, with the incidence typically peaking in the fourth and fifth decades of life. Within this demographic, the elbow joint is clinically involved in 20% to 70% of cases. The disease presents across a wide spectrum of severity, ranging from mild, episodic synovitis to catastrophic, end-stage osteoarticular destruction that renders the upper extremity virtually functionless.
The fundamental histopathologic hallmark of RA is prolonged, aggressive synovitis. This inflammatory cascade is driven by a complex interplay of B lymphocytes, CD4+ T cells, mononuclear phagocytes, and fibroblasts, which collectively produce a deluge of cytokines, including Tumor Necrosis Factor-alpha (TNF-α), Interleukin-1 (IL-1), and Interleukin-6 (IL-6). This results in the formation of a hypertrophic, hypervascular synovial pannus that invades and enzymatically degrades articular cartilage. Subsequent subchondral cyst formation, marginal erosions, and progressive ligamentous attenuation culminate in gross joint instability and the end-stage arthropathy we are addressing today.
Pathogenesis and Natural History
The natural history of the rheumatoid elbow is characterized by a relentless progression from predominantly soft tissue inflammation to catastrophic osseous destruction. Initially, synovial hyperplasia and pannus formation correlate with the classic clinical presentation of a boggy, hyperemic, and exquisitely painful joint with a restricted arc of motion. As the hyperplastic synovia expands, it physically distends the joint capsule and enzymatically degrades the stabilizing ligamentous structures, particularly the medial collateral ligament (MCL) complex and the lateral ulnar collateral ligament (LUCL).
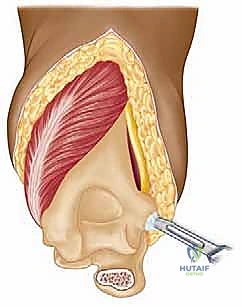
As the disease advances into its later stages, the mechanical integrity of the joint is profoundly compromised. The destruction of the annular ligament and the proximal radioulnar joint often leads to radial head instability, typically manifesting as anterior or anterolateral subluxation. This radial head migration further disrupts the kinematics of the elbow, leading to accelerated wear of the capitellum and the ulnohumeral articulation. The resulting instability is not merely functional but structural, making joint-preserving procedures like synovectomy futile in the late stages.
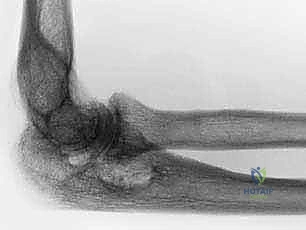
Furthermore, the systemic nature of the inflammatory cascade means that local bone quality is invariably poor. Osteoclastogenesis is heavily upregulated by the RANK/RANKL pathway, which is stimulated by the inflammatory milieu. This results in profound periarticular osteopenia and subchondral cystic changes, complicating implant fixation and increasing the risk of intraoperative and postoperative periprosthetic fractures. Understanding this pathogenesis is critical for the arthroplasty surgeon, as it dictates the necessity for semiconstrained implant designs and meticulous cementation techniques.
Clinical Presentation and Diagnostic Studies
The classic clinical presentation of a patient requiring TEA for RA involves a swollen, tender, and warm elbow with a severely diminished and painful arc of motion. Patients frequently report a progressive decline in upper extremity function, particularly in tasks requiring bringing the hand to the mouth or face, which requires a functional arc of flexion and supination. Constitutional complaints are common, and it is imperative to remember that 90% of these patients exhibit concomitant hand and wrist involvement, while 80% have ipsilateral shoulder pathology, highlighting the polyarticular nature of the disease.
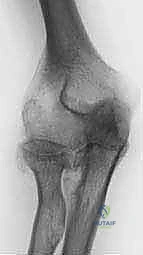
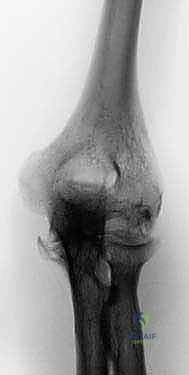
Diagnostic imaging begins with high-quality, orthogonal anteroposterior (AP) and lateral radiographs of the elbow. These are fundamental for assessing the degree of osteopenia, the extent of joint space narrowing, and the presence of subchondral architectural changes. Advanced imaging, such as MRI or CT, is generally reserved for cases where specific soft tissue impingement, subtle periprosthetic fractures (in revision scenarios), or complex cavitary bone defects require three-dimensional quantification prior to surgical intervention.
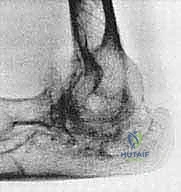
Special consideration must be given to patients with Juvenile Rheumatoid Arthritis (JRA), or Still's disease. JRA patients are typically younger at the time of presentation for arthroplasty, often exhibit profoundly stiff elbows with severe osseous destruction, and have a higher propensity for bony ankylosis. Their medullary canals are frequently hypoplastic, requiring specialized, smaller-diameter implants and meticulous preoperative templating to avoid catastrophic intraoperative diaphyseal fractures.
Mayo Radiographic Classification System
The Mayo Radiographic Classification System remains the gold standard for categorizing the severity of rheumatoid involvement in the elbow. This system is invaluable not only for monitoring longitudinal disease progression but also for correlating radiographic findings with clinical symptomatology and guiding surgical decision-making.
Grade I disease is characterized by clinically evident synovitis in a radiographically normal-appearing joint, save for mild to moderate periarticular osteopenia and impressive soft tissue swelling. Grade II involves early loss of the ulnohumeral and radiocapitellar joint spaces, though the macroscopic subchondral architecture remains largely intact. At these stages, medical management and potentially arthroscopic synovectomy are the mainstays of treatment.
Grade III is marked by a complete loss of joint space. The active synovitis has often "burned out," leaving behind a stiff, fibrotic joint. Grade IIIA indicates maintained bony architecture, while Grade IIIB denotes the onset of significant bone loss and structural deformity. Grade IV represents severe, catastrophic bony destruction with profound functional instability, which is the classic indication for Total Elbow Arthroplasty. Finally, Grade V denotes spontaneous bony ankylosis of the ulnohumeral joint, a phenomenon most frequently observed in the JRA population, requiring complex takedown and reconstruction.
Detailed Surgical Anatomy and Biomechanics
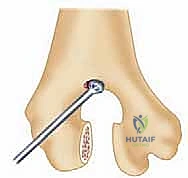
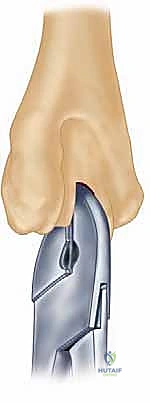
A profound mastery of the osseous architecture and ligamentous constraints of the elbow is the foundation of successful arthroplasty. The elbow is a highly congruent, inherently stable hinge-like joint, but its kinematics are far more complex than a simple uniaxial hinge. The distal humerus features a 30-degree anterior tilt relative to the humeral shaft, which is critical for allowing full terminal flexion without impingement of the coronoid process against the anterior humeral cortex.
The proximal ulna, specifically the greater sigmoid notch, exhibits a reciprocal 30-degree posterior angulation relative to the ulnar diaphysis. This architectural relationship must be meticulously recreated during the preparation of the osseous beds and the implantation of the prosthetic components. Failure to restore the native joint line and these critical angles will result in altered kinematics, restricted range of motion, and accelerated polyethylene wear or component loosening.
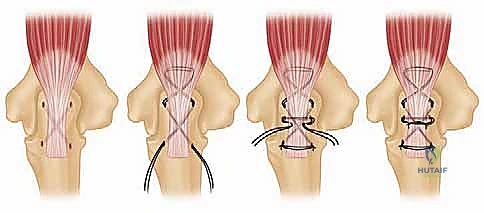
The ligamentous constraints, primarily the MCL and LCL complexes, dictate the stability of the native joint. In the rheumatoid elbow, these structures are universally attenuated or entirely incompetent. The MCL, specifically its anterior bundle, is the primary restraint to valgus stress, while the LUCL restrains posterolateral rotatory instability. Because these soft-tissue stabilizers cannot be relied upon in the RA patient, the arthroplasty surgeon must depend on the intrinsic stability of the implant design to prevent postoperative dislocation.
Kinematics and Biomechanical Considerations in Arthroplasty
The kinematics of the human elbow involve a complex, moving instantaneous center of rotation (ICOR). During the arc of flexion and extension, the axis of rotation shifts slightly, accompanied by a subtle degree of axial rotation and varus-valgus toggle. Early, rigidly constrained hinge implants failed catastrophically because they did not permit this coupled motion, leading to massive stress transfer to the cement-bone interface and rapid aseptic loosening.
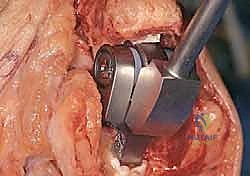
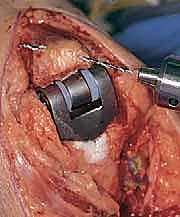
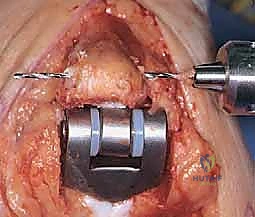
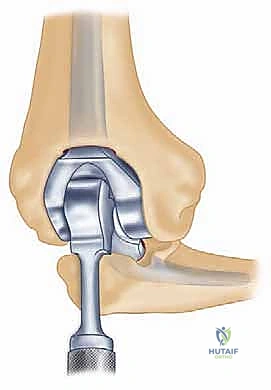
Modern semiconstrained (linked) implants were developed to address this biomechanical reality. These devices feature a "sloppy hinge" mechanism—typically a pin and locking ring assembly—that permits approximately 7 degrees of varus-valgus play and 7 degrees of axial rotation. This design brilliantly dissipates forces away from the intramedullary stems and into the surrounding soft tissue envelope, drastically reducing the incidence of implant loosening while providing the necessary stability for a ligamentously incompetent rheumatoid elbow.
Unconstrained (unlinked) implants, which rely entirely on the native soft tissues for stability, are generally contraindicated in the severe rheumatoid elbow. The compressive forces across the joint during normal activities of daily living can reach up to three times body weight. Without competent collateral ligaments to guide these forces and maintain articular congruity, unlinked components in an RA patient will inevitably subluxate or dislocate, leading to catastrophic failure.
Neurovascular Topography Relevant to Exposure
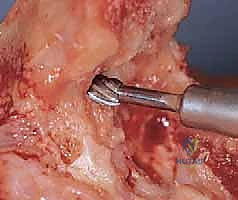
The surgical approach to the elbow places several critical neurovascular structures at risk, demanding meticulous anatomical awareness. The ulnar nerve is the most vulnerable structure during the posterior approach. It courses through the cubital tunnel, posterior to the medial epicondyle, and is tethered by the arcuate ligament of Osborne. In the rheumatoid patient, the nerve is often encased in inflammatory tissue and may be subluxating, making careful, early identification and neurolysis imperative.
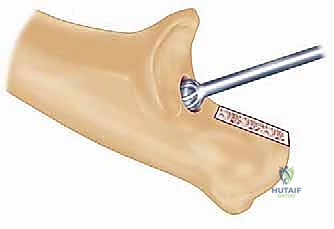
The radial nerve, while not routinely exposed during a standard primary TEA, is at significant risk during the preparation of the humeral medullary canal and the placement of the humeral component. The nerve spirals around the posterior humerus in the spiral groove, piercing the lateral intermuscular septum approximately 10 cm proximal to the radiocapitellar joint. Overzealous reaming, cement extravasation, or cortical perforation laterally can result in devastating radial nerve palsy.
The median nerve and brachial artery lie anterior to the brachialis muscle. While protected by this muscular barrier during a posterior approach, they are vulnerable to injury from retractor placement over the anterior capsule, particularly when excising the radial head or performing an anterior capsulectomy. Furthermore, heterotopic ossification or severe anterior osteophytes can displace these structures, requiring extreme caution during anterior osseous resections.
Exhaustive Indications and Contraindications
The decision to proceed with Total Elbow Arthroplasty in the rheumatoid patient is a complex calculus weighing the severity of symptoms against the functional demands of the patient and the inherent risks of the procedure. The primary indication is advanced (Mayo Grade III or IV) rheumatoid arthritis characterized by intractable pain, severe restriction in range of motion, and profound functional instability that has proven refractory to comprehensive medical management and joint-preserving surgical interventions.
In the modern era of highly effective biologic disease-modifying antirheumatic drugs (DMARDs), the incidence of patients progressing to catastrophic Grade IV destruction has decreased. However, for those who do progress, or for those who present with neglected disease, TEA remains a transformative procedure. It is particularly indicated for older, low-demand patients who require a functional upper extremity for basic activities of daily living, such as personal hygiene and feeding.
It is crucial to emphasize that TEA is a salvage procedure with strict postoperative limitations. Patients must be willing and able to adhere to a lifetime lifting restriction, typically capped at 5 to 10 pounds (2.2 to 4.5 kg) for a single event, and 1 to 2 pounds for repetitive lifting. Failure to adhere to these restrictions will inevitably lead to accelerated polyethylene wear, aseptic loosening, and catastrophic mechanical failure of the prosthesis.
Absolute and Relative Contraindications
Patient selection is arguably the most critical determinant of long-term success in TEA. The surgeon must rigorously evaluate the patient for both absolute and relative contraindications to avoid devastating complications.
| Contraindication Category | Specific Condition | Clinical Rationale |
|---|---|---|
| Absolute | Active local or systemic infection | High risk of immediate periprosthetic joint infection (PJI), which is exceptionally difficult to eradicate in the elbow. |
| Absolute | Upper extremity paralysis / Flail arm | Lack of motor control will lead to immediate instability and inability to utilize the reconstructed joint. |
| Absolute | Inability to comply with restrictions | Non-compliance with the 5 lb lifetime lifting restriction ensures premature mechanical failure and aseptic loosening. |
| Relative | Remote history of infection | Requires meticulous preoperative workup (ESR, CRP, aspiration) to ensure complete eradication prior to implantation. |
| Relative | Severe soft tissue compromise | Inadequate soft tissue coverage increases the risk of wound breakdown, hardware exposure, and subsequent deep infection. |
| Relative | Young age / High physical demand | High-demand activities will lead to rapid implant failure. Alternative salvage procedures (e.g., arthrodesis) may be considered, though poorly tolerated in the upper extremity. |
Differential Diagnosis and Medical Optimization
While the diagnosis of end-stage RA is often clear in the context of polyarticular disease, the surgeon must maintain a broad differential diagnosis. Conditions such as Calcium Pyrophosphate Deposition Disease (CPPD), advanced primary Osteoarthritis, Psoriatic Arthritis, and Systemic Lupus Erythematosus can present with similar degrees of joint destruction. Differentiating these is crucial, as the systemic management and perioperative optimization strategies differ significantly.
Medical optimization is a multidisciplinary effort requiring close collaboration with the patient's rheumatologist. The perioperative management of DMARDs and biologic agents is a delicate balance between preventing disease flare-ups and minimizing the risk of surgical site infection. Traditional DMARDs like Methotrexate are often continued perioperatively, as studies have shown no significant increase in infection risk.
However, biologic agents, particularly TNF-α inhibitors (e.g., Infliximab, Etanercept), profoundly suppress the immune system and are associated with a marked increase in postoperative PJI. Current guidelines strongly recommend withholding these agents for at least one to two dosing cycles prior to surgery, and resuming them only after complete wound healing has occurred, typically 14 to 21 days postoperatively. Furthermore, patients on chronic corticosteroid therapy require stress-dose steroids intraoperatively to prevent acute adrenal crisis.
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the blueprint for a successful Total Elbow Arthroplasty. The margin for error in the elbow is exceedingly small, and failure to anticipate anatomical anomalies or bone stock deficiencies will lead to intraoperative disasters. The planning phase begins with a comprehensive radiographic assessment and the use of digital templating software to estimate implant sizing and alignment.
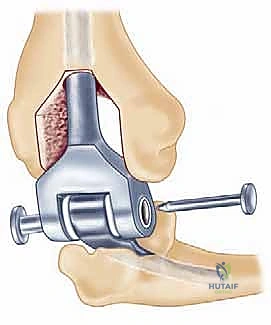
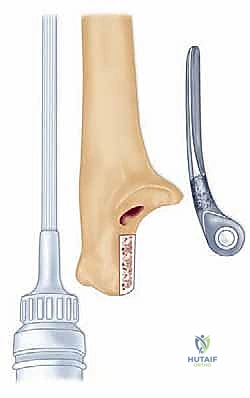
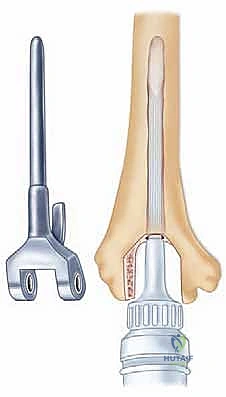
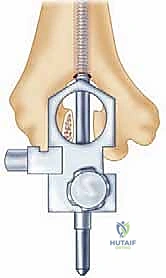
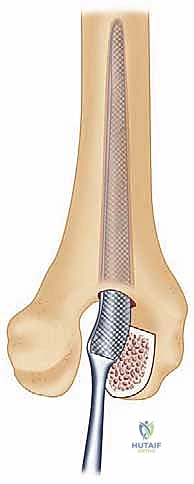
Radiographic templating must account for magnification, typically standardized at 115% to 120%. The surgeon evaluates the AP and lateral radiographs to assess the humeral bow, the diameter of the medullary canals of both the humerus and ulna, and the degree of bone loss at the articular surfaces. In rheumatoid patients, particularly those with JRA, the medullary canals can be pencil-thin or entirely obliterated by sclerotic bone, necessitating the availability of specialized pediatric or micro-sized implants and flexible reamers.
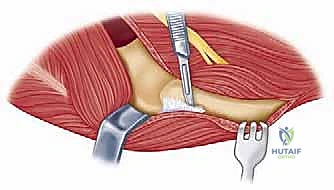
Furthermore, the surgeon must plan the level of the joint line. The center of rotation of the implant must closely approximate the native anatomical axis. On the lateral radiograph, the axis of the capitellum and trochlea should be identified, and the template should be positioned such that the hinge of the prosthesis aligns with this axis. Failure to restore the joint line will alter the resting tension of the flexor and extensor mechanisms, leading to weakness and restricted motion.
Cervical Spine and Ipsilateral Joint Assessment
A critical, yet frequently overlooked, component of preoperative planning in the RA patient is the evaluation of the cervical spine. Up to 90% of patients with advanced RA have cervical spine involvement, and approximately 30% exhibit significant atlantoaxial subluxation or subaxial instability. The manipulation of the neck during endotracheal intubation can cause catastrophic spinal cord compression if this instability is unrecognized.
Therefore, dynamic flexion-extension lateral radiographs of the cervical spine are mandatory before any surgical intervention requiring general anesthesia. If the anterior atlantodental interval (ADI) exceeds 3mm, or if the patient exhibits myelopathic signs, an MRI must be obtained, and clearance from a spine surgeon is required. In severe cases, cervical spine stabilization must precede the elbow arthroplasty.
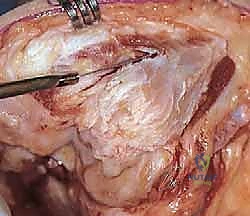
Additionally, the entire ipsilateral upper extremity must be evaluated. Limitations in forearm rotation may stem from pathology at the distal radioulnar joint (DRUJ) or wrist, rather than the elbow. If an ipsilateral total shoulder arthroplasty (TSA) has been performed or is anticipated, the surgeon must carefully plan the length of the humeral stem for the TEA. Using a standard-length TEA stem may create a devastating stress riser between the two implants, leading to an interprosthetic fracture. In these scenarios, a shorter (e.g., 4-inch) humeral implant with a cement restrictor is strongly recommended.
Implant Selection and Tribology
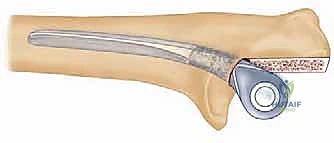
As previously discussed, modern TEA implants are broadly categorized as linked (semiconstrained) or unlinked. For the rheumatoid patient, the linked, semiconstrained prosthesis is the undisputed gold standard. The intrinsic stability provided by the pin-and-locking-ring mechanism compensates for the universally compromised collateral ligaments and the extensive soft-tissue releases often required to correct severe flexion contractures.
The tribology of modern linked implants typically involves a highly polished cobalt-chrome alloy articulating against ultra-high-molecular-weight polyethylene (UHMWPE) bushings. These bushings are designed to absorb the varus-valgus and rotational stresses, preventing these forces from being transmitted to the cement-bone interface. The surgeon must ensure that the implant system chosen has a robust track record of survivorship and offers a wide variety of stem lengths and diameters to accommodate the unpredictable rheumatoid anatomy.
Patient Positioning and Operating Room Setup
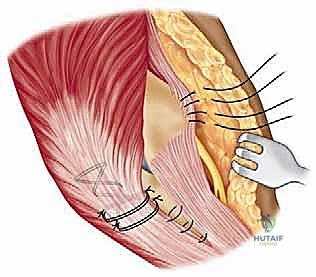
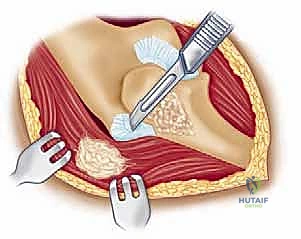
Proper patient positioning is essential for optimal surgical exposure and the prevention of intraoperative complications. The patient is placed in the supine position on a standard operating table. A rolled towel or a specialized scapular bump is placed beneath the ipsilateral scapula. This maneuver protracts the scapula and allows the shoulder to fall posteriorly, dramatically improving access to the posterior aspect of the elbow when the arm is brought across the chest.
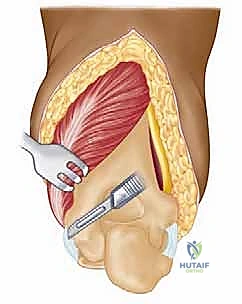
A sterile tourniquet is applied high on the brachium. The entire upper extremity, from the fingertips to the axilla, is meticulously prepped and draped in a standard sterile fashion. The arm is then brought across the patient's chest, allowing the elbow to rest in a
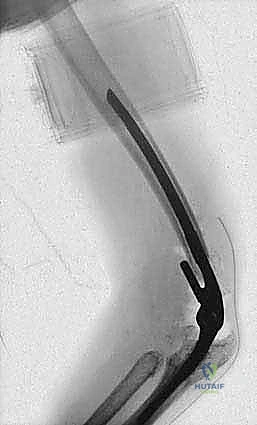
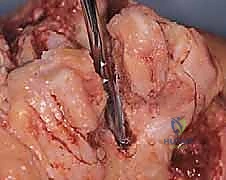
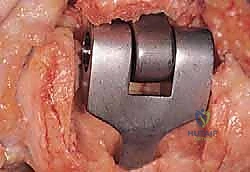
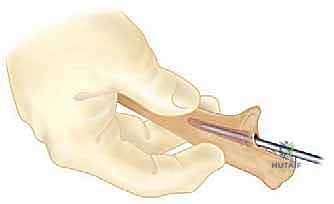
Clinical & Radiographic Imaging Archive