Elbow Arthroplasty Masterclass: Reconstructing Posttraumatic Elbows
Key Takeaway
This masterclass guides you through surgical management of posttraumatic elbow conditions, focusing on interposition arthroplasty and total elbow replacement. We'll meticulously cover patient assessment, imaging, precise intraoperative techniques, including ulnar nerve management, collateral ligament reconstruction, and hinged external fixator application. Learn critical anatomy, common pitfalls, and postoperative care for optimal outcomes in these challenging cases.
Comprehensive Introduction and Patho-Epidemiology
Welcome to the operating theater, colleagues. Today, we confront one of the most technically demanding and unforgiving arenas in orthopaedic surgery: the reconstruction of the posttraumatic elbow. This is not merely an exercise in fracture fixation; it is a masterclass in restoring a complex, highly constrained, and functionally critical articulation after it has been devastated by high-energy trauma, failed prior osteosyntheses, or chronic, attritional instability. These cases demand a meticulous preoperative strategy, flawless intraoperative execution, and a profound, three-dimensional understanding of elbow kinematics, osseous anatomy, and soft-tissue balancing.
When we evaluate posttraumatic conditions of the elbow, we are addressing a broad, heterogeneous spectrum of disorders that manifest as a direct consequence of previous injury. The epidemiology of elbow arthroplasty has shifted dramatically over the past two decades. While rheumatoid arthritis was historically the primary indication for total elbow arthroplasty (TEA), the advent of disease-modifying antirheumatic drugs (DMARDs) has precipitously decreased this cohort. Consequently, posttraumatic arthritis, distal humeral nonunions, and chronic instability now represent the predominant, and arguably the most challenging, indications for elbow reconstruction.
Posttraumatic arthritis of the elbow is a primary pathology characterized by the accelerated mechanical degradation of the articular cartilage. Unlike primary osteoarthritis, which is relatively rare in the elbow, posttraumatic arthritis is driven by altered joint kinematics, articular incongruity, and the sequelae of intra-articular fractures. This primary cartilage loss is rarely an isolated entity. We invariably encounter secondary, confounding pathologies: profound capsular contractures, intra-articular loose bodies, and extensive heterotopic ossification (HO). These elements synergistically contribute to the patient's severe pain, mechanical block to motion, and profound functional disability.
Furthermore, we frequently encounter the catastrophic biomechanical failure of the distal humerus, manifesting as a recalcitrant nonunion or a frankly flail elbow. When a distal humeral fracture fails to unite—often due to comminution, devascularization, or inadequate initial fixation—the patient is left with a dysfunctional, unstable extremity. The very fulcrum of the upper limb is lost, resulting in a dissociation of the forearm from the brachium. In these scenarios, where osteosynthesis is precluded by massive bone loss, avascularity, or poor patient physiology, reconstruction via total elbow arthroplasty transitions from an elective procedure to an essential, limb-salvaging intervention.
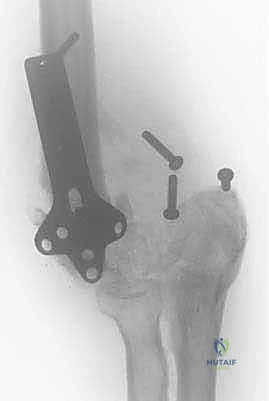
Chronic ligamentous instability and recurrent dislocations present another distinct patho-epidemiological challenge. In the osteopenic or elderly patient, persistent instability inevitably accelerates articular degeneration due to abnormal, eccentric loading and repetitive microtrauma across the radiocapitellar and ulnohumeral articulations. The treatment algorithm for these diverse posttraumatic conditions must be highly individualized. A rigid, dogmatic approach will fail. We must meticulously weigh the specific underlying pathoanatomy, the patient's physiological age, their functional demands, and their adherence to stringent postoperative restrictions. A high-demand, thirty-year-old laborer requires a fundamentally different reconstructive philosophy than a sedentary, seventy-five-year-old patient with a flail elbow.
Detailed Surgical Anatomy and Biomechanics
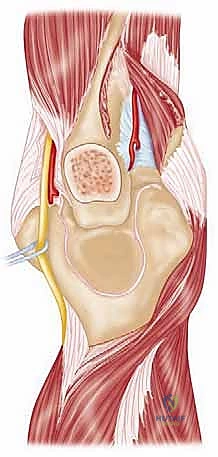
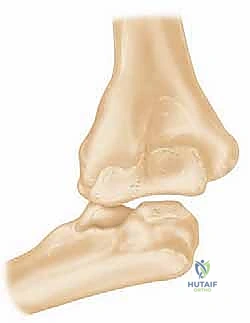
To successfully reconstruct the posttraumatic elbow, the surgeon must possess an intimate, highly technical understanding of its anatomy and biomechanics. The elbow is a highly constrained, complex hinge joint, comprising three distinct articulations: the ulnohumeral, radiocapitellar, and proximal radioulnar joints. The ulnohumeral joint provides the primary intrinsic osseous stability. The distal humerus projects anteriorly at an angle of approximately 30 degrees relative to the humeral shaft, while the greater sigmoid notch of the ulna is oriented 30 degrees posteriorly. This reciprocal angulation is critical for accommodating the soft tissue bulk of the arm during terminal flexion and must be perfectly recreated during prosthetic implantation to restore a functional arc of motion.
The kinematic axis of rotation of the elbow is a central concept in arthroplasty design and surgical execution. The flexion-extension axis is essentially collinear, passing through the center of the trochlea and the center of the capitellum. It is internally rotated approximately 3 to 5 degrees relative to the transepicondylar axis and is in 4 to 8 degrees of valgus relative to the longitudinal axis of the humerus. Failure to accurately reproduce this anatomical axis of rotation during TEA results in eccentric loading of the polyethylene bushings, accelerated wear, and premature aseptic loosening. The implants must be positioned to respect this native axis, particularly in unlinked or convertible systems where the soft tissues bear the brunt of the stabilizing forces.
The soft tissue envelope, specifically the collateral ligament complexes, forms the secondary line of defense against instability. The medial ulnar collateral ligament (MUCL), particularly its anterior bundle, is the primary restraint to valgus stress. The lateral collateral ligament (LCL) complex, specifically the lateral ulnar collateral ligament (LUCL), is the primary restraint to varus and posterolateral rotatory instability. In the posttraumatic setting, these ligamentous structures are frequently attenuated, contracted, or entirely obliterated by prior trauma and multiple surgical interventions. This reality heavily influences implant selection; the unreliability of the soft tissue envelope in posttraumatic elbows is the primary reason that semi-constrained, linked prostheses are overwhelmingly favored over unlinked designs in this patient population.
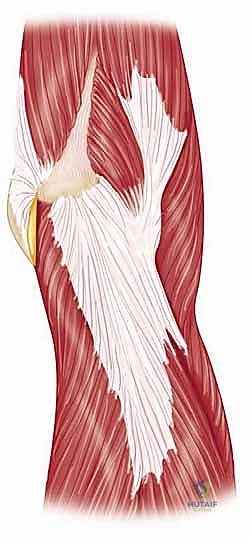
Finally, the musculotendinous units crossing the elbow dictate both function and surgical approach. The triceps brachii is the sole extensor of the elbow, and its preservation or meticulous repair is paramount to a successful outcome. Triceps insufficiency is a devastating complication of elbow arthroplasty, severely limiting the patient's ability to perform activities of daily living, particularly reaching overhead or pushing up from a chair. Furthermore, the ulnar nerve's perilous course through the cubital tunnel makes it highly vulnerable during posterior approaches. In the posttraumatic elbow, the nerve is often encased in scar tissue or distorted by previous transpositions, demanding meticulous, microscopic dissection and neurolysis before any osseous preparation begins.
Exhaustive Indications and Contraindications
The decision to proceed with operative intervention in the posttraumatic elbow requires a rigorous risk-benefit analysis. When conservative measures—such as activity modification, judicious use of NSAIDs, intra-articular corticosteroid injections, and static progressive splinting—have been exhausted, surgical reconstruction is indicated. However, the choice of procedure must be precisely tailored to the patient's unique biological and functional profile. We primarily consider two distinct reconstructive pathways: Interposition Arthroplasty and Total Elbow Arthroplasty (TEA).
Interposition arthroplasty is a biologic resurfacing technique designed to provide a smooth gliding surface while preserving the patient's native bone stock. The ideal candidate is a young, high-demand patient (typically under 60 years of age) with severe posttraumatic arthritis who is unwilling or unable to adhere to the strict permanent lifting restrictions mandated by TEA. These patients usually present with a painful, stiff elbow but possess an intact, functional motor unit and adequate bone stock. Achilles tendon allograft is the preferred interpositional material due to its robust nature and the ability to utilize the attached calcaneal bone block for collateral ligament reconstruction, which is often necessary in these complex cases.
Conversely, Total Elbow Arthroplasty is reserved for older, lower-demand patients, or those with catastrophic bone loss where biological reconstruction is impossible. The classic indications for TEA in the posttraumatic setting include distal humeral nonunions in the elderly, flail elbows, and severe posttraumatic arthritis in patients who are willing to accept a permanent lifelong lifting restriction (typically 1 pound repetitive, 5 pounds single lift). TEA provides highly predictable pain relief and restoration of functional motion, but it is fundamentally a salvage procedure with a finite lifespan. The semi-constrained, linked implant designs provide immediate intrinsic stability, bypassing the need for competent collateral ligaments.
Contraindications must be strictly respected to avoid catastrophic failures. Absolute contraindications for both procedures include active local or systemic infection, a non-functional neuromuscular envelope (e.g., profound paralysis of the biceps and triceps), and a patient who is cognitively unable to comply with postoperative rehabilitation protocols. Relative contraindications for TEA include young age, high-demand occupations or hobbies (e.g., heavy manual labor, contact sports), and inadequate soft tissue coverage that might preclude wound healing over the prosthesis.
| Reconstructive Option | Primary Indications | Absolute Contraindications | Relative Contraindications |
|---|---|---|---|
| Interposition Arthroplasty | Young, high-demand patients (<60 yrs); Posttraumatic arthritis with preserved bone stock; Patients refusing TEA restrictions. | Active infection; Gross instability unamenable to ligament reconstruction; Severe bone loss; Neuropathic joint. | Moderate bone loss; Pre-existing severe triceps weakness; Unrealistic patient expectations regarding pain relief. |
| Total Elbow Arthroplasty (TEA) | Elderly, low-demand patients (>65 yrs); Distal humerus nonunion/flail elbow; Severe bone loss; Failed interposition arthroplasty. | Active infection; Neuropathic joint (Charcot); Inadequate soft tissue coverage; Non-compliant patient. | Age < 60 years; High-demand occupation/hobbies; History of prior deep infection (requires 2-stage approach). |
Pre-Operative Planning, Templating, and Patient Positioning
Preoperative planning for posttraumatic elbow reconstruction is an exhaustive process that begins with a meticulous clinical evaluation. We must document the history of the initial trauma, the specifics of all prior surgical interventions, and any history of infection. The physical examination must quantify the exact arc of active and passive motion. A functional arc is generally considered to be 30 to 130 degrees of flexion-extension, with 50 degrees of pronation and supination. We must meticulously map all previous surgical incisions, as the presence of multiple, intersecting scars severely compromises the vascularity of the skin flaps and dictates our surgical approach. A rigorous neurological examination, with specific attention to the ulnar nerve's motor and sensory distribution, is mandatory.
Radiographic evaluation is the cornerstone of our surgical blueprint. High-quality, orthogonal anteroposterior (AP) and lateral radiographs are essential, though obtaining a true AP view is often confounded by severe flexion contractures. These initial films allow us to assess the overall alignment, the presence of retained hardware, the extent of joint space narrowing, and the presence of gross deformities or nonunions.
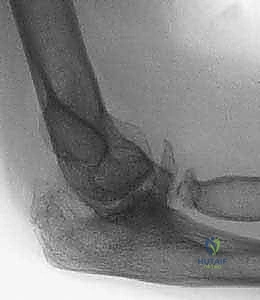
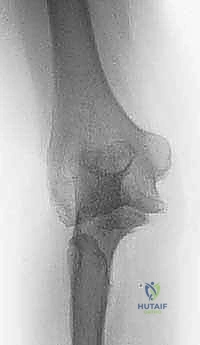
However, plain radiographs are insufficient for the complex posttraumatic elbow. Advanced imaging, specifically a fine-cut Computed Tomography (CT) scan with three-dimensional (3D) reconstructions, is absolutely critical. The CT scan allows for precise quantification of bone loss, evaluation of the medullary canal diameter (which may be sclerotic or obliterated by prior trauma), and the exact mapping of heterotopic ossification that must be excised to restore motion.
Digital templating is performed on the CT multiplanar reformats to determine the optimal implant size, estimate the required depth of the humeral and ulnar stems, and anticipate the need for bone grafting or custom, long-stem implants to bypass areas of cortical deficiency. The surgeon must enter the operating room with a primary plan and at least two contingency plans.
Patient positioning is a critical early step. The procedure is typically performed with the patient in the lateral decubitus position, with the operative arm draped over a well-padded bolster or suspended using a specialized arm holder. This position allows gravity to assist with elbow flexion and provides excellent, unobstructed access to the posterior aspect of the elbow. A sterile tourniquet is applied high on the brachium to ensure a bloodless field during the critical phases of dissection and cementation. The entire upper extremity must be prepped and draped free to allow for full intraoperative manipulation and assessment of kinematics.
Step-by-Step Surgical Approach and Fixation Technique
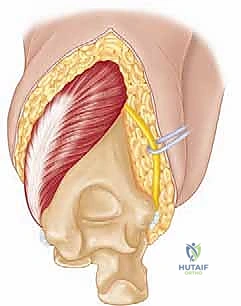
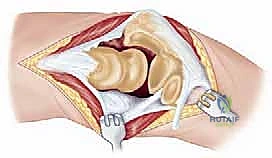
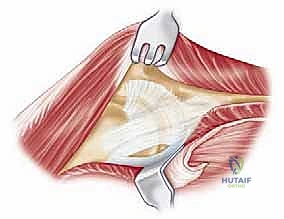
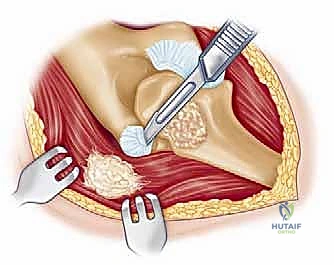
The surgical execution of a total elbow arthroplasty in the posttraumatic setting is a demanding sequence of precise maneuvers. We utilize a universal posterior midline incision, incorporating prior incisions where possible to avoid narrow skin bridges. Full-thickness fasciocutaneous flaps are elevated, preserving the vascular supply to the skin. The first and most critical deep structure to identify is the ulnar nerve. In the revision or posttraumatic setting, the nerve is frequently encased in dense perineural fibrosis. A meticulous neurolysis is performed from the arcade of Struthers proximally to the two heads of the flexor carpi ulnaris distally. The nerve is mobilized and typically transposed anteriorly into a subcutaneous pocket to protect it during the subsequent osseous preparation and from the heat of polymerization during cementation.
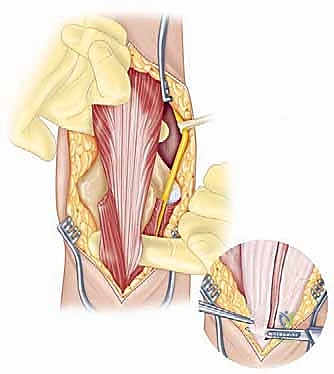
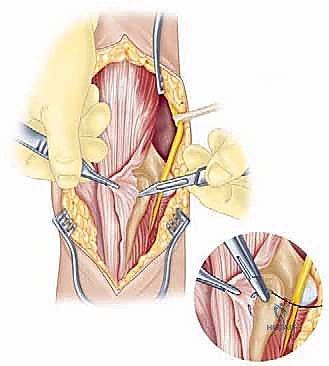
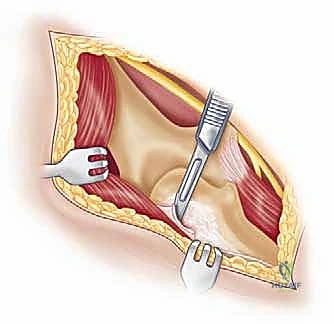
Management of the triceps mechanism is the next major decision point. Depending on the patient's anatomy and the surgeon's preference, a Bryan-Morrey approach (reflecting the triceps from medial to lateral), a triceps-reflecting anconeus pedicle (TRAP) approach, or a triceps-sparing approach may be utilized. In cases of severe contracture or ankylosis, reflecting the triceps provides the most extensile exposure. The collateral ligaments are released from their epicondylar origins, and the joint is dislocated. The radial head is typically excised to facilitate exposure of the proximal ulna and to prevent postoperative radiocapitellar impingement, which is a common source of persistent pain.
Preparation of the osseous structures requires meticulous attention to alignment. The distal humerus is prepared first. The medullary canal is identified—often requiring a high-speed burr if obscured by sclerosis—and sequentially broached. The distal humerus is then resected using cutting jigs that reference the transepicondylar axis to ensure appropriate rotation. The proximal ulna is subsequently prepared. The entry point is critical; it must be precisely at the base of the coronoid process, tracking down the anatomical axis of the ulnar shaft. Eccentric reaming of the ulna can lead to cortical perforation or malalignment of the ulnar component, resulting in catastrophic implant failure.

Trial components are inserted, and the elbow is taken through a full range of motion. We assess for impingement, soft tissue tension, and tracking. Once satisfied, the canals are prepared for cementation using pulsatile lavage, epinephrine-soaked sponges for hemostasis, and a medullary restrictor. Third-generation cementing techniques are mandatory. Polymethylmethacrylate (PMMA) cement is injected in a retrograde fashion using a cement gun to minimize voids. The definitive implants are inserted, and the articulation is linked using the manufacturer-specific locking pin and bushing mechanism.
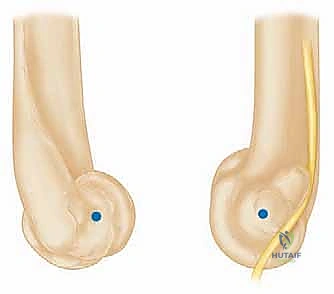
Closure must be exceptionally robust. If the triceps was reflected, it is repaired using heavy, non-absorbable transosseous sutures passed through drill holes in the olecranon. This repair must be secure enough to withstand early, gentle active range of motion. A deep suction drain is placed, and the soft tissues are closed in multiple, meticulous layers to eliminate dead space and minimize the risk of hematoma formation and subsequent infection.

Complications, Incidence Rates, and Salvage Management
The complication profile for elbow arthroplasty in the posttraumatic setting is significantly higher than for primary interventions in rheumatoid arthritis. The compromised soft tissue envelope, poor bone stock, and the presence of prior hardware all contribute to a hostile surgical environment. Surgeons undertaking these procedures must be intimately familiar with the diagnosis and management of these complex failure modes.
Aseptic loosening and bushing wear represent the most common long-term complications, particularly in younger, more active posttraumatic patients. The semi-constrained nature of linked implants transfers
Clinical & Radiographic Imaging Archive