Detailed Patient Presentation and Mechanism of Injury
A 63-year-old male is brought to the emergency department via emergency medical services (EMS) following a high-velocity diving accident. The patient was attending a family gathering and dove headfirst into the shallow end of a residential swimming pool, sustaining a sudden, violent hyperextension injury to his cervical spine upon impact with the pool floor. Immediate bystander intervention prevented drowning, and EMS arrived to find the patient conscious but complaining of severe neck pain, profound weakness in his upper extremities, and dense numbness in his hands. He was appropriately immobilized with a rigid cervical collar and placed on a rigid backboard prior to transport.
Upon arrival at the Level I trauma center, the patient is hemodynamically stable, though mildly hypertensive, which is a common physiologic response to acute pain and anxiety, but must also be monitored for signs of autonomic dysreflexia or impending neurogenic shock. The primary ATLS survey reveals an intact airway, clear bilateral breath sounds, and adequate perfusion. However, his secondary survey is notable for a distinct and striking neurologic deficit. He reports a burning, dysesthetic sensation radiating down both arms into his hands, accompanied by a profound inability to utilize his hands for fine motor tasks. Interestingly, he reports no significant complaints regarding his lower extremities and was able to move his legs spontaneously at the scene.
The mechanism of injury here is the classic hallmark of Central Cord Syndrome (CCS). In older patients, the cervical spine often harbors pre-existing, asymptomatic degenerative changes, including anterior osteophyte formation and posterior ligamentum flavum hypertrophy. When the neck is forcibly hyperextended, the spinal canal diameter is acutely narrowed. The spinal cord is consequently compressed in a "pincer" mechanism—squeezed anteriorly by the hard osteophytic ridges and intervertebral disc complexes, and posteriorly by the buckling, hypertrophied ligamentum flavum.
This acute compression leads to immediate mechanical deformation of the central gray matter and the medial aspects of the surrounding white matter tracts. The resulting cascade of primary mechanical injury is rapidly followed by secondary injury mechanisms, including microvascular hemorrhage, localized ischemia, excitotoxicity, and profound intramedullary edema. The patient's demographic profile—a male in his sixth decade with likely pre-existing cervical spondylosis—is the archetypal presentation for this specific incomplete spinal cord injury pattern.
Comprehensive Clinical Examination Findings
A meticulous neurologic examination is paramount in the acute setting to establish the baseline American Spinal Injury Association (ASIA) Impairment Scale (AIS) grade. On motor examination, the patient demonstrates a striking dissociation between his upper and lower extremities. He possesses 5/5 strength in his deltoids (C5) and elbow flexors (C5, C6). However, his strength drops to 4/5 in the elbow extensors (C7), wrist extensors (C6, C7), and finger flexors (C8). Intrinsics of the hand (T1) are profoundly weak, graded at 2/5. Conversely, his lower extremity motor examination is entirely normal, with 5/5 strength in hip flexion, knee extension, ankle dorsiflexion, long toe extension, and ankle plantarflexion.
Sensory examination reveals preserved light touch and pinprick sensation in the lower extremities and torso. However, there is a distinct decrease in sensation to light touch in the C6 through T1 dermatomes bilaterally, most pronounced in the palmar aspect of both hands. Proprioception and vibration sense, mediated by the dorsal columns, remain largely intact throughout, which is a common finding in CCS due to the peripheral location of these tracts within the spinal cord architecture. Perineal sensation is intact, and voluntary anal sphincter contraction is present, confirming that this is an incomplete spinal cord injury.
Reflex testing in the acute phase of spinal cord injury can be variable due to the transient phenomenon of spinal shock. In this patient, deep tendon reflexes in the upper extremities are diminished (1+ at the biceps and brachioradialis, absent at the triceps). Lower extremity reflexes are normoactive (2+ at the patella and Achilles). Pathologic upper motor neuron signs, such as Hoffmann's sign or the Babinski reflex, are absent at presentation but may manifest days to weeks later as spinal shock resolves and spasticity sets in. The bulbocavernosus reflex is intact, indicating that the period of absolute spinal shock is either resolving or was not profound enough to abolish sacral reflexes.
To understand this clinical presentation, one must possess a deep understanding of the cross-sectional neuroanatomy of the spinal cord. The motor deficit in Central Cord Syndrome is directly attributable to injury to the lateral corticospinal tract. Crucially, the fibers within this tract are somatotopically organized. The fibers innervating the cervical musculature (upper extremities) are located most medially, directly adjacent to the central gray matter. The fibers innervating the thoracic, lumbar, and sacral regions are layered progressively outward toward the lateral periphery of the cord. Therefore, a central intramedullary injury disproportionately damages the medial cervical fibers, resulting in severe upper extremity weakness (particularly the distal musculature, which has dense monosynaptic corticospinal innervation) while sparing the laterally situated lumbosacral fibers.
Advanced Imaging and Diagnostics
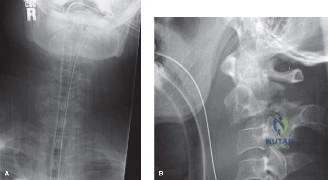
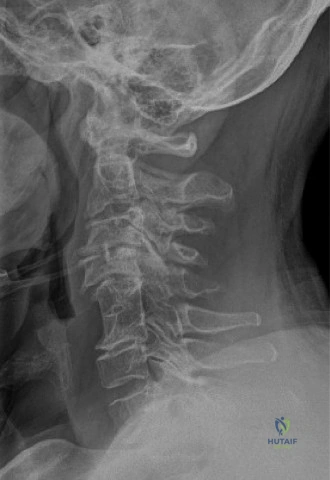
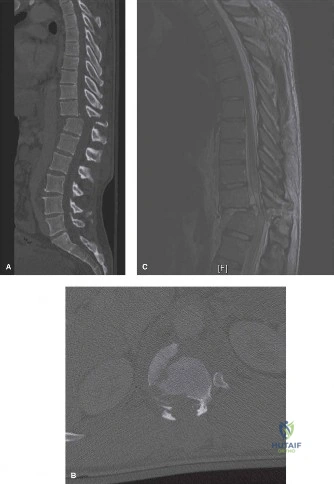
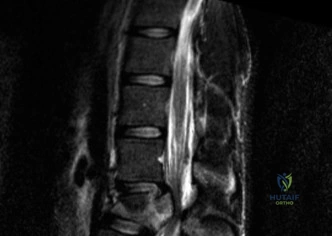
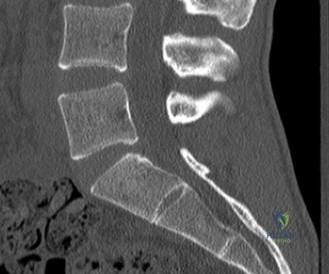
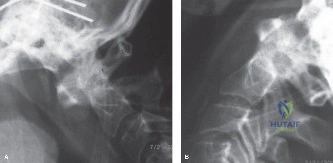
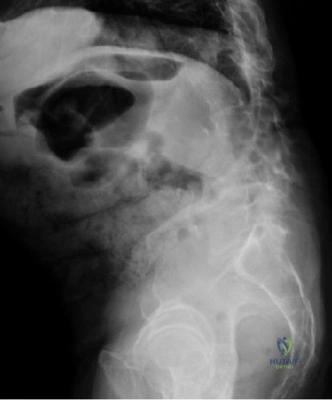
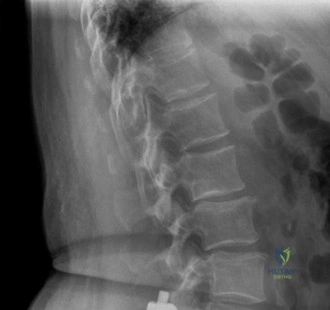
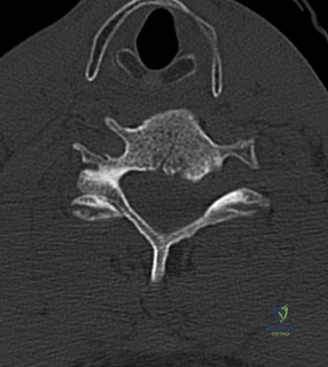
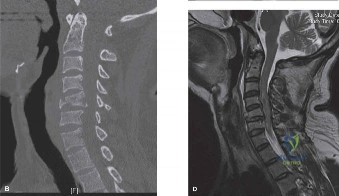
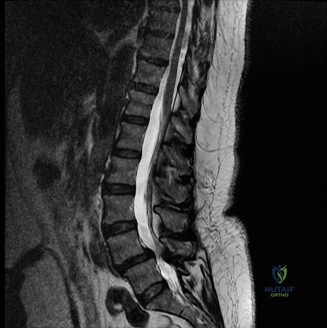
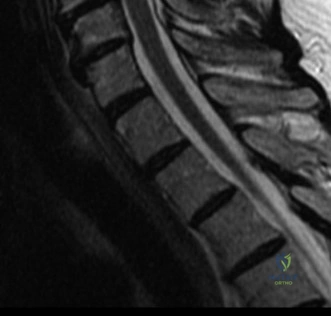
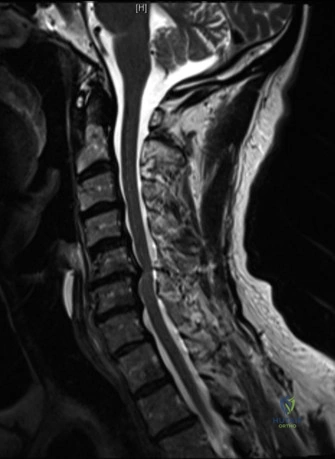
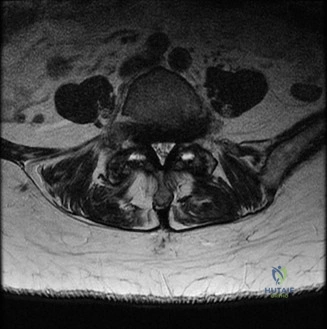
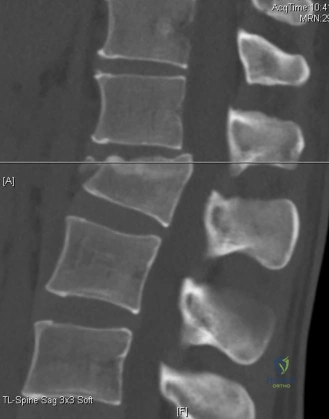
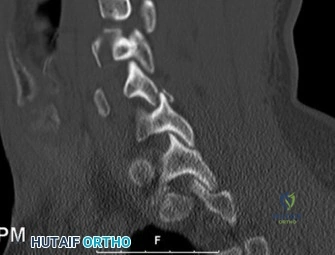
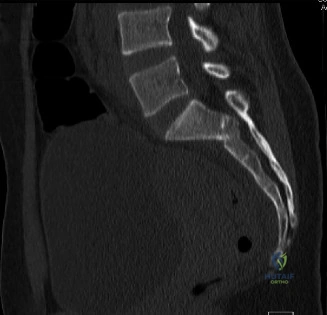
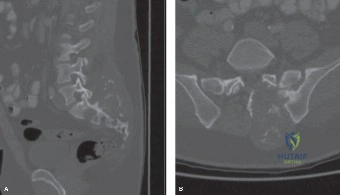
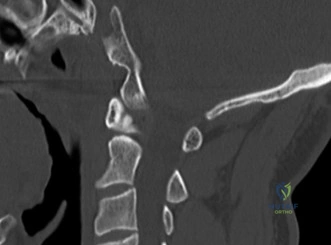
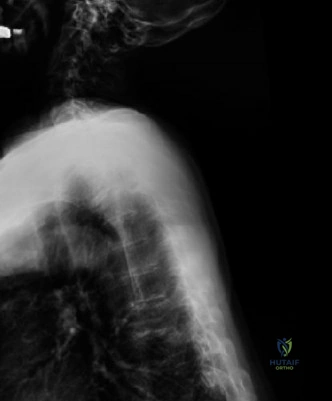
The diagnostic workup for acute spinal cord injury mandates a multimodal imaging approach. Initial plain radiographs of the cervical spine (AP, lateral, and odontoid views) are obtained. The lateral radiograph reveals significant multilevel cervical spondylosis, most prominent from C4 to C7. There is a loss of the normal cervical lordosis, disc space narrowing, and large anterior marginal osteophytes. The Torg-Pavlov ratio, calculated by dividing the sagittal diameter of the spinal canal by the sagittal diameter of the corresponding vertebral body, is less than 0.8 at multiple levels, indicating baseline developmental or acquired cervical stenosis.
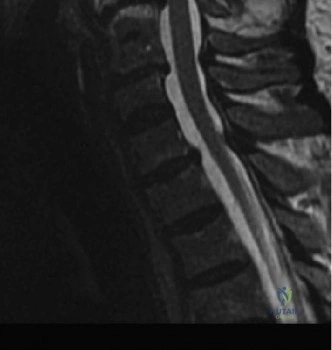
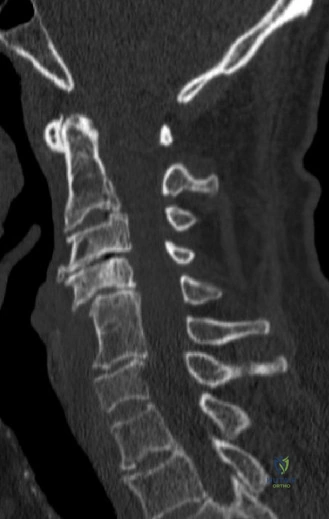
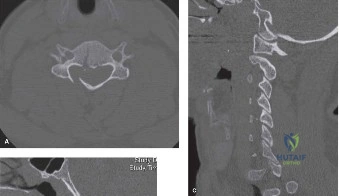
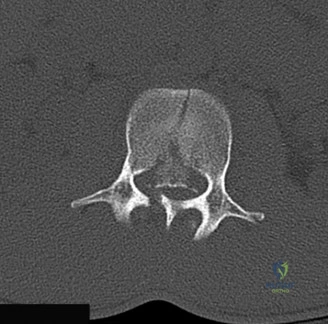
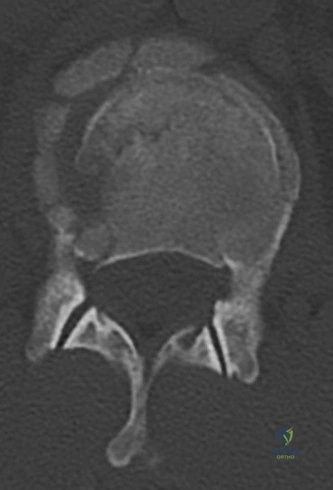
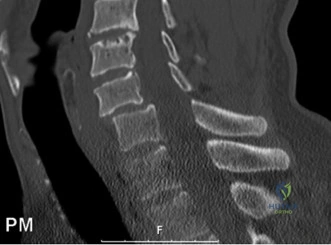
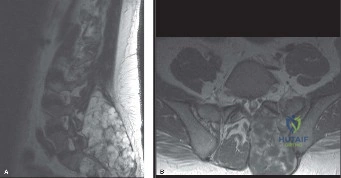
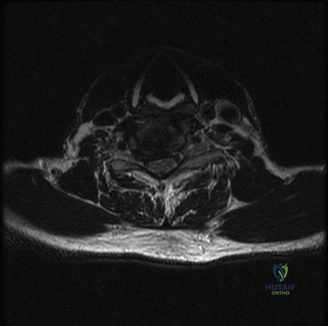
Following plain films, a non-contrast computed tomography (CT) scan of the cervical spine with sagittal and coronal reconstructions is mandatory. The CT scan is highly sensitive for identifying occult osseous injuries, such as subtle facet fractures, lamina fractures, or lateral mass injuries that might be missed on plain radiographs. In this patient, the CT confirms the absence of acute fractures or dislocations. However, it provides excellent bony detail of the central canal stenosis, demonstrating severe uncovertebral joint hypertrophy and facet arthropathy contributing to the narrowing. Ossification of the posterior longitudinal ligament (OPLL) is ruled out on the CT scan.
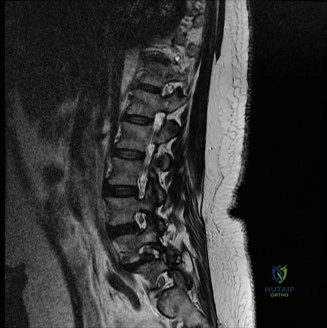
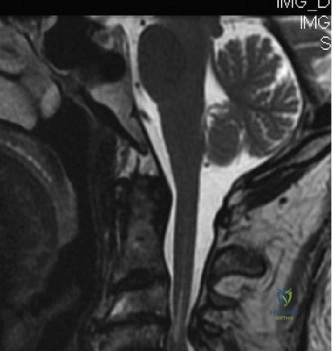
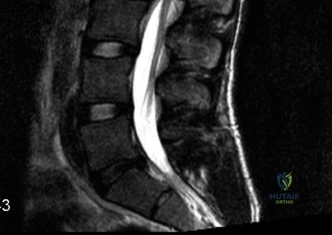
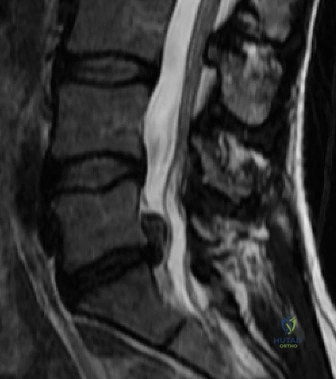
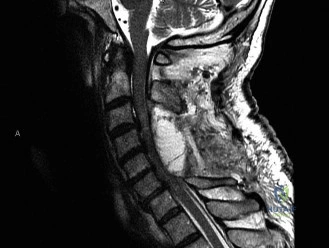
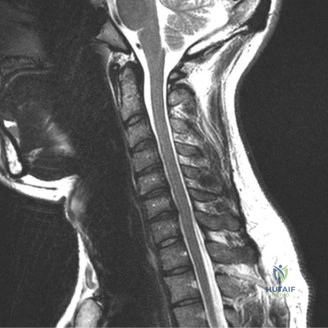
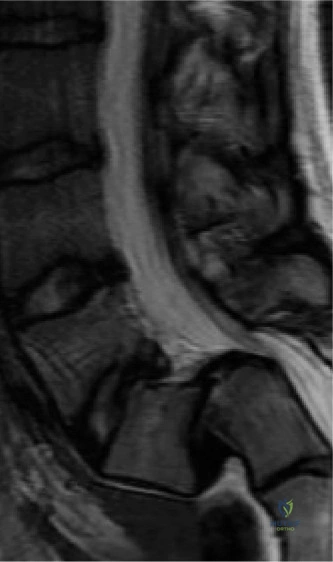
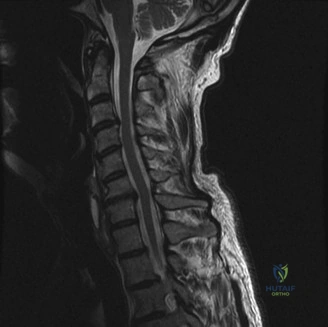
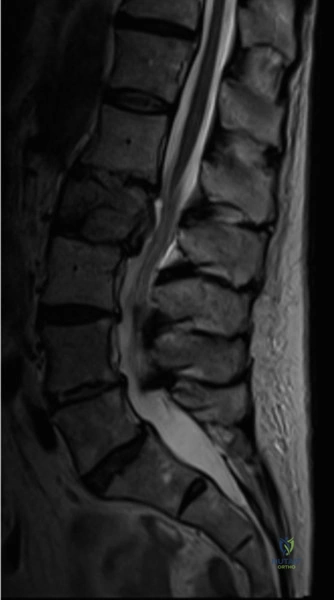
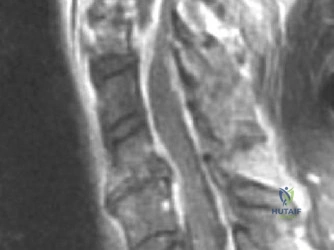
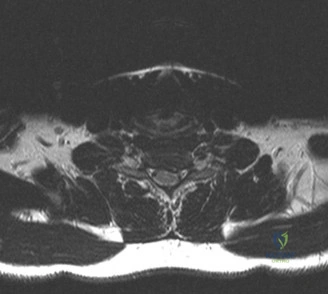
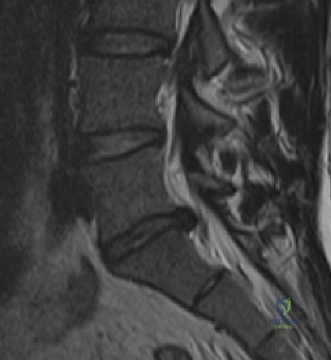
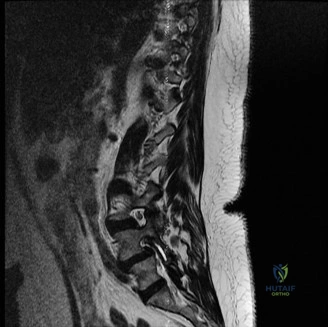
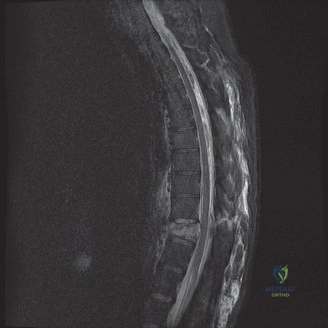
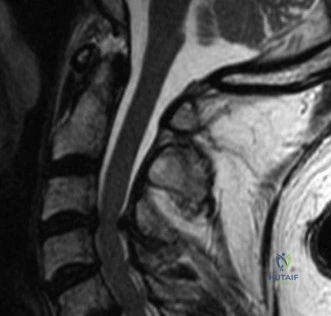
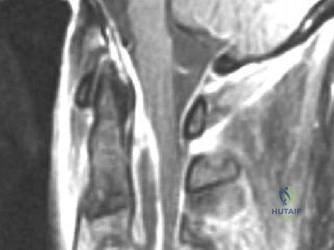
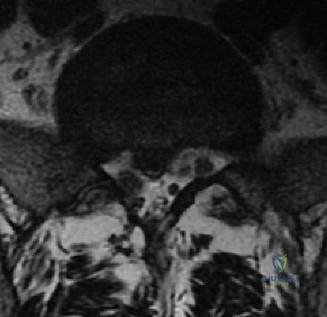
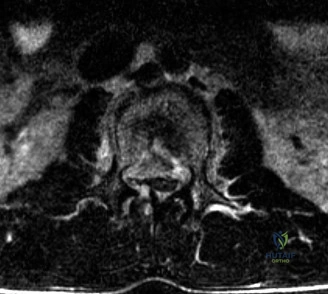
Magnetic Resonance Imaging (MRI) without contrast is the gold standard for evaluating the soft tissues, ligamentous structures, and the spinal cord parenchyma itself. The T2-weighted sagittal sequences reveal a hyperintense signal within the central substance of the spinal cord extending from C4 to C6, indicative of acute intramedullary edema. Crucially, there is no evidence of a hypointense core within this hyperintense region on T2 or gradient-echo (GRE) sequences, suggesting the absence of significant hematomyelia (intramedullary hemorrhage). The presence of hemorrhage is a known predictor of poor neurologic recovery, so its absence here is a favorable prognostic indicator.
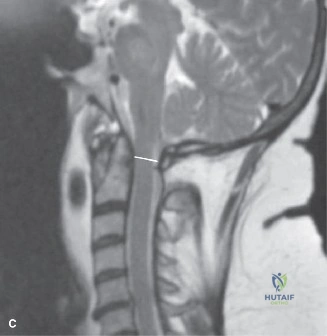
In addition to the parenchymal changes, the MRI clearly delineates the pathoanatomy of the compression. The intervertebral discs at C4-C5 and C5-C6 exhibit broad-based posterior bulges, while the ligamentum flavum is thickened and infolded posteriorly. The anterior subarachnoid space is completely effaced at these levels. Concurrently with imaging, the patient is admitted to the surgical intensive care unit (SICU) for strict hemodynamic monitoring. An arterial line is placed to ensure continuous blood pressure monitoring, with strict parameters set to maintain a Mean Arterial Pressure (MAP) greater than 85 mmHg. This is critical to maximize perfusion to the watershed areas of the injured spinal cord and minimize the expansion of the ischemic penumbra.
Exhaustive Differential Diagnosis
When evaluating a patient with an acute neurologic deficit following spinal trauma, the orthopedic surgeon must rapidly differentiate between the various incomplete spinal cord injury syndromes. Each syndrome presents with a unique constellation of motor and sensory deficits based on the specific neuroanatomic tracts involved. Central Cord Syndrome is the most common, accounting for approximately 9% of all traumatic spinal cord injuries, but it must be distinguished from Anterior Cord Syndrome, Brown-Séquard Syndrome, and Posterior Cord Syndrome.
Anterior Cord Syndrome typically results from a hyperflexion injury or vascular compromise to the anterior spinal artery. It involves damage to the anterior two-thirds of the spinal cord, encompassing the anterior and lateral corticospinal tracts (motor) and the spinothalamic tracts (pain and temperature). Patients present with complete motor paralysis and loss of pain and temperature sensation below the level of injury, but crucially, they retain proprioception and vibratory sense, which are mediated by the spared dorsal columns. This syndrome carries the worst prognosis for functional recovery among the incomplete spinal cord syndromes.
Brown-Séquard Syndrome is a spinal cord hemisection, classically resulting from penetrating trauma (e.g., a stab wound) or asymmetric compression (e.g., a massive unilateral disc herniation). It presents with a distinct crossed neurologic deficit. Patients exhibit ipsilateral loss of motor function (corticospinal tract) and proprioception/vibration (dorsal columns), combined with contralateral loss of pain and temperature sensation (spinothalamic tract) beginning one to two levels below the lesion. This syndrome generally has a favorable prognosis for ambulatory recovery.
Posterior Cord Syndrome is exceedingly rare in trauma and is more commonly associated with medical conditions such as neurosyphilis (tabes dorsalis) or vitamin B12 deficiency. It involves isolated damage to the dorsal columns. Patients present with a profound loss of proprioception, two-point discrimination, and vibratory sense below the level of injury, leading to a severe sensory ataxia. Motor function and pain/temperature sensation remain intact.
Differential Diagnosis Table for Incomplete Spinal Cord Syndromes
| Syndrome | Primary Mechanism | Motor Deficit (Corticospinal) | Sensory Deficit (Spinothalamic) | Sensory Deficit (Dorsal Columns) | Prognosis for Ambulation |
|---|---|---|---|---|---|
| Central Cord | Hyperextension in spondylotic spine | Upper extremities > Lower extremities | Variable loss of pain/temperature | Usually preserved | Good (Lower extremities recover first) |
| Anterior Cord | Hyperflexion, Anterior Spinal Artery ischemia | Complete paralysis below lesion | Complete loss of pain/temperature below lesion | Preserved proprioception and vibration | Poor (Worst prognosis of incomplete syndromes) |
| Brown-Séquard | Hemisection (Penetrating trauma, lateral mass) | Ipsilateral paralysis | Contralateral loss of pain/temperature | Ipsilateral loss of proprioception/vibration | Excellent (Best prognosis for ambulation) |
| Posterior Cord | Direct posterior blow, medical etiologies | Preserved | Preserved | Complete loss of proprioception/vibration | Fair (Limited by profound sensory ataxia) |
Complex Surgical Decision Making and Classifications
The management of acute Central Cord Syndrome remains one of the most hotly debated topics in orthopedic spine surgery. Historically, based on the seminal work by Richard Schneider in the 1950s, CCS was treated strictly non-operatively. Schneider posited that surgery in the setting of an acutely swollen, edematous spinal cord would only exacerbate the neurologic injury. However, modern advancements in microsurgical techniques, neuroanesthesia, and intraoperative neuromonitoring have radically shifted the paradigm toward surgical intervention, particularly when there is evidence of ongoing, persistent spinal cord compression.
The timing of surgical decompression is a critical decision point. The STASCIS (Surgical Timing in Acute Spinal Cord Injury Study) trial demonstrated that early decompression (within 24 hours) for acute spinal cord injury resulted in improved neurologic outcomes compared to delayed decompression. While the STASCIS trial included all types of SCI, recent specific analyses of CCS patients suggest that early surgery is beneficial, particularly for those with profound motor deficits or those who exhibit neurologic deterioration. Conversely, in patients with very mild deficits or rapid spontaneous improvement, a delayed approach (after the acute edema subsides) or a non-operative approach may still be considered. In our 63-year-old patient with significant hand weakness and persistent cord compression on MRI, surgical intervention is strongly indicated.
The choice of surgical approach—anterior, posterior, or combined—depends heavily on the patient's specific pathoanatomy. If the compression is focal (e.g., a single-level traumatic disc herniation) and the patient has preserved cervical lordosis, an Anterior Cervical Discectomy and Fusion (ACDF) is appropriate. However, in cases of multilevel developmental stenosis, extensive OPLL, or multilevel spondylosis with posterior ligamentum flavum buckling, a posterior approach is generally favored.
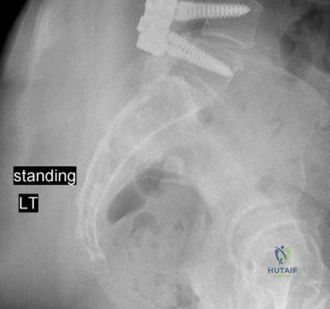
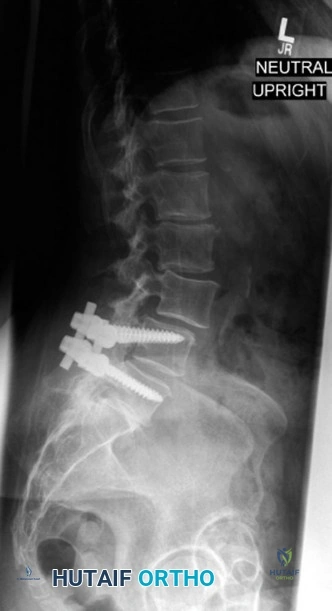
In this specific case, the patient has multilevel stenosis (C4-C7) driven by both anterior osteophytes and posterior infolding. A posterior cervical laminectomy and instrumented fusion is the optimal choice. While a laminoplasty could be considered to preserve motion, the patient's pre-existing loss of cervical lordosis is a relative contraindication for laminoplasty, as the spinal cord may not adequately drift backward away from the anterior osteophytes in a kyphotic or straight spine. Furthermore, an instrumented fusion prevents post-laminectomy kyphosis and provides immediate biomechanical stability.
Step-by-Step Surgical Technique and Intervention
The patient is brought to the operating room and placed under general endotracheal anesthesia. Given the unstable neurologic status and the hyperextension mechanism of injury, awake fiberoptic intubation is performed to avoid any manipulation or extension of the cervical spine during airway securement. Baseline somatosensory evoked potentials (SSEPs) and motor evoked potentials (MEPs) are obtained. The patient is then carefully turned prone onto a Jackson spinal table, with the head secured in Mayfield skeletal tongs. The neck is positioned in a neutral alignment, avoiding both hyperflexion (which stretches the cord) and hyperextension (which recreates the injury mechanism).
A standard midline posterior cervical incision is made from C3 to T1. Subperiosteal dissection is meticulously performed to expose the spinous processes, laminae, and lateral masses of C3 through C7. Extreme care is taken to preserve the muscular attachments to the C2 spinous process (to prevent postoperative kyphosis and axial neck pain) and the C7/T1 attachments where possible. Hemostasis is achieved using bipolar electrocautery.
Prior to performing the laminectomy, the lateral mass screws are placed to minimize manipulation of the unroofed spinal cord. The Magerl technique is utilized for screw trajectory. The starting point is 1 mm medial and 1 mm cephalad to the center of the lateral mass. The trajectory is 15 degrees cranial (parallel to the facet joint) and 15 to 25 degrees lateral. This outward trajectory is critical to avoid injury to the vertebral artery, which runs in the foramen transversarium anterior to the lateral mass, and the exiting nerve root. Screws measuring 3.5 mm in diameter and 14 mm in length are typically placed bilaterally from C4 to C6, with pedicle screws placed at C7 due to the small size and transitional anatomy of the C7 lateral mass.
Once the instrumentation is safely placed, the decompression begins. A high-speed burr (such as a 3 mm matchstick burr) is used to create bilateral troughs at the junction of the lamina and the lateral mass (the medial aspect of the facet joint). The troughs are deepened until the inner cortical margin is breached. A micro-curette and Kerrison rongeurs are then used to carefully release the ligamentum flavum cranially and caudally. The laminae and spinous processes of C4, C5, and C6 are then removed en bloc. This technique prevents the central canal from being compromised by instruments pushing down on the edematous cord, which can happen with piecemeal laminectomy.
Following the en bloc removal, the dura is inspected. It should appear pulsatile and fully expanded. Prophylactic bilateral foraminotomies are performed at C4-C5 to decompress the C5 nerve roots, mitigating the risk of postoperative C5 palsy, which can occur as the spinal cord drifts posteriorly. The surgical field is thoroughly irrigated. Pre-contoured titanium rods are laid into the screw heads and secured with set screws. Decortication of the lateral masses is performed, and local autograft mixed with allograft bone matrix is packed into the decorticated gutters to facilitate arthrodesis. The wound is closed in multiple layers over a subfascial drain to prevent postoperative epidural hematoma.
Strict Post-Operative Protocol and Rehabilitation Stages
Immediately following surgery, the patient is transferred back to the surgical intensive care unit. The strict MAP goals (greater than 85 mmHg) established preoperatively are maintained for a total of 5 to 7 days post-injury to ensure maximal perfusion to the recovering spinal cord. Neurologic examinations are performed hourly. The subfascial drain is carefully monitored for output and is typically removed on postoperative day 1 or 2, once output falls below 30 cc per shift.

Early mobilization is a cornerstone of the postoperative protocol. The patient is fitted with a rigid cervical collar (e.g., Miami J collar) to be worn when out of bed for 6 to 12 weeks, primarily for comfort and secondary stabilization while the bony fusion mass consolidates. Physical therapy (PT) and occupational therapy (OT) are consulted on postoperative day 1. Given the specific deficits of Central Cord Syndrome, OT plays a critical role. While the lower extremities often regain function rapidly—allowing the patient to ambulate relatively early—the upper extremities, particularly the fine motor skills of the hands, lag significantly behind.
The rehabilitation stages for CCS follow a predictable pattern of recovery based on the somatotopic organization of the corticospinal tract. Recovery typically occurs in a distal-to-proximal fashion regarding the spinal cord tracts (meaning lower extremity recovery precedes upper extremity recovery). The typical sequence of recovery is:
1. Lower extremity strength and ambulatory capacity.
2. Bladder and bowel function.
3. Proximal upper extremity strength (shoulders and elbows).
4. Distal upper extremity strength and fine motor dexterity (hands).
Long-term rehabilitation focuses heavily on neuroplasticity. Functional electrical stimulation (FES) may be utilized to prevent muscle atrophy in the hands and forearms. Spasticity management becomes crucial in the subacute and chronic phases, as upper motor neuron signs develop. Medications such as baclofen or tizanidine, and potentially botulinum toxin injections for focal contractures, may be required. Neuropathic pain, presenting as burning or tingling in the upper extremities, is common and is managed with gabapentin, pregabalin, or tricyclic antidepressants.
High-Yield Clinical Pearls and Pitfalls
Diagnostic Pitfall: The "Walking Quadriplegic"
A major diagnostic pitfall in the emergency department is missing the diagnosis of Central Cord Syndrome in an ambulatory patient. Because the lower extremities are often spared or minimally affected, an elderly patient who sustains a fall and walks into the ED complaining of "numb hands" or "weak grip" may be misdiagnosed with bilateral peripheral nerve compression or a simple cervical sprain. The orthopedic surgeon must maintain a high index of suspicion and perform a thorough intrinsic hand muscle examination on all patients with hyperextension mechanisms.
Anatomic Pearl: Somatotopic Organization
The classic board examination question regarding CCS tests the surgeon's knowledge of the lateral corticospinal tract. Remember the somatotopic arrangement: the cervical fibers are located medially, and the sacral fibers are located laterally. This anatomic reality is the sole reason why the upper extremities are disproportionately affected compared to the lower extremities in a central intramedullary lesion.
Surgical Pitfall: C5 Nerve Root Palsy
A well-documented complication of posterior cervical decompression (both laminectomy and laminoplasty) is the postoperative C5 nerve root palsy. As the spinal cord is untethered from anterior compression, it drifts posteriorly. The C5 nerve root, which has a short, horizontal course, can become tethered and stretched, leading to severe deltoid and biceps weakness. To mitigate this pitfall, surgeons should routinely perform prophylactic C4-C5 foraminotomies during the posterior decompression to allow the nerve root room to translate posteriorly without tension.
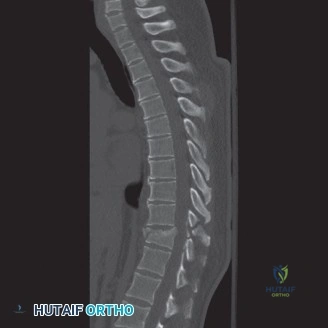
Imaging Pearl: Edema vs. Hemorrhage
When reviewing the preoperative MRI, pay close attention to the T2-weighted and GRE sequences. A diffuse, hyperintense signal on T2 indicates edema, which has a reasonable potential for neurologic recovery. However, a central core of hypointensity on T2 or "blooming" artifact on GRE indicates hematomyelia (intramedullary hemorrhage). The presence of hematomyelia is a devastating prognostic sign, indicating irreversible tissue necrosis, and correlates with permanent, severe neurologic deficits regardless of surgical intervention. Accurate interpretation of these MRI findings is essential for setting realistic expectations with the patient and their family.
Clinical & Radiographic Imaging Archive
Detailed Chapters & Topics
Dive deeper into specialized chapters regarding spine-cases-2
