Anterior Thoracic Corpectomy: An Intraoperative Masterclass for Complex Spinal Pathology
Key Takeaway
This masterclass guides fellows through anterior thoracic corpectomy, a vital procedure for complex spinal pathologies including tumors, trauma, and severe disc herniations. We meticulously cover patient positioning, intricate anatomical dissection, precise vertebral body resection, and robust anterior column reconstruction. Emphasizing critical neurovascular protection and optimal hardware placement, this session provides granular, real-time insights into achieving superior patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
The anterior thoracic corpectomy remains one of the most demanding, yet fundamentally rewarding, procedures within the armamentarium of the complex spine surgeon. This intervention is not merely an exercise in osseous resection; it is a definitive, biomechanically sound strategy aimed at restoring neurological function, achieving robust spinal column stability, and providing definitive eradication or palliation for a myriad of complex ventral pathologies. The thoracic spine, characterized by its inherent rigidity conferred by the rib cage and sternal complex, requires significant kinetic energy to sustain traumatic injury. Conversely, addressing pathologies within this region demands significant surgical expertise, meticulous preoperative planning, and an intimate understanding of the surrounding visceral and neurovascular anatomy.
Historically, posterior-only approaches to ventral thoracic pathology, such as laminectomy, were associated with catastrophic neurological outcomes due to the manipulation of the spinal cord within a constrained canal and the failure to address the primary ventral compressive vector. The evolution of the anterior approach has revolutionized the management of these lesions. By accessing the spine through the thoracic cavity, surgeons can achieve direct visualization of the ventral dura, allowing for margin-free tumor resection, thorough debridement of osteomyelitis, and the removal of retropulsed bone fragments without placing traction on the tenuous thoracic spinal cord.
The patho-epidemiology of conditions necessitating an anterior thoracic corpectomy is diverse. The spinal column represents the most frequent site of skeletal metastasis, with the thoracic spine being disproportionately affected due to its extensive volume and the valveless extradural venous plexus of Batson, which facilitates retrograde hematogenous dissemination from primary visceral malignancies (e.g., breast, lung, prostate). Furthermore, primary spinal infections, such as pyogenic vertebral osteomyelitis and tuberculous spondylitis (Pott’s disease), frequently target the well-vascularized subchondral bone of the thoracic vertebral bodies, leading to progressive kyphotic deformity and epidural abscess formation. In the realm of trauma, high-energy burst fractures frequently result in anterior and middle column failure, necessitating anterior column reconstruction to prevent post-traumatic kyphosis and delayed neurological deterioration.
Understanding these epidemiological trends is crucial for the orthopedic and neurosurgical spine specialist. The decision to proceed with an anterior corpectomy must be weighed against the physiological toll of a thoracotomy, particularly in patients with compromised pulmonary reserve or extensive metastatic disease. However, when executed with precision, the anterior thoracic corpectomy provides unmatched access for ventral decompression and the placement of load-sharing interbody constructs that biomechanically out-perform posterior-only short-segment fixations in the setting of severe anterior column deficiency.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of thoracic surgical anatomy is the absolute prerequisite for safe and effective anterior spinal surgery. The thoracic vertebral bodies are characteristically heart-shaped, possessing pedicles that are morphologically distinct from their lumbar counterparts—being larger superoinferiorly than mediolaterally. The medial cortex of the thoracic pedicle is the thickest, but critically, there is a virtual absence of epidural space between the posterior longitudinal ligament (PLL) and the ventral dura in the thoracic spine. This anatomical reality dictates that any dorsal displacement of pathology, or iatrogenic manipulation during surgery, translates directly into spinal cord compression.
The costovertebral and costotransverse articulations serve as the gateway to the lateral aspect of the vertebral body. Understanding the intricate ligamentous attachments of the rib head to the vertebral body and the transverse process is essential for gaining access to the neural foramen and the pedicle. The rib head articulates with the demifacets of two adjacent vertebral bodies and the intervening intervertebral disc. Resection of the rib head is a non-negotiable step in exposing the posterolateral margin of the vertebral body and identifying the anatomical landmarks necessary for safe decompression of the spinal canal.
The neurovascular relationships in the anterior thoracic cavity are unforgiving. The segmental vessels (arteries and veins) course transversely across the waist of the vertebral bodies, deep to the sympathetic chain and parietal pleura, before entering the neural foramen. These vessels must be meticulously isolated, ligated, and divided to prevent catastrophic hemorrhage and to clear the surgical corridor. The sympathetic chain runs vertically along the anterolateral aspect of the vertebral bodies; inadvertent injury to this structure, particularly in the upper thoracic spine, can precipitate Horner’s syndrome or profound autonomic dysregulation.
Biomechanically, the thoracic spine is a highly stable construct, functioning in conjunction with the rib cage to protect the cardiopulmonary viscera. This inherent stability, however, is compromised when the anterior and middle columns are destroyed by tumor, infection, or trauma. The thoracic spine naturally exists in a state of kyphosis. Pathological destruction of the anterior vertebral body exacerbates this kyphosis, shifting the instantaneous axis of rotation anteriorly and placing immense tensile stress on the posterior ligamentous complex. An anterior corpectomy and structural reconstruction directly counteract this pathological moment arm, providing a load-sharing strut that restores the anatomical sagittal profile and creates an optimal biomechanical environment for solid arthrodesis.
Exhaustive Indications and Contraindications
The decision algorithm for performing an anterior thoracic corpectomy requires a meticulous synthesis of the patient's neurological status, oncological prognosis, systemic health, and the specific biomechanical demands of the lesion. This procedure is the gold standard for progressive myelopathy secondary to anterior compressive lesions that are anatomically inaccessible or unsafe to address via a posterior or posterolateral approach. Direct anterior decompression avoids the catastrophic risk of spinal cord manipulation inherent in posterior approaches to ventral pathology.
In the context of spinal trauma, anterior corpectomy is highly indicated for fractures with significant anterior spinal cord compression, particularly those featuring retropulsed bone fragments that have breached the posterior vertebral body wall and are actively compressing the ventral dura, resulting in incomplete neurological deficits. For oncological cases, whether metastatic or primary thoracic tumors, the anterior approach facilitates direct access for circumferential decompression of the thecal sac, allows for margin-free en bloc resection in selected primary tumors, and permits robust anterior column stabilization.
Similarly, in cases of severe vertebral osteomyelitis or discitis, the anterior approach provides the most direct route for radical debridement of necrotic, infected bone and the placement of structural allograft or titanium cages to restore height and alignment. Sequestered disc herniations, particularly those that have migrated posterior to the vertebral body, are heavily calcified, or are located centrally within the narrow thoracic canal, are also prime indications. Attempting to remove a calcified, central thoracic disc via a posterior approach carries an unacceptably high risk of iatrogenic paraplegia.
Despite its powerful utility, the anterior thoracic corpectomy is not without significant contraindications. Severe pulmonary compromise (e.g., advanced COPD, pulmonary fibrosis) is a major relative, and sometimes absolute, contraindication due to the necessity of single-lung ventilation and the physiological insult of a thoracotomy. Furthermore, patients with profound medical comorbidities who cannot tolerate the hemodynamic shifts associated with major intracavitary surgery are poor candidates. In oncological scenarios, a predicted life expectancy of less than 3 to 6 months may shift the paradigm toward palliative posterior decompression and stabilization rather than a morbid anterior resection, unless intractable pain or acute neurological decline mandates intervention.
Indications and Contraindications Summary
| Category | Primary Indications | Relative / Absolute Contraindications |
|---|---|---|
| Trauma | Burst fractures with severe ventral cord compression; Retropulsed fragments causing myelopathy; Anterior/middle column failure. | Polytrauma with severe pulmonary contusions (relative); Complete, irreversible spinal cord injury >72 hours (relative). |
| Oncology | Radioresistant metastatic tumors (e.g., Renal, Thyroid) with epidural cord compression; Primary bone tumors requiring en bloc resection. | Expected survival < 3 months; Extensive multilevel contiguous disease precluding single-level reconstruction. |
| Infection | Pyogenic osteomyelitis with epidural abscess and structural collapse; Tuberculous spondylitis (Pott's disease) with kyphosis. | Active, uncontrolled sepsis with hemodynamic instability; Inability to tolerate prolonged general anesthesia. |
| Degenerative | Calcified, central, or giant thoracic disc herniations; Severe OPLL causing ventral cord tethering and myelopathy. | Purely radicular symptoms amenable to less invasive procedures; Severe osteoporosis compromising hardware purchase. |
| Systemic | Need for definitive anterior column support in revision pseudarthrosis. | Severe COPD/pulmonary fibrosis; Uncorrectable coagulopathy; Prior extensive ipsilateral thoracotomy (relative). |
Pre-Operative Planning, Templating, and Patient Positioning
The foundation of a successful anterior thoracic corpectomy is laid entirely in the preoperative phase. A multidisciplinary approach, often involving a thoracic access surgeon, neuro-monitoring technicians, and specialized anesthesiologists, is mandatory. Advanced imaging is the cornerstone of our operative blueprint. High-resolution Magnetic Resonance Imaging (MRI) is the primary diagnostic modality, providing unparalleled visualization of neural compression, the integrity of the posterior longitudinal ligament, and the extent of soft tissue or epidural tumor extension.
Computed Tomography (CT) is equally critical for delineating osseous anatomy. Fine-cut CT scans allow the surgeon to assess the degree of osteolysis, evaluate pedicle morphology, and meticulously template the size of the anticipated corpectomy defect. This templating dictates the selection of our structural graft—whether an expandable titanium cage, a static mesh cylinder, or a structural allograft—and the length of the anterolateral fixation plate. In cases where MRI is contraindicated or degraded by hardware artifact, CT-Myelography remains an invaluable adjunct for precisely defining the limits of subarachnoid block and ventral cord compression. Furthermore, preoperative angiography may be indicated to identify the Artery of Adamkiewicz, particularly for lesions between T9 and T12, to minimize the risk of devastating postoperative anterior spinal artery syndrome.
Patient positioning and anesthetic setup require obsessive attention to detail. The procedure is typically performed with the patient in the lateral decubitus position. For lesions from T4 to T11, a left-sided approach is generally preferred to mobilize the aorta, which is structurally more robust and easier to retract than the thin-walled inferior vena cava on the right. However, for lesions at the thoracolumbar junction (T12-L1), a right-sided approach is often selected to avoid the liver and the complexities of taking down the diaphragm on the left. The anesthesiologist will utilize a double-lumen endotracheal tube to allow for selective deflation of the ipsilateral lung, providing the necessary working space within the thoracic cavity.
Intraoperative neuromonitoring, encompassing both Somatosensory Evoked Potentials (SSEPs) and Motor Evoked Potentials (MEPs), is standard of care. Baseline signals must be obtained prior to positioning and continuously monitored throughout the case. The patient is secured on a radiolucent beanbag, with an axillary roll placed to protect the dependent brachial plexus. The ipsilateral arm is elevated and supported to allow unimpeded access to the lateral chest wall. Fluoroscopy is utilized prior to prepping and draping to localize the target vertebral level and plan the exact trajectory of the thoracotomy incision, ensuring that the surgical corridor aligns perfectly with the pathology.
Step-by-Step Surgical Approach and Fixation Technique
Thoracotomy and Initial Exposure
The procedure commences with a meticulously planned posterolateral thoracotomy. The skin incision typically follows the contour of the rib that corresponds to the vertebral level above the pathology (e.g., resecting the 7th rib for a T8 corpectomy) to provide a trajectory that looks down onto the superior endplate of the target vertebra. The latissimus dorsi and serratus anterior muscles are divided, and the designated rib is identified. The periosteum of the rib is stripped using a Doyen elevator, and the rib is resected to be utilized later as autogenous bone graft. The pleural cavity is then entered through the rib bed.
Once the pleural space is accessed, the anesthesiologist deflates the ipsilateral lung. A self-retaining rib retractor is deployed to maintain exposure. The deflated lung is gently packed away anteriorly and inferiorly using moist laparotomy sponges and a malleable retractor. This maneuver exposes the posterior mediastinum and the parietal pleura draping over the vertebral column.
The parietal pleura is incised longitudinally over the target vertebral body and the adjacent discs. Blunt dissection is utilized to mobilize the pleura and the underlying connective tissue. The great vessels—the aorta on the left or the azygos vein on the right—are gently swept anteriorly. The sympathetic chain is identified, mobilized, and protected.
Rib Head Resection and Segmental Vessel Ligation
The critical transition from the thoracic cavity to the spinal canal involves the resection of the rib head and the management of the segmental vasculature. The costovertebral and costotransverse articulations of the rib corresponding to the target vertebra are identified. Using a Cobb elevator and electrocautery, the strong radiate and costotransverse ligaments are divided.

Once the ligaments are released, the rib head is resected using a high-speed burr or a Leksell rongeur. This resection must be flush with the vertebral body to expose the lateral aspect of the pedicle and the neural foramen. This maneuver is essential for defining the posterior limit of the corpectomy and safely locating the spinal canal.
Simultaneously, the segmental vessels must be addressed. These vessels run transversely across the mid-portion of the vertebral body. They are meticulously dissected, doubly ligated with non-absorbable sutures or surgical clips, and transected. Failure to adequately secure these vessels can result in profound hemorrhage that obscures the surgical field and compromises patient hemodynamics.
Execution of the Corpectomy
With the vertebral body fully exposed and the vascular supply controlled, the corpectomy begins. The intervertebral discs above and below the pathological vertebra are incised with a scalpel and removed using pituitary rongeurs and curettes. This defines the superior and inferior boundaries of the resection.
Using a high-speed matchstick or round cutting burr, the surgeon begins to excavate the cancellous bone of the vertebral body. The resection proceeds from anterior to posterior, and from the superior endplate to the inferior endplate. The lateral cortex on the contralateral side is preserved if possible, acting as a protective barrier to the contralateral neurovascular structures.
As the burring deepens toward the spinal canal, the bone bleeds significantly; this is controlled with bone wax and hemostatic agents. The surgeon intentionally leaves a thin shell of the posterior vertebral body wall intact during the initial excavation. This posterior wall serves to protect the ventral dura from the high-speed burr.
Neural Decompression and Reconstruction
The final, most delicate phase of the resection involves the removal of the posterior cortical wall and the posterior longitudinal ligament (PLL). Using micro-curettes, Kerrison rongeurs, and diamond burrs under direct illumination (and often magnification), the posterior wall is thinned and carefully elevated away from the dura. The epidural venous plexus is encountered here and must be managed with bipolar electrocautery and hemostatic matrix.
Once the posterior wall and PLL are completely removed, the ventral thecal sac is exposed and decompressed. The surgeon must confirm that the dura is fully expanded and pulsatile, and that no residual compressive pathology remains within the epidural space.

Following successful decompression, the focus shifts to reconstruction. The cartilaginous endplates of the adjacent superior and inferior vertebrae are meticulously prepared with curettes and rasps to expose bleeding subchondral bone, optimizing the environment for arthrodesis. The defect is measured using calipers.
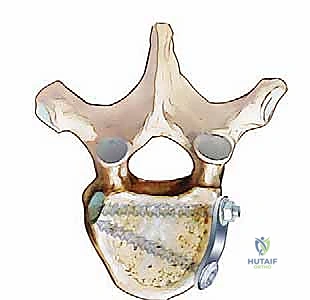
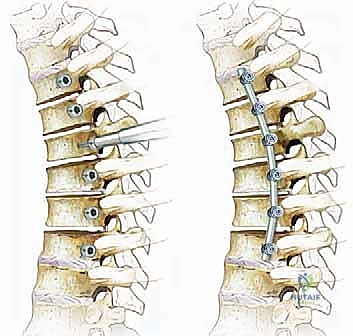
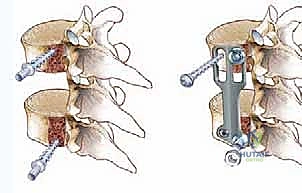
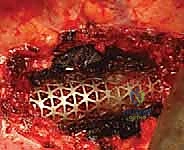
An expandable titanium cage or a pre-cut structural allograft, packed with the autograft harvested from the rib and corpectomy site, is carefully impacted into the defect. If an expandable cage is utilized, it is expanded in situ to achieve a tight friction fit and restore vertebral height and sagittal alignment.

To secure the construct and provide immediate biomechanical stability, an anterolateral locking plate is applied. The plate is positioned over the lateral aspect of the adjacent intact vertebral bodies, and bicortical screws are placed to lock the construct in place. The final construct is verified with intraoperative fluoroscopy to ensure optimal hardware placement and spinal alignment.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, the anterior thoracic corpectomy carries a formidable complication profile. The intersection of orthopedic spine surgery and thoracic surgery inherently exposes the patient to both neurological and cardiopulmonary risks. Spinal cord ischemia or direct mechanical injury remains the most devastating complication. Ischemic injury can occur due to the sacrifice of critical radiculomedullary arteries (e.g., Artery of Adamkiewicz) or secondary to systemic hypotension during the procedure. Continuous MEP and SSEP monitoring is critical; a sudden loss of signals demands immediate cessation of manipulation, elevation of mean arterial pressure, and potentially the administration of high-dose corticosteroids.
Vascular injuries, particularly to the aorta, inferior vena cava, or azygos vein, represent acute intraoperative emergencies. Aortic injury, though rare (incidence <1%), carries a high mortality rate and requires immediate primary repair, often necessitating the urgent assistance of a vascular or cardiothoracic surgeon. Pulmonary complications are significantly more common, with atelectasis, pneumonia, and pleural effusions occurring in up to 15-20% of patients. Inadequate closure of the pleura or injury to the thoracic duct can result in a chylothorax, a persistent leak of lymphatic fluid that requires prolonged chest tube drainage, dietary modifications (medium-chain triglycerides), and occasionally surgical ligation of the thoracic duct.
Hardware-related complications, including cage subsidence, plate pullout, and eventual pseudarthrosis, are late-term failures that compromise the biomechanical integrity of the reconstruction. Subsidence is particularly common in osteoporotic bone or when endplate preparation has been overly aggressive, breaching the strong subchondral bone. Salvage management for hardware failure typically requires a secondary posterior approach for long-segment pedicle screw fixation to bypass the failed anterior construct and restore stability.
Complications and Salvage Strategies
| Complication | Estimated Incidence | Prevention Strategy | Salvage / Management Strategy |
|---|---|---|---|
| Spinal Cord Injury / Paraplegia | 1.0% - 2.5% | Continuous MEP/SSEP monitoring; Avoid hypotension; Gentle tissue handling. | Induce hypertension (MAP > 85-90 mmHg); Immediate removal of hardware if compressing dura; High-dose steroids (controversial). |
| Major Vascular Injury (Aorta/IVC) | < 1.0% | Meticulous blunt dissection; Direct visualization before instrument use; Careful retractor placement. | Immediate direct pressure; Urgent intraoperative vascular surgery consultation; Primary suture repair or patch angioplasty. |
| Pulmonary Complications (Pneumonia/Atelectasis) | 10% - 20% | Preoperative pulmonary optimization; Early postoperative mobilization; Aggressive incentive spirometry. | Broad-spectrum antibiotics; Bronchoscopy for mucus plugging; Prolonged ventilatory support if necessary. |
| Chylothorax | 1.0% - 3.0% | Identify and ligate thoracic duct if visualized; Careful dissection near the diaphragm on the left. | Prolonged chest tube drainage; TPN or medium-chain triglyceride diet; Re-exploration and duct ligation if output >1L/day. |
| Hardware Failure / Subsidence | 5.0% - 10.0% | Preserve subchondral bone during endplate prep; Use appropriately sized footprints; Treat osteoporosis. | Supplemental posterior spinal fusion with pedicle screws; Revision anterior surgery for cage replacement (rarely preferred). |
| Cerebrospinal Fluid (CSF) Leak | 2.0% - 5.0% | Careful use of diamond burr near dura; Avoid pulling on adherent PLL or tumor capsule. | Primary watertight dural repair (difficult anteriorly); Fibrin glue/sealants; Lumbar subarachnoid drain placement. |
Phased Post-Operative Rehabilitation Protocols
The postoperative management of the anterior thoracic corpectomy patient is intensive and requires a highly coordinated, multidisciplinary approach. Immediately following surgery, patients are typically transferred to the Surgical Intensive Care Unit (SICU) for continuous hemodynamic and neurological monitoring. Extubation is usually achieved in the operating room or shortly after arrival in the ICU, provided the patient meets standard respiratory parameters and has not experienced massive fluid shifts or airway edema.
A thoracostomy tube (chest tube) is routinely left in place to evacuate postoperative hemothorax and pneumothorax. Chest tube management is critical; output is monitored hourly, and the tube is generally maintained on water seal or low continuous wall suction until drainage decreases to less than 150-200 cc per 24-hour period, and a chest radiograph confirms full lung re-expansion. Pain management is a significant hurdle, given the morbid nature of a thoracotomy. Multimodal analgesia, often incorporating patient-controlled analgesia (PCA), intercostal nerve blocks, or epidural catheters placed by the anesthesia team, is essential to facilitate deep breathing and prevent atelectasis.
Mobilization is initiated as early as postoperative day one. Physical and occupational therapy are consulted immediately. Depending on the bone quality and the rigidity of the anterior construct, the surgeon may prescribe a Thoracolumbosacral Orthosis (TLSO) brace for use when the patient is out of bed, typically for a duration of 6 to 12 weeks. The brace serves as an adjunct to internal fixation, limiting extreme ranges of motion and providing proprioceptive feedback while osseous fusion occurs.
Long-term rehabilitation focuses on restoring core strength, improving pulmonary capacity, and safely returning the patient to their baseline activities of daily living. Serial upright radiographs are obtained at 6 weeks, 3 months, 6 months, and 1 year to monitor for hardware position, cage subsidence, and the progression of the interbody arthrodesis. In oncological cases, postoperative radiation or systemic chemotherapy is coordinated with the medical oncology team, typically delayed until early wound healing is confirmed (usually 2 to 4 weeks postoperatively) to prevent wound dehiscence.
Summary of Landmark Literature and Clinical Guidelines
The clinical guidelines and surgical rationale underpinning the anterior thoracic corpectomy are deeply rooted in decades of landmark orthopedic and neurosurgical literature. Historically, the management of ventral thoracic pathology was fraught with high morbidity. The pivotal work by Hodgson and Stock in the 1950s and 60s regarding the anterior approach for tuberculous spondylitis (Pott’s disease) laid the anatomical and technical groundwork for modern anterior spinal surgery, demonstrating that direct debridement and structural grafting yielded superior clinical outcomes compared to posterior laminectomy.
In the realm of spinal oncology, the Weinstein-Boriani-Biagini (WBB) surgical staging system and the Tomita classification have been instrumental in guiding the surgical approach. These frameworks emphasize that tumors occupying the anterior compartments of the vertebral body are best accessed via an anterior or combined approach to achieve oncological margins and protect the spinal cord. Furthermore, the Spine Oncology Study Group (SOSG) developed the Spinal Instability Neoplastic Score (SINS), which helps identify patients with impending or actual biomechanical failure who require structural stabilization, often via an anterior corpectomy and cage reconstruction.
For spinal trauma, the Thoracolumbar Injury Classification and Severity Score (TLICS) provides an evidence-based algorithm for operative intervention. While many burst fractures can be managed with posterior-only percutaneous or open constructs, literature by authors such as Kaneda and McAfee has extensively validated the anterior approach for severe burst fractures with significant canal compromise and anterior column comminution. Their studies demonstrated that anterior decompression and stabilization effectively restore canal diameter, maintain sagittal alignment, and yield excellent long-term neurological recovery rates, cementing the anterior thoracic corpectomy as a masterclass procedure in the armamentarium of the modern spine surgeon.
