Total En Bloc Spondylectomy: A Masterclass in Oncologic Spinal Resection
Key Takeaway
This masterclass details Total En Bloc Spondylectomy (TES), a meticulous technique for complete spinal tumor resection. Fellows will learn preoperative planning, precise patient positioning, and granular intraoperative steps for en bloc laminectomy and corpectomy. We cover critical anatomy, neurovascular risks, instrument use, and strategies for managing potential pitfalls and postoperative complications, ensuring comprehensive oncologic control and spinal stability.
Comprehensive Introduction and Patho-Epidemiology
Total En Bloc Spondylectomy (TES) represents one of the most intellectually demanding and technically unforgiving procedures in the armamentarium of the modern spine surgeon. Historically, the surgical management of primary vertebral malignancies and aggressive solitary metastases was fraught with compromise. Surgeons frequently relied upon intralesional curettage or piecemeal excision, techniques that, while seemingly less morbid in the short term, fundamentally violate the core tenets of oncologic surgery. The primary disadvantage of these historical approaches is the extraordinarily high risk of microscopic and macroscopic tumor cell contamination within the surgical bed. Because it is virtually impossible to reliably demarcate neoplastic tissue from healthy bone and epidural soft tissue during a piecemeal resection, residual tumor burden is almost inevitable. This invariably leads to catastrophic local recurrence, neurological compromise, and diminished overall survival.
The paradigm shifted dramatically with the pioneering work of Dr. Katsuro Tomita and his colleagues, who conceptualized and operationalized the TES procedure. The philosophical underpinning of TES is the achievement of a true en bloc resection—excising the entire tumor-bearing vertebral segment, inclusive of its primary mass and any satellite microlesions within that defined anatomical compartment, as a single, contiguous, unviolated specimen. By treating the vertebra as an isolated oncologic compartment, the surgeon aims to achieve wide or, at minimum, marginal surgical margins. This radical departure from intralesional debulking is paramount for minimizing local recurrence, preserving long-term neurological function, and maximizing the patient's overall survival trajectory.
Understanding the patho-epidemiology of the lesions amenable to TES is critical for appropriate application. Primary malignant bone tumors of the spine, such as chordomas, chondrosarcomas, Ewing sarcomas, and osteosarcomas, are relatively rare but exhibit aggressive local behavior. Chordomas, for instance, are notorious for their relentless local tissue destruction and high recurrence rates if the pseudocapsule is breached during resection. Similarly, aggressive benign lesions like giant cell tumors (Campanacci Grade III) or aneurysmal bone cysts with extensive osseous destruction can behave with pseudomalignant local aggression, justifying an en bloc approach to prevent inevitable recurrence.
Furthermore, the role of TES in metastatic spine disease has evolved. While systemic therapy and stereotactic radiosurgery (SRS) have revolutionized the treatment of spinal metastases, there remains a highly selected cohort of patients with solitary, radioresistant metastatic lesions (e.g., renal cell carcinoma, thyroid carcinoma, melanoma) who possess a prolonged life expectancy and excellent performance status. In these highly specific clinical scenarios, TES can offer a potential cure or a profound extension of progression-free survival, effectively transforming a palliative situation into a definitive oncologic intervention. The decision to proceed requires a multidisciplinary consensus, weighing the significant physiological insult of the surgery against the biological behavior of the specific tumor histology.
Detailed Surgical Anatomy and Biomechanics
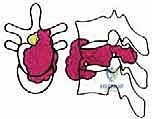
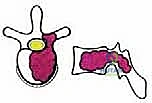
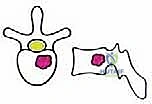
Before embarking on a TES, the surgeon must possess an encyclopedic understanding of spinal anatomy, specifically conceptualizing the vertebra as an independent oncologic unit governed by natural anatomical barriers. The success of an en bloc resection hinges entirely on the surgeon's ability to navigate outside these barriers without breaching the tumor capsule. The anatomical structures surrounding the vertebra exhibit varying degrees of resistance to neoplastic invasion, dictating the feasibility of achieving clear surgical margins.
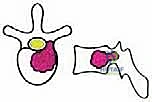
We categorize these structures into strong and weak anatomical barriers. Strong barriers are robust, avascular, or dense fibrous tissues that effectively resist tumor penetration, providing excellent planes for extralesional dissection. The Anterior Longitudinal Ligament (ALL) is a thick, formidable fibrous band that typically contains anterior tumor extension. Superiorly and inferiorly, the cartilaginous endplates and the annulus fibrosus of the intervertebral discs serve as highly effective barriers against vertical tumor spread into adjacent vertebral bodies. Preserving these structures during the osteotomy phase is critical to maintaining an intact oncologic margin.
Conversely, weak anatomical barriers offer minimal resistance to tumor progression and are frequently compromised early in the disease process. The Posterior Longitudinal Ligament (PLL), while a theoretical barrier, is significantly thinner than the ALL and is easily breached by aggressive neoplasms, leading to epidural extension and spinal cord compression. Similarly, the lateral periosteum of the vertebral body is remarkably thin; tumors readily penetrate this layer to invade the paravertebral musculature, the psoas, or the thoracic cavity. During dissection, extreme caution must be exercised when mobilizing tissues adjacent to these weak barriers to avoid inadvertent intralesional entry.
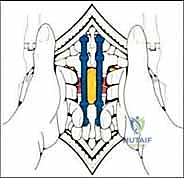
Neurovascular anatomy presents the most formidable challenge during TES. The spinal cord must be meticulously protected throughout the 360-degree mobilization of the tumor-bearing segment. The segmental arteries, which arise from the aorta and supply both the vertebral body and the spinal cord via radiculomedullary branches (most notably the Artery of Adamkiewicz), must be identified, isolated, and ligated bilaterally. Preoperative angiography is indispensable for mapping this vascular network. Biomechanically, the excision of an entire vertebral segment creates massive instability. The surgeon must be prepared to reconstruct the anterior column with a structural cage or allograft capable of withstanding immense axial loads, while simultaneously restoring the posterior tension band with rigid, multi-level pedicle screw instrumentation to prevent catastrophic hardware failure or post-operative kyphosis.

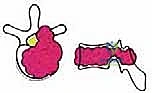
Exhaustive Indications and Contraindications
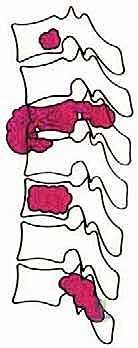
Patient selection is the absolute determinant of success or failure in TES. The physiological toll of this procedure is immense, and it must only be offered to patients whose oncologic prognosis justifies the perioperative risk. We primarily consider TES for Enneking Stage I (inactive) or II (active) primary malignant tumors, and Stage III (aggressive) benign tumors where intralesional excision carries an unacceptably high recurrence rate. For metastatic disease, indications are strictly limited to isolated, solitary lesions in patients with a highly favorable prognostic profile, typically defined by a long anticipated life expectancy, absence of visceral metastases, and an Eastern Cooperative Oncology Group (ECOG) Performance Status of 0 to 1.

To systematize decision-making, we rely heavily on Tomita's Surgical Classification of Spinal Tumors, which categorizes lesions based on their anatomical growth pattern. TES is highly recommended for Type 3 (lamina extension), Type 4 (spinal canal extension), and Type 5 (paravertebral extension) lesions. In these configurations, the tumor is often anatomically amenable to en bloc resection while permitting the preservation of critical neurovascular structures. The procedure is relatively indicated for Type 1 (vertebral body only) and Type 2 (pedicle extension) lesions, though less morbid alternatives such as stereotactic radiosurgery or isolated corpectomy must be carefully weighed against the morbidity of a total spondylectomy.

Contraindications to TES are absolute and must be rigorously respected to avoid catastrophic outcomes. Type 7 lesions, characterized by multiple vertebral involvement or skip lesions, are an absolute contraindication for en bloc resection; these patients are better served by systemic therapies, palliative decompression, or hospice care. Furthermore, extensive invasion of major anterior vascular structures (such as the aorta or inferior vena cava) that precludes safe mobilization, or bilateral encasement of the spinal cord by a radioresistant tumor, generally renders an en bloc margin impossible and contraindicates the procedure.
| Clinical Parameter | Indications for TES | Contraindications for TES |
|---|---|---|
| Tumor Pathology | Primary malignancies (Chordoma, Chondrosarcoma); Aggressive benign (GCT); Solitary radioresistant metastasis | Multi-level metastatic disease; Highly radiosensitive tumors (Myeloma, Lymphoma) |
| Tomita Classification | Types 3, 4, 5 (and highly selected 1, 2, 6) | Type 7 (Multiple vertebral involvement) |
| Patient Status | ECOG 0-1; KPS > 80; Life expectancy > 1-2 years | ECOG > 2; Poor physiological reserve; Widespread visceral metastases |
| Anatomical Limits | Resectable epidural extension; Mobilizable great vessels | Unresectable aortic/IVC invasion; Bilateral cord encasement |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous, obsessive preoperative planning is the cornerstone of a successful TES. The surgeon must mentally execute the entire operation before making the incision, anticipating every anatomical challenge and strategizing the approach. Comprehensive imaging is non-negotiable. High-resolution Computed Tomography (CT) provides exquisite detail of bony destruction, cortical breaches, and is essential for precise templating of pedicle screws and planning the exact trajectory of the osteotomies. Magnetic Resonance Imaging (MRI) is the gold standard for evaluating soft tissue involvement, defining the extent of epidural tumor, assessing spinal cord compression, and visualizing the integrity of the anatomical barriers (ALL, PLL, and discs).
A critical, often underappreciated step is preoperative spinal angiography. This study maps the segmental arterial supply to the tumor and identifies the Artery of Adamkiewicz to mitigate the risk of anterior spinal artery syndrome. Once the feeding vessels are identified, preoperative embolization is performed, typically 24 to 48 hours prior to surgery. The segmental arteries immediately above and below the affected vertebra, along with the primary tumor feeders, are selectively embolized using particles or coils. This intervention dramatically reduces intraoperative blood loss—a major cause of morbidity in highly vascular tumors like renal cell metastases or aneurysmal bone cysts—allowing the surgeon to operate in a clear, controlled field.
Patient positioning requires exactitude. The patient is placed in the prone position on a specialized radiolucent frame, most commonly the Relton-Hall four-poster frame or an equivalent Jackson table setup. The primary objective is to allow the abdomen to hang completely free. Any abdominal compression increases intra-abdominal pressure, which retrogradely engorges the epidural venous plexus (Batson's plexus). Engorged epidural veins turn a delicate spinal cord mobilization into a hemorrhagic nightmare. By ensuring a free abdomen, venous ooze is minimized, vastly improving visualization during the critical phases of dural separation.
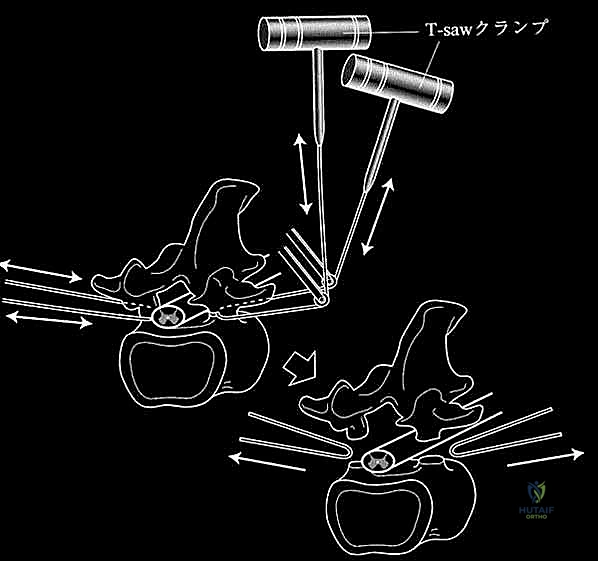
Intraoperative neuromonitoring is mandatory. Continuous Somatosensory Evoked Potentials (SSEPs) and Motor Evoked Potentials (MEPs) provide real-time feedback regarding the functional integrity of the spinal cord. Baseline signals must be established before positioning, and any degradation of signals during tumor manipulation, vessel ligation, or spinal column reconstruction must be immediately communicated to the surgical team. A C-arm fluoroscopy unit is positioned for seamless AP and lateral imaging to confirm vertebral levels, guide pedicle screw insertion, and verify the trajectory of the T-saw or osteotomes during the en bloc resection.
Step-by-Step Surgical Approach and Fixation Technique
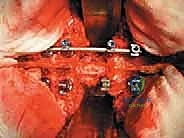
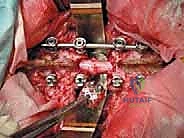
The surgical execution of TES is dictated by the tumor's location and extent. For lesions above L4 without massive anterior vascular encasement, a single posterior approach is preferred, allowing 360-degree resection through one incision. The procedure begins with a robust posterior exposure. A midline incision is extended three levels above and below the pathology. Subperiosteal dissection of the paraspinal musculature is performed with extreme care; if a prior biopsy tract exists, it is circumscribed and resected en bloc with the posterior elements to prevent tumor seeding. Pedicle screws are placed multiple levels above and below the tumor to establish the foundation for the eventual posterior tension band reconstruction.
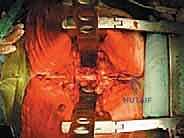
The Posterior Dissection and Osteotomy
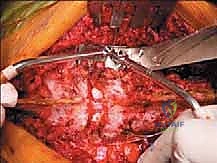
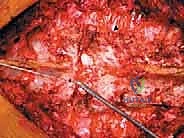
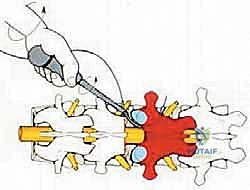
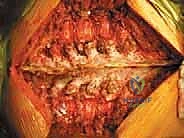
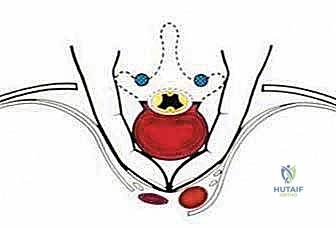
The critical phase of separating the posterior elements from the anterior column begins with the pediculotomy. Using a high-speed burr or a Tomita T-saw (a flexible, multi-stranded wire saw), the pedicles of the tumor-bearing vertebra are transected. If the tumor involves the pedicles, the osteotomy must be extended wider into the adjacent healthy bone to maintain an oncologic margin. Following pediculotomy, a wide laminectomy is performed on the adjacent superior and inferior vertebrae to expose the dura mater and the exiting nerve roots. The nerve roots at the level of the tumor are meticulously isolated and, depending on the spinal level (e.g., thoracic spine), may be ligated and divided to facilitate complete mobilization of the dural sac.
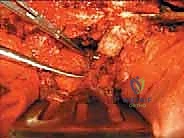
Anterior Mobilization and Corpectomy
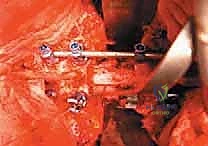
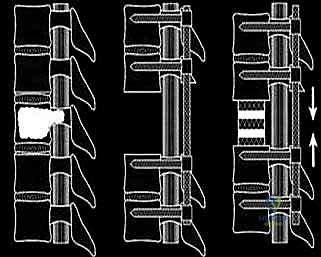
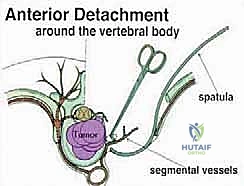
Once the posterior elements are removed and the dura is entirely free, attention turns to the anterior column. Working through the posterior approach, blunt dissection is carried out laterally along the vertebral body, gently sweeping the pleura (in the thoracic spine) or the psoas muscle (in the lumbar spine) away from the lateral periosteum. The segmental vessels are identified, double-ligated, and divided. A specialized spatulated retractor is carefully advanced anterior to the vertebral body to protect the aorta and vena cava. The intervertebral discs above and below the tumor are then incised. Using cobb elevators, osteotomes, or a T-saw passed anteriorly, the entire tumor-bearing vertebral body is detached from the adjacent endplates and delivered en bloc through the posterior incision.

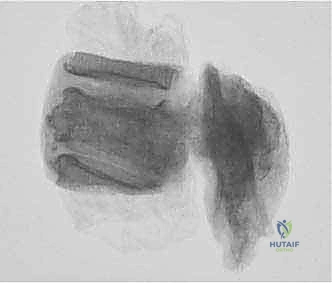
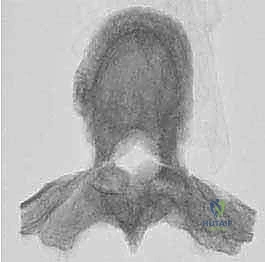
Reconstruction and Stabilization
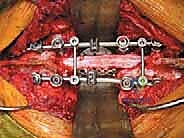
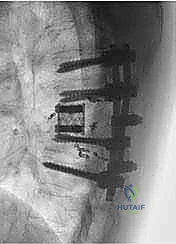
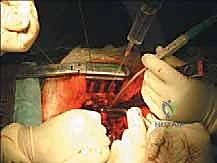
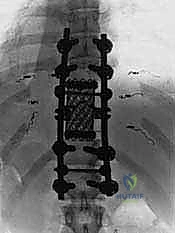
The resulting massive structural void must be meticulously reconstructed. An expandable titanium cage or a customized structural allograft is sized and inserted into the anterior defect. It is imperative that the endplates of the adjacent healthy vertebrae are meticulously prepared to prevent cage subsidence while avoiding violation of the subchondral bone, which could compromise structural integrity. Once the anterior column support is perfectly positioned, compressive forces are applied through the posterior pedicle screw construct. This locks the anterior cage in place and restores the physiological lordosis or kyphosis of the spine. Two rigid rods are secured, and cross-links are added to maximize torsional rigidity.

Complications, Incidence Rates, and Salvage Management
The magnitude of a Total En Bloc Spondylectomy intrinsically carries a high complication profile. Surgeons undertaking this procedure must be adept not only at its execution but also at the rapid identification and management of catastrophic intraoperative and postoperative events. Massive hemorrhage is the most immediate threat, particularly during the anterior mobilization phase if the epidural venous plexus is engorged or if a segmental vessel is avulsed near its aortic origin. Meticulous preoperative embolization significantly mitigates this risk, but the surgical team must have rapid transfusion protocols, cell salvage technology, and advanced hemostatic agents immediately available.
Neurological injury is a devastating complication, occurring either through direct mechanical trauma during dural mobilization or secondary to ischemic insult following the sacrifice of critical radiculomedullary arteries. Continuous neuromonitoring is our primary defense. If MEPs or SSEPs drop during vessel clamping, the surgeon must immediately unclamp the vessel and elevate the mean arterial pressure to enhance collateral perfusion. Dural tears are also common, given the intimate adherence of some tumors to the dura mater. Any durotomy must be primarily repaired with non-absorbable suture and reinforced with a dural sealant and fascial patch to prevent postoperative cerebrospinal fluid (CSF) fistulas, which can lead to meningitis or wound breakdown.
Mechanical complications, including instrumentation failure, cage subsidence, and pseudoarthrosis, are significant long-term risks due to the massive biomechanical stresses placed on the reconstructed segment. Meticulous endplate preparation and the use of multi-rod constructs can reduce these risks. Furthermore, surgical site infections (SSI) are notably high in this population, exacerbated by prolonged operative times, extensive soft tissue dissection, and the immunocompromised state of many oncologic patients. Aggressive prophylactic antibiotics, meticulous dead-space management, and the use of intrawound vancomycin powder are standard preventive measures.

| Complication Category | Specific Complication | Estimated Incidence | Salvage / Management Strategy |
|---|---|---|---|
| Vascular/Hemorrhagic | Massive intraoperative bleeding | 15 - 30% | Pre-op embolization; Cell saver; Rapid massive transfusion protocol; Direct packing |
| Neurological | Spinal cord ischemia/injury | 5 - 15% | Maintain MAP > 85 mmHg; Intraoperative neuromonitoring; Avoid blind retractor placement |
| Dural/CSF | Incidental durotomy / CSF leak | 10 - 20% | Primary watertight suture repair; Dural sealants; Lumbar drain placement if refractory |
| Mechanical/Hardware | Cage subsidence / Rod fracture | 10 - 25% | Multi-rod posterior constructs; Meticulous endplate preparation; Revision surgery for pseudoarthrosis |
Phased Post-Operative Rehabilitation Protocols
The postoperative management of a TES patient is as critical as the surgical execution, requiring a highly coordinated, multidisciplinary approach. Immediately following surgery, the patient is transferred to the Surgical Intensive Care Unit (SICU) for rigorous hemodynamic and neurological monitoring. Mean arterial pressure (MAP) is strictly maintained above 85 mmHg to ensure adequate spinal cord perfusion, particularly in the setting of ligated segmental vessels. The patient is typically kept on flat bed rest for the first 24 to 48
Clinical & Radiographic Imaging Archive