Lumbar Spinal Decompression: An Intraoperative Masterclass for Fellows
Key Takeaway
This masterclass guides fellows through lumbar spinal decompression, addressing neurogenic claudication and radiculopathy. We cover comprehensive anatomy, precise patient positioning, and meticulous intraoperative execution using various approaches like laminotomy, laminectomy, and microdecompression. Critical pearls, potential pitfalls, and structured postoperative management are detailed to ensure optimal patient outcomes and surgical mastery.
Comprehensive Introduction and Patho-Epidemiology
Fellows, welcome to the operating theater. Today, we are addressing a ubiquitous yet highly nuanced pathology: lumbar spinal stenosis. This masterclass is designed to provide you with a comprehensive, exhaustive understanding of the disease process, transitioning from foundational pathoanatomy to the granular nuances of surgical execution and postoperative care. Our primary operative objective is the complete, safe, and definitive decompression of the neural elements, thereby restoring neurologic function and dramatically improving our patients' quality of life. The surgical management of lumbar spinal stenosis is a cornerstone of orthopedic spine surgery, requiring both a profound understanding of biomechanics and meticulous surgical technique.
Lumbar spinal stenosis arises predominantly from the inexorable degenerative changes inherent to the aging process, classically described by the Kirkaldy-Willis degenerative cascade. These biomechanical alterations lead to a progressive narrowing of the spinal canal, the subarticular zones (lateral recesses), and the neural foramina. This volumetric reduction physically compresses the delicate neurologic tissues and their microvascular supply. The underlying pathophysiologic mechanism involves a slow, gradual compromise of blood supply—specifically, the restriction of arterial inflow and the engorgement of venous outflow—to the traversing and exiting nerve roots. Furthermore, the free flow of cerebrospinal fluid, which provides vital nutritional support within the common dural sac, is mechanically obstructed.
Clinically, this ischemic and mechanical compromise manifests in highly predictable patterns. Patients typically present with neurogenic claudication in approximately 70% of cases. This is characterized by bilateral or unilateral leg pain, numbness, or weakness that is reliably exacerbated by walking or prolonged standing. Crucially, these symptoms are relieved by sitting or lumbar flexion (the "shopping cart sign"), which transiently increases the cross-sectional area of the spinal canal. This phenomenon occurs because the compromised microvasculature cannot meet the increased metabolic demands of the nerve roots during activity, leading to the accumulation of noxious metabolic byproducts.
Alternatively, monoradiculopathy is observed in roughly 15% of cases, where symptoms are strictly confined to a single nerve root distribution, almost always due to focal, severe compression in the lateral recess or foramen. The remaining 15% of patients present with a complex combination of both claudication and radiculopathy. Foraminal stenosis, specifically accounting for 10% to 15% of cases, is best diagnosed clinically by severe, unrelenting monoradiculopathy of an exiting nerve root. Radiographically, we meticulously evaluate this on parasagittal MRI imaging or CT sagittal reconstructions, looking for the obliteration of perineural fat.
Detailed Surgical Anatomy and Biomechanics
Before we even consider making an incision, a profound, three-dimensional understanding of the lumbar spine's anatomy and the pathogenesis of stenosis is absolutely paramount. You must be able to visualize the structures beneath the skin before the knife touches the patient.
The Functional Vertebral Unit
Observe the classic functional vertebral unit, often referred to as the three-joint complex. This is the fundamental biomechanical building block of the spine. The anterior column consists of the vertebral bodies and the intervertebral disc, serving as the primary load-bearing structures. The intervertebral disc provides critical cushioning and flexibility; however, degenerative changes here, such as desiccation, loss of disc height, and annular bulging, are significant anterior contributors to central and lateral recess stenosis.
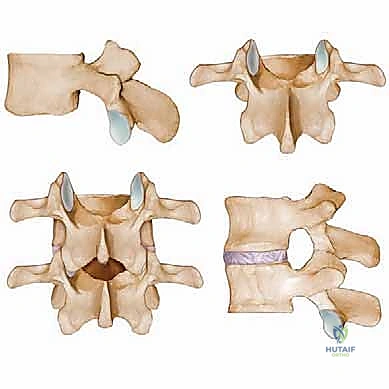
Posteriorly, the pedicles connect the vertebral body to the posterior elements, acting as the structural pillars of the neural arch. The laminae form the posterior roof of the spinal canal, protecting the thecal sac. Hypertrophy of the lamina itself, particularly in cases of congenital short-pedicle stenosis, can act as a direct, unyielding compressive force. The facet joints, or zygapophyseal joints, are formed by the articulation of the superior articular process of the caudal vertebra and the inferior articular process of the cephalad vertebra. Degenerative hypertrophy, capsular thickening, and osteophyte formation at these joints are the most common causes of subarticular and foraminal stenosis.
Connecting these bony elements is the ligamentum flavum, a robust, highly elastic ligament spanning the interlaminar space. In the degenerative spine, loss of disc height leads to buckling and infolding of the ligamentum flavum. Concurrently, it undergoes true hypertrophy and fibrotic transformation. This thickened, buckled ligament is frequently the primary dorsal compressive pathology in central canal stenosis. Understanding the spatial relationship between the disc anteriorly, the facets laterally, and the ligamentum flavum posteriorly is essential for executing a precise, targeted decompression.
Muscular and Neurovascular Considerations
Our standard posterior approach primarily involves dissecting through the midline dorsolumbar fascia and subsequently elevating the paraspinal musculature. The multifidus muscle is intimately associated with the spinous processes and laminae; we must meticulously elevate it from medial to lateral using subperiosteal dissection to minimize denervation and ischemic muscle injury. For the intertransverse or Wiltse approach, we must accurately identify the avascular cleavage plane between the multifidus medially and the longissimus laterally.
Neurovascular risks are omnipresent during this procedure. The dura mater and the traversing/exiting nerve roots are the primary structures we are attempting to liberate. Meticulous, controlled technique is required to prevent iatrogenic dural tears or direct thermal/mechanical nerve root injury. A dural tear can lead to devastating complications, including persistent cerebrospinal fluid leaks, pseudomeningocele formation, intracranial hypotension, and a significantly increased risk of deep surgical site infection. Furthermore, a rich, valveless epidural venous plexus (Batson's plexus) surrounds the thecal sac. These veins can bleed profusely when decompressed, rapidly obscuring the surgical field. Careful hemostasis with bipolar electrocautery and proactive patient positioning to keep the abdomen free are critical to mitigating venous engorgement.
Exhaustive Indications and Contraindications
Patient selection is arguably the most critical determinant of surgical success. Decompression is a highly effective procedure, but only when applied to the correct clinical scenario. We must rigorously evaluate each patient against established indications and contraindications.
Indications for Surgical Intervention
The primary indication for lumbar spinal decompression is debilitating neurogenic claudication or radiculopathy that has proven refractory to a comprehensive, prolonged course of conservative management. This conservative regimen should typically span a minimum of 12 weeks and include targeted physical therapy, non-steroidal anti-inflammatory medications, activity modification, and potentially fluoroscopically guided epidural steroid injections.
Absolute indications for emergent or urgent surgical intervention include the development of cauda equina syndrome—characterized by saddle anesthesia, bowel or bladder dysfunction, and profound bilateral lower extremity weakness—or the presence of a rapidly progressive, profound motor deficit, such as a foot drop. In these scenarios, the ischemic window of the neural elements is closing rapidly, and immediate decompression is mandatory to prevent permanent, irreversible neurologic catastrophe.
Absolute and Relative Contraindications
Conversely, isolated lumbar decompression is contraindicated in several specific clinical scenarios. Absolute contraindications include active systemic or local spinal infections (unless the decompression is part of an irrigation and debridement procedure) and severe medical comorbidities that render the patient unfit for general anesthesia.
Relative contraindications revolve primarily around the presence of underlying spinal instability. If a patient presents with significant mechanical back pain coupled with dynamic instability on flexion-extension radiographs (e.g., a mobile degenerative spondylolisthesis with greater than 3mm of translation), an isolated decompression may exacerbate the instability, leading to failed back surgery syndrome. In such cases, a concomitant instrumented fusion is generally required. Furthermore, patients with predominant, severe axial back pain without significant radicular or claudicatory symptoms are generally poor candidates for isolated decompression, as the procedure is designed to relieve leg symptoms, not mechanical back pain.
| Category | Specific Clinical Scenarios | Clinical Rationale / Management Strategy |
|---|---|---|
| Primary Indications | Refractory neurogenic claudication; Persistent radiculopathy | Failure of >12 weeks of structured conservative care; Significant quality of life impairment. |
| Emergent Indications | Cauda equina syndrome; Rapidly progressive motor deficit | Requires immediate surgical decompression to salvage neurologic function and prevent permanent paralysis/incontinence. |
| Absolute Contraindications | Active systemic infection; Unacceptable anesthetic risk | Medical optimization required; Surgery deferred until medically stable. |
| Relative Contraindications | Dynamic overt instability; Predominant axial back pain | High risk of iatrogenic destabilization; Concomitant spinal fusion typically indicated. |
Pre-Operative Planning, Templating, and Patient Positioning
Fellows, meticulous preoperative planning is entirely non-negotiable. It dictates our surgical strategy, anticipates anatomical anomalies, and is the primary bulwark against intraoperative complications.
Advanced Imaging and Surgical Templating
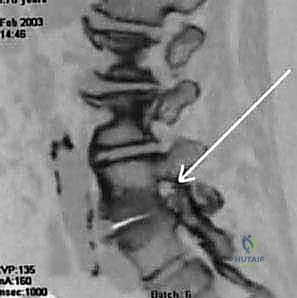
Magnetic Resonance Imaging (MRI) remains the gold standard and the imaging modality of choice for diagnosing and anatomically classifying spinal canal stenosis. It excels at visualizing soft tissue contributors to stenosis, including desiccated discs, synovial cysts, hypertrophied ligamentum flavum, and the precise morphology of the thecal sac and nerve roots. We systematically evaluate the axial and sagittal sequences to classify the stenosis based on its vertical extent, the specific regions involved (central, lateral recess, foraminal), and its severity (mild, moderate, severe).
Computed Tomography (CT) Myelography, while invasive, offers unparalleled, high-resolution visualization of the osseous anatomy and the dynamic nature of the stenosis. It is particularly indispensable in patients with contraindications to MRI (e.g., incompatible pacemakers), patients with severe scoliosis where MRI slices may miss the orthogonal plane of the canal, or in revision cases where extensive epidural fibrosis obscures the MRI interpretation. Plain radiographs, including upright anteroposterior, lateral, and dynamic flexion-extension views, are mandatory to assess for global coronal and sagittal balance, and to rule out dynamic instability such as degenerative spondylolisthesis or lateral listhesis.
Patient Positioning and Operating Room Setup
Proper patient positioning is the first step of the operation and sets the stage for a successful decompression. We utilize prone positioning on a specialized radiolucent spinal frame, typically a Jackson table with a Wilson frame or specialized chest and hip pads.

The hips and knees must be gently flexed. This crucial maneuver serves two primary purposes: first, it decreases the natural lumbar lordosis, which physically opens the interlaminar spaces and provides a wider, safer trajectory to the spinal canal. Second, it places the posterior ligamentous complex on stretch, facilitating our interlaminar approach. Most importantly, the abdomen must hang completely free and uncompressed. Any intra-abdominal pressure will be transmitted directly to the inferior vena cava, forcing venous return through the epidural Batson's plexus. This leads to massive epidural venous engorgement, resulting in torrential intraoperative bleeding that obscures the surgical field and increases the risk of neural injury.
Once positioned, meticulous padding is applied to all bony prominences and peripheral nerves. The eyes must be checked to ensure zero direct pressure, preventing catastrophic ischemic optic neuropathy. The arms are positioned with the shoulders abducted less than 90 degrees to prevent brachial plexus traction injuries, and the ulnar nerves are carefully padded at the cubital tunnel. Finally, precise fluoroscopic localization is performed using a spinal needle to definitively mark the operative levels, a critical step to absolutely eliminate the risk of wrong-site surgery.
Step-by-Step Surgical Approach and Fixation Technique
Now, fellows, we transition to the operative execution. This is where anatomical knowledge meets surgical precision.
Incision, Exposure, and Soft Tissue Dissection
Following standard sterile prep and drape, I make a precise, direct midline posterior skin incision over the targeted levels. For a single-level pathology, this typically extends approximately 1.5 to 2 inches, spanning from the cephalad aspect of the superior spinous process to the caudal aspect of the inferior spinous process. Using electrocautery, we sharply divide the subcutaneous tissues down to the thick, pearlescent dorsolumbar fascia, maintaining strict hemostasis throughout.
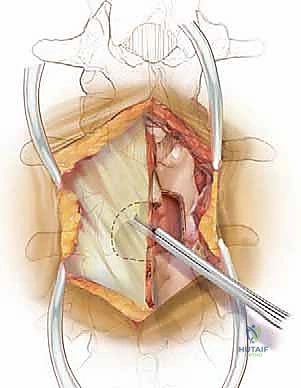
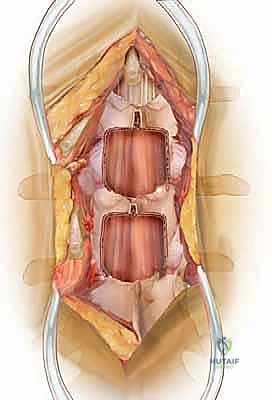
The dorsolumbar fascia is incised precisely in the avascular midline raphe. Using a Cobb elevator, we perform a meticulous subperiosteal dissection of the paraspinal muscles—primarily the multifidus—off the spinous processes and laminae. We sweep the muscle laterally, progressing to the medial border of the facet joints bilaterally. It is imperative to stay strictly subperiosteal; straying into the muscle belly causes unnecessary bleeding, denervation, and postoperative pain. Once adequate exposure is achieved, self-retaining retractors (such as Gelpi or cerebellar retractors) are placed deeply and securely to maintain the operative corridor.
The Interlaminar Window and Bony Decompression
With the posterior elements exposed, we bring in the operative microscope or utilize high-powered surgical loupes with a dedicated headlight. The magnification and illumination are critical for the delicate work ahead. Our standard workhorse approach is the interlaminar window, utilized for the vast majority of central and lateral recess stenosis cases.
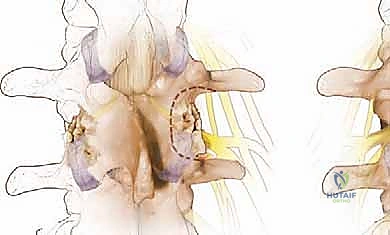
We begin the bony decompression with a high-speed pneumatic burr utilizing a 3mm or 4mm matchstick diamond bit. Under constant, copious irrigation to prevent thermal necrosis of the bone and underlying dura, we systematically thin the caudal aspect of the superior lamina and the cephalad aspect of the inferior lamina. We use a gentle, sweeping "painting" motion, avoiding downward pressure. The goal is to thin the lamina to a translucent cortical shell. Once thinned, we use a small angled curette to separate the ligamentum flavum from the undersurface of the bone, and then carefully resect the remaining bony shell using a 2mm or 3mm Kerrison rongeur.
Medial Facetectomy and Foraminotomy
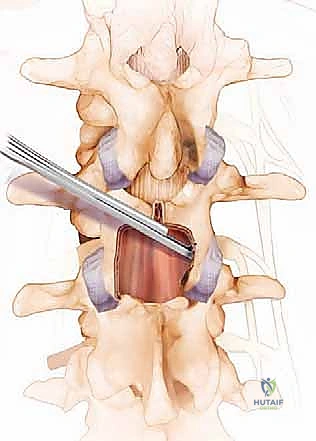
After the central laminotomy is complete, we must address the lateral recess and the neural foramina. This requires a medial facetectomy. Using the Kerrison rongeur or the high-speed burr, we carefully resect the medial aspect of the superior articular process, which forms the dorsal roof of the lateral recess.
CRITICAL PEARL: You must strictly limit your medial facetectomies to less than 50% of the facet joint bilaterally. Resecting more than 50% of the pars interarticularis or the facet joint compromises the structural integrity of the functional vertebral unit, leading to an unacceptably high risk of postoperative iatrogenic instability and spondylolisthesis, which would subsequently necessitate a revision fusion surgery.
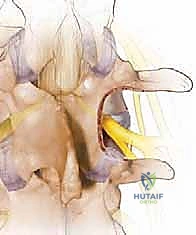
Once the medial facet is resected, the hypertrophied ligamentum flavum is exposed in its entirety. Using a combination of curettes and Kerrison rongeurs, the ligament is carefully detached from its lateral and superior insertions and excised en bloc or piecemeal. Only after the ligament is removed do we clearly visualize the traversing nerve root in the lateral recess and the exiting nerve root in the foramen. We confirm adequate decompression by passing a blunt nerve hook (such as a Woodson elevator) along the course of the nerve root; it should pass freely without resistance out into the extraforaminal zone.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, complications can and do occur. A master surgeon is defined not just by their ability to execute the procedure, but by their capacity to anticipate, recognize, and definitively manage these adverse events.
Intraoperative Complications
The most common intraoperative complication during lumbar decompression is an incidental durotomy, or dural tear, occurring in approximately 3% to 10% of primary cases and up to 20% in revision surgeries due to epidural fibrosis. If a tear occurs, it must be recognized immediately. The primary management is a watertight, primary repair using a 4-0 or 5-0 non-absorbable suture (e.g., Nurolon or Prolene) in a running or locked fashion. Following repair, a Valsalva maneuver to 40 cm H2O is performed by anesthesia to confirm a watertight seal. We frequently augment the repair with a synthetic dural sealant or a fibrin glue matrix, and occasionally a fascial or fat graft.
Nerve root injury is a devastating but rare complication (less than 1%). It typically occurs due to overzealous retraction, thermal injury from the burr, or direct laceration with a Kerrison rongeur. To prevent this, always ensure the footplate of the Kerrison is visibly clear of neural tissue before biting. Epidural bleeding from Batson's plexus can be brisk; it is managed systematically with bipolar electrocautery, hemostatic agents (such as Gelfoam soaked in thrombin, or flowable hemostatic matrices), and gentle tamponade with neurosurgical cottonoids.
Postoperative Complications
Postoperatively, patients must be monitored for the development of an epidural hematoma. While rare (less than 0.5%), a compressive hematoma presents with out-of-proportion pain and new-onset, progressive neurologic deficits. This is a surgical emergency requiring immediate return to the operating room for evacuation.
Surgical site infections occur in 1% to 2% of cases. Superficial infections can often be managed with targeted oral antibiotics, but deep infections extending to the fascial layer or epidural space necessitate prompt surgical irrigation and debridement followed by prolonged intravenous antibiotic therapy. Long-term complications include iatrogenic instability (if the >50% facet rule is violated) and adjacent segment disease, both of which may require future fusion procedures.
| Complication | Estimated Incidence | Prevention and Salvage Management |
|---|---|---|
| Incidental Durotomy | 3% - 10% (Primary) | Avoid plunging with instruments. Primary repair with 4-0 suture, Valsalva check, dural sealant augmentation. |
| Epidural Hematoma | < 0.5% | Meticulous hemostasis prior to closure. Emergent surgical evacuation if progressive neurologic deficit occurs. |
| Surgical Site Infection | 1% - 2% | Strict sterile technique, prophylactic antibiotics. Deep infections require urgent surgical I&D and IV antibiotics. |
| Iatrogenic Instability | 2% - 5% | Preserve >50% of the facet joint and pars interarticularis. Requires revision instrumented spinal fusion. |
Phased Post-Operative Rehabilitation Protocols
The surgery is only half the battle; a structured, phased rehabilitation protocol is essential to maximize functional outcomes and ensure long-term success.
Immediate Postoperative Phase
In the immediate postoperative period (Days 0-14), the focus is on early mobilization, adequate pain control, and complication prevention. Patients are typically mobilized on the day of surgery or postoperative day one with the assistance of physical therapy. We emphasize early ambulation to mitigate the risks of deep vein thrombosis (DVT), pulmonary embolism, and postoperative atelectasis.
Pain management transitions rapidly from intravenous to oral analgesics, utilizing a multimodal approach including acetaminophen, NSAIDs (if no fusion was performed), muscle relaxants, and short courses of oral opioids. Patients are instructed in proper body mechanics, specifically avoiding repetitive bending, lifting (greater than 10 pounds), and twisting (the "BLT" restrictions) to allow the paraspinal musculature and fascial incisions to heal. Wound care involves keeping the incision clean and dry until the sutures or staples are removed at two weeks.
Subacute and Long-Term Rehabilitation
Once the soft tissues have achieved primary healing (Weeks 2-6), patients transition to a formal, outpatient physical therapy program. The initial focus is on neural mobilization and gentle core stabilization exercises. We aim to activate the transversus abdominis and multifidus muscles to provide dynamic stability to the lumbar spine, compensating for any minor structural alterations caused by the decompression.
From weeks 6 to 12, the rehabilitation protocol intensifies, focusing on progressive resistance training, cardiovascular conditioning (such as stationary cycling or aquatic therapy), and functional restoration. The ultimate goal is to return the patient to their baseline activities of daily living and recreational pursuits without restriction. Patients are educated that while the radicular leg pain typically resolves rapidly, the numbness and motor weakness may take several months to improve, and the axial back pain will require ongoing core maintenance.
Summary of Landmark Literature and Clinical Guidelines
To practice evidence-based medicine, fellows must be intimately familiar with the landmark literature that guides our decision-making in lumbar spinal stenosis.
The Spine Patient Outcomes Research Trial (SPORT), led by Weinstein et al., is the most comprehensive prospective study evaluating the surgical versus non-operative management of lumbar spinal stenosis. The SPORT data definitively demonstrated that patients with symptomatic spinal stenosis treated with surgical decompression experienced significantly greater improvement in pain, function, and quality of life compared to those treated non-operatively, and this surgical advantage was maintained at the 4-year and 8-year follow-up intervals.
Similarly, the Maine Lumbar Spine Study, a highly respected prospective cohort study, corroborated these findings, showing that surgical patients had greater improvements in leg pain, back pain, and functional status at 10 years compared to the conservative management cohort.
Current clinical guidelines from the North American Spine Society (NASS) strongly recommend surgical decompression for patients with severe, symptomatic lumbar spinal stenosis who have failed a trial of conservative care. Furthermore, the guidelines emphasize that for patients with pure stenosis and no evidence of dynamic instability or significant deformity, isolated decompression without fusion is the gold standard, offering excellent clinical outcomes with lower morbidity, shorter operative times, and reduced healthcare costs compared to decompression with concomitant fusion. As surgeons, we must lean on this robust literature to guide our hands and counsel our patients effectively.
