Posterior Thoracic & Lumbar Spine Approaches: An Intraoperative Masterclass
Key Takeaway
Join our intraoperative masterclass on posterior thoracic and lumbar spine approaches. We meticulously cover surgical anatomy, patient positioning, and step-by-step execution for both midline and paraspinal techniques. Learn to identify critical landmarks, manage neurovascular risks, and master instrument use. This guide includes extensive pearls, pitfalls, and post-operative considerations for optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues, to the operating theater. Today, we delve into the intricacies of posterior thoracic and lumbar spine approaches, undeniably the foundational cornerstone of modern spinal surgery. The posterior approach is the workhorse of the spine surgeon’s armamentarium, offering unparalleled access to the posterior neural elements, the facet complexes, and the pedicles for rigid instrumentation. This is not merely an exercise in making a linear incision; it is a highly choreographed sequence of anatomical dissection that requires a profound understanding of three-dimensional regional anatomy, the anticipation of biomechanical consequences, and the execution of each step with absolute precision and purpose. Mastery of this approach is the prerequisite for treating the vast majority of spinal pathologies encountered in clinical practice.
Historically, the posterior approach has evolved from radical, muscle-stripping exposures that often left patients with debilitating iatrogenic flat-back syndrome and paraspinal muscle necrosis, to the refined, anatomically respectful techniques we employ today. Early iterations of posterior spinal surgery prioritized bony exposure above all else, frequently sacrificing the dynamic stabilizing function of the erector spinae and multifidus musculature. Today, our understanding of spino-pelvic biomechanics dictates a tissue-sparing philosophy. We recognize that the posterior tension band is not expendable; it is a critical component of sagittal balance and long-term functional recovery. The evolution of our surgical technique reflects a paradigm shift from purely structural mechanical fixation to biological preservation.
The epidemiology of spinal disorders dictating this approach is vast, encompassing degenerative disc disease, spinal stenosis, adult spinal deformity, traumatic fractures, and oncological resections. As our aging population continues to expand, the incidence of degenerative lumbar spondylosis and concomitant central canal or lateral recess stenosis has surged, making mastery of this approach more critical than ever for the practicing orthopedic surgeon. Furthermore, the rising prevalence of adult spinal deformity (ASD) demands a surgical approach capable of facilitating complex multi-level osteotomies and the placement of robust, rigid instrumentation to restore global sagittal and coronal alignment. The posterior approach remains the gold standard for achieving these ambitious reconstructive goals.
Furthermore, the patho-epidemiology of spinal disorders necessitates an approach that can be tailored to the specific pathology at hand. A simple microdiscectomy requires a vastly different exposure magnitude compared to a multi-level pedicle subtraction osteotomy (PSO) for rigid sagittal imbalance. Therefore, the modern spine surgeon must not only know how to expose the spine but must understand the physiological cost of that exposure. Every millimeter of lateral dissection, every second of self-retaining retractor ischemia, and every vessel coagulated carries implications for postoperative recovery, adjacent segment biomechanics, and long-term functional outcomes. Let us scrub in and examine the comprehensive mastery required for these procedures.
Detailed Surgical Anatomy and Biomechanics
Before a scalpel is even requested, a comprehensive, three-dimensional understanding of the regional anatomy is absolutely paramount. This anatomical knowledge serves as your intraoperative roadmap, guiding every dissection plane, facilitating rapid exposure, and, most importantly, protecting vital neural and vascular structures from iatrogenic injury. The posterior spine is a complex layered environment where osteology, myology, and neurology intersect.
Superficial and Intermediate Myofascial Layers
The posterior approach begins with an appreciation of the superficial landmarks and the initial myofascial layers. Proximally, the prominent spinous processes of C7 (vertebra prominens) and T1 serve as highly reliable starting points for clinical level determination in the upper thoracic spine. Distally, the intercrestal line—an imaginary horizontal line connecting the superior-most aspects of the iliac crests—consistently approximates the L4-L5 interspace or the L4 spinous process. While these gross determinations are crucial for initial skin incision planning, it is a rigid axiom of spine surgery that intraoperative fluoroscopy must always be utilized to provide definitive anatomical confirmation prior to any irreversible surgical step. Relying solely on palpation is a documented pathway to wrong-level surgery.
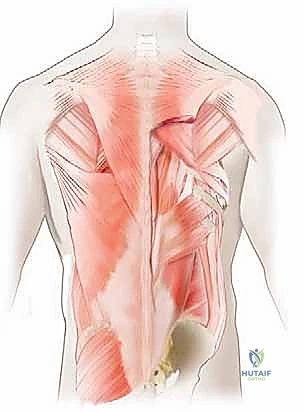
Beneath the subcutaneous adipose tissue lies the thoracolumbar fascia, a complex, multi-layered aponeurotic structure that plays a critical role in load transfer between the spine, pelvis, and lower extremities. The musculature beneath this fascia is organized into three distinct functional layers. The superficial layer includes the trapezius, latissimus dorsi, rhomboid major and minor, and levator scapulae. These muscles are appendicular in function, primarily governing shoulder and upper extremity kinematics. Their innervation is derived from anterior rami and cranial nerves (e.g., the spinal accessory nerve for the trapezius), meaning they are not at risk of denervation during a standard posterior midline approach.
Progressing deeper, the intermediate layer consists of the serratus posterior superior and inferior, as well as the levatores costarum. These are accessory muscles of respiration. Similar to the superficial layer, their innervation is derived from anterior rami (intercostal nerves), keeping them relatively safe during our midline dissection. During a standard posterior midline approach, these superficial and intermediate layers are largely reflected laterally as a single musculofascial unit, revealing the true paraspinal musculature beneath. Meticulous hemostasis during the division of the thoracolumbar fascia minimizes dead space and reduces the incidence of postoperative seroma formation.
Deep Paraspinal Musculature and Neurovascular Supply
The deep layer is the critical anatomical zone we interact with directly. It is comprised of the erector spinae group (iliocostalis, longissimus, spinalis), the transversospinalis group (semispinalis, multifidus, rotatores), and the deepest segmental muscles (interspinales and intertransversarii). The multifidus is of particular importance in the lumbar spine; its fibers originate from the spinous processes and insert onto the mammillary processes of vertebrae two to four levels below. Preserving the multifidus and its neurovascular supply is paramount to preventing postoperative paraspinal atrophy and chronic axial back pain. The surgeon must utilize a strict subperiosteal dissection technique to elevate these muscles off the bony elements without violating their intramuscular substance.
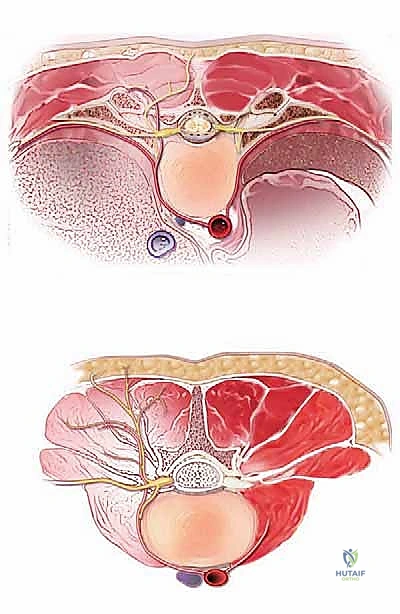
These deep muscles receive their nervous supply segmentally from the posterior dorsal rami of the spinal nerves. The medial branch of the dorsal ramus innervates the multifidus and the facet joints, while the lateral branch supplies the longissimus and iliocostalis. This segmental innervation provides a degree of redundancy, allowing us to elevate and retract these muscles without causing widespread denervation, provided the retraction is intermittent and not excessively forceful. Prolonged, high-pressure retraction is a known cause of ischemic necrosis and denervation of the multifidus, leading to poor functional outcomes despite a technically successful decompression or fusion. Retractors should be relaxed every 30 to 45 minutes to allow for tissue reperfusion.
Vascularly, the deep muscle layers are supplied by dorsal branches of the segmental arteries, which originate directly from the aorta. These vessels pierce the intertransverse ligaments and enter our operative field. They can be a source of significant, persistent bleeding if not meticulously identified and controlled with bipolar electrocautery. Furthermore, the extensive valveless epidural venous plexus (Batson's plexus) communicates freely with the basivertebral veins and the systemic circulation. Intra-abdominal pressure directly correlates with engorgement of this plexus; thus, meticulous patient positioning to allow the abdomen to hang free is a critical hemostatic maneuver that cannot be overstated.
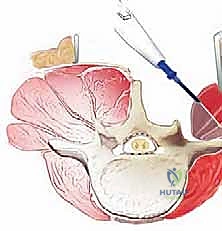
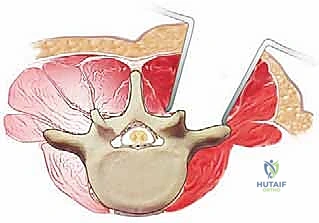
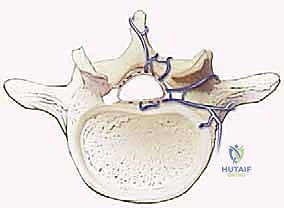
Osteology and Ligamento-Capsular Complexes
As the subperiosteal dissection progresses laterally, the osteology of the posterior elements becomes the primary focus. The spinous processes, laminae, pars interarticularis, and facet joints form the protective osseous roof over the neural elements. The facet joint capsules possess a distinct, shiny white, fibrous appearance. It is a fundamental rule that unless a specific segment is targeted for arthrodesis, extreme care must be taken to avoid violating these capsular fibers. Iatrogenic disruption of a facet capsule at an adjacent segment is a primary driver of adjacent segment disease (ASD) and subsequent iatrogenic instability, often necessitating revision surgery.

The ligamentum flavum, with its characteristic yellow appearance due to a high elastin content, connects the laminae of adjacent vertebrae. Its fibers run in a predominantly cephalad-caudad direction. Understanding its complex attachments is key for safe and efficient decompression. The cephalad end has a broad insertion from the base of the spinous process extending to the anterior surface of the superior lamina. The caudad end inserts onto the superior edge of the inferior lamina. Hypertrophy of this ligament, combined with facet arthropathy and disc bulging, represents the classic triad contributing to central canal and lateral recess stenosis.
Special anatomical considerations must be noted depending on the patient demographic and specific spinal level. At the L5-S1 interspace, the interlaminar window is naturally widened, and the posterior bony anatomy (spina bifida occulta) may be incompletely formed. This anatomical variation necessitates extreme caution during the initial midline dissection to prevent inadvertent plunging into the spinal canal and catastrophic injury to the thecal sac. Conversely, in pediatric patients undergoing deformity correction, the spinous process apophyses have not yet fused. During dissection, the surgeon must carefully split this cartilaginous apophysis down to the bone and elevate it in continuity with the paraspinal musculature to preserve future growth potential and minimize postoperative hematoma.
Exhaustive Indications and Contraindications
The decision to utilize a posterior approach to the thoracic and lumbar spine is dictated by a careful synthesis of the patient's clinical presentation, radiographic findings, and overall physiological reserve. Patient selection is arguably the most critical determinant of surgical success. A technically flawless operation executed for the wrong indication will invariably yield a poor clinical outcome, leading to the dreaded "failed back surgery syndrome." The surgeon must act as an astute diagnostician, ensuring that the patient's subjective complaints correlate precisely with objective imaging and neurophysiological data.
Ideal candidates for posterior decompression and/or instrumented fusion are those presenting with refractory neurogenic claudication, progressive radicular motor deficits, or mechanical back pain associated with gross radiographic instability (e.g., isthmic or degenerative spondylolisthesis) that has failed exhaustive non-operative management. Non-operative modalities, including targeted physical therapy, non-steroidal anti-inflammatory drugs (NSAIDs), and fluoroscopically guided epidural steroid injections, should typically be trialed for a minimum of 6 to 12 weeks. The exception to this rule is the presence of an acute neurological emergency, such as cauda equina syndrome, progressive myelopathy, or an acute, profound motor deficit, which mandates emergent surgical decompression.
Conversely, the surgeon must be acutely aware of contraindications. Active systemic infection, uncorrected coagulopathies, and severe medical comorbidities that preclude safe induction of general anesthesia are absolute contraindications. Relative contraindications require nuanced clinical judgment. For instance, severe osteoporosis significantly increases the risk of pedicle screw pullout, subsidence, and construct failure. In such cases, the surgeon must evaluate bone mineral density via DEXA scan or measure Hounsfield units on a preoperative CT. If surgery is unavoidable, the operative plan must incorporate cement augmentation of screws, the use of expandable pedicle screws, or extending the fusion construct to incorporate multiple points of fixation to distribute biomechanical stress.
Furthermore, the psychological profile of the patient cannot be ignored. Active substance abuse, severe untreated psychiatric illness, and secondary gain issues (such as pending litigation or workers' compensation claims) represent significant relative contraindications. These factors have been repeatedly shown in the orthopedic literature to negatively impact postoperative rehabilitation and patient-reported outcome measures (PROMs). A multidisciplinary approach, often involving pain psychology and physical medicine and rehabilitation (PM&R), is essential for optimizing these complex patients prior to offering surgical intervention.
| Category | Indications for Posterior Approach | Contraindications |
|---|---|---|
| Degenerative | Spinal stenosis with neurogenic claudication, degenerative spondylolisthesis, massive/recurrent disc herniations. | Predominantly axial back pain without instability (relative), severe osteoporosis without optimization (relative). |
| Trauma | Unstable burst fractures, flexion-distraction injuries (Seatbelt/Chance), fracture-dislocations. | Stable compression fractures without neurological deficit, isolated spinous process fractures. |
| Deformity | Adult degenerative scoliosis, adolescent idiopathic scoliosis (AIS), Scheuermann's kyphosis, sagittal imbalance. | Fixed anterior bony ankylosis requiring anterior release first (relative), inability to tolerate massive blood loss. |
| Oncology/Infection | Epidural spinal cord compression (ESCC) from metastasis, primary bone tumors, epidural abscesses requiring evacuation. | Terminal illness with life expectancy < 3 months (relative), active systemic sepsis precluding surgery. |
| General/Systemic | Need for multi-level rigid posterior instrumentation and deformity correction. | Uncorrected coagulopathy, medical unfitness for prone positioning and general anesthesia (absolute). |
Pre-Operative Planning, Templating, and Patient Positioning
Proper preoperative planning and meticulous patient positioning are non-negotiable elements of spine surgery. They optimize surgical exposure, minimize intraoperative complications, dictate the efficiency of the procedure, and represent the foundational steps toward a successful clinical outcome. The modern spine surgeon does not "figure it out" in the operating room; the entire procedure is mentally executed and digitally templated long before the first incision is made.
Advanced Imaging and Pre-Surgical Templating
Before the patient ever enters the operating room, the surgeon must have exhaustively reviewed all relevant imaging modalities. Standard weight-bearing anteroposterior (AP) and lateral radiographs, including full-length 36-inch cassettes, are essential for assessing global spino-pelvic alignment. Parameters such as Pelvic Incidence (PI), Pelvic Tilt (PT), Lumbar Lordosis (LL), and Sagittal Vertical Axis (SVA) must be calculated. A fundamental principle of modern deformity surgery is ensuring that any planned fusion restores the critical PI-LL mismatch to within 10 degrees, thereby minimizing the risk of postoperative sagittal imbalance and adjacent segment breakdown.
Advanced imaging, including thin-cut Computed Tomography (CT) and Magnetic Resonance Imaging (MRI), provides the granular detail required for surgical execution. CT is invaluable for templating the appropriate size, length, and trajectory of pedicle screws. It allows the surgeon to identify anatomical variations such as dysplastic pedicles, anomalous vertebral artery routing in the upper thoracic spine, or spina bifida occulta. Furthermore, preoperative Hounsfield unit measurements on CT can reliably predict regional bone mineral density, guiding the decision to use fenestrated screws for cement augmentation.
MRI remains the gold standard for evaluating the neural elements, assessing the degree of central and foraminal stenosis, and identifying the presence of perineural cysts or conus medullaris abnormalities. The surgeon must correlate the MRI findings with the patient's specific radicular symptoms to ensure the planned decompression is targeted and sufficient. In complex revision cases, a preoperative CT myelogram may be necessary if metallic artifact from previous instrumentation obscures the MRI, allowing for accurate visualization of the thecal sac and exiting nerve roots amidst the hardware.
Patient Positioning and Operating Room Setup
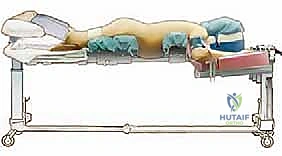
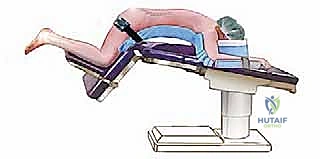
Positioning the patient prone for a posterior spine procedure is a critical, high-risk phase of the operation that requires the coordinated effort of the entire surgical and anesthesia team. The patient is typically intubated on the transport stretcher and then carefully log-rolled onto the operating table while maintaining strict spinal precautions. For multi-level fusions and deformity corrections, an open Jackson-style radiolucent frame is preferred. This specialized table allows for unobstructed AP and lateral fluoroscopic imaging throughout the procedure and facilitates the dynamic restoration of lumbar lordosis by manipulating the patient's hip extension.
A rigorous checklist for prone positioning must be strictly adhered to. The cervical spine must be maintained in a neutral alignment to prevent hyperextension injuries, which can stretch the brachial plexus or compromise cerebral venous outflow. The upper extremities are positioned on padded arm boards at 90 degrees or less of shoulder abduction and forward flexion. This specific angulation minimizes the likelihood of rotator cuff impingement and reduces traction on the brachial plexus. Crucially, the axilla must be completely clear of any resting pressure to prevent devastating compressive brachial plexopathies.
The chest and iliac crests are supported by specialized pads, ensuring that the abdomen hangs entirely free. As previously discussed, abdominal compression increases intra-abdominal pressure, which translates directly to the epidural venous plexus, resulting in catastrophic intraoperative bleeding. Finally, strict attention must be paid to the eyes and face. Direct pressure on the globes can lead to Postoperative Visual Loss (POVL), a devastating complication caused by central retinal artery occlusion or ischemic optic neuropathy. The head must be supported in a specialized foam prone-view helmet, and the eyes must be checked repeatedly throughout the case by the anesthesia team.
Step-by-Step Surgical Approach and Fixation Technique
The execution of the posterior approach requires a methodical, step-wise progression. Each phase of the operation builds upon the success of the previous one. From the initial skin incision to the final tightening of the set screws, the surgeon must maintain an unwavering focus on anatomical landmarks, hemostasis, and the protection of neural elements.
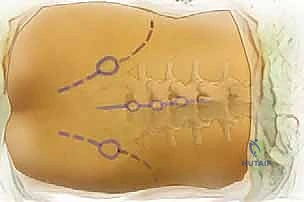
Incision and Subperiosteal Dissection
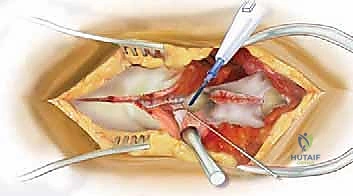
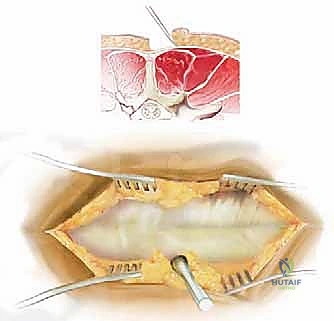
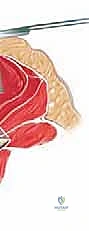
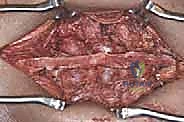
The procedure begins with a precise midline incision, utilizing the spinous processes as a palpable guide. The incision is carried down through the subcutaneous tissues to the thoracolumbar fascia. Hemostasis is achieved meticulously with electrocautery. The fascial incision is made strictly in the midline, directly over the tips of the spinous processes, dividing the supraspinous ligament. A Cobb elevator or a Bovie electrocautery device is then used to initiate the subperiosteal dissection. The key to a bloodless and tissue-sparing exposure is staying strictly beneath the periosteum, sweeping the multifidus and erector spinae musculature laterally off the spinous processes and laminae.
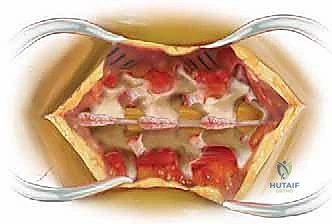
The lateral extent of the exposure is dictated by the goals of the surgery. For a simple laminectomy, exposure to the medial aspect of the facet joints is sufficient. However, for a posterolateral instrumented fusion, the dissection must be carried laterally over the facet joints to expose the transverse processes. This lateral exposure is critical for providing an adequate bed for the bone graft and for identifying the starting points for pedicle screw insertion. During this lateral dissection, the surgeon must identify and cauterize the dorsal branches of the segmental vessels to prevent pooling of blood in the lateral gutters.
Once adequate exposure is achieved, self-retaining retractors (such as a Weitlaner, cerebellar, or specialized spine retractors like the McCulloch or Taylor) are placed. It is imperative to place the retractor blades carefully beneath the muscle mass, avoiding direct pressure on the facet capsules of adjacent, non-fused levels. As previously noted, the surgeon must be mindful of retractor-induced ischemia. Periodically releasing the tension on the retractors during a long case allows for reperfusion of the paraspinal musculature, significantly reducing the risk of postoperative
Clinical & Radiographic Imaging Archive