Spine Cases: Consider the Patient for Accurate Diagnosis
Key Takeaway
Discover the latest medical recommendations for Spine Cases: Consider the Patient for Accurate Diagnosis. Spondylolysis in adolescent athletes, like c the patient, is a common cause of low back pain, often linked to repetitive hyperextension activities and involving a pars fracture or stress reaction. Initial treatment typically consists of 3-6 months of nonoperative care, including a rigid lumbar brace (e.g., Boston flexion type) to manage pain and support healing, while avoiding extension exercises.
Detailed Patient Presentation and Mechanism of Injury
The patient initially presented to our orthopedic clinic as a 15-year-old high school football linebacker complaining of an insidious onset of lower back pain that had been progressively worsening over the preceding six months. As is highly characteristic of adolescent athletes engaged in collision sports, his pain was initially mild, intermittent, and exclusively present at the conclusion of football games or heavy tackling drills. However, the temporal profile of his symptoms escalated, transitioning into a more frequent and persistent dull ache that began to interfere with his daily activities and athletic performance. He explicitly denied any objective weakness, numbness, or tingling in his bilateral lower extremities at the time of his initial presentation. He did, however, report a deep, non-radiating pain localized to the left posterior buttock during periods of peak symptom intensity, which is a classic manifestation of sclerotomal referred pain originating from the posterior spinal elements rather than true radiculopathy.
To fully appreciate the etiology of this patient's condition, one must critically analyze the biomechanical demands placed upon a football linebacker. The position requires explosive bursts of speed, sudden changes in direction, and, most importantly, repetitive hyperextension combined with axial loading and rotational torque. When an athlete assumes a tackling stance and subsequently drives upward and backward, the lumbar spine is forced into extreme extension. This repetitive cyclical loading concentrates immense shear stress directly across the pars interarticularis—the narrow isthmus of bone connecting the superior and inferior articular processes of the facet joints. In the skeletally immature adolescent, this region is particularly vulnerable to fatigue failure. The repetitive microtrauma overwhelms the osteoblastic reparative capacity of the bone, leading to a stress reaction and, ultimately, a frank stress fracture, clinically recognized as spondylolysis.
Despite a comprehensive initial consultation wherein the diagnosis of bilateral L5 pars defects was established and a strict non-operative protocol was recommended, the patient and his family unfortunately refused the proposed treatment plan. He was lost to follow-up and continued to participate in high-impact sports without any bracing or activity modification. This non-compliance set the stage for the natural progression of the disease process. The untreated bilateral pars fractures failed to achieve bony union, resulting in a fibrous pseudoarthrosis. Over time, the structural integrity of the posterior tension band was compromised, allowing the anterior shear forces—driven by the patient's body weight and pelvic morphology—to cause progressive anterior translation of the L5 vertebral body over the S1 sacral promontory.
Five years later, the patient, now a 20-year-old collegiate athlete, presented back to our clinic with a drastically altered clinical picture. He was now experiencing severe, unrelenting, and constant pain in his lower back that severely limited his mobility. Crucially, the pain character had evolved; he now reported sharp, shooting, electric-like pain radiating from his lower back, deep into his left posterior buttock, and extending down the lateral aspect of his left thigh and calf. This transition from purely axial mechanical pain to a profound radicular symptom complex indicates that the underlying pathology had progressed from a localized bony defect to a structural instability causing direct neural compression. The progressive anterior slip of L5 on S1 had led to dynamic foraminal stenosis, trapping the exiting L5 nerve root against the fibrocartilaginous mass of the pars nonunion and the superior endplate of S1.
Comprehensive Clinical Examination Findings
Upon his return to the clinic at age 20, a rigorous and systematic physical examination of the spine and lower extremities was conducted. Observation of the patient's standing posture immediately revealed several compensatory mechanisms characteristic of advanced isthmic spondylolisthesis. He exhibited a classic Phalen-Dickson sign, characterized by a knee-flexed, hip-flexed gait with a profoundly flattened lumbar lordosis and a vertical sacrum. This postural adaptation is primarily driven by severe hamstring spasm, a reflexive response attempting to posteriorly tilt the pelvis to reduce the lumbosacral shear angle and stabilize the unstable L5-S1 segment. When viewed from the sagittal plane, a distinct "step-off" deformity was palpable at the lumbosacral junction. The spinous process of L5 remained posterior (attached to the loose neural arch), while the anterior column had translated forward, creating a noticeable structural void above the sacrum.
Provocative clinical testing further localized the pathology. The single-leg hyperextension test (Stork test), which was likely positive during his initial presentation at age 15, was now exquisitely painful and practically impossible for the patient to perform due to severe mechanical instability and paraspinal muscle guarding. Palpation over the L5-S1 facet joints and the midline supraspinous ligament elicited focal tenderness. Furthermore, assessing the patient's range of motion revealed a severe restriction in forward flexion, limited not by true articular stiffness, but by the profound tightness of the hamstring musculature. Extension of the lumbar spine exacerbated his radicular symptoms, a hallmark sign of dynamic foraminal narrowing occurring as the posterior disc height collapses and the pars defect compresses the exiting nerve root.
The neurological examination was pivotal in delineating the extent of his pathology. Unlike his initial presentation five years prior, the patient now exhibited objective neurological deficits localizing to the left L5 nerve root. Manual muscle testing revealed a grade 4/5 weakness in the left extensor hallucis longus (EHL) and mild weakness in ankle dorsiflexion, indicating motor compromise of the L5 distribution. Sensory examination demonstrated a distinct area of hypesthesia and dysesthesia over the lateral aspect of the left calf and the dorsum of the left foot, perfectly mapping to the L5 dermatome. Deep tendon reflexes were assessed; the patellar reflex (L4) and Achilles reflex (S1) were symmetric and graded 2+ bilaterally, further isolating the lesion to the L5 nerve root specifically, which does not have a primary reflex associated with it in standard clinical testing.
To ensure a comprehensive evaluation and rule out concurrent hip pathology—which frequently mimics lumbosacral radiculopathy—a thorough hip examination was performed. The FABER (Flexion, Abduction, External Rotation) and FADIR (Flexion, Adduction, Internal Rotation) tests were negative for groin pain, effectively ruling out intra-articular hip joint pathology such as femoroacetabular impingement or labral tears. Additionally, palpation of the greater trochanter and sacroiliac joints did not reproduce his primary symptom complex. The culmination of these physical findings—a palpable step-off, profound hamstring tightness, and an isolated left L5 radiculopathy—painted a definitive clinical picture of a progressive isthmic spondylolisthesis with concomitant foraminal stenosis.
Advanced Imaging and Diagnostics
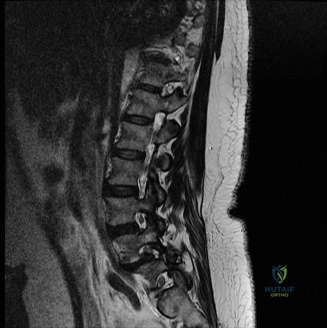
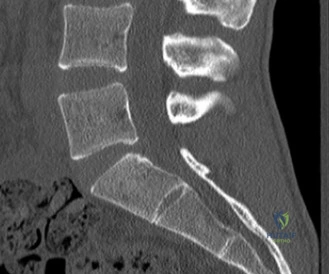
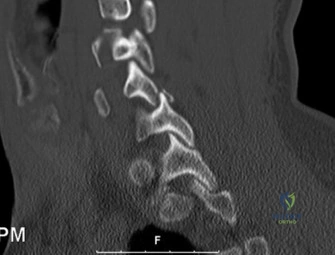
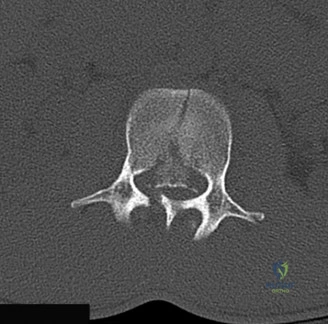
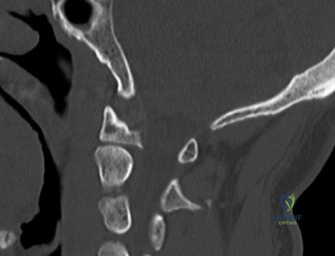
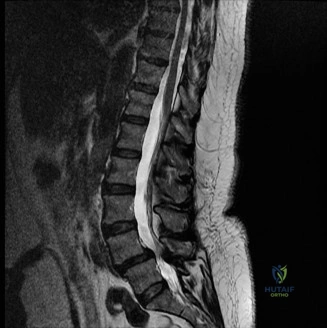
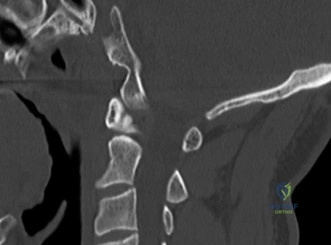
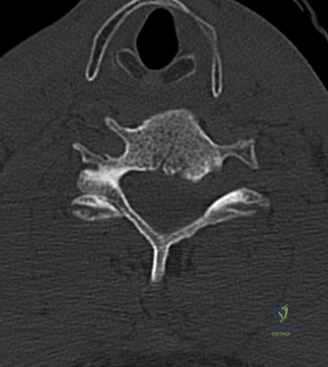
The diagnostic imaging workup for this patient spanned his initial presentation at age 15 and his subsequent return at age 20, providing a textbook illustration of the natural history of untreated isthmic spondylolysis. During his initial visit, standard weight-bearing anteroposterior (AP), lateral, and bilateral oblique radiographs of the lumbar spine were obtained. The lateral radiograph demonstrated a normal lumbar lordosis and preserved L5-S1 disc space height, with no evidence of anterior translation (spondylolisthesis). However, the oblique radiographs revealed the pathognomonic "collar on the Scotty dog" sign. The "Scotty dog" represents the posterior elements of the vertebra, where the nose is the transverse process, the eye is the pedicle, the front leg is the inferior articular process, and the neck is the pars interarticularis. A radiolucent line across the neck confirmed the presence of bilateral L5 pars defects.
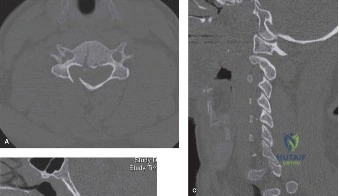
Had the patient been compliant with follow-up during his initial presentation, advanced imaging such as a Single Photon Emission Computed Tomography (SPECT) scan or a Magnetic Resonance Imaging (MRI) scan with Short Tau Inversion Recovery (STIR) sequences would have been indicated. These modalities are highly sensitive for detecting acute stress reactions and marrow edema in the pars interarticularis before a frank fracture line becomes visible on plain radiographs. An MRI at that early stage would have likely shown high signal intensity in the bilateral L5 pars regions on T2-weighted STIR images, confirming an active, metabolically "hot" lesion that possessed a high potential for healing with rigid bracing.
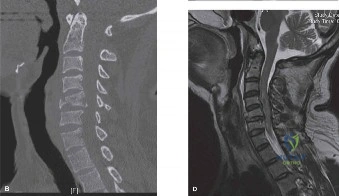
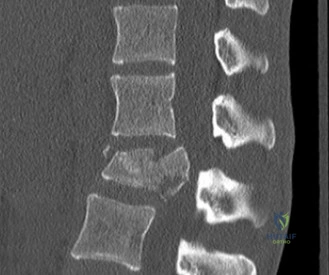
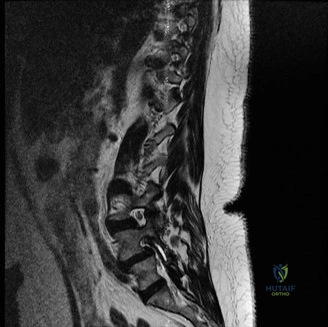
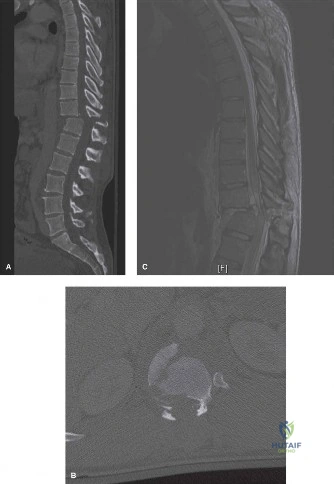
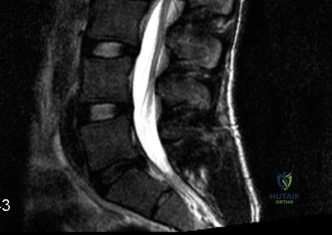
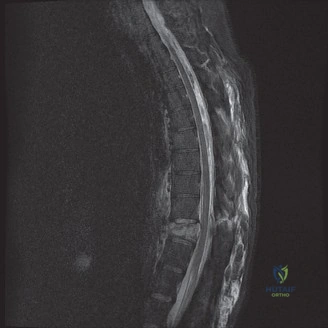
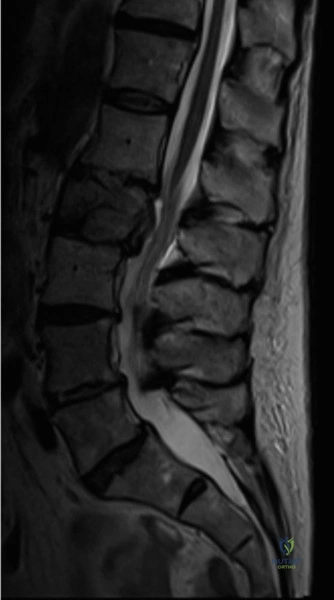
Upon his return five years later, the imaging profile was drastically different. Standing lateral radiographs now revealed a Meyerding Grade 2 spondylolisthesis at L5-S1, indicating that the L5 vertebral body had translated anteriorly by 25% to 50% over the sacral promontory. Furthermore, there was a noticeable collapse of the L5-S1 intervertebral disc space, signifying advanced degenerative changes secondary to the altered biomechanics and chronic instability. We also meticulously measured the patient's spinopelvic parameters on a standing 36-inch scoliosis cassette. His Pelvic Incidence (PI) was measured at 65 degrees (high), which mathematically dictates a higher Sacral Slope (SS) and Pelvic Tilt (PT). A high PI is a known biomechanical risk factor for the progression of spondylolisthesis, as it creates a steeper lumbosacral shear angle, placing immense anterior translational force across the compromised L5-S1 segment.
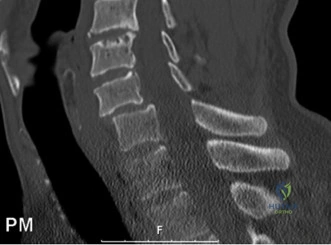
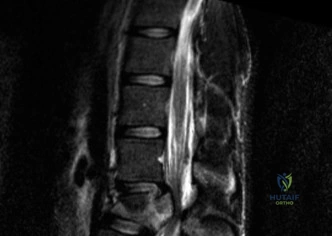
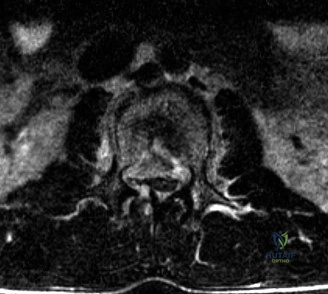
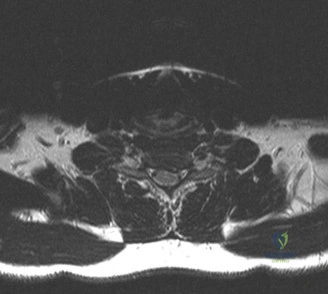
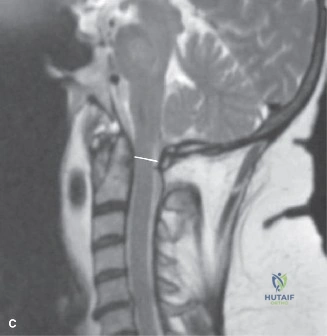
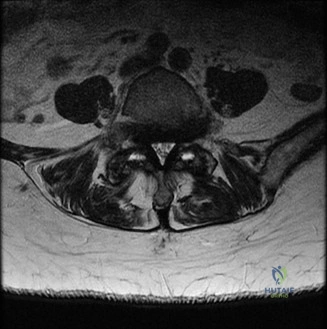
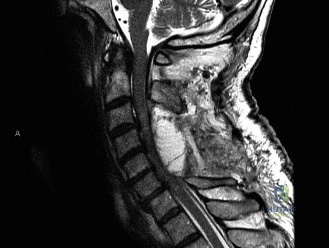
To evaluate the neural elements and the soft tissue structures, a comprehensive MRI of the lumbar spine without contrast was obtained. The T2-weighted sagittal and axial sequences confirmed the Grade 2 anterolisthesis and demonstrated severe bilateral foraminal stenosis at L5-S1. The exiting L5 nerve roots were severely compressed between the hypertrophied, fibrocartilaginous pseudarthrosis tissue of the pars defect dorsally, and the bulging, degenerated L5-S1 disc and superior endplate of S1 ventrally. Furthermore, the MRI revealed Modic Type II changes (fatty replacement of the subchondral bone marrow) in the adjacent vertebral endplates, confirming chronic biomechanical stress and discogenic degeneration. The central canal remained relatively patent, which is characteristic of isthmic spondylolisthesis, as the posterior neural arch is left behind during the anterior translation of the vertebral body.
Exhaustive Differential Diagnosis
When evaluating a young athlete presenting with acute or subacute lower back pain, the orthopedic surgeon must maintain a broad and highly suspicious differential diagnosis. The adolescent spine is structurally unique, possessing open apophyses and highly elastic ligamentous structures, making it prone to specific injury patterns distinct from those seen in adults. While mechanical back pain from musculoligamentous strain is common, it is a diagnosis of exclusion. The clinician must systematically rule out more insidious pathologies, including infectious processes (discitis, osteomyelitis), neoplastic conditions (osteoid osteoma, osteoblastoma, Ewing sarcoma), and inflammatory arthropathies (ankylosing spondylitis, juvenile idiopathic arthritis). An osteoid osteoma, for instance, often presents with severe, unrelenting night pain that is dramatically relieved by non-steroidal anti-inflammatory drugs (NSAIDs), a clinical feature absent in our patient.
Focusing on structural and mechanical etiologies, the primary differential diagnosis for this patient's initial presentation centered around the various classifications of spondylolisthesis and discogenic pathology. The Wiltse classification of spondylolisthesis categorizes the condition into dysplastic, isthmic, degenerative, traumatic, and pathologic types. Dysplastic spondylolisthesis involves congenital anomalies of the upper sacrum or the neural arch of L5, often leading to severe slips in early childhood. Degenerative spondylolisthesis, driven by facet joint osteoarthritis and ligamentum flavum hypertrophy, is almost exclusively seen in older adults (typically >50 years old) and most commonly affects the L4-L5 level. Traumatic spondylolisthesis requires a high-energy mechanism resulting in an acute fracture of the neural arch other than the pars. Therefore, the insidious onset of pain in an adolescent athlete with repetitive hyperextension perfectly aligns with the isthmic subtype.
Another critical differential to consider in the adolescent population is a herniated nucleus pulposus (HNP). While less common in teenagers than in adults, acute disc herniations can occur in adolescent athletes, particularly those involved in heavy weightlifting or contact sports. However, a lumbar herniated disc typically presents with acute, severe lower extremity radicular pain (sciatica) that often overshadows the axial back pain. Furthermore, disc herniations frequently exacerbate with lumbar flexion (which increases intradiscal pressure) rather than extension. Our patient's initial symptoms were exclusively axial back pain worsened by extension, making a disc herniation highly unlikely at the age of 15. It was only after five years of progressive instability and structural collapse that radicular symptoms emerged.
Below is a comprehensive comparative table outlining the key differentiating features of the most common pathologies considered in this clinical scenario:
| Feature | Isthmic Spondylolysis/Listhesis | Degenerative Disc Disease (DDD) | Herniated Nucleus Pulposus (HNP) | Lumbar Spinal Stenosis |
|---|---|---|---|---|
| Typical Age of Onset | 10 - 20 years (Adolescents) | > 40 years | 30 - 50 years | > 60 years |
| Primary Mechanism | Repetitive hyperextension/shear | Age-related desiccation/wear | Acute axial load / hyperflexion | Degenerative cascade/hypertrophy |
| Pain Characteristics | Axial LBP, worse with extension | Dull, aching LBP, worse with sitting | Sharp, shooting radicular pain (sciatica) | Neurogenic claudication, heavy legs |
| Provocative Maneuvers | Positive Stork test (extension) | Pain with lumbar flexion/loading | Positive Straight Leg Raise (SLR) | Pain relieved by sitting/flexion (shopping cart sign) |
| Key Imaging Findings | Pars defect (Scotty dog), anterior slip | Disc space narrowing, osteophytes | Focal disc protrusion/extrusion on MRI | Central/foraminal narrowing, ligamentum flavum hypertrophy |
| Neurological Deficits | Often absent early; L5 radiculopathy late | Rare, unless severe collapse | Common, follows specific dermatome | Bilateral, multilevel, patchy deficits |
Complex Surgical Decision Making and Classifications
The surgical decision-making process for this patient was highly nuanced, heavily influenced by his delayed presentation, the progression of his structural deformity, and the onset of profound neurological deficits. During his initial presentation at age 15 with an isolated pars stress fracture and no evidence of spondylolisthesis, the gold standard of care was non-operative management. This typically involves a rigid anti-lordotic brace (such as a Boston overlapping brace) worn for 23 hours a day for 3 to 6 months, coupled with absolute cessation of offending sports activities. The goal of bracing in flexion is to reduce the shear forces across the pars interarticularis, theoretically allowing the osteogenic potential of the adolescent bone to bridge the fracture gap. Extension exercises are strictly contraindicated during this phase, as they exacerbate the pathological forces.
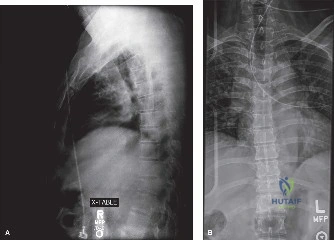
When the patient returned five years later with a Meyerding Grade 2 isthmic spondylolisthesis, severe L5 radiculopathy, and MRI evidence of L5-S1 disc degeneration, the treatment paradigm shifted entirely to surgical intervention. The Meyerding classification grades the severity of the slip based on the percentage of anterior translation of the superior vertebral body over the inferior one: Grade 1 (0-25%), Grade 2 (26-50%), Grade 3 (51-75%), Grade 4 (76-100%), and Spondyloptosis (>100%). For a symptomatic Grade 2 slip that has failed conservative management (or in this case, progressed due to non-compliance), surgical stabilization is definitively indicated. The primary goals of surgery are twofold: first, to decompress the compromised neural elements (the L5 nerve roots), and second, to achieve a solid bony arthrodesis to halt further progression and eliminate mechanical back pain.
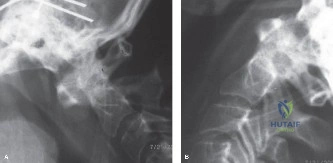
We considered several surgical options. A direct pars repair (such as the Buck, Scott, or Morscher techniques) was immediately ruled out. While pars repairs are excellent motion-preserving options for young patients with isolated pars defects and normal intervertebral discs, they are absolutely contraindicated in the presence of a spondylolisthesis greater than Grade 1 or when there is MRI evidence of disc degeneration. Attempting a pars repair in this patient would fail to address the underlying instability and the degenerated disc, inevitably leading to continued pain and hardware failure. An isolated decompression (Gill laminectomy) without fusion was also rejected. Resecting the loose neural arch removes the compressive fibrocartilaginous tissue but further destabilizes the spine. In an active 20-year-old, an isolated Gill procedure carries an unacceptably high risk of catastrophic postoperative slip progression.

Therefore, the definitive surgical plan was a posterior decompression and an L5-S1 instrumented fusion. The addition of an interbody fusion—specifically a Transforaminal Lumbar Interbody Fusion (TLIF)—was heavily debated and ultimately selected. A TLIF offers several distinct biomechanical advantages in this scenario. First, it allows for direct, thorough bilateral decompression of the L5 nerve roots by completely removing the facet joints and the compressive pars tissue. Second, placing a structural cage in the anterior column restores the collapsed disc height, which indirectly decompresses the neural foramina and helps restore regional lumbar lordosis. Finally, a 360-degree fusion (anterior interbody plus posterior posterolateral fusion) significantly increases the overall arthrodesis rate compared to an isolated posterolateral gutter fusion, a crucial consideration in a young, highly active patient with a high pelvic incidence.
Step-by-Step Surgical Technique and Intervention
The patient was brought to the operating room and placed under general endotracheal anesthesia. Following the induction of anesthesia, a comprehensive neuromonitoring array was applied, including Somatosensory Evoked Potentials (SSEPs), Motor Evoked Potentials (MEPs), and spontaneous Electromyography (sEMG) for the bilateral lower extremities, with a specific focus on the L5 and S1 nerve roots. The patient was then carefully rolled prone onto a radiolucent Jackson spinal table. Meticulous attention was paid to patient positioning; the hips and knees were slightly flexed to relax the psoas and hamstring musculature, while the abdomen was allowed to hang free to minimize intra-abdominal pressure and reduce epidural venous bleeding. The lumbar spine was positioned to optimize and maintain physiological lordosis.
A standard midline longitudinal incision was made extending from L4 to S2. Subperiosteal dissection was meticulously performed using Bovie electrocautery and Cobb elevators to strip the paraspinal musculature off the spinous processes, laminae, and facet joints of L5 and S1 bilaterally. Exposure was carried out laterally to the tips of the transverse processes of L5 and the sacral alae. Upon exposure, the classic anatomical derangement of isthmic spondylolisthesis was immediately evident. The spinous process and lamina of L5 (the "Gill body") were highly mobile, floating freely due to the bilateral pars interarticularis nonunions. A massive hypertrophic mass of fibrocartilaginous pseudarthrosis tissue was identified at the site of the pars defects, severely compressing the underlying dura and the exiting L5 nerve roots.
The decompression phase commenced with the resection of the Gill body. The ligamentum flavum was detached from the superior border of the L5 lamina and the inferior border of the L4 lamina. Using a combination of Leksell rongeurs and a high-speed matchstick burr, the loose L5 lamina was carefully excised. We then turned our attention to the neural foramina. The hypertrophic fibrocartilaginous mass at the pars defect was meticulously resected using Kerrison rongeurs. A bilateral complete facetectomy of L5-S1 was performed to facilitate the TLIF approach and ensure absolute decompression of the L5 nerve roots. The L5 roots were traced laterally well into the extraforaminal zone to ensure they were completely free from any tethering or compression. Neuromonitoring remained stable throughout this critical phase.
Following decompression, pedicle screws were placed bilaterally into the L5 and S1 pedicles using anatomical landmarks and fluoroscopic guidance. The pedicles were cannulated with a gearshift probe, palpated with a ball-tip feeler to ensure intraosseous trajectory, and tapped before screw insertion. For the TLIF, the L5-S1 disc space was accessed unilaterally on the more symptomatic left side. A thorough discectomy was performed, and the cartilaginous endplates were meticulously scraped down to bleeding subchondral bone using specialized rasps and curettes to optimize the fusion bed. The anterior disc space was packed with autologous local bone graft mixed with cellular allograft. A lordotic polyetheretherketone (PEEK) interbody cage, also packed with graft, was then impacted into the anterior third of the disc
Clinical & Radiographic Imaging Archive