Anterior Lumbar Interbody Fusion & Corpectomy: A Masterclass in Spinal Reconstruction
Key Takeaway
This masterclass provides an exhaustive, real-time guide to Anterior Lumbar Interbody Fusion (ALIF) and Corpectomy. Fellows will learn intricate surgical anatomy, meticulous patient positioning, and granular intraoperative execution, from initial incision to final hardware placement. We'll cover critical decision-making, instrument handling, and strategies for managing potential complications, ensuring a thorough understanding of this complex spinal reconstructive procedure.
Comprehensive Introduction and Patho-Epidemiology
Alright, fellows, gather 'round. Welcome to the operating theater. Today, we are tackling an Anterior Lumbar Interbody Fusion (ALIF), potentially escalating to a full corpectomy, for a patient suffering from severe, recalcitrant degenerative disc disease (DDD) and gross spinal instability. This procedure is far more sophisticated than merely placing a titanium or PEEK cage into a void; it is a masterclass in restoring native spinal biomechanics, indirectly decompressing compromised neural elements, and achieving a robust, lasting arthrodesis. Pay close attention to the nuances we discuss today, because in anterior spinal reconstruction, every millimeter and every anatomical detail dictates the line between a triumphant clinical outcome and a catastrophic complication.
Let us begin by thoroughly understanding the "why" behind this formidable procedure. Lumbar disc degeneration is an inexorable, progressive, and largely age-related physiological process. However, it is frequently accelerated and exacerbated by a confluence of factors, including post-traumatic disc herniations, micro-fractures of the vertebral endplates, occupational mechanical overload, and profound genetic predispositions. Pathologically, this cascade manifests as a precipitous loss of disc height, deleterious biochemical alterations within the nucleus pulposus and annulus fibrosus, and ultimately, a complete failure of the intervertebral segment's normal biomechanical behavior. The anterior column, which typically bears 80% of the axial load in the lumbar spine, becomes functionally incompetent.
Understanding Symptomatic Degenerative Disc Disease
While radiographic disc degeneration is ubiquitous in aging populations and frequently asymptomatic, in a specific subset of patients, these morphological changes become a profound source of debilitating pain, crossing the threshold into what we clinically term "degenerative disc disease." The precise pathophysiological divergence between symptomatic presentation and asymptomatic degeneration remains somewhat elusive, but it invariably involves a complex intersection of structural mechanical failure, neurochemical sensitization, and localized inflammatory cascades. The intervertebral disc, normally an avascular and aneural structure in its inner layers, becomes neovascularized and innervated by nociceptive nerve fibers growing inward from the outer annulus and posterior longitudinal ligament.

Key pathological manifestations are readily apparent on advanced imaging and during direct surgical visualization. Osteophyte formation—occurring anteriorly, laterally, or posteriorly—is a classic Wolff's Law response to abnormal segmental motion, often leading to acquired foraminal or central canal stenosis. Endplate changes, famously categorized by Modic, range from acute inflammatory edema (Modic Type 1) to chronic fatty replacement (Modic Type 2) and eventual dense sclerosis (Modic Type 3). Furthermore, Schmorl nodes and cystic cavities compromise the structural integrity of the subchondral bone. As the disc space collapses, the posterior facet-joint complex experiences abnormal shear forces, leading to arthropathy, capsular hypertrophy, and a profound loss of physiological motion.
The etiology of this discogenic pain specifically describes nociception originating directly from the degenerating disc itself, independent of neural compression. This pain is classically exacerbated by axial loading—such as prolonged sitting or standing with a forward-flexed posture (increasing intradiscal pressure)—and is often temporarily relieved by recumbency or positions that unload the anterior column. The mechanisms driving this include a reduced nutrient supply via the endplates, a steep decline in viable chondrocyte-like cells, and a catastrophic loss of matrix proteins, proteoglycans, and hydrophilic capacity, leading to severe desiccation. Increased degradative enzyme activity (matrix metalloproteinases) and fatigue failure of the annular collagen matrix further compound the structural collapse. Notably, chronic nicotine exposure is a proven detrimental factor, inducing microvascular ischemia and accelerating these degenerative mechanisms.
Detailed Surgical Anatomy and Biomechanics
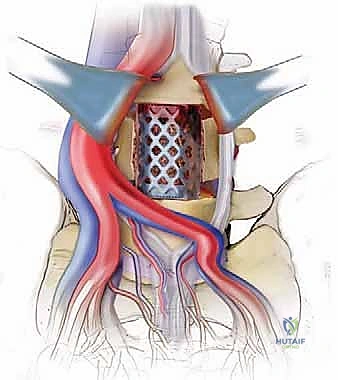
Before we even contemplate making an incision, a profound, three-dimensional understanding of the regional anatomy is absolutely paramount. We are operating in the anterior retroperitoneal space, a treacherous anatomical corridor rich with critical, life-sustaining neurovascular structures. The anterior approach affords us unparalleled access to the entire footprint of the intervertebral disc, allowing for the insertion of massive, hyperlordotic interbody grafts that maximize surface area for fusion while simultaneously restoring sagittal balance and indirectly unkinking the ligamentum flavum posteriorly.

Osteology and Ligamentous Architecture
The vertebral bodies themselves are biomechanical marvels, composed of a trabecular cancellous bone core encased within a strong, dense cortical rim. The cartilaginous endplates are the crucial interface for our fusion construct. Our surgical preparation must meticulously remove the hyaline cartilage to expose bleeding subchondral bone without violating the structural integrity of the endplate itself; breaching this cortical boundary invites catastrophic cage subsidence. The intervertebral disc, as we know, consists of the outer annulus fibrosus—with its highly organized, alternating radial collagen lamellae—encasing the gelatinous, proteoglycan-rich inner nucleus pulposus.
The ligamentous stabilizers of the anterior column must be carefully managed. The Anterior Longitudinal Ligament (ALL) is a robust, fibrous band running continuously along the anterior aspect of the vertebral bodies from the atlas down to the sacrum. In a standard ALIF, we must boldly incise and mobilize the ALL to access the disc space, often utilizing a wide, rectangular annulotomy. Conversely, the Posterior Longitudinal Ligament (PLL) lies on the posterior aspect of the vertebral bodies, forming the anterior boundary of the spinal canal. While we typically aim to preserve the PLL to act as a tension band and protect the dura, a thorough, radical discectomy must invariably reach its anterior surface to ensure complete decompression and maximum graft volume.
Neurovascular Structures: The Retroperitoneal Minefield
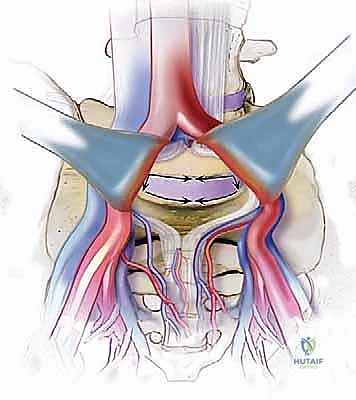
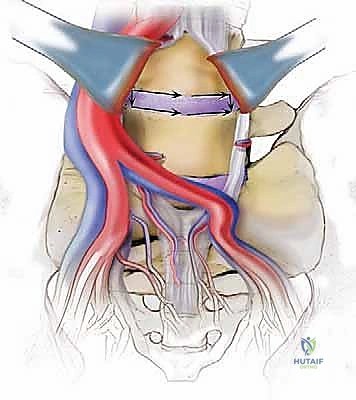
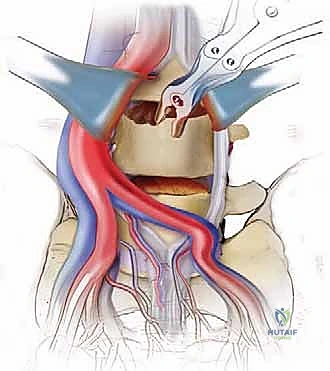
The great vessels represent our primary concern and the most significant source of intraoperative morbidity. The abdominal aorta and the Inferior Vena Cava (IVC) dominate the prevertebral space. The aorta typically descends slightly to the left of the midline, while the IVC lies to the right. At approximately the L4-L5 disc space, the aorta bifurcates into the common iliac arteries, and just inferior to this, the common iliac veins converge to form the IVC. The left common iliac vein is of particular surgical significance; it frequently crosses directly anterior to the L5-S1 disc space from left to right, making it highly vulnerable to traction injury or direct laceration during exposure.
Mobilization of these great vessels requires meticulous identification and ligation of the lumbar segmental vessels. These vessels arise from the posterior aspects of the aorta and IVC at roughly the midpoint of each vertebral body, coursing horizontally and posteriorly. To safely retract the great vessels across the midline (especially at L4-L5 and above), these tethering segmental arteries and veins—particularly the ascending lumbar vein and the formidable iliolumbar vein at L5—must be definitively isolated, ligated, and divided. Failure to respect these vessels can lead to avulsion directly from the IVC or aorta, resulting in rapid, massive, and potentially fatal hemorrhage.
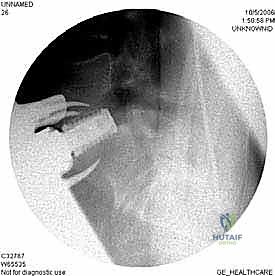
Beyond the vasculature, the autonomic and somatic nerve plexuses demand our utmost respect. The superior hypogastric sympathetic plexus descends anterolaterally over the lower lumbar vertebral bodies and the aortic bifurcation. Injury to this delicate, spiderweb-like plexus, particularly through aggressive electrocautery or rough blunt dissection, can precipitate retrograde ejaculation in male patients—a devastating functional complication. The genitofemoral nerve, which pierces and runs superficially along the anterior surface of the psoas major muscle, provides sensory innervation to the groin and medial thigh. Prolonged retraction of the psoas can induce a neurapraxia of this nerve, causing distressing postoperative numbness or neuralgia.
Exhaustive Indications and Contraindications
Patient selection is the cornerstone of successful anterior spinal surgery. The primary indication for an ALIF remains severe, concordant discogenic back pain that has proven entirely refractory to a minimum of six months of exhaustive, comprehensive nonoperative management. This conservative regimen must have included targeted physical therapy, non-steroidal anti-inflammatory drugs (NSAIDs), rigorous activity modification, weight loss optimization, and judicious use of epidural steroid or facet injections. When these modalities fail, and the patient's quality of life is unacceptably compromised, surgical intervention becomes a rational consideration.
Deformity, Instability, and Corpectomy Indications
Beyond isolated DDD, the anterior approach is a powerful tool in the armamentarium for adult spinal deformity correction. ALIF is unparalleled in its ability to restore lumbar lordosis, particularly at the L4-L5 and L5-S1 segments, which naturally account for over 60% of total lumbar lordosis. By utilizing hyperlordotic cages (often 12 to 30 degrees), we can dramatically correct sagittal imbalance, shifting the patient's C7 plumb line posteriorly and reducing the immense muscular energy required to stand upright. Furthermore, ALIF is highly indicated for the treatment of low-grade isthmic or degenerative spondylolisthesis, where the anterior cage provides immediate structural reduction and a massive surface area for arthrodesis.
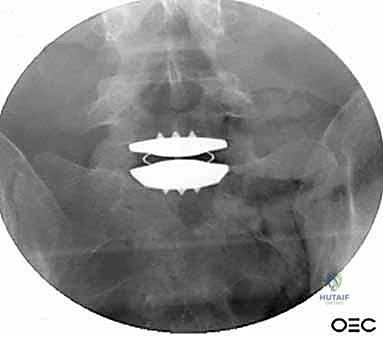
In more catastrophic scenarios—such as severe burst fractures compromising the anterior column, destructive primary or metastatic osseous tumors, or advanced pyogenic vertebral osteomyelitis/discitis—a simple discectomy is insufficient. These pathologies necessitate a formal corpectomy. A corpectomy involves the radical resection of the entire diseased vertebral body and adjacent discs, followed by the insertion of a structural strut graft or an expandable titanium cage to reconstruct the anterior column spanning multiple levels. The anterior approach provides direct, unhindered access for complete debridement of infected or neoplastic tissue.
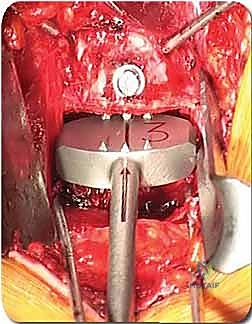
Absolute and Relative Contraindications
While highly effective, the anterior approach is not universally applicable. Absolute contraindications include active intra-abdominal sepsis or peritonitis, which would inevitably contaminate the spinal hardware. Severe, uncorrectable coagulopathies also preclude this highly vascular surgery. Relative contraindications require intense preoperative scrutiny. Patients with severe aortoiliac occlusive disease or heavily calcified, "porcelain" aortas are at extreme risk for plaque embolization or vessel rupture during retraction; in such cases, a vascular surgeon's consultation is mandatory, and alternative posterior approaches should be strongly considered.
| Category | Specific Indications | Specific Contraindications |
|---|---|---|
| Degenerative | Refractory Discogenic Pain, Degenerative Scoliosis, Foraminal Stenosis (Indirect Decompression) | Isolated severe central canal stenosis without instability (better suited for posterior decompression) |
| Structural | Isthmic/Degenerative Spondylolisthesis (Grade 1-2), Flatback Syndrome | High-grade spondylolisthesis (Grade 3+) where anterior reduction is impossible |
| Pathological | Vertebral Osteomyelitis, Discitis, Primary/Metastatic Tumors (requiring Corpectomy) | Active intra-abdominal infection, severe peritoneal adhesions from prior surgeries |
| Vascular/Anatomical | Need for massive anterior column support | Heavily calcified aorta/iliac vessels, prior retroperitoneal radiation therapy |
Pre-Operative Planning, Templating, and Patient Positioning
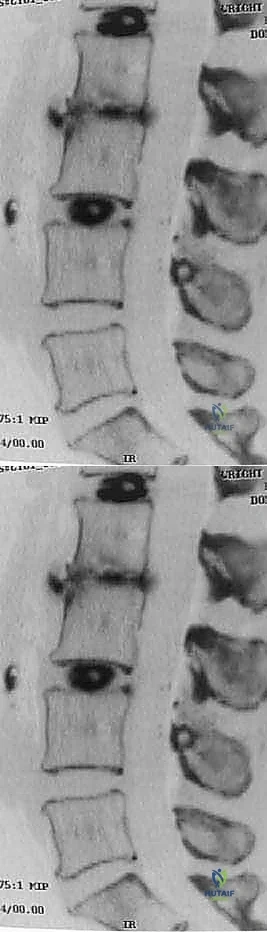
Meticulous preoperative planning is the invisible foundation upon which surgical success is built. We rely heavily on a multimodal imaging approach to dictate our surgical strategy. Standing plain radiographs—including anteroposterior, lateral, and dynamic flexion-extension views—are critical for assessing baseline sagittal alignment, overall disc height loss, and identifying occult, dynamic instability that might not be visible on supine imaging. These films also allow us to measure the pelvic incidence and calculate the necessary lordotic correction required to achieve spinopelvic harmony.
Advanced Imaging and Implant Templating
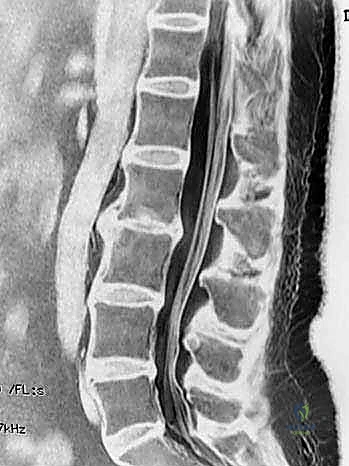
Magnetic Resonance Imaging (MRI) is the gold standard for evaluating the soft tissue elements. T2-weighted sagittal and axial sequences provide exquisite detail regarding disc hydration—where a "black disc" signifies severe desiccation and degeneration—as well as the status of the neural foramina and central canal. We carefully analyze the MRI for Modic changes, which correlate strongly with discogenic pain. In complex revision cases or when precise bony architecture is needed, a fine-cut Computed Tomography (CT) scan is invaluable for assessing endplate integrity, facet joint arthropathy, and the presence of bridging osteophytes that may impede distraction.
Templating the interbody implant is a critical exercise. Using digital templating software overlaid on lateral radiographs or CT sagittal reconstructions, we estimate the required height, width, depth, and lordotic angle of the final cage. The goal is to maximize the footprint of the implant to distribute axial loads evenly across the dense apophyseal ring of the endplate. A critical pearl: an oversized implant can lead to catastrophic over-distraction, inducing iatrogenic neural stretch injuries or fracturing the vertebral endplate, which guarantees cage subsidence. Conversely, an undersized implant fails to restore foraminal height, provides inadequate stability, and significantly increases the risk of pseudarthrosis.
Patient Positioning and Operating Room Setup
Patient positioning dictates the ease of our exposure and the quality of our fluoroscopic imaging. The patient is placed supine on a radiolucent operating table. A critical maneuver involves placing an inflatable bladder or a firm, 1-inch-thick foam pad transversely under the lumbar spine. This "bump" dynamically accentuates lumbar lordosis, opening the anterior disc spaces and facilitating easier access and cage insertion. The patient's arms must be carefully padded and tucked across the chest or positioned superiorly, completely out of the lateral fluoroscopic field of view.
The surgical team's orchestration is equally vital. Typically, an access surgeon (a general or vascular surgeon specialized in anterior spine exposures) stands on the patient's left side, utilizing the natural avascular plane to mobilize the peritoneum and great vessels from left to right. The orthopedic spine surgeon stands on the right, ready to execute the discectomy and reconstruction once the exposure is secured. Continuous, unhindered access for the C-arm fluoroscopy machine in both AP and lateral planes must be confirmed before the patient is prepped and draped.
Step-by-Step Surgical Approach and Fixation Technique
The surgical approach begins with either a transverse Pfannenstiel incision (ideal for L5-S1) or a longitudinal midline/paramedian incision (better for multi-level or higher lumbar pathology). We incise the anterior rectus sheath and mobilize the rectus abdominis muscle laterally. We then carefully incise the posterior rectus sheath and transversalis fascia to enter the retroperitoneal space. This space is a delicate, areolar plane that can be bluntly developed using sweeping motions with a sponge stick or the surgeon's fingers. The entire peritoneal sac, containing the intra-abdominal viscera and the ureter, is swept medially and superiorly, protecting the bowel from direct manipulation and minimizing the risk of postoperative ileus.
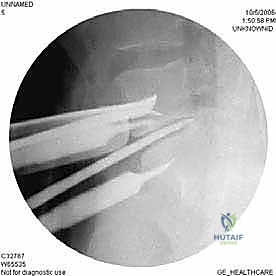
Vascular Mobilization and Discectomy
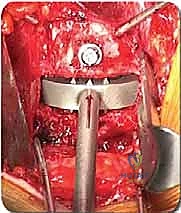
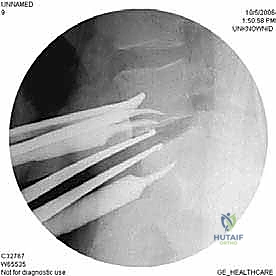
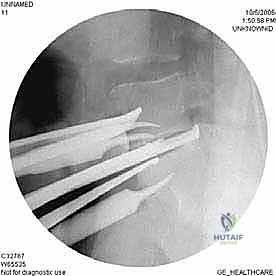
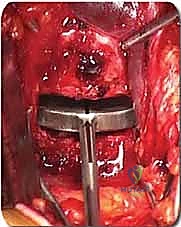
Once in the retroperitoneum, the psoas major muscle laterally and the great vessels medially serve as our primary landmarks. At L5-S1, the approach is typically straight down between the bifurcation of the great vessels. We must meticulously identify the middle sacral artery and vein, which course directly over the L5-S1 disc, and ligate them using clips or bipolar electrocautery. At L4-L5, the exposure is more complex. We must mobilize the left common iliac vein and artery from left to right. This often requires identifying, ligating, and dividing the iliolumbar vein, which tightly tethers the common iliac vein to the spine. Once the vessels are safely retracted using specialized, smooth-bladed retractor blades, the shimmering white fibers of the Anterior Longitudinal Ligament (ALL) are exposed.
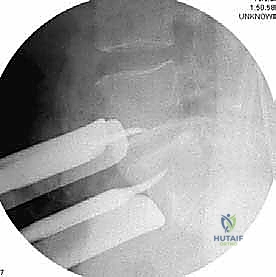
The discectomy is initiated with a wide, box-shaped annulotomy using a long-handled scalpel. We utilize a combination of pituitary rongeurs, straight and angled curettes, and Cobb elevators to aggressively evacuate the nucleus pulposus and the inner annulus. The goal is a radical, complete discectomy extending posteriorly to the PLL and laterally to the medial borders of the psoas muscles. Endplate preparation is the most critical step for achieving arthrodesis. We use ring curettes and rasps to meticulously scrape away the cartilaginous endplates until we reach punctate bleeding from the subchondral bone. We must be exceptionally careful not to breach the cortical endplate, as this structural rim is essential for supporting the interbody cage.
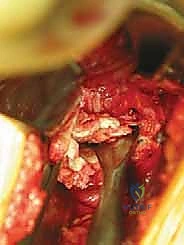
Implantation, Corpectomy Reconstruction, and Fixation
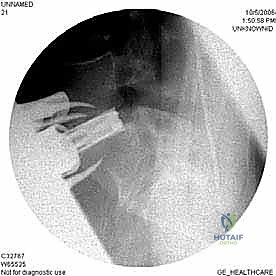
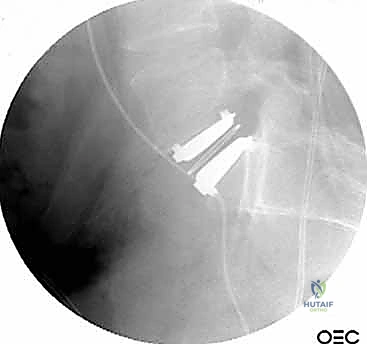
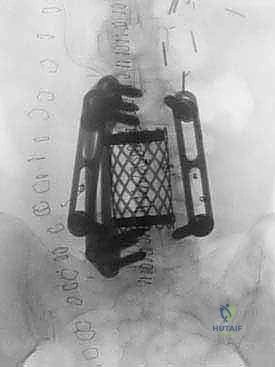
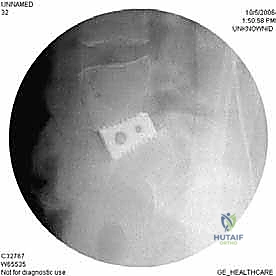
With the disc space prepared, we utilize sequential trial spacers under lateral fluoroscopic guidance to determine the optimal cage size. We assess for adequate distraction, restoration of disc height, and appropriate lordosis without over-tensioning the annulus. The definitive cage—whether made of rough-surfaced titanium, PEEK, or a structural allograft—is densely packed with osteoinductive and osteoconductive materials. This typically includes a combination of local autograft bone (harvested from anterior osteophytes), cancellous allograft chips, and often a biologic enhancer such as Recombinant Human Bone Morphogenetic Protein-2 (rhBMP-2) applied to an absorbable collagen sponge. The cage is then impacted into the disc space under continuous fluoroscopic monitoring until it is perfectly recessed just deep to the anterior vertebral cortical margin.
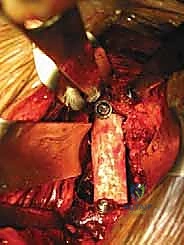
In cases requiring a corpectomy, the procedure is significantly amplified. After the adjacent discs are removed, the vertebral body itself is resected using a high-speed burr, rongeurs, and osteotomes. We typically aim to preserve the extreme lateral walls and, if oncologically safe, the posterior cortical wall overlying the dura to protect the neural elements. Once the corpectomy defect is created, an expandable titanium cage or a massive structural strut graft is inserted and expanded to engage the superior and inferior endplates, restoring the anterior column's height and structural integrity.

Regardless of whether an ALIF or a corpectomy is performed, anterior column reconstruction often requires supplemental fixation to ensure immediate biomechanical stability. This can be achieved via an anterior titanium plate secured with bicortical screws into the adjacent vertebral bodies, or via integrated screws that pass directly through the anterior flange of the interbody cage. In many cases, particularly multi-level reconstructions or cases with significant instability, we will flip the patient prone and supplement the anterior construct with posterior pedicle screw instrumentation to create a rigid, 360-degree fusion construct.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, anterior lumbar surgery carries a unique and potentially severe complication profile. Vascular injury is the most dreaded intraoperative event. Lacerations to the left common iliac vein or the IVC can result in catastrophic, life-threatening hemorrhage within seconds. The incidence of major vascular injury is reported to be between 1% and 5%, heavily dependent on the surgeon's experience and the patient's anatomy. If a venous tear occurs, the immediate response must be direct, firm pressure with a sponge stick to control the bleeding. Blind clamping is strictly forbidden, as it will invariably enlarge the tear. The vessel must be carefully isolated, and the defect repaired primarily with 5-0 or
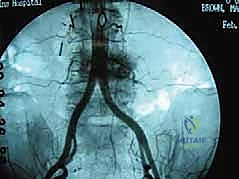
Clinical & Radiographic Imaging Archive