Acute Total Elbow Arthroplasty for Comminuted Distal Humerus Fractures: A Masterclass
Key Takeaway
Join us in the OR for an immersive masterclass on acute total elbow arthroplasty for complex distal humerus fractures. We'll meticulously cover patient selection, detailed anatomy, preoperative planning, and step-by-step intraoperative execution, emphasizing critical techniques for ulnar nerve protection and bone preparation. Learn to navigate common pitfalls and optimize outcomes for this challenging procedure in physiologically elderly, low-demand patients.
Comprehensive Introduction and Patho-Epidemiology
The management of comminuted distal humerus fractures in the physiologically elderly patient represents one of the most formidable challenges in modern orthopedic traumatology. Historically, open reduction and internal fixation (ORIF) has been the gold standard, driven by the imperative to restore native articular congruity and preserve joint kinematics. However, the bimodal epidemiological distribution of these fractures reveals a rapidly expanding cohort of geriatric patients presenting with osteoporotic bone and complex, multi-fragmentary fracture patterns, frequently classified as AO/OTA 13-C3. In this specific demographic, the pursuit of anatomical reduction via ORIF is frequently thwarted by inadequate bone stock, leading to an unacceptably high incidence of catastrophic hardware failure, screw cut-out, non-union, and rapid post-traumatic arthrosis.
Acute total elbow arthroplasty (TEA) has emerged not merely as a salvage procedure, but as a highly efficacious, primary therapeutic intervention for unreconstructable distal humerus fractures in the elderly. The paradigm shift towards acute TEA is supported by its predictable capacity to restore immediate joint stability, alleviate pain, and permit early mobilization, thereby mitigating the profound morbidities associated with prolonged immobilization in the geriatric population. By bypassing the necessity for fracture union to achieve joint stability, a semi-constrained TEA provides a durable, functional articulation that dramatically outpaces the functional outcomes of compromised ORIF attempts in osteoporotic bone.
It is critical to distinguish total elbow arthroplasty from distal humeral hemiarthroplasty. While acute elbow hemiarthroplasty has been described in recent literature as an innovative attempt to preserve the native ulna and radial head, it remains an experimental procedure. The complex, highly congruent topography of the native radiocapitellar and ulnohumeral articulations makes hemiarthroplasty biomechanically unpredictable, often leading to rapid contralateral cartilage wear. Consequently, distal humerus hemiarthroplasty is not FDA-approved for this indication and should not be considered for general clinical application. Our focus remains unequivocally on the established, evidence-based application of linked, semi-constrained total elbow arthroplasty.
This masterclass chapter provides an exhaustive, step-by-step surgical blueprint for performing an acute total elbow arthroplasty. We will dissect the nuanced preoperative planning, meticulous soft-tissue handling, complex intraoperative decision-making, and precise biomechanical reconstruction required to execute this procedure successfully. The mastery of this technique demands a profound understanding of elbow anatomy, an appreciation for the limitations of geriatric bone healing, and a rigorous adherence to biomechanical principles to ensure long-term implant survivorship.
Detailed Surgical Anatomy and Biomechanics
A profound, three-dimensional understanding of elbow anatomy is the absolute prerequisite for executing a successful total elbow arthroplasty. The elbow is a highly constrained, complex hinge joint composed of three distinct articulations: the ulnohumeral, radiocapitellar, and proximal radioulnar joints.
Osteology and Articular Geometry
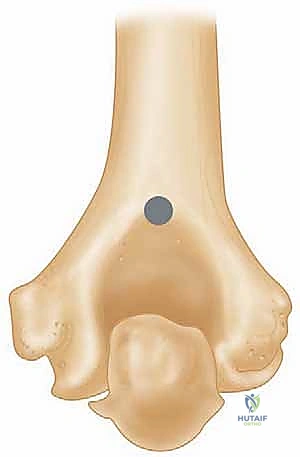
The distal humerus is architecturally conceptualized as a structural triangle, consisting of a medial column, a lateral column, and the transverse articular segment (the trochlea and capitellum) acting as the base. In a highly comminuted trauma setting, this entire triangular architecture is obliterated. The trochlea, which articulates with the greater sigmoid notch of the ulna, is spool-shaped and is oriented with approximately 30 degrees of anterior angulation and 3 to 8 degrees of valgus relative to the humeral shaft. Reproducing this precise anatomical axis of rotation during prosthetic implantation is critical; failure to do so results in eccentric loading of the polyethylene bushings, accelerated wear, and premature aseptic loosening.
The proximal ulna features the olecranon posteriorly and the coronoid process anteriorly, encompassing the greater sigmoid notch. The articular surface of the ulna is not perfectly congruent with the trochlea, possessing a central non-articular bare area that must be respected during preparation. The radial head articulates with the capitellum and is vital for load transfer across the elbow, particularly in valgus stress. In the context of a semi-constrained TEA, the radial head is frequently resected to facilitate exposure and prevent impingement against the humeral component, though some modern implant designs allow for its retention if the lateral column is partially intact.
Biomechanically, linked semi-constrained implants have revolutionized TEA for trauma. Older, rigid-hinge designs failed catastrophically due to the transmission of immense torsional and varus/valgus stresses directly to the bone-cement interface. Modern semi-constrained designs incorporate a "sloppy hinge" mechanism, allowing for 7 to 10 degrees of varus/valgus and rotational laxity. This critical design feature dissipates stress away from the fixation interfaces and transfers it to the surrounding soft tissue envelope, dramatically reducing the incidence of aseptic loosening while providing sufficient intrinsic stability in the absence of functional collateral ligaments.
Ligamentous Stabilizers and Kinematics
The primary static stabilizers of the elbow are the medial collateral ligament (MCL) complex and the lateral collateral ligament (LCL) complex. The anterior bundle of the MCL is the primary restraint to valgus stress, while the lateral ulnar collateral ligament (LUCL) is the primary restraint to varus and posterolateral rotatory instability. In the setting of an acute, highly comminuted distal humerus fracture requiring TEA, these ligamentous structures are frequently avulsed from their epicondylar origins or must be surgically detached to resect the non-viable articular fragments.
Because a linked TEA provides intrinsic stability through its mechanical coupling, the anatomical repair of the collateral ligaments is not strictly necessary for joint stability, which is a major advantage over unlinked implants or ORIF. However, preserving the epicondyles and their attached soft tissues (the common flexor and extensor origins) is highly desirable when feasible. Reattaching these muscle masses to the triceps fascia or the epicondylar remnants helps restore dynamic stability, improves grip strength, and provides a robust soft-tissue envelope to protect the implant from infection.
The triceps brachii is the primary dynamic extensor of the elbow and is the most critical soft-tissue structure managed during the surgical approach. The continuity of the triceps mechanism must be meticulously preserved or robustly repaired. Triceps insufficiency following TEA is a devastating complication that severely limits the patient's ability to perform activities of daily living, such as pushing up from a chair. The choice of surgical approach—whether triceps-reflecting, triceps-splitting, or a triceps-sparing technique—dictates the specific postoperative rehabilitation constraints required to protect the extensor mechanism during the critical healing phase.
Neurovascular Topography
The ulnar nerve is the structure at greatest risk during both the traumatic event and the subsequent surgical intervention. It courses through the posterior compartment of the arm, piercing the medial intermuscular septum at the arcade of Struthers, approximately 8 cm proximal to the medial epicondyle. It then transits behind the medial epicondyle within the cubital tunnel, tethered by Osborne's fascia, before entering the forearm between the two heads of the flexor carpi ulnaris (FCU). In comminuted fractures, the nerve is frequently displaced, contused, or entrapped by medial column fragments.
The radial nerve spirals around the posterior humerus in the spiral groove, piercing the lateral intermuscular septum approximately 10 cm proximal to the lateral epicondyle to enter the anterior compartment. While less directly exposed during a standard posterior approach to the elbow, it is at risk during proximal dissection or when addressing extensive humeral shaft comminution. The surgeon must remain acutely aware of its trajectory, particularly when placing retractors on the anterior aspect of the lateral humerus.
The median nerve and brachial artery course anterior to the brachialis muscle and are generally well-protected from posterior surgical approaches. However, severe traumatic displacement of the distal humerus anteriorly can tent or lacerate these structures. A meticulous preoperative neurovascular examination is paramount. Any deficits must be documented, and the surgical plan must incorporate deliberate steps for neurolysis, protection, and potential anterior transposition of the ulnar nerve to prevent iatrogenic injury or postoperative compressive neuropathy.
Exhaustive Indications and Contraindications
Patient selection is the cornerstone of success in acute total elbow arthroplasty. The decision to abandon joint preservation in favor of arthroplasty requires a nuanced assessment of the patient's physiological age, functional demands, bone quality, and fracture morphology.
Defining the Ideal Candidate
The quintessential candidate for an acute TEA is a physiologically elderly patient (typically >65 years of age) presenting with a low-demand lifestyle and an AO/OTA 13-C3 comminuted distal humerus fracture. In these patients, the presence of severe osteoporosis renders internal fixation constructs inherently unstable. The inability to achieve rigid anatomical reduction precludes early mobilization, leading to profound joint stiffness and functional devastation. TEA offers these patients immediate stability and the ability to initiate range of motion exercises within days of surgery.
Beyond acute trauma, patients with pre-existing inflammatory arthropathies, such as rheumatoid arthritis, who sustain a distal humerus fracture are exceptional candidates for primary TEA. These patients already possess compromised articular cartilage and attenuated periarticular soft tissues, making ORIF highly prone to failure and subsequent post-traumatic arthritis. Furthermore, patients presenting with pathological fractures secondary to metastatic disease or severe metabolic bone disorders are also appropriately managed with TEA, as it provides immediate palliative stabilization and functional restoration without relying on compromised biological healing.
Another critical indication is the presence of an unreconstructable fracture pattern. Even in a moderately aged patient, if the articular surface is pulverized into osteochondral fragments too small to hold a screw (frequently termed a "bag of bones"), any attempt at ORIF is doomed to fail. In these specific scenarios, the surgeon must objectively evaluate the fracture intraoperatively; if anatomical reduction and stable fixation are impossible, pivoting to an acute TEA is the most prudent and evidence-based clinical decision.
Absolute and Relative Contraindications
Despite its profound benefits, TEA is not a panacea and carries strict contraindications that must be respected to avoid catastrophic outcomes. The absolute contraindications are primarily biological and infectious.
| Category | Specific Contraindications | Clinical Rationale |
|---|---|---|
| Absolute | Active local or systemic infection | Implantation of hardware in an infected bed guarantees periprosthetic joint infection (PJI), necessitating explantation. |
| Absolute | Paralysis of the elbow musculature | Without a functioning triceps or biceps (e.g., severe brachial plexus injury), the implant serves no functional purpose and is prone to instability. |
| Absolute | Severe soft tissue compromise | Inadequate soft tissue coverage (e.g., massive degloving) precludes wound closure, leading to exposed hardware and inevitable infection. |
| Relative | High physical demands / Young age | TEA implants have a strict lifetime lifting restriction (5-10 lbs). High-demand use leads to rapid aseptic loosening and catastrophic mechanical failure. |
| Relative | Isolated single-column fractures | These can typically be managed successfully with ORIF, preserving the native joint and avoiding the lifelong limitations of an arthroplasty. |
| Relative | Neuropathic (Charcot) arthropathy | The lack of protective proprioception leads to rapid, massive destruction of the bone-cement interface and implant failure. |
A significant relative contraindication is the young, high-demand patient. The polyethylene bushings and the bone-cement interfaces of current TEA designs are not engineered to withstand repetitive heavy lifting or high-impact activities. Patients must adhere to a strict lifetime lifting restriction (typically no more than 5 pounds repetitively, or 10 pounds for a single event). Implanting a TEA in a 40-year-old manual laborer will almost certainly result in early mechanical failure, aseptic loosening, and a highly complex revision scenario with massive bone loss. In such cases, even a suboptimal ORIF or an elbow arthrodesis may be preferable.
Pre-Operative Planning, Templating, and Patient Positioning
The success of a total elbow arthroplasty is largely determined before the first incision is made. Meticulous preoperative planning allows the surgeon to anticipate anatomical distortions, select the appropriate implant constraints, and formulate a strategy for soft tissue management.
Advanced Imaging and Digital Templating
Standard anteroposterior (AP) and lateral radiographs of the elbow are the initial diagnostic modality. Due to severe pain and comminution, these are often obtained through a splint.
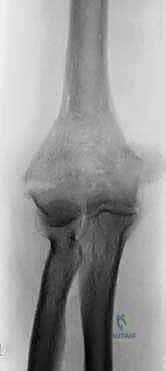
Traction views, administered under gentle sedation, can occasionally help delineate the major fracture fragments by utilizing ligamentotaxis. Crucially, the surgeon must also obtain bilateral wrist radiographs. A fall onto an outstretched hand can transmit axial force through the forearm, resulting in a concomitant distal radius fracture or a disruption of the distal radioulnar joint (DRUJ) and interosseous membrane—the classic Essex-Lopresti lesion. If an Essex-Lopresti lesion is identified, radial head resection during the TEA is strictly contraindicated, as it will lead to proximal migration of the radius and chronic wrist pain; a radial head prosthesis must be incorporated into the reconstruction.
A Computed Tomography (CT) scan with three-dimensional reconstructions is mandatory for all comminuted intra-articular fractures. The CT scan defines the exact geometry of the articular comminution, the integrity of the humeral columns, and the proximal extent of the fracture lines into the humeral diaphysis. This information is critical for templating.
Digital templating is performed on both the AP and lateral planes to determine the optimal size of the humeral and ulnar stems. The surgeon must carefully assess the degree of humeral bone loss. Standard TEA implants can accommodate up to 2 cm of distal humeral bone loss. If the fracture extends further proximally, resulting in greater than 2 cm of loss, an implant with an extended anterior flange is required. This flange rests against the anterior cortex of the intact proximal humerus, restoring the anatomical joint line, ensuring appropriate triceps tension, and preventing catastrophic rotational failure of the humeral component.
Operating Room Setup and Patient Positioning
Patient positioning must optimize surgical exposure while allowing for intraoperative fluoroscopy without compromising the sterile field. The two primary options are the supine position and the lateral decubitus position.
For this masterclass, we advocate for the supine position with the arm draped freely across the chest. A large, sterile bump or rolled towel is placed under the ipsilateral scapula to elevate the shoulder, and a padded Mayo stand or a specialized arm holder is positioned across the patient's torso. This setup allows the surgeon to stand on the operative side, providing unhindered access to the posterior elbow while an assistant manipulates the limb through full ranges of flexion, extension, pronation, and supination.
Alternatively, the lateral decubitus position with the arm suspended over a bolster provides excellent posterior exposure and utilizes gravity to retract the anterior soft tissues. However, it can be more cumbersome for intraoperative fluoroscopy. Regardless of the chosen position, a sterile tourniquet is applied high on the brachium. The entire upper extremity is prepped and draped to allow access from the mid-humerus to the wrist. The C-arm fluoroscopy unit is brought in from the contralateral side, ensuring that perfect AP and lateral views can be obtained seamlessly throughout the procedure to verify implant alignment and seating.
Step-by-Step Surgical Approach and Fixation Technique
The execution of the surgical approach requires a delicate balance between achieving adequate exposure of the joint and preserving the critical soft tissue envelope, particularly the extensor mechanism and the ulnar nerve.
Incision, Flap Elevation, and Triceps Management
A straight, midline longitudinal incision is made over the posterior aspect of the elbow, beginning approximately 5 to 7 cm proximal to the olecranon tip and extending 5 to 7 cm distally along the ulnar crest.
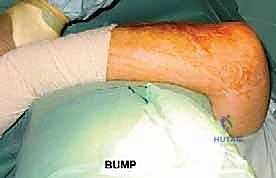
Crucially, the incision must gently curve slightly radially or ulnarly around the tip of the olecranon to prevent a painful scar directly over the weight-bearing prominence.
Following the skin incision, full-thickness fasciocutaneous flaps are elevated medially and laterally using the "flat knife" technique.
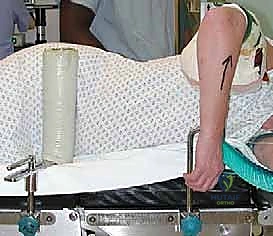
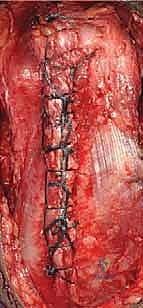
The scalpel blade is held parallel to the deep fascia, preserving the subcutaneous vascular plexus. Hemostasis is meticulously maintained with electrocautery to prevent postoperative hematoma, which is a significant risk factor for infection. The medial and lateral borders of the triceps are identified and defined.
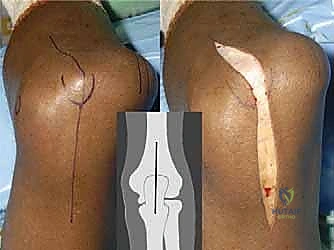
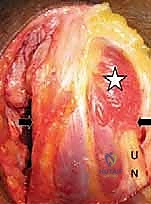
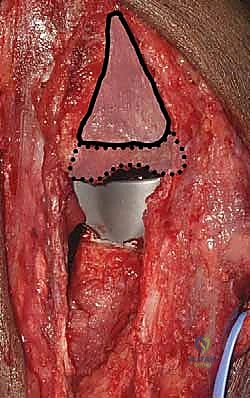
For acute trauma, a triceps-reflecting approach (such as the Bryan-Morrey approach) or a Triceps-Reflecting Anconeus Pedicle (TRAP) approach is preferred. These approaches maintain the continuity of the triceps insertion onto the periosteum of the proximal ulna, allowing for a robust, bone-to-bone or tendon-to-bone repair at closure. The triceps is elevated from medial to lateral off the posterior humerus, exposing the olecranon fossa and the comminuted distal humerus.
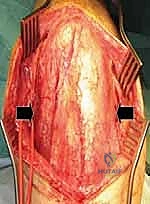
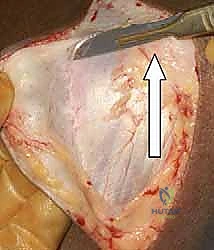
Ulnar Nerve Management and Joint Exposure
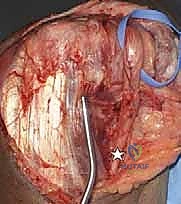
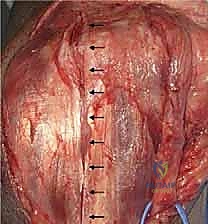
Before proceeding with deep bony resection, the ulnar nerve must be definitively identified, neurolysed, and protected.
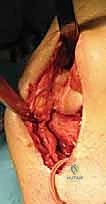
The nerve is located at the medial border of the triceps, proximal to the medial epicondyle. The fascia overlying the cubital tunnel is incised, and the nerve is traced proximally to the arcade of Struthers, which is released.
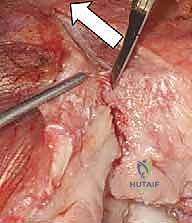
Distally, the nerve is followed between the two heads of the FCU, and the aponeurosis is divided to prevent kinking.
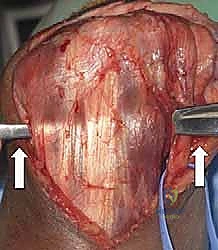
A vessel loop is placed around the nerve for continuous identification. Depending on the implant design and the degree of medial column disruption, the nerve may be left in situ or subjected to a formal anterior subcutaneous transposition. In trauma settings with massive swelling and altered anatomy, anterior transposition is frequently favored to remove the nerve from the inflammatory bed of the arthroplasty and prevent postoperative tethering.
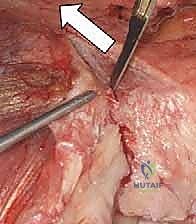
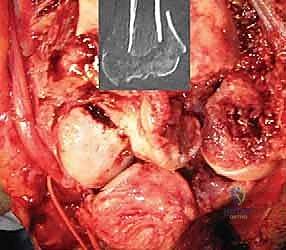
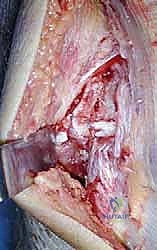
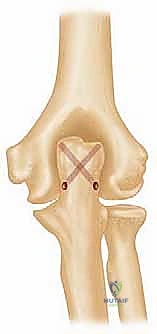
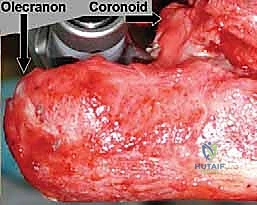
With the nerve protected, the joint is fully exposed. The comminuted articular fragments of the trochlea and capitellum are sharply excised.
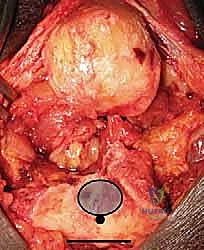
If the medial and lateral epicondyles are intact, every effort is made to preserve them, along with their respective collateral ligaments and flexor/extensor origins. However, in an AO 13-C3 fracture, these are often non-viable. If resected, heavy non-absorbable sutures are placed through the common flexor and extensor tendons for later repair to the triceps fascia.
Humeral Preparation and Implantation
Attention is turned to the humeral canal. The medullary canal is identified, often requiring a high-speed burr to clear metaphyseal cancellous bone and open the diaphysis.
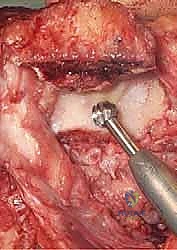
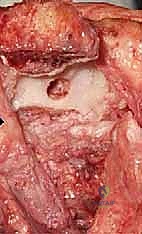
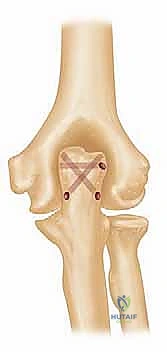
Sequential reaming and broaching are performed to appropriately size the humeral stem. It is imperative to maintain the anatomical alignment; the humeral component must be seated with the axis of rotation matching the native trochlea (typically 3-8 degrees of valgus and neutral rotation relative to the epicondylar axis).
A trial humeral component is inserted. If there is significant distal bone loss, an implant with an anterior flange is utilized. The flange must sit flush against the anterior humeral cortex to provide rotational stability and prevent posterior subsidence. A bone graft, often harvested from the excised articular fragments, can be placed behind the anterior flange to encourage osteointegration and restore bone stock.

Ulnar Preparation and Articular Reconstruction
The proximal ulna is then addressed. The tip of the olecranon is often resected to facilitate access to the medullary canal.
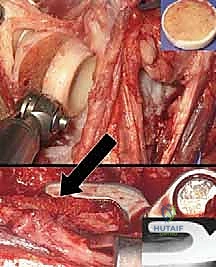
The ulnar canal is notoriously difficult to navigate due to its inherent bow and the dense cortical bone of the proximal diaphysis. A high-speed burr or an awl is used to locate the canal, and sequential broaching is performed.
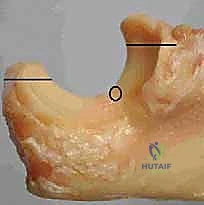
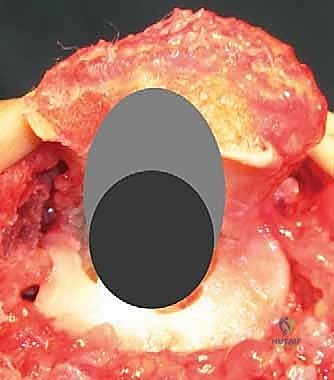
The surgeon must be vigilant to avoid cortical perforation or fracture of the proximal ulna during this step, which is a known and disastrous complication.
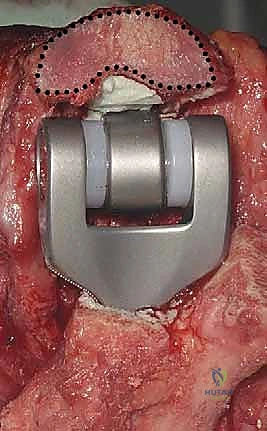
Once both canals are prepared, trial components are inserted, and the joint is linked. The elbow is taken through a full range of motion to assess for impingement, stability, and appropriate soft tissue tension. The radial head is evaluated; if it impinges on the humeral component during flexion or pronation/supination, it is resected.
Following trialing, the canals are thoroughly irrigated with pulsatile lavage and dried. Cement restrictors are placed distally in both canals. Polymethylmethacrylate (PMMA) bone cement is mixed and injected in a retrograde fashion using a cement gun to ensure a uniform mantle without voids.
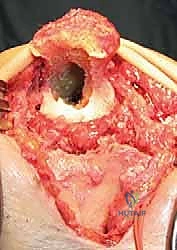
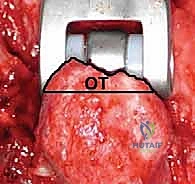
The definitive humeral and ulnar components are inserted and held rigidly in the correct anatomical alignment until the cement polymerizes.