Elbow Arthrodesis: A Masterclass in Salvage Reconstruction for Severe Elbow Pathology
Key Takeaway
Elbow arthrodesis is a rarely performed salvage procedure for severe joint destruction or instability. This masterclass details comprehensive preoperative planning, meticulous surgical technique, including step-cut osteotomy and locking plate fixation, and crucial intraoperative pearls. Fellows will gain insights into optimal fusion angles, neurovascular protection, and postoperative management, ensuring successful outcomes for challenging elbow pathologies.
Comprehensive Introduction and Patho-Epidemiology
Elbow arthrodesis represents the ultimate salvage reconstruction within the armamentarium of the orthopedic surgeon. Historically, this procedure was frequently employed to manage the devastating sequelae of poliomyelitis and tuberculous arthritis. However, the epidemiological landscape has shifted dramatically over the past several decades. Today, the modern orthopedic surgeon is most likely to consider elbow arthrodesis in the setting of catastrophic post-traumatic joint destruction, intractable chronic osteomyelitis, severe soft-tissue compromise, or as a final recourse following the catastrophic failure of a total elbow arthroplasty (TEA) where revision is anatomically or biologically impossible. It is a procedure born of necessity rather than preference, deployed when all other satisfactory surgical options for a severely compromised elbow have been exhausted.
The fundamental objective of an elbow arthrodesis is to convert a painful, unstable, or chronically infected joint into a painless, stable, and functional fulcrum. This is achieved at the profound cost of radiocapitellar and ulnohumeral motion. Unlike the hip, knee, or ankle—where arthrodesis can often yield a highly functional, albeit altered, gait—the elbow functions primarily as a dynamic positioning crane for the hand. It allows the hand to interact with the environment, facilitating both personal hygiene and extrapersonal activities. Consequently, the functional disability resulting from an elbow arthrodesis is significantly greater than that of major lower extremity fusions. The loss of the elbow's arc of motion demands extraordinary compensatory flexibility and strength from the ipsilateral shoulder, wrist, and cervical spine.
Because of the severe functional implications, the decision to proceed with an elbow arthrodesis must be approached with immense clinical trepidation and exhaustive patient counseling. The patient must possess a clear understanding that the procedure will permanently alter their ability to perform activities of daily living (ADLs). The surgeon must evaluate the patient not merely as a localized joint pathology, but as a complex biomechanical system. Excellent ipsilateral shoulder function, a stable and mobile wrist, and, most importantly, a functional hand are absolute prerequisites. If the hand itself is severely compromised or insensate, the primary rationale for fusing the elbow—to position the hand for function—is entirely negated, rendering the procedure functionally useless.
Furthermore, the biomechanical environment of the elbow presents unique challenges to achieving a successful osseous union. The long lever arms of the forearm and the arm generate massive bending, shear, and torsional forces across the fusion site. These forces, coupled with the relatively small cross-sectional area of bony contact between the distal humerus and proximal ulna, contribute to historically high rates of nonunion. Therefore, modern surgical techniques demand meticulous joint preparation, robust internal fixation constructs (often utilizing locked plating technology), and the liberal application of autologous bone grafting to overcome these hostile biomechanical forces and achieve a durable, lifelong fusion.
Detailed Surgical Anatomy and Biomechanics
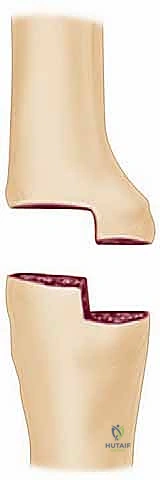
A profound understanding of the surgical anatomy and the unique biomechanical forces acting upon the elbow is essential for executing a successful arthrodesis. The osseous anatomy of the distal humerus is characterized by two distinct columns—the medial and lateral columns—which diverge from the humeral shaft to form a triangular structure supporting the articular segment (trochlea and capitellum). In the setting of severe pathology or failed TEA, these columns are often severely compromised, osteopenic, or entirely absent. The proximal ulna, comprising the olecranon, trochlear notch, and coronoid process, provides the opposing articular surface. During arthrodesis, the complex congruency of these surfaces must be aggressively decorticated to expose bleeding, cancellous bone. The radial head, while anatomically part of the joint, often acts as an impediment to achieving broad ulnohumeral contact and is frequently excised to facilitate optimal bony apposition and alignment.
The muscular anatomy surrounding the posterior elbow dictates our surgical approach. The triceps brachii, inserting broadly onto the olecranon, is the primary extensor and must be carefully managed. A longitudinal split of the triceps tendon is generally preferred as it preserves the medial and lateral continuity of the extensor mechanism, allowing for a robust repair during closure. Distally, the surgical interval is defined by the anconeus muscle laterally and the flexor carpi ulnaris (FCU) medially. Dissection within this interval exposes the posterior capsule and the underlying ulnohumeral articulation. Preservation of the surrounding muscular envelope is critical not only for maintaining vascularity to the surgical site but also for providing soft-tissue coverage over bulky internal fixation hardware, thereby reducing the risk of postoperative infection and wound breakdown.
The neurovascular anatomy, particularly the ulnar nerve, represents the most significant hazard during posterior elbow surgery. The ulnar nerve traverses the posterior aspect of the medial epicondyle within the cubital tunnel before passing between the two heads of the FCU. In the setting of revision surgery, trauma, or chronic infection, the normal anatomical planes are frequently obliterated by dense, unyielding scar tissue. The nerve may be tethered, encased in heterotopic ossification, or anatomically distorted. It is a fundamental principle of this procedure that the ulnar nerve must be identified in a zone of normal, unscarred anatomy—typically proximally in the arm—and meticulously traced distally into the zone of injury. Failure to adequately identify, mobilize, and protect the ulnar nerve can result in catastrophic iatrogenic injury, leading to profound motor and sensory deficits in the hand, further compounding the patient's functional disability.
Biomechanically, the elbow is subjected to immense forces that actively antagonize the fusion process. The forearm acts as a long lever arm, amplifying any force applied to the hand or wrist and transmitting it directly to the ulnohumeral junction. When the elbow is fused, particularly at angles between 70 and 90 degrees, the fusion site is subjected to extreme bending moments during lifting or weight-bearing activities. Furthermore, pronation and supination of the forearm generate significant torsional stresses across the fusion mass. To counteract these forces, the fixation construct must be exceptionally rigid. Posterior plating utilizes the tension band principle, converting the tensile forces on the posterior cortex during attempted flexion into compressive forces across the arthrodesis site. However, reliance on a single plate may be insufficient in cases of severe bone loss; thus, dual plating strategies or the addition of robust interfragmentary compression screws are often necessary to achieve immediate biomechanical stability and promote primary bone healing.
Exhaustive Indications and Contraindications
The decision algorithm for elbow arthrodesis requires a meticulous balancing of the patient's pathology, functional demands, and available bone stock. The overarching indication is a severely compromised elbow joint where motion preservation is impossible, and stability is paramount. The most common driver in modern practice is severe joint destruction secondary to trauma or infection. Highly comminuted, intra-articular fractures of the distal humerus in young, high-demand laborers may necessitate primary arthrodesis if the joint is deemed irreparable and the patient's occupational demands preclude the use of a total elbow arthroplasty (TEA), which would inevitably fail under heavy loading. Similarly, the sequelae of septic arthritis or chronic, intractable osteomyelitis can leave the joint devoid of articular cartilage and structurally unsound, making eradication of the infection and subsequent fusion the only viable limb-salvage strategy.
Another critical indication is the catastrophic failure of a total elbow arthroplasty. As the volume of primary TEA procedures has increased, so too has the incidence of complex revisions. When a TEA fails due to deep periprosthetic joint infection, massive aseptic loosening, or catastrophic periprosthetic fracture resulting in unmanageable bone loss, revision arthroplasty may be technically impossible. In these scenarios, explantation of the hardware, placement of an antibiotic spacer (if infected), and subsequent conversion to an elbow arthrodesis becomes the definitive salvage pathway. Furthermore, chronic, severe elbow instability that is entirely refractory to ligamentous reconstruction, dynamic external fixation, or bracing may also warrant arthrodesis to provide the patient with a stable, albeit stiff, upper extremity.
Conversely, the contraindications to elbow arthrodesis are equally stringent and must be respected to prevent catastrophic surgical failure. The absolute contraindication is the absence of a functional hand. The sole purpose of the elbow is to position the hand in space; fusing the elbow in a patient with an insensate, paralyzed, or severely mangled hand provides no functional benefit and exposes the patient to unnecessary surgical risk. Additionally, massive, uncontained bone loss that precludes the achievement of stable bony contact between the humerus and ulna, even with the use of massive structural allografts, renders the procedure physically impossible. In such extreme cases of bone loss, alternative salvage procedures, such as a flail elbow (resection arthroplasty) or, in the most dire circumstances, amputation, must be considered.
Severe soft-tissue compromise also represents a major hurdle. An inadequate soft-tissue envelope that cannot provide robust coverage over the necessary internal fixation hardware guarantees postoperative wound breakdown and deep infection. If local or free flap coverage is not feasible, arthrodesis is contraindicated. Finally, the status of the ipsilateral joints is paramount. A patient with severe, restrictive pathology in the ipsilateral shoulder, wrist, or cervical spine will be unable to compensate for the loss of elbow motion. Fusing the elbow in a patient who already lacks shoulder elevation or wrist mobility will effectively lock their hand in a useless position, devastating their ability to perform even the most basic activities of daily living.
| Category | Specific Conditions / Parameters | Clinical Rationale and Considerations |
|---|---|---|
| Primary Indications | Severe Post-traumatic Arthrosis | Ideal for young, heavy laborers where TEA is guaranteed to fail due to high functional demands. |
| Chronic/Intractable Infection | Sequelae of septic arthritis, TB, or osteomyelitis where joint eradication is necessary for cure. | |
| Failed Total Elbow Arthroplasty (TEA) | Salvage for massive bone loss, periprosthetic infection, or recurrent instability precluding revision TEA. | |
| Paralytic / Flail Elbow | Severe brachial plexus injury or polio sequelae requiring a stable fulcrum for hand function. | |
| Irreparable Instability | Chronic, multi-directional instability refractory to all ligamentous or osseous reconstructive efforts. | |
| Absolute Contraindications | Non-functional Ipsilateral Hand | Fusing the elbow to position a useless hand offers zero functional benefit to the patient. |
| Massive, Unreconstructable Bone Loss | Inability to achieve ulnohumeral contact even with structural grafts guarantees nonunion. | |
| Inadequate Soft Tissue Envelope | Lack of coverage for bulky hardware leads to inevitable wound breakdown and deep infection. | |
| Stiff Ipsilateral Shoulder or Wrist | Patient cannot utilize compensatory motion, leading to profound and debilitating functional loss. | |
| Relative Contraindications | Advanced Age / Low Demand | Older, low-demand patients may be better served by resection arthroplasty or constrained TEA. |
| Active, Untreated Systemic Infection | Must eradicate systemic bacteremia and optimize host biology prior to complex reconstructive surgery. | |
| Severe Patient Non-compliance | Inability to adhere to strict post-operative immobilization and rehabilitation protocols. |
Pre-Operative Planning, Templating, and Patient Positioning
The success of an elbow arthrodesis is inextricably linked to exhaustive, meticulous preoperative planning. This is not a procedure that can be improvised intraoperatively. The surgeon must begin with a comprehensive clinical evaluation, focusing intently on the soft-tissue envelope. Prior incisions, traumatic scars, skin grafts, and areas of adherence are mapped carefully. The vascular status of the limb must be unequivocally established; any signs of arterial insufficiency necessitate vascular surgery consultation prior to proceeding. If the posterior soft tissues are deemed inadequate to cover robust internal fixation, early collaboration with plastic surgery is mandatory to plan for local rotational flaps (e.g., latissimus dorsi) or free tissue transfer. The surgeon must also quantify the exact range of motion of the ipsilateral shoulder, wrist, and hand, as these will dictate the patient's postoperative functional envelope.

Determining the optimal angle of fusion is arguably the most critical and highly individualized aspect of preoperative planning. There is no universally correct angle; it must be tailored to the patient's specific anatomy, hand dominance, occupation, and primary functional goals. Generally, a dominant arm in a male laborer is fused at approximately 90 degrees to maximize strength for lifting and carrying. Conversely, female patients or those prioritizing extrapersonal activities (driving, keyboarding) may prefer a more extended angle, typically between 40 and 70 degrees. For patients prioritizing personal hygiene (feeding, facial care), a more flexed position between 90 and 110 degrees is required. To eliminate guesswork, the surgeon should employ a preoperative casting trial. The patient's arm is placed in a rigid fiberglass cast at the proposed angle for 1 to 3 weeks. This invaluable trial allows the patient to experience the profound limitations of the fused joint and actively participate in confirming or adjusting the final surgical angle before it becomes permanent.
Advanced imaging is non-negotiable. While standard anteroposterior and lateral radiographs provide a baseline understanding of joint destruction, high-resolution Computed Tomography (CT) with 3D reconstructions is essential. CT imaging allows the surgeon to precisely quantify the extent of bone loss, evaluate the structural integrity of the remaining medial and lateral humeral columns, and template the size and contour of the required internal fixation plates. If an underlying infection is suspected, particularly in cases of prior trauma or failed TEA, a comprehensive infectious workup must be completed. This includes serum inflammatory markers (ESR, CRP), joint aspiration for cell count and advanced cultures, and potentially an Indium-111 labeled leukocyte scan to localize active osteomyelitis. The surgical plan must include contingencies for massive autologous bone grafting, typically requiring preparation of the anterior or posterior iliac crest.
In the operating theater, precise patient positioning is the foundation of a successful exposure. The patient is typically placed in the lateral decubitus position, heavily padded to prevent pressure neuropathies, with the operative arm draped free over a specialized arm holder or a padded post. This position allows for unobstructed access to the posterior elbow, facilitates gravity-assisted retraction, and permits unimpeded access to the posterior iliac crest for bone graft harvesting if required. A sterile tourniquet is applied as high as possible on the brachium to maximize the surgical field. The limb is exsanguinated, and the tourniquet is inflated to provide the bloodless field necessary for meticulous neurovascular dissection and precise osseous preparation. The surgeon must ensure that a sterile goniometer, high-speed burrs, flexible osteotomes, and a comprehensive large-fragment locking plate system (including a plate press) are immediately available on the back table prior to incision.
Step-by-Step Surgical Approach and Fixation Technique
Surgical Approach and Soft Tissue Dissection
The surgical approach begins with a generous, straight posterior longitudinal incision, typically centered over the olecranon and extending proximally into the distal arm and distally along the subcutaneous border of the ulna. If previous surgical scars exist, they should be incorporated or crossed at 90-degree angles to minimize the risk of skin necrosis. Full-thickness fasciocutaneous flaps are elevated medially and laterally. It is imperative to maintain the thickness of these flaps by dissecting directly on the deep fascia, preserving the delicate subdermal vascular plexus. Once the flaps are elevated, the triceps mechanism is exposed. A midline longitudinal split of the triceps tendon and muscle belly is performed, extending distally to the olecranon insertion. This split is then carefully deepened, elevating the medial and lateral halves of the triceps subperiosteally off the distal humerus to expose the posterior aspect of the joint.
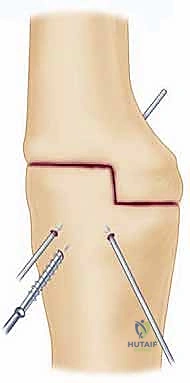
The most critical soft-tissue maneuver is the identification, neurolysis, and protection of the ulnar nerve. The nerve must be located proximally, superior to the medial epicondyle, in virgin tissue before it enters the scarred, distorted cubital tunnel. Using vessel loops, the nerve is gently mobilized and traced distally through the two heads of the flexor carpi ulnaris. In cases of severe deformity or planned massive hardware placement, an anterior subcutaneous or submuscular transposition of the ulnar nerve is highly recommended to remove it from the posterior zone of injury and prevent hardware-induced neuropathy. Throughout the procedure, the nerve must be protected with moist sponges and gentle retraction, avoiding any excessive tension that could lead to a traction neuropraxia.
Joint Preparation and Osseous Resection
With the posterior joint fully exposed and the neurovascular structures protected, the focus shifts to osseous preparation. The goal is to create broad, flat, highly vascularized bony surfaces that can be compressed against one another. All remaining articular cartilage, fibrous tissue, and sclerotic bone must be aggressively removed. A high-speed burr or an oscillating saw is utilized to decorticate the trochlea, capitellum, and the corresponding surfaces of the greater sigmoid notch of the ulna. To maximize the surface area for fusion, the radial head is routinely excised. This allows the proximal ulna to be translated laterally and seated deeply into the prepared distal humerus, enhancing bony contact and mechanical stability.
To further optimize the biological environment for fusion, the surgeon should employ the "fish-scaling" technique. Using a sharp, flexible osteotome, multiple small, overlapping cortical cuts are made into the subchondral bone of both the humerus and the ulna. This technique significantly increases the surface area, opens the Haversian canals, and releases osteoprogenitor cells and bone marrow into the fusion site. If significant bone voids exist after debridement, particularly in cases of failed TEA, these defects must be meticulously packed with autologous cancellous bone graft, typically harvested from the iliac crest. The graft acts as an osteoconductive scaffold and provides the necessary osteoinductive factors to bridge the osseous defects.
Internal Fixation and Construct Assembly
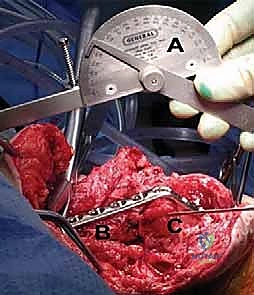
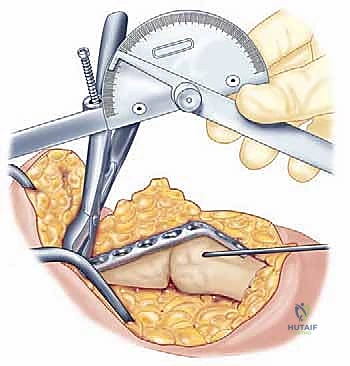
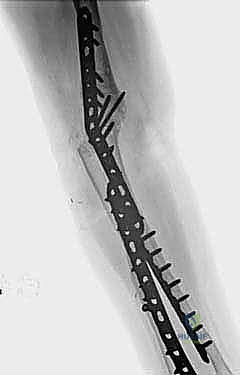
The biomechanical success of the arthrodesis hinges entirely on the rigidity of the internal fixation construct. The current gold standard involves the use of heavy, large-fragment (4.5-mm) locking compression plates (LCP) applied to the posterior aspect of the humerus and ulna. The elbow is positioned at the pre-determined, templated angle, and this angle is strictly verified using a sterile intraoperative goniometer. Provisional fixation is achieved using multiple stout Kirschner wires or a strategically placed articulated tension device. The 4.5-mm narrow LCP is then meticulously contoured using a heavy-duty plate press. The plate must bridge the posterior humerus, navigate the complex bend of the chosen fusion angle, and sit flush against the posterior border of the ulna.
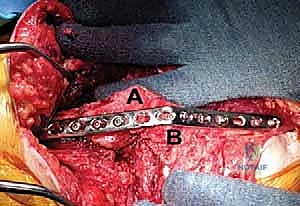
Once the plate is perfectly contoured, it is secured to the humerus and ulna. It is critical to achieve dynamic compression across the arthrodesis site before committing to locking screws. This is typically accomplished by utilizing the eccentric drilling technique in the dynamic compression holes of the plate, or by placing independent lag screws across the ulnohumeral interface prior to plate application. These interfragmentary lag screws provide massive compressive forces, maximizing bony apposition. Once compression is achieved, the construct is neutralized and rigidly stabilized by filling the remaining plate holes with locking screws.
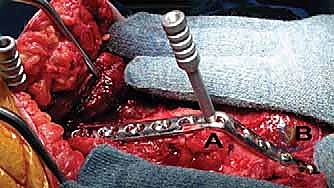
In cases of profound osteopenia, massive bone loss, or highly unstable configurations, a single posterior plate may be biomechanically insufficient. In these challenging scenarios, the surgeon must employ a dual-plating technique. A second plate, often a 3.5-mm reconstruction plate or a pre-contoured distal humerus plate, is applied orthogonally—typically along the medial or lateral column of the humerus and extending down the ulnar shaft. This orthogonal construct provides supreme torsional and bending stability, significantly mitigating the high forces generated by the long lever arm of the forearm. Copious amounts of bone graft are packed around the fusion mass and hardware before the triceps is meticulously repaired and the wound is closed in layers over a closed suction drain.

Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique and rigid internal fixation, elbow arthrodesis remains fraught with a high incidence of postoperative complications. The hostile biomechanical environment, characterized by massive cantilever forces and torsional stresses, constantly challenges the integrity of the fusion mass. Furthermore, the patient population requiring this salvage procedure often presents with compromised local biology, poor soft-tissue envelopes, and a history of multiple prior surgical interventions. The surgeon must be acutely aware of these risks, counsel the patient extensively preoperatively, and possess the technical expertise to manage these complications when they inevitably arise.
The most formidable and frequent complication is nonunion or pseudoarthrosis. Historically, nonunion rates have been reported as high as 15% to 30%, though modern locked plating techniques and the aggressive use of autologous bone grafting have improved these outcomes. Nonunion typically presents as persistent, localized pain at the surgical site, hardware failure (screw breakage or plate fracture), and progressive loss of the established fusion angle. The primary cause is inadequate mechanical stability, failure to achieve broad bony contact, or biological failure due to infection or poor vascularity. Management of a symptomatic nonunion requires a complex revision surgery. This involves complete hardware explantation, aggressive debridement of the fibrous pseudoarthrosis back to bleeding bone, massive structural or cancellous bone grafting (often requiring a vascularized fibular graft in severe cases), and the application of an even more rigid, often orthogonal, dual-plating construct.
Infection, both superficial and deep, is another devastating complication, particularly in patients undergoing arthrodesis for a previously infected total elbow arthroplasty or severe open trauma. The posterior elbow has a notoriously thin soft-tissue envelope, and the introduction of bulky internal fixation hardware creates a significant risk for wound breakdown and subsequent bacterial colonization. Deep periprosthetic infection presents with erythema, drainage, escalating pain, and elevated systemic inflammatory markers. Eradication of a deep infection requires urgent surgical intervention, consisting of radical irrigation and debridement, potential removal of all hardware (if stability is compromised or if the infection is chronic), placement of antibiotic-impregnated cement spacers, and a prolonged course of culture-directed intravenous antibiotics. In catastrophic cases where the infection cannot be controlled, or if massive, unmanageable bone loss occurs, the ultimate salvage procedure is amputation above the elbow.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage Strategy / Management |
|---|---|---|---|
| Nonunion / Pseudoarthrosis | 10% - 25% | Inadequate fixation, poor bone stock, massive cantilever forces, smoking, prior infection. | Revision open reduction internal fixation (ORIF) with dual orthogonal plating, aggressive decortication, and massive autologous bone grafting (iliac crest or vascularized fibula). |
| Deep Surgical Site Infection | 5% - 15% | Poor soft tissue envelope, prior septic arthritis, failed infected TEA, prolonged operative time. | Radical irrigation and debridement, hardware removal if loose, antibiotic spacers, prolonged IV antibiotics, potential flap coverage. Ultimate salvage: Amputation. |
| Ulnar Neuropathy | 5% - 10% | Iatrogenic traction during exposure, entrapment in scar tissue, compression from hardware. | Prompt surgical exploration, neurolysis, and anterior subcutaneous or submuscular transposition of the ulnar nerve away from the hardware. |
| Hardware Failure | 5% - 15% | Fatigue failure of plates/screws secondary to delayed union or nonunion. | Hardware removal, reassessment of bone quality, revision fixation with heavier or dual constructs, and bone grafting. |
| Wound Breakdown / Necrosis | 10% - 20% | Thin posterior skin flaps, multiple prior incisions, bulky underlying hardware. | Local wound care, negative pressure wound therapy (VAC). If deep structures/hardware exposed: urgent plastic surgery consultation for local or free flap coverage. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following an elbow arthrodesis is a delicate balance between protecting the fragile internal fixation construct and preventing devastating stiffness in the adjacent joints. Immediately following surgery, the operative arm is immobilized in a well-padded, rigid posterior splint or a bivalved fiberglass cast at the exact angle of fusion achieved intraoperatively. The arm must be kept strictly elevated above the level of the heart for the first 48 to 72 hours to minimize edema, which is critical for protecting the precarious posterior soft-tissue envelope and preventing wound dehiscence. Pain management is typically multimodal, utilizing regional anesthesia blocks placed preoperatively, supplemented with oral narcotics and anti-inflammatories.
During the initial phase of healing (Weeks 0 to 6), the primary focus is absolute protection of the arthrodesis site. The elbow remains continuously immobilized. However, active and passive range of motion exercises for the ipsilateral shoulder, wrist, and hand must commence on postoperative day one. The patient is instructed to perform aggressive finger flexion and extension, wrist circumduction, and shoulder pendulums and elevations within their pain tolerance. This early mobilization is absolutely critical; the patient will rely entirely on these adjacent joints to compensate for the fused elbow. Any loss of motion in the shoulder or wrist during this phase will exponentially compound their final functional disability. The patient is strictly instructed to maintain non-weight-bearing status on the operative extremity.
The intermediate phase (Weeks 6 to 12) begins once radiographic evidence of early bridging callus is observed. The rigid cast may be transitioned to a custom-molded thermoplastic splint, which can be removed for hygiene and gentle skin care. The patient may begin light, progressive activities of daily living using the operative arm, strictly limiting lifting to less than one to two pounds. Strengthening exercises for the shoulder girdle and wrist musculature are intensified. The final phase of rehabilitation (Months 3 to 6 and beyond) is initiated only after definitive clinical and radiographic union is confirmed, typically demonstrating trabecular bone crossing the ulnohumeral interface. The splint is discontinued, and the patient is gradually cleared for progressive resistance training and a return to heavier occupational tasks. Maximum medical improvement is often not reached until 12 to 18 months postoperatively, as the patient continues to adapt to their altered biomechanics and develop new compensatory movement strategies.
Summary of Landmark Literature and Clinical Guidelines
The academic literature surrounding elbow arthrodesis is characterized by small, retrospective case series, reflecting the rarity of the procedure in modern orthopedic practice. However, several landmark studies have significantly shaped our current surgical algorithms, particularly regarding the optimal position of fusion and the evolution of fixation techniques. Early literature, heavily influenced by the sequelae of tuberculosis and poliomyelitis, focused primarily on achieving stability at the expense of all else. As the indications shifted toward post-traumatic reconstruction and failed arthroplasty, the literature began to emphasize the profound functional consequences of the fusion angle.
A seminal study by
