Carpal Tunnel Decompression: What to Expect from Surgery

Key Takeaway
Here are the crucial details you must know about Carpal Tunnel Decompression: What to Expect from Surgery. Carpal tunnel decompression is a surgical procedure designed to relieve median nerve compression neuropathy at the wrist. This intervention, a form of peripheral nerve surgery, creates more space for the median nerve, addressing the root cause of symptoms. It is primarily performed to alleviate discomfort and restore nerve function, often under local anesthesia.
Comprehensive Introduction and Patho-Epidemiology
Principles of Peripheral Nerve Surgery
The fundamental tenets of peripheral nerve surgery dictate a meticulous, atraumatic approach to the delicate neural architecture and its surrounding vascular supply. Whether addressing the median nerve at the wrist or the ulnar nerve at the cubital tunnel, the overarching goal remains consistent: the restoration of normal axonal transport and the mitigation of intraneural microvascular ischemia. The peripheral nerve is a highly dynamic structure, reliant on continuous anterograde and retrograde axoplasmic flow to maintain cellular viability and target organ function. Surgical intervention, therefore, must not only relieve extrinsic compression but also respect the intrinsic epineurial and perineurial blood supply. The vasa nervorum, comprising a complex plexus of segmental vessels, is highly susceptible to traction and thermal injury during dissection. Consequently, the surgeon must employ magnification, utilize sharp dissection over blunt avulsion, and maintain strict hemostasis using bipolar electrocautery at the lowest effective settings to prevent iatrogenic thermal necrosis.
Furthermore, the timing of surgical intervention is dictated by the precise pathophysiological stage of nerve compression, classically categorized by Seddon and Sunderland. In cases of simple neurapraxia, characterized by localized demyelination without axonal disruption, decompression yields rapid and often complete recovery. However, in prolonged compressive neuropathies leading to axonotmesis or early neurotmesis, the potential for recovery is inversely proportional to the duration of compression and the distance to the target motor endplates. The principles of peripheral nerve surgery also mandate a thorough understanding of the "double crush" phenomenon, wherein a proximal subclinical compression (e.g., cervical radiculopathy) renders the distal nerve segment highly susceptible to symptomatic entrapment. Failure to recognize and address these concurrent pathologies invariably leads to suboptimal surgical outcomes and persistent patient dissatisfaction.

Pathophysiology of Compressive Neuropathy
The pathophysiology of compressive neuropathy, particularly within the constrained osteofibrous boundaries of the carpal tunnel, is fundamentally rooted in a cascade of microvascular compromise and subsequent mechanical deformation. The normal resting pressure within the carpal tunnel ranges from 2 to 10 mm Hg. In patients with Carpal Tunnel Syndrome (CTS), this baseline pressure is frequently elevated above 30 mm Hg, a threshold known to impede epineurial venous return. This venous congestion initiates a vicious cycle of localized edema, further elevating intracompartmental pressure and eventually compromising arterial inflow. The resulting ischemia disrupts the ATP-dependent sodium-potassium pumps within the axonal membrane, leading to the early clinical manifestations of paresthesia and intermittent numbness, particularly exacerbated by nocturnal wrist flexion which further spikes intracanalicular pressures.
As the duration and severity of compression progress, the ischemic environment induces structural changes within the nerve. Schwann cells undergo metabolic distress, leading to localized segmental demyelination. This demyelination manifests electrodiagnostically as prolonged distal latencies and decreased conduction velocities. If the mechanical insult persists, the inflammatory cascade stimulates the proliferation of epineurial and perineurial fibroblasts, resulting in intraneural fibrosis. This fibrotic scarring restricts the normal longitudinal excursion of the median nerve during wrist and finger motion, causing repetitive traction micro-trauma. Ultimately, prolonged ischemia and mechanical tethering lead to Wallerian degeneration of the axons, clinically presenting as fixed sensory deficits, constant numbness, and profound atrophy of the thenar musculature (specifically the abductor pollicis brevis and opponens pollicis).
Epidemiology and Demographics
Carpal Tunnel Syndrome stands as the most ubiquitous entrapment neuropathy in the upper extremity, representing a significant source of morbidity and economic burden globally. Epidemiological data indicates a prevalence of approximately 3% to 6% in the general adult population, with an incidence rate that continues to climb in industrialized nations. The demographic distribution exhibits a pronounced bimodal peak, primarily affecting individuals in their late 40s to early 60s, with a secondary, smaller peak occurring in the elderly population over the age of 70. There is a distinct sexual dimorphism associated with the condition, as females are affected at a rate approximately three to four times higher than their male counterparts. This discrepancy is largely attributed to the comparatively smaller cross-sectional area of the female carpal tunnel, combined with hormonal fluctuations associated with pregnancy, menopause, and the use of oral contraceptives, which can influence systemic fluid retention and tenosynovial volume.
The etiology of CTS is predominantly idiopathic, though a myriad of systemic and occupational risk factors have been definitively linked to its pathogenesis. Endocrine disorders, most notably diabetes mellitus and hypothyroidism, significantly predispose individuals to peripheral nerve entrapment due to compromised microcirculation and mucopolysaccharide deposition within the perineurium, respectively. Inflammatory arthritides, such as rheumatoid arthritis, frequently cause proliferative tenosynovitis of the flexor tendons, directly encroaching upon the limited space within the carpal canal. Occupationally, repetitive forceful manual labor, the use of vibratory tools, and prolonged extremes of wrist posture have been heavily implicated in the development of CTS. Despite the common societal association, rigorous epidemiological studies have failed to establish a definitive, independent causal link between standard computer keyboard usage and the onset of clinical Carpal Tunnel Syndrome, suggesting that ergonomic factors must be considered in conjunction with intrinsic anatomical and physiological predispositions.
Detailed Surgical Anatomy and Biomechanics
Osteofibrous Boundaries of the Carpal Tunnel
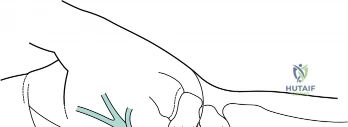
A profound, three-dimensional understanding of the carpal tunnel's osteofibrous architecture is the absolute prerequisite for safe and effective surgical decompression. The carpal tunnel is an elliptical, inextensible conduit situated at the volar aspect of the wrist. Its floor and lateral walls are formed by the deep concave arch of the carpal bones. Proximally, the ulnar border is defined by the pisiform, while the radial border is marked by the tubercle of the scaphoid. Distally, the boundaries transition to the hook of the hamate on the ulnar side and the crest of the trapezium on the radial side. This bony trough is converted into a closed tunnel by the robust transverse carpal ligament (TCL), also known as the flexor retinaculum, which spans the volar aspect of the carpal arch.
The TCL is a dense, highly organized band of connective tissue, measuring approximately 1.5 to 2.0 mm in thickness and 3 to 4 cm in length. It is continuous proximally with the deep investing fascia of the forearm and the palmar carpal ligament, and distally it merges with the palmar aponeurosis. The canal itself is densely packed, containing exactly ten structures: the four tendons of the flexor digitorum superficialis (FDS), the four tendons of the flexor digitorum profundus (FDP), the tendon of the flexor pollicis longus (FPL), and the median nerve. The median nerve is typically the most superficial structure within the tunnel, lying directly beneath the TCL, making it highly vulnerable to compression against this rigid ligamentous roof when tenosynovial volume increases. The spatial constraints are so precise that an increase in the volume of the contents by merely a few milliliters, or a slight dynamic alteration in the shape of the boundaries during wrist flexion or extension, drastically elevates intracanalicular pressure.

The Median Nerve and its Variations
The median nerve enters the forearm between the two heads of the pronator teres and courses distally deep to the FDS. As it approaches the wrist, it emerges from beneath the radial margin of the FDS muscle belly to lie between the tendons of the FDS and the flexor carpi radialis (FCR). Just proximal to the carpal tunnel, the nerve gives off the palmar cutaneous branch (PCB). The PCB is of paramount surgical importance; it arises from the radial-volar aspect of the median nerve approximately 5 cm proximal to the wrist crease, travels distally between the palmaris longus (PL) and FCR, and passes superficial to the TCL to provide sensation to the base of the thenar eminence. An improperly placed surgical incision, particularly one that strays too far radially, risks catastrophic iatrogenic neuroma formation of the PCB.
Within the carpal tunnel, the median nerve typically divides into its terminal sensory and motor branches at the distal margin of the TCL. However, the branching pattern of the recurrent motor branch to the thenar musculature is highly variable and represents a critical hazard during decompression. The Lanz classification categorizes these variations into four main types. Type I (Standard) features an extraligamentous, recurrent branching pattern (approximately 46-90% of cases). Type II exhibits a subligamentous origin, where the branch arises within the tunnel and courses beneath the TCL before turning radially. Type III is the transligamentous variant, where the motor branch pierces directly through the substance of the TCL; this variant is at extreme risk of transection during both open and endoscopic releases. Type IV involves an accessory branch arising proximal to the carpal tunnel. The prudent surgeon must operate under the assumption that an anomalous branch is present until visual confirmation dictates otherwise.
Biomechanics of the Transverse Carpal Ligament
The transverse carpal ligament is not merely a passive restraint; it is a critical biomechanical structure that contributes significantly to the stability and kinematics of the carpal arch. By tethering the thenar and hypothenar eminences, the TCL acts as a tie-rod, maintaining the concavity of the carpal arch and preventing splaying of the carpal bones under axial loads. Complete surgical transection of the TCL, while necessary for nerve decompression, inherently alters this biomechanical equilibrium. Post-operatively, patients often experience an increase in the transverse dimension of the carpal arch, which can translate clinically into a phenomenon known as "pillar pain"—a deep, aching discomfort localized to the thenar and hypothenar eminences during grasping activities.
Furthermore, the TCL serves as a crucial pulley for the extrinsic flexor tendons. It prevents bowstringing of the FDS and FDP tendons during active wrist flexion, thereby maximizing their mechanical advantage and preserving grip strength. Following TCL release, there is a transient, and sometimes permanent, alteration in flexor tendon kinematics. The flexor tendons displace volarly, increasing their moment arm but simultaneously decreasing the total excursion available for digit flexion. This biomechanical shift explains the ubiquitous, albeit usually temporary, reduction in grip and pinch strength observed in the immediate months following carpal tunnel surgery. Surgeons must counsel patients that while paresthesias may resolve rapidly, maximal grip strength may take upwards of six to twelve months to fully recover as the regional anatomy adapts to the released ligamentous constraints.

Exhaustive Indications and Contraindications
Clinical Diagnosis and Electrodiagnostic Criteria
The diagnosis of Carpal Tunnel Syndrome is primarily clinical, predicated on a meticulous history and physical examination. Patients classically present with nocturnal paresthesias, clumsiness, and numbness in the radial three-and-a-half digits. Provocative testing is essential: Phalen’s test (sustained wrist flexion) and the Durkan carpal compression test boast high sensitivity and specificity. The presence of a positive Tinel’s sign at the wrist crease further corroborates the diagnosis. Advanced physical findings, such as static two-point discrimination greater than 5 mm or visible atrophy of the abductor pollicis brevis, indicate severe, long-standing compression and portend a less predictable surgical recovery.
While the diagnosis is clinical, electrodiagnostic studies (Electromyography and Nerve Conduction Velocity - EMG/NCV) are the gold standard for confirming the diagnosis, stratifying severity, and ruling out proximal entrapments (e.g., cervical radiculopathy, pronator syndrome). Pathognomonic NCV findings include a prolonged distal sensory latency (typically > 3.5 ms) and a prolonged distal motor latency (typically > 4.5 ms). In severe cases, needle EMG may reveal fibrillations, positive sharp waves, or decreased motor unit recruitment in the thenar musculature, signifying active axonal loss and denervation. Additionally, high-resolution neuromuscular ultrasound is increasingly utilized as a complementary diagnostic modality; a median nerve cross-sectional area (CSA) exceeding 10 to 12 mm² at the level of the pisiform is highly indicative of pathological swelling secondary to compression.
Surgical Indications
The decision to proceed with carpal tunnel decompression is dictated by the severity of symptoms, the duration of the pathology, and the patient's response to conservative management. Surgical intervention is unequivocally indicated in patients presenting with acute carpal tunnel syndrome, often secondary to distal radius fractures, perilunate dislocations, or acute hemorrhagic events within the tunnel. In these scenarios, emergent decompression is required to prevent irreversible ischemic necrosis of the median nerve. For chronic CTS, surgery is indicated when patients exhibit recalcitrant symptoms that have failed a dedicated trial of non-operative modalities, including nocturnal rigid splinting, non-steroidal anti-inflammatory drugs (NSAIDs), and corticosteroid injections.
Furthermore, surgical decompression is strongly recommended as a first-line treatment for patients presenting with severe clinical or electrodiagnostic findings. The presence of constant numbness, objective sensory loss, or thenar muscle atrophy represents impending or active neurotmesis; delaying surgery in these cases risks permanent functional impairment. Electrodiagnostically, absent sensory nerve action potentials (SNAPs) or severe motor denervation on EMG are absolute indications for prompt surgical release. The surgeon must clearly communicate to the patient that in cases of profound preoperative atrophy, the primary goal of surgery is to arrest the progression of nerve damage and alleviate pain, rather than to guarantee the complete restoration of muscle bulk or fine tactile sensation.

Absolute and Relative Contraindications
While carpal tunnel release is a highly successful procedure, meticulous patient selection is paramount to avoid catastrophic outcomes and patient dissatisfaction. Absolute contraindications are rare but include active, uncontrolled local infection at the surgical site (e.g., cellulitis, purulent tenosynovitis) and severe, uncorrectable coagulopathies that would preclude safe hemostasis. Additionally, an incorrect diagnosis—such as attributing symptoms entirely to CTS when a severe C6/C7 cervical radiculopathy is the primary driver of the pathology—constitutes an absolute contraindication to isolated carpal tunnel release, as the procedure will fail to resolve the patient's symptoms.
Relative contraindications require careful preoperative optimization and nuanced clinical judgment. Patients with mild, intermittent symptoms and normal electrodiagnostic studies should generally be managed conservatively, as the risks of surgery may outweigh the marginal benefits. Severe psychiatric comorbidities or a history of complex regional pain syndrome (CRPS) in the ipsilateral extremity represent significant relative contraindications, as surgical trauma can trigger a severe, debilitating flare of sympathetically mediated pain. Furthermore, patients with profound, long-standing systemic neuropathies (e.g., end-stage diabetic peripheral neuropathy) must be counseled extensively; while decompression may relieve superimposed focal entrapment, it will not reverse the underlying systemic axonal degradation, and postoperative recovery will be markedly protracted and incomplete.
| Category | Indications for Decompression | Contraindications (Absolute & Relative) |
|---|---|---|
| Clinical | Refractory symptoms > 3-6 months; Thenar atrophy; Constant numbness | Active local infection (Absolute); Uncontrolled CRPS (Relative) |
| Diagnostic | Prolonged motor/sensory latencies; Denervation on EMG; Ultrasound CSA > 12mm² | Normal EMG/NCS with mild symptoms (Relative); Misdiagnosis (Absolute) |
| Trauma/Acute | Acute CTS post-fracture/dislocation; Compartment syndrome of the hand | Uncorrectable coagulopathy (Absolute) |
| Systemic | Failure of corticosteroid injections; Progressive loss of hand dexterity | End-stage diffuse polyneuropathy (Relative - requires tempered expectations) |
Pre-Operative Planning, Templating, and Patient Positioning
Pre-Operative Assessment and Optimization
Meticulous preoperative planning begins with a comprehensive review of the patient's medical history, focusing on factors that influence wound healing and neuro-recovery. Glycemic control must be optimized in diabetic patients, as elevated HbA1c levels correlate directly with increased surgical site infection rates and diminished potential for axonal regeneration. Anticoagulant and antiplatelet medications must be managed in accordance with current perioperative guidelines. While routine carpal tunnel release can often be performed safely without cessation of low-dose aspirin, more potent anticoagulants (e.g., Warfarin, DOACs) may necessitate a brief perioperative pause or bridging therapy, depending on the patient's cardiovascular risk profile and the surgeon's preference for regional versus local anesthesia.
The physical examination immediately prior to surgery must confirm the surgical site and the laterality of the procedure. The surgeon should carefully palpate and mark the anatomical landmarks: the pisiform, the hook of the hamate, the tubercle of the scaphoid, and the crest of the trapezium. The course of the palmaris longus (if present) and the flexor carpi radialis should be identified to estimate the trajectory of the median nerve and its palmar cutaneous branch. In cases of revision surgery or anticipated complex anatomy (e.g., severe rheumatoid tenosynovitis, prior trauma), advanced imaging such as MRI may be warranted to evaluate the extent of scarring, the presence of space-occupying lesions (e.g., lipomas, ganglion cysts), and the precise morphological status of the median nerve.

Anesthesia Considerations and Tourniquet Use
The anesthetic approach to carpal tunnel decompression has evolved significantly, with a strong modern paradigm shift towards Wide Awake Local Anesthesia No Tourniquet (WALANT). The WALANT technique utilizes a mixture of 1% lidocaine with 1:100,000 epinephrine, buffered with sodium bicarbonate (typically in a 10:1 ratio) to mitigate the burning sensation during infiltration. This approach eliminates the risks associated with general anesthesia, bypasses the need for preoperative fasting, and avoids the ischemic pain generated by a proximal pneumatic tourniquet. Crucially, WALANT allows the surgeon to assess active digit flexion intraoperatively, confirming complete release of the TCL and ensuring that the flexor tendons glide freely without impingement.
Despite the advantages of WALANT, traditional anesthetic modalities remain highly relevant. Intravenous regional anesthesia (Bier block) or axillary brachial plexus blocks are excellent options for patients who are highly anxious, needle-phobic, or when concurrent procedures (e.g., trigger finger release, local flap coverage) are planned. When utilizing these regional techniques or general anesthesia, a proximal arm tourniquet is typically inflated to 250 mm Hg to provide a bloodless surgical field. The surgeon must be cognizant of tourniquet time; while carpal tunnel release is expeditious, prolonged tourniquet inflation (> 2 hours) can induce iatrogenic neurapraxia and exacerbate the preexisting ischemic insult to the median nerve. Therefore, efficient surgical execution and prompt tourniquet deflation are imperative.
Patient Positioning and Operating Room Setup
Optimal patient positioning is critical for surgical ergonomics and exposure. The patient is placed supine on the operating table with the operative extremity extended onto a radiolucent hand table. The shoulder is abducted to approximately 45 to 60 degrees, avoiding excessive abduction that could place traction on the brachial plexus. The forearm is supinated, and the hand is secured using a lead hand or specialized retractor system. A rolled towel or a dedicated wrist bump is placed dorsal to the radiocarpal joint to maintain the wrist in approximately 30 degrees of extension. This position optimally tensions the transverse carpal ligament, drawing it superficially and facilitating a safer, more controlled transection.
The operating room setup should prioritize the surgeon's visualization of the microscopic anatomy. Loupe magnification (minimum 2.5x to 3.5x) and a dedicated headlight are non-negotiable requirements for identifying anomalous nerve branches and ensuring complete hemostasis. The surgical tray must be equipped with delicate soft tissue instruments, including Senn retractors, Ragnell retractors, tenotomy scissors, and a Freer elevator. Bipolar electrocautery must be readily available and tested prior to the incision. For endoscopic procedures, the video tower should be positioned directly across from the surgeon, ensuring a direct line of sight without ergonomic strain, and the endoscope must be white-balanced and focused prior to insertion into the carpal canal.

Step-by-Step Surgical Approach and Fixation Technique
Open Carpal Tunnel Release Technique
The standard open carpal tunnel release remains the gold standard, offering unparalleled visualization of the neural and vascular anatomy. The incision is planned along Kaplan’s cardinal line or slightly ulnar to the longitudinal axis of the radial border of the ring finger. This placement is critical: it remains ulnar to the palmaris longus tendon, safely avoiding the palmar cutaneous branch of the median nerve, and radial to the hypothenar eminence, protecting the ulnar neurovascular bundle. A 3 to 4 cm longitudinal incision is made through the skin and subcutaneous fat. Meticulous hemostasis is achieved using bipolar electrocautery. The palmar aponeurosis is identified and longitudinally incised, revealing the distinct, transversely oriented fibers of the transverse carpal ligament beneath.
Dissection proceeds through the TCL, utilizing a combination of sharp scalpel dissection and specialized instruments such as a carpal tunnel director or a blunt-tipped tenotomy scissor. The release must be carried distally to the level of the superficial palmar arch, ensuring the distal fascial bands are completely divided. Proximally, the release extends through the antebrachial fascia, approximately 2 cm proximal to the distal wrist crease, to ensure the nerve is free from any proximal fascial tethering. Once the ligament is entirely transected, the median nerve is visually inspected. The surgeon must confirm the absence of a transligamentous motor branch and verify that the nerve demonstrates a normal hourglass deformity (a hallmark of chronic compression) rather than an acute intraoperative laceration. The floor of the tunnel is inspected for space-occupying lesions. Closure is typically limited to the skin using non-absorbable horizontal mattress sutures or a running subcuticular closure, intentionally leaving the TCL open to decompress the canal.
Endoscopic Carpal Tunnel Release Technique
Endoscopic carpal tunnel release (ECTR) was developed to minimize palmar scarring and accelerate postoperative recovery. The two most prevalent techniques are the single-portal (Agee) and the two-portal (Chow) methods. In the single-portal technique, a small 1-1.5 cm transverse incision is made proximal to the wrist crease, between the FCR and FCU tendons. The antebrachial fascia is incised, and a synovial elevator is used to clear the undersurface of the TCL, creating a working space. The endoscope/blade assembly is then introduced into the carpal tunnel, deep to the TCL and superficial to the median nerve and flexor tendons. This establishes the "safe zone."
The surgeon must meticulously visualize the transverse fibers of the TCL on the monitor. The distal edge of the ligament and the intervening fat pad must be clearly identified to avoid injury to the superficial palmar arch. Once the anatomy is unequivocally confirmed, the retrograde blade is deployed, and the ligament is divided from distal to proximal under direct endoscopic visualization. The two-portal technique involves an additional distal incision in the palm to facilitate the passage of a trocar and cannula system, allowing for visualization from both ends of the tunnel. Regardless of the specific endoscopic technique, the fundamental requirement is absolute visual clarity. If the anatomy is obscured by tenosynovitis, bleeding, or anatomical variants, the surgeon must immediately convert to an open procedure to prevent catastrophic iatrogenic injury.

Ulnar Nerve Decompression Considerations
While the median nerve is the primary focus of carpal tunnel surgery, the principles of decompression are directly applicable to the ulnar nerve, which frequently requires concurrent or isolated release. At the wrist, the ulnar nerve passes through Guyon's canal, an osteofibrous tunnel bounded by the pisiform, the hook of the hamate, the volar carpal ligament, and the transverse carpal ligament. Decompression of Guyon's canal requires a localized incision over the hypothenar eminence, carefully protecting the ulnar artery and releasing the volar carpal ligament and the hypothenar muscle fascia to free the superficial sensory and deep motor branches.
More proximally, ulnar nerve decompression at the elbow (cubital tunnel syndrome) is the second most common upper extremity entrapment neuropathy. The nerve is vulnerable as it passes posterior to the medial epicondyle beneath the arcuate ligament (Osborne's fascia). Surgical management ranges from simple in situ decompression—releasing the arcuate ligament and the fascia of the flexor carpi ulnaris—to complex anterior transposition techniques (subcutaneous, intramuscular, or submuscular). Transposition is typically reserved for cases of nerve subluxation, severe valgus deformity of the elbow, or revision surgery. The critical surgical pearl in ulnar nerve surgery is the preservation of the medial antebrachial cutaneous nerve branches during the approach and the meticulous protection of the segmental vascular supply to prevent devascularization of the transposed nerve.
Complications, Incidence Rates, and Salvage Management
Intraoperative Nerve and Vascular Injuries
The most devastating complications associated with carpal tunnel decompression are iatrogenic injuries to the neurovascular structures. The incidence of major nerve laceration is exceedingly low (less than 0.5%), but the consequences are profound. The structures most at risk include the recurrent motor branch of the median nerve, the palmar cutaneous branch, and the common digital nerves. Injury to the recurrent motor branch, particularly an unrecognized transligamentous variant, results in devastating loss of thenar opposition. If a nerve transection is identified intraoperatively, immediate microsurgical primary epineurial repair using 8-0 or 9-0 nylon is mandatory.
Vascular injuries primarily involve the superficial palmar arch or the ulnar artery. The superficial palmar arch crosses the palm just distal to the distal edge of the TCL. Aggressive distal dissection, particularly during blind endoscopic release or excessive traction during open release, can avulse or lacerate this critical vessel. Management requires immediate proximal and distal control, followed by microsurgical repair or, if collateral flow via the deep arch is confirmed to be robust (via Allen's test), meticulous ligation. Hematoma formation within the tightly constrained palmar space can lead to a localized compartment syndrome, necessitating emergent return to the operating room for evacuation and hemostasis.

Postoperative Pain Syndromes and Pillar Pain
Pillar pain is the most frequently encountered postoperative complication, affecting up to 25-30% of patients following both open and endoscopic releases. It is characterized by a deep, aching tenderness over the thenar and hypothenar eminences, exacerbated by direct pressure or heavy grasping. The etiology is multifactorial, likely involving the biomechanical alteration of the carpal arch following TCL transection, localized edema, and irritation of the microscopic nerve endings within the palmar fascia. Pillar pain is generally self-limiting but can persist for 3 to 6 months. Management is conservative, focusing on desensitization therapies, silicone gel padding, and targeted NSAID use.
A more severe, albeit less common, pain complication is Complex Regional Pain Syndrome (CRPS) Type I or Type II. CRPS presents with out-of-proportion burning pain, hyperalgesia, allodynia, and profound autonomic dysregulation (vasomotor instability, sudomotor changes, and trophic skin alterations). Early recognition is critical. Treatment requires an aggressive, multidisciplinary approach involving intensive physical therapy, neuropathic pain modulators (e.g., gabapentin, pregabalin), high-dose oral corticosteroids, and early referral to a pain management specialist for sympathetic ganglion blocks. Delayed treatment of CRPS invariably leads to permanent joint stiffness and severe functional impairment of the entire upper extremity.
Recurrent Carpal Tunnel Syndrome and Revision Surgery
Recurrent or persistent Carpal Tunnel Syndrome represents a complex clinical challenge, with incidence rates reported between 1% and 7%. The most common cause of persistent symptoms (no relief following the index procedure) is an incomplete release of the transverse carpal ligament, typically at its most distal extent.
Clinical & Radiographic Imaging Archive