Mastering Nerve Injury in Continuity: An Intraoperative Guide to Neurolysis and Split Repair
Key Takeaway
Welcome to the OR, fellows. Today, we're tackling nerve injuries in continuity—a complex challenge demanding precision. We'll meticulously dissect the nuances of diagnosis, patient selection, and surgical execution. From comprehensive neurolysis to intricate split repair, this masterclass will equip you with the advanced techniques and critical decision-making skills essential for optimizing patient outcomes in these delicate cases.
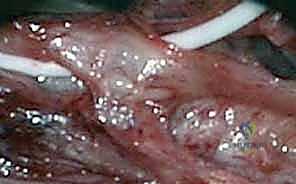
The management of peripheral nerve injuries in continuity represents one of the most formidable challenges in modern reconstructive microsurgery. Unlike a frank transection where the surgical objective is an immediate, tension-free end-to-end coaptation, an injury in continuity presents a complex, often deceptive, macroscopic appearance. The nerve trunk remains outwardly intact, yet internally, the axonal architecture and connective tissue scaffolding may be profoundly disrupted. The surgeon is tasked with a delicate intraoperative interrogation: distinguishing viable, regenerating fascicles from fibrotic, non-conductive neuromatous tissue. This chapter provides an exhaustive, evidence-based roadmap for navigating the neuroma-in-continuity, emphasizing the critical role of intraoperative electrophysiology, meticulous internal neurolysis, and the highly specialized technique of split repair.
Comprehensive Introduction and Patho-Epidemiology
A nerve injury in continuity signifies a structural or functional interruption of axonal conduction where the gross macroscopic integrity of the epineurium remains preserved. This clinical entity most frequently arises from traction, crush, or severe compressive forces, rather than sharp lacerations. The epidemiological profile is heavily skewed toward high-energy trauma, including motor vehicle collisions, industrial accidents, and complex fracture-dislocations (e.g., brachial plexus avulsions, radial nerve palsies associated with humeral shaft fractures). Understanding the precise mechanism of injury is paramount, as it dictates the longitudinal extent of intraneural damage, which often far exceeds the visually apparent zone of injury.

The pathogenesis of these lesions is intrinsically linked to the biomechanical properties of the peripheral nerve. As longitudinal stretch is applied, the undulating axons and their delicate endoneurial tubes lose their slack and are subjected to tensile forces. The neural elements—specifically the axolemma and endoneurial microvessels—are highly vulnerable to stretch and fail long before the robust, collagen-dense epineurium ruptures. This differential mechanical failure leads to intraneural hemorrhage, edema, and subsequent fibroblast proliferation, culminating in a dense, disorganized scar known as a neuroma-in-continuity. The extent of this internal scarring directly dictates the potential for spontaneous recovery and forms the basis of our clinical classification systems.
To systematically approach these injuries, we rely on the seminal classifications established by Seddon and subsequently expanded by Sunderland. Sunderland’s five-degree classification system remains the gold standard, as it correlates the microanatomical level of disruption with the anticipated clinical prognosis.
-
Sunderland Type I (Neurapraxia): Represents a localized conduction block secondary to focal demyelination or ion channel dysfunction, typically due to mild compression or stretch. Macroscopically and microscopically, the axonal continuity and connective tissue stroma are completely preserved. Crucially, Wallerian degeneration does not occur. Recovery is spontaneous, rapid, and complete, typically within days to weeks.

-
Sunderland Type II (Axonotmesis): Involves physical disruption of the axon and myelin sheath, but the endoneurial tube remains intact. Distal to the injury, the axon undergoes Wallerian degeneration, while the proximal stump forms a growth cone. Because the endoneurial conduit is preserved, regenerating axons are perfectly guided back to their original target organs. Recovery is slow (approximately 1 mm/day) but typically complete.
-
Sunderland Type III: Marks a critical threshold where both the axon and the endoneurial tube are disrupted, but the perineurium enclosing the fascicle remains intact. The loss of the endoneurial guide rails leads to intrafascicular scarring and axonal misdirection. Regenerating axons may innervate inappropriate targets (e.g., motor axons reaching sensory receptors), resulting in a clinically evident, often incomplete, and disorganized recovery.
-
Sunderland Type IV: Represents a severe internal disorganization. The axons, endoneurium, and perineurium are all destroyed, leaving only a thickened, scarred epineurium. The fascicular architecture is entirely obliterated, replaced by a dense neuroma-in-continuity. Spontaneous, functional regeneration across this fibrotic barrier is impossible. Surgical resection and grafting are absolutely mandated.
-
Sunderland Type V (Neurotmesis): A complete macroscopic transection of the nerve trunk. While not technically an injury in continuity, it represents the terminal end of the injury spectrum, requiring immediate or early surgical coaptation to bridge the physical gap.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of peripheral nerve microanatomy and its surrounding vascular networks is the prerequisite for executing complex neurolysis and split repairs. The peripheral nerve is not a simple biological wire; it is a highly organized, dynamic, and vascularized organ. The functional unit is the axon, enveloped by Schwann cells that produce the insulating myelin sheath in myelinated fibers. These axons are bundled within the endoneurium, a delicate matrix of type I and type III collagen fibers and glycosaminoglycans. The endoneurial space is tightly regulated, maintaining a specific ionic milieu essential for action potential propagation.
Multiple endoneurial tubes are grouped together to form a fascicle, which is circumscribed by the perineurium. The perineurium is a biomechanically robust, multilayered structure composed of specialized perineurial cells possessing tight junctions. This structure functions as the blood-nerve barrier, protecting the intraneural environment from systemic fluctuations and toxins. The perineurium also provides the primary tensile strength of the peripheral nerve, allowing it to withstand physiological stretching across joints. During internal neurolysis, the surgeon must exercise extreme caution to avoid violating the perineurium, as iatrogenic breach leads to axonal herniation and catastrophic scarring.
The entire nerve trunk is enveloped by the epineurium, which is subdivided into the internal (interfascicular) epineurium and the external epineurium. The internal epineurium acts as a cushion, allowing fascicles to glide against one another during limb movement. The external epineurium defines the outer boundary of the nerve and anchors it to the surrounding fascial beds via the mesoneurium. The vascular supply to the nerve, the vasa nervorum, consists of an extrinsic system (segmental vessels entering via the mesoneurium) and an intrinsic system (longitudinal capillary networks within the epineurium, perineurium, and endoneurium). Surgical mobilization must be judicious; stripping the nerve over extensive lengths destroys the extrinsic supply, forcing the nerve to rely solely on the precarious intrinsic network, potentially inducing ischemic necrosis and exacerbating fibrotic scarring.
Exhaustive Indications and Contraindications
The decision-making algorithm for a nerve injury in continuity hinges on the precise timing of the injury, the clinical trajectory of recovery, and the findings of serial electrodiagnostic testing. The overarching principle is to allow sufficient time for spontaneous regeneration (Sunderland I-III injuries) while not delaying surgical intervention so long that the distal motor endplates undergo irreversible atrophy and fibrosis. The critical window of opportunity for surgical intervention typically lies between 3 and 6 months post-injury for closed, high-energy traction or crush injuries.
The primary indication for surgical exploration of a nerve in continuity is the absolute failure of clinical or electrophysiological progression at the 3-to-4-month mark. If serial clinical examinations reveal no advancing Tinel's sign, and electromyography (EMG) demonstrates persistent fibrillation potentials without nascent motor unit action potentials (MUAPs) in the most proximal muscles innervated by the injured nerve, spontaneous recovery is deemed highly unlikely. In these scenarios, the nerve is trapped in a Sunderland IV state, and surgical resection of the neuroma with subsequent grafting is indicated. Furthermore, severe, intractable neuropathic pain or a rapidly progressing neurological deficit in the setting of a known compressive lesion (e.g., expanding hematoma or severe fracture displacement) serves as an absolute indication for urgent exploration.
Contraindications to early exploration of closed injuries include documented clinical improvement, advancing sensory or motor function, and early signs of reinnervation on EMG. Operating prematurely on a Sunderland II or III injury risks iatrogenic damage to regenerating axons, effectively converting a recovering lesion into a surgical neurotmesis. Additionally, severe physiological instability, active local infection, or an uncooperative patient unable to adhere to complex postoperative rehabilitation protocols represent significant contraindications. Late presentations (greater than 12–18 months post-injury) present a relative contraindication for proximal nerve repairs directed at distal musculature, as the motor endplates are likely irrecoverably degenerated; in such cases, palliative tendon transfers or free functional muscle transfers are preferred.
| Indication | Contraindication |
|---|---|
| Closed traction/crush injury with no clinical/EMG recovery at 3-4 months | Documented clinical or EMG evidence of progressive reinnervation |
| Open injuries with suspected high-grade continuity lesions (e.g., gunshot wounds) | Premature timing (< 3 months) in closed injuries without baseline establishment |
| Intractable, debilitating neuropathic pain localized to a neuroma-in-continuity | Active local or systemic infection |
| Rapidly progressive neurological deficit in the setting of known trauma | Late presentation (> 18 months) for proximal nerve to distal motor targets |
| Intraoperative identification of a non-conducting neuroma (via NAP) | Severe medical comorbidities precluding prolonged microsurgery |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the cornerstone of a successful surgical outcome. The clinical examination must be exhaustive, documenting precise Medical Research Council (MRC) muscle grades and mapping sensory deficits using two-point discrimination and Semmes-Weinstein monofilaments. The exact anatomical location of the advancing Tinel's sign must be measured from fixed bony landmarks and recorded at each visit to calculate the rate of regeneration. This clinical data provides the baseline against which intraoperative findings will be judged.
Electrodiagnostic studies (EMG and Nerve Conduction Studies) are indispensable. An initial baseline study is typically obtained at 3 to 4 weeks post-injury to confirm Wallerian degeneration (evident by fibrillation potentials and positive sharp waves in denervated muscle). A follow-up study at 12 weeks is critical; the presence of nascent MUAPs indicates early axonal reinnervation long before clinical movement is detectable, prompting continued observation. Conversely, electrical silence in the target muscles strongly advocates for surgical exploration. Advanced imaging modalities, particularly High-Resolution Ultrasound (HRUS) and Magnetic Resonance Neurography (MRN), have revolutionized our preoperative templating. HRUS allows for real-time, dynamic assessment of fascicular continuity and neuroma cross-sectional area, while MRN can vividly demonstrate T2 hyperintensity indicative of downstream denervation edema and precise neuroma localization.
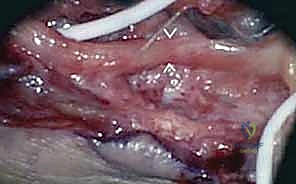
Patient positioning in the operating room must facilitate wide, unhindered access to the injured nerve, its proximal and distal normal segments, and potential autograft donor sites (most commonly the bilateral sural nerves). For upper extremity cases, the patient is typically positioned supine with the arm extended on a radiolucent hand table. A sterile tourniquet is applied but inflated only judiciously, as prolonged ischemia can confound intraoperative electrophysiological testing. The surgical microscope must be draped and balanced prior to the incision, and the intraoperative nerve monitoring equipment (bipolar stimulating and recording electrodes) must be calibrated and confirmed functional by the neurophysiologist.
Step-by-Step Surgical Approach and Fixation Technique
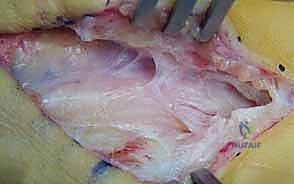
The surgical approach to a neuroma-in-continuity demands meticulous, atraumatic microsurgical technique. The fundamental principle is to initiate the dissection in pristine, unscarred anatomical planes both proximal and distal to the zone of injury. By identifying the normal nerve trunk first, the surgeon can trace it toward the epicenter of the neuroma, carefully dissecting it free from the surrounding fibrotic bed. This external neurolysis involves incising the overlying fascia and meticulously dividing the compressive epineurial scar tissue, freeing the nerve circumferentially while strictly preserving the longitudinal vasa nervorum.
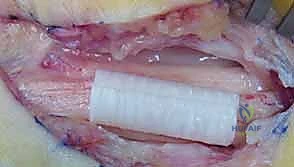
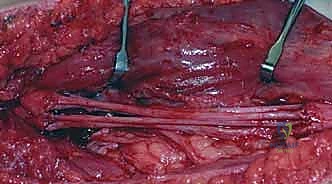
Once the neuroma is completely mobilized, the most critical phase of the operation commences: the acquisition of Intraoperative Nerve Action Potentials (NAPs). This technique, pioneered by Kline, is the definitive arbiter of whether a neuroma-in-continuity requires resection or merely neurolysis. The nerve is suspended on bipolar stimulating electrodes proximally and recording electrodes distally across the neuroma. A supramaximal stimulus is applied. If a reproducible NAP is recorded distally, it definitively proves that a functionally significant number of axons (typically >3000-4000 myelinated fibers) have successfully traversed the scar tissue. In this scenario, external neurolysis alone is sufficient, and the nerve is left intact.
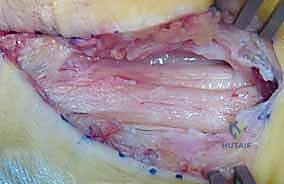
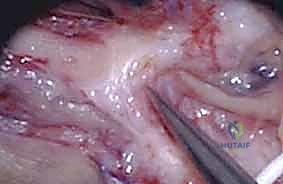
Conversely, the absence of a transmitted NAP across the neuroma at the 3-to-4-month mark indicates a Sunderland IV injury. The dense intraneural scar is an insurmountable barrier to regeneration. The surgeon must then proceed with resection of the neuroma back to healthy, pouting fascicles proximally and distally, followed by reconstruction. However, the clinical reality is rarely binary. Often, the nerve exhibits a mixed injury pattern, where some fascicles conduct a NAP while others are completely fibrosed. This necessitates the highly advanced technique of internal neurolysis and split repair.
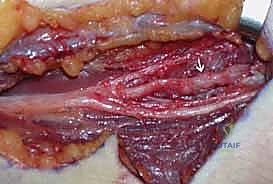
During a split repair, the external epineurium is longitudinally incised under high microscopic magnification. Using micro-scissors and jewelers' forceps, the surgeon delicately dissects the internal epineurium, separating the individual fascicular groups. Each fascicular group is then independently interrogated with the NAP electrodes.
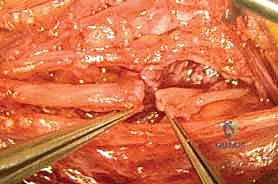
The functioning fascicles (those conducting a NAP) are carefully neurolysed and preserved. The non-functioning, fibrotic fascicular groups are sharply resected using a diamond knife or micro-scissors until healthy, unscarred axonal tissue is visualized.
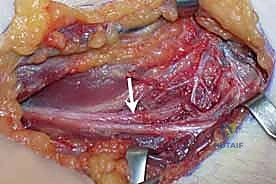
This selective resection creates a segmental defect specifically in the non-viable portion of the nerve.

To bridge this defect, autologous nerve grafting is the gold standard. The sural nerve is the most common donor, harvested via a minimally invasive step-cut technique or endoscopic approach to minimize donor-site morbidity.

The harvested graft is reversed (to prevent axonal branching into dead-end donor branches) and cut into cables matching the length of the defect.
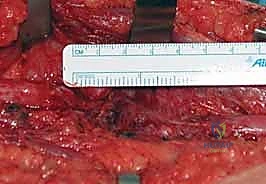
These cables are meticulously coapted to the proximal and distal stumps of the resected fascicles using 9-0 or 10-0 nylon sutures placed strictly in the epineurium or perineurium, ensuring a tension-free repair. Fibrin glue may be used to augment the repair and reduce the required number of sutures, thereby minimizing foreign body reaction and subsequent scarring.
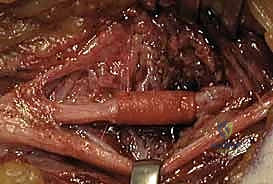
Complications, Incidence Rates, and Salvage Management
Despite flawless microsurgical execution, the reconstruction of nerve injuries in continuity carries a high risk of complications, primarily dictated by the biological limitations of axonal regeneration. The most devastating complication is the failure of functional motor recovery. This occurs when regenerating axons fail to reach the motor endplates before irreversible muscle fibrosis ensues (typically 18-24 months post-injury). The incidence of failure is directly proportional to the proximal level of the injury and the length of the required graft; brachial plexus reconstructions have significantly higher failure rates than distal forearm repairs.
Neuropathic pain and the development of a symptomatic recurrent neuroma at the proximal coaptation site are also significant concerns. Patients may develop Complex Regional Pain Syndrome (CRPS), characterized by severe allodynia, hyperalgesia, and autonomic dysfunction. Management of CRPS requires a multidisciplinary approach involving aggressive pharmacological therapy (gabapentinoids, tricyclic antidepressants), sympathetic blocks, and intensive desensitization therapy. If a painful focal neuroma recurs, surgical revision with transposition of the nerve end into deep muscle or bone may be required.
When primary nerve reconstruction fails, or when the patient presents outside the biological window for nerve repair, the surgeon must pivot to salvage procedures. Tendon transfers are the workhorse of salvage management, utilizing redundant, functionally intact muscle-tendon units to restore critical lost functions (e.g., transferring the Pronator Teres to the Extensor Carpi Radialis Brevis for radial nerve palsy). In cases of massive tissue loss or global plexus avulsions, free functional muscle transfers (e.g., Gracilis transfer innervated by the spinal accessory nerve or intercostal nerves) or joint arthrodesis (e.g., wrist or shoulder fusion) provide durable, albeit limited, functional restoration.
| Complication | Estimated Incidence | Prevention / Salvage Management |
|---|---|---|
| Failure of functional motor recovery | 15% - 40% (Depends on injury level) | Minimize graft length; early intervention; salvage with tendon transfers or free functional muscle transfer. |
| Symptomatic Neuroma Recurrence | 5% - 10% | Tension-free coaptation; adequate resection of initial scar; salvage via neuroma excision and intra-muscular/intra-osseous transposition. |
| Complex Regional Pain Syndrome (CRPS) | 2% - 5% | Meticulous hemostasis; early postoperative mobilization; multidisciplinary pain management, sympathetic ganglion blocks. |
| Donor Site Morbidity (Sural Nerve) | 10% - 20% (Numbness, neuroma) | Endoscopic harvest; burying the proximal stump of the donor nerve deep in the calf musculature. |
| Joint Contracture (Secondary to paralysis) | 10% - 30% | Aggressive passive range of motion protocols during the denervation phase; dynamic splinting; surgical capsular release if rigid. |
Phased Post-Operative Rehabilitation Protocols
The surgical repair is merely the first step in a prolonged, arduous journey toward functional recovery. The postoperative rehabilitation protocol is strictly phased, balancing the need to protect the fragile microsurgical coaptation with the imperative to prevent joint contractures and promote cortical neuroplasticity. The rehabilitation team, led by specialized hand and upper extremity therapists, is integral to maximizing the surgical outcome.
Phase I: Immobilization and Protection (Weeks 0-3)
Immediately postoperatively, the limb is immobilized in a custom orthosis designed to eliminate all tension across the repair site. For example, a median nerve repair at the wrist requires a dorsal blocking splint with the wrist in slight flexion. During this phase, absolute protection of the coaptation is paramount. The biological glue and delicate 9-0 sutures are the only structures holding the fascicles together while initial fibrin cross-linking and early fibroplasia occur. However, passive range of motion of all uninvolved joints is initiated immediately to prevent secondary stiffness.
Phase II: Early Mobilization and Gliding (Weeks 3-8)
As the tensile strength of the repair increases, the immobilization is gradually weaned. The focus shifts to controlled, protected nerve gliding exercises. The objective is to prevent the repaired nerve from becoming tethered to the surrounding surgical bed by dense adhesions. Synergistic joint movements are employed to gently slide the nerve longitudinally without applying direct stretch to the coaptation site. Edema management and scar massage are aggressively pursued to soften the surgical site.
Phase III: Motor and Sensory Re-education (Months 3-24+)
This is the longest and most demanding phase, commencing when clinical or electromyographic signs of reinnervation appear. As nascent motor units reach the target muscles, biofeedback and gravity-eliminated exercises are utilized to strengthen the profoundly weak musculature. Concurrently, sensory re-education protocols (such as Dellon's protocol) are instituted. Because regenerating axons often misdirect (e.g., a thumb sensory axon innervating the index finger), the somatosensory cortex receives confusing signals. Sensory re-education involves structured, repetitive tactile stimuli paired with visual feedback, forcing the brain to remap and correctly interpret the new, altered peripheral inputs.
Summary of Landmark Literature and Clinical Guidelines
The modern surgical approach to nerve injuries in continuity is built upon a foundation of rigorous anatomical and clinical research. Sir Sydney Sunderland’s seminal texts in the mid-20th century established the microanatomical classification that remains universally utilized today, fundamentally linking intraneural pathology to clinical prognosis. His work underscored the critical importance of the perineurium
