Masterclass in Primary Peripheral Nerve Repair: Hand, Wrist, and Forearm Transections
Key Takeaway
This masterclass guides fellows through primary nerve repair for transections in the hand, wrist, and forearm. We cover essential surgical anatomy, meticulous intraoperative technique, and critical decision-making for optimal patient outcomes. From initial incision to precise epineurial or fascicular repair, every micro-step is detailed, emphasizing tension-free approximation and vital post-operative care.
Comprehensive Introduction and Patho-Epidemiology
The management of complete peripheral nerve transections in the upper extremity remains one of the most formidable challenges in orthopedic and hand surgery. The primary objective of surgical intervention extends far beyond the mere structural reapproximation of severed nerve ends; it is the meticulous facilitation of a biological environment conducive to axonal regeneration, ultimately aiming to restore the patient's sensory perception, motor function, and overall interaction with their environment. Precision, patience, and a profound comprehension of nerve biology are paramount. The surgeon must operate with the understanding that the goal is not simply anatomical continuity, but the maximization of useful functional recovery in a highly complex neurovascular network.
Epidemiologically, traumatic peripheral nerve injuries of the upper extremity disproportionately affect young, active individuals, frequently resulting from penetrating trauma such as glass lacerations, knife wounds, or industrial accidents involving heavy machinery. The socioeconomic burden of these injuries is staggering, often leading to prolonged disability, loss of employment, and significant psychological distress. The median and ulnar nerves are particularly vulnerable at the level of the wrist and distal forearm due to their superficial anatomical course, while the radial nerve is frequently compromised in the context of humeral shaft fractures or penetrating trauma to the proximal forearm. Understanding the mechanism of injury—whether sharp transection, crush, avulsion, or blast—is critical, as it directly dictates the zone of injury and the appropriate timing and technique for surgical repair.
The pathophysiology of nerve injury and subsequent healing is fundamentally distinct from other musculoskeletal tissues. Upon complete transection (Sunderland Grade V / Seddon Neurotmesis), the structural continuity of the axon, endoneurium, perineurium, and epineurium is entirely disrupted. Immediately following the insult, the nerve segment distal to the injury undergoes Wallerian degeneration. This is an active, highly orchestrated biological process where the distal axonal segments and myelin debris are phagocytosed and cleared by invading macrophages and resident Schwann cells. Concurrently, Schwann cells proliferate and align within the preserved basal lamina tubes, forming the bands of Büngner. These bands release vital neurotrophic factors (such as Nerve Growth Factor, NGF) and surface adhesion molecules, essentially preparing the distal stump to act as a hospitable, chemotactic conduit for regenerating axons.
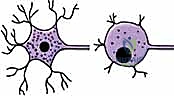
Simultaneously, the proximal neuronal cell body undergoes a profound metabolic shift known as chromatolysis. The cell body swells, the nucleus migrates peripherally, and the Nissl granules (rough endoplasmic reticulum) disperse and dissolve as the neuron shifts its synthetic machinery from neurotransmitter production to the massive upregulation of structural proteins required for axonal elongation. Regenerating axonal sprouts, or growth cones, emerge from the proximal stump and navigate across the repair site toward the empty endoneurial tubes of the distal stump. This antegrade transport and elongation occur at a remarkably slow rate of approximately 1 to 4 millimeters per day, representing the absolute rate-limiting step in clinical nerve regeneration. If the nerve is left unrepaired, or if the repair is fraught with tension or scar tissue, these proliferating proximal axons will fail to cross the gap, instead forming a disorganized, exquisitely painful, and functionally useless tangle of nerve fibers known as a terminal neuroma.
Detailed Surgical Anatomy and Biomechanics
Before a surgical incision is ever planned, the operating surgeon must possess an intimate, three-dimensional understanding of peripheral nerve micro- and macro-anatomy. A peripheral nerve is a highly organized, anisotropic, and delicate structure, possessing a complexity that far exceeds a simple electrical conduit. It is a dynamic organ that must withstand tension, compression, and excursion during normal physiological joint movement.
Microscopic Architecture and Its Surgical Relevance
The fundamental functional unit of the peripheral nervous system is the axon, the cytoplasmic extension of the neuronal cell body responsible for the transmission of action potentials. All peripheral axons are intimately enveloped by Schwann cells. In myelinated fibers, these cells wrap concentrically around the axon to produce the myelin sheath, a lipid-rich insulating layer that dramatically increases the velocity of electrical conduction. The periodic interruptions in this myelin sheath, termed the nodes of Ranvier, facilitate saltatory conduction, allowing depolarization to rapidly "jump" from node to node. Myelinated fibers range significantly in diameter (2 to 22 µm), with larger diameters correlating directly with faster conduction velocities.
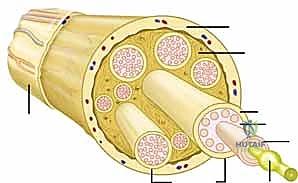
Moving outward, the endoneurium constitutes the most delicate connective tissue layer, forming a thin, supportive sleeve around each individual axonal fiber and its associated Schwann cells. It provides a critical microenvironment and a blood-nerve barrier essential for regeneration. Groups of these endoneurium-encased axons are subsequently bundled together to form a fascicle, which represents the smallest functional unit of the nerve visible to the naked eye during microsurgery. The fascicle is circumscribed by the perineurium, a robust, multi-layered membrane composed of specialized perineurial cells and longitudinally arranged collagen fibrils. The perineurium provides immense tensile strength to the nerve and acts as a formidable diffusion barrier, maintaining the specialized internal milieu required for axonal function.
Fascicles are not randomly distributed; they exhibit a highly specific topographic arrangement within the peripheral nerve. This fascicular segregation into distinct motor and sensory groupings is a foundational concept for aligning a sectioned nerve prior to repair. Accurate identification and alignment of these functional groups significantly maximize the probability of motor axons regenerating into motor pathways and sensory axons into sensory pathways. Surrounding the groups of fascicles is the epineurium, the outermost connective tissue layer that forms the superstructure of the entire nerve. Composed of longitudinally oriented collagen fibers, fibroblasts, and an intrinsic vascular network (vasa nervorum), the epineurium constitutes 60% to 85% of the nerve's cross-sectional area and is the primary layer engaged during a standard epineurial repair. Finally, the paraneurium (or mesoneurium) is the loose, areolar adventitial tissue surrounding the epineurium. It houses the extrinsic segmental blood supply and functions as the nerve's gliding apparatus, permitting excursion within its anatomical bed. Preservation of the mesoneurium during surgical mobilization is critical to preventing devascularization and subsequent ischemic fibrosis.
Macro-Anatomy and Neurovascular Relationships in the Upper Extremity
Operating within the hand, wrist, or forearm requires mastery of the specific anatomical courses and neurovascular relationships of the median, ulnar, and radial nerves. Each nerve presents distinct surgical risks and topographical nuances that must be respected. The median nerve traverses the arm and enters the forearm between the two heads of the pronator teres, coursing distally between the flexor digitorum superficialis (FDS) and flexor digitorum profundus (FDP). At the wrist, it becomes highly superficial before passing through the rigid confines of the carpal tunnel. It provides critical motor innervation to the forearm pronators, wrist flexors (excluding the flexor carpi ulnaris), and the thenar intrinsic muscles, while supplying sensation to the radial palm and the palmar aspect of the radial three-and-a-half digits. Its superficial location at the volar wrist makes it highly susceptible to sharp lacerations, often accompanied by injury to the adjacent median artery and palmaris longus tendon.
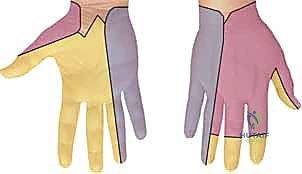
The ulnar nerve descends the medial aspect of the arm, passing posterior to the medial epicondyle through the cubital tunnel, a common site of compression. It enters the forearm between the two heads of the flexor carpi ulnaris (FCU) and courses distally alongside the ulnar artery. At the wrist, it enters the hand through Guyon's canal. The ulnar nerve is the primary motor nerve of the hand, innervating the hypothenar muscles, all interossei, the adductor pollicis, and the ulnar two lumbricals, making its injury devastating to fine motor dexterity and grip strength. It provides sensation to the ulnar border of the hand and the ulnar one-and-a-half digits.
The radial nerve spirals around the posterior humerus within the spiral groove, making it highly vulnerable to humeral shaft fractures. As it crosses the elbow anterior to the lateral epicondyle, it bifurcates into the superficial sensory branch and the deep motor branch (the posterior interosseous nerve, PIN). The PIN dives deep into the supinator muscle via the arcade of Frohse, providing motor innervation to all wrist and finger extensors. The superficial branch descends under the brachioradialis to provide sensation to the dorsal-radial aspect of the hand. Surgical approaches to the proximal radius must meticulously identify and protect the PIN, as iatrogenic transection during fracture fixation or radial head arthroplasty is a known and severe complication.
Exhaustive Indications and Contraindications
The decision to perform a primary nerve repair hinges on a multitude of factors, including the mechanism of injury, the timing of presentation, the degree of wound contamination, and the overall physiological status of the patient. Primary repair, defined as repair undertaken within the first 48 to 72 hours post-injury, is the gold standard for sharp, clean transections where the zone of injury is minimal and the nerve ends can be approximated without tension.
Delayed primary repair (within 1 to 3 weeks) may be indicated in cases where the initial wound is contaminated, requires serial debridement, or when the exact extent of nerve contusion cannot be accurately demarcated at the time of the initial presentation. This delay allows the nerve ends to undergo initial fibrotic changes, making the epineurium slightly more robust for suturing, while still capitalizing on the optimal biological window for Schwann cell activity and neurotrophic factor expression. Secondary repair (after 3 weeks) is typically reserved for blunt trauma, avulsion injuries, or delayed presentations, often necessitating the use of nerve grafts due to the inevitable retraction of the nerve stumps and the necessity to resect terminal neuromas back to healthy fascicular tissue.
| Parameter | Indications for Primary/Delayed Primary Repair | Contraindications for Acute Primary Repair |
|---|---|---|
| Mechanism of Injury | Sharp lacerations (glass, knife, scalpel); clean, focal transections. | Severe crush injuries; massive avulsion injuries; high-velocity ballistic trauma. |
| Wound Bed Status | Clean, well-vascularized soft tissue bed; minimal contamination. | Gross contamination; active infection; severe soft tissue loss requiring free flaps. |
| Nerve Gap | Ends can be approximated completely tension-free (usually < 1-2 cm after minimal mobilization). | Large segmental defects where tension-free coaptation is impossible without grafting. |
| Patient Status | Hemodynamically stable; able to tolerate prolonged microsurgery; compliant with rehab. | Polytrauma with life-threatening injuries (damage control orthopedics takes precedence). |
| Timing | Presentation within 0-21 days of injury. | Late presentations (> 3-4 weeks) where stump retraction necessitates grafting. |
Contraindications to acute primary repair must be strictly respected to prevent catastrophic failure. Attempting to primarily repair a nerve that has suffered a massive longitudinal stretch or crush injury is biologically futile, as the intraneural architecture is damaged far beyond the visible transection site. Suture placement in such ischemic, contused tissue will inevitably lead to dehiscence or dense fibrotic scarring that blocks axonal crossing. Furthermore, any repair performed under tension is doomed to fail; tension induces localized ischemia at the coaptation site, leading to massive collagen deposition and a mechanical block to regeneration. In scenarios where a tension-free primary repair is impossible, the surgeon must be prepared to tag the nerve ends to adjacent fascial planes to prevent further retraction and plan for a delayed reconstruction utilizing autologous nerve grafts or nerve conduits.
Pre-Operative Planning, Templating, and Patient Positioning
Successful peripheral nerve surgery demands exhaustive preoperative evaluation and meticulous operative planning. The clinical examination is the cornerstone of this process, serving to precisely localize the lesion, quantify the functional deficit, and establish a baseline against which postoperative recovery will be measured. The sensory examination must be systematic, mapping the exact autonomous zones of the median, ulnar, and radial nerves. While static two-point discrimination (2PD) is a classic metric, it primarily measures innervation density and is often entirely absent in acute transections. Semmes-Weinstein monofilament testing provides a more sensitive threshold measurement of cutaneous pressure sensation. The Tinel sign—elicited by gently percussing along the course of the nerve—is invaluable; a strongly positive Tinel sign at the site of injury confirms the location of the proximal stump, and its distal progression over subsequent months is the primary clinical indicator of advancing axonal regeneration.
Motor testing must isolate specific muscles to prevent misinterpretation due to trick movements or overlapping innervation. The Medical Research Council (MRC) grading system (M0 to M5) remains the standard for documenting motor strength. For the median nerve, the abductor pollicis brevis (APB) is isolated by asking the patient to palmar abduct the thumb against resistance. For the ulnar nerve, the first dorsal interosseous (abduction of the index finger) and the adductor pollicis (Froment's sign) are tested. Radial nerve function is assessed via wrist extension (extensor carpi radialis longus/brevis) and retropulsion of the thumb (extensor pollicis longus). Furthermore, the surgeon must carefully document any associated injuries, particularly vascular compromise, tendon lacerations, or fractures, as the sequence of repair (typically bone, then tendon, then vessels, then nerves) must be established preoperatively.
Electrodiagnostic studies (EMG and NCV) have a limited role in the hyperacute setting of a sharp transection, as Wallerian degeneration takes 7 to 10 days to manifest electromyographically, and distal nerve segments may remain excitable for up to 72 hours post-injury. However, in closed injuries, stretch injuries, or delayed presentations, baseline electrodiagnostics at 3 to 4 weeks are critical to differentiate between neurapraxia (which will recover spontaneously), axonotmesis, and neurotmesis (which require surgery). Imaging modalities such as high-resolution ultrasound are increasingly utilized to rapidly visualize nerve discontinuity, retraction gaps, and associated foreign bodies (e.g., glass shards) in the emergency department.
Patient positioning and operating room setup are critical for microsurgical success. The patient is typically positioned supine with the affected extremity extended on a radiolucent hand table. A well-padded pneumatic tourniquet is applied to the proximal arm. The use of a high-quality operating microscope with dual-viewing capabilities is non-negotiable for definitive nerve repair; surgical loupes (minimum 3.5x to 4.5x magnification) are sufficient for the initial exposure and mobilization but are inadequate for precise fascicular alignment and epineurial suturing. Meticulous hemostasis is maintained using bipolar electrocautery, and a sterile background material (often a blue or green silastic sheet) is prepared to place behind the nerve ends to enhance visual contrast during the repair.
Step-by-Step Surgical Approach and Fixation Technique
The surgical approach to a transected peripheral nerve must be extensile, respecting the principles of safe anatomical exposure while allowing for adequate mobilization of the nerve stumps. Incisions should incorporate the traumatic wound but must be extended proximally and distally using Brunner-type zigzag or mid-lateral incisions to prevent flexion contractures across joints. The procedure is performed under tourniquet control to ensure a bloodless field during the initial dissection. The uninjured proximal and distal portions of the nerve are identified first in virgin tissue planes before tracing them into the zone of injury and hematoma.
Once the nerve ends are identified, they must be meticulously mobilized to gain length. This involves dissecting the nerve from its surrounding paraneurial bed over several centimeters. However, the surgeon must balance the need for length with the risk of devascularization; excessive stripping of the mesoneurium will destroy the segmental blood supply, leading to ischemic fibrosis of the repair site. Gentle traction is applied, and the surrounding fascial attachments are carefully released.
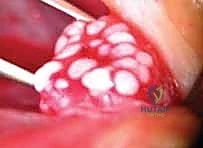
Preparation of the nerve stumps is arguably the most critical step determining biological success. The traumatized, contused ends must be resected back to entirely healthy, viable tissue. Using a fresh surgical blade or specialized nerve scissors against a wooden tongue depressor, the surgeon sequentially "bread-loafs" the nerve ends by making 1-millimeter step-cuts until normal fascicular architecture is visualized. Healthy fascicles will "mushroom" or pout out from the surrounding epineurium, and pinpoint bleeding from the vasa nervorum should be observed upon tourniquet deflation. Failure to resect back to healthy tissue guarantees that the regenerating axons will encounter a dense fibrotic scar barrier.
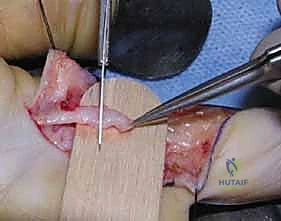
Alignment of the proximal and distal stumps relies on careful observation of surface topography and internal architecture. The surgeon matches the longitudinal epineurial blood vessels, which act as excellent surface markers. Internally, the size, shape, and grouping of the fascicles are meticulously matched. In major mixed nerves like the median or ulnar, identifying the distinct motor and sensory fascicular groups is paramount.

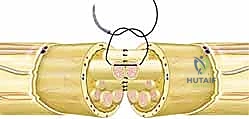
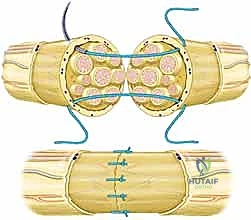
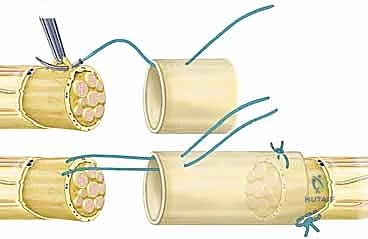
The repair technique is executed under the operating microscope using micro-instruments and 8-0 or 9-0 monofilament nylon sutures. An epineurial repair is the standard technique for most acute transections. The first two sutures are placed exactly 180 degrees apart to serve as stay sutures, perfectly aligning the rotational axis of the nerve. These sutures grasp only the robust epineurium, avoiding penetration into the delicate fascicles.

Once the primary stay sutures are tied, the anterior epineurium is closed with interrupted sutures placed precisely to coapt the edges without causing bunching or overriding. The stay sutures are then used to rotate the nerve 180 degrees, exposing the posterior aspect, which is similarly repaired. The goal is a watertight epineurial closure that contains the fascicles and prevents axonal escape, which would lead to neuroma formation. Group fascicular repair, involving suturing the perineurium of matched fascicle bundles, may be indicated in distal injuries where motor and sensory branches are clearly separated, but it carries a higher risk of intraneural scarring and iatrogenic trauma if not performed expertly.
The absolute cardinal rule of peripheral nerve surgery is that the repair must be completely tension-free. If, after mobilization, the nerve ends cannot be coapted with an 8-0 suture without the suture tearing through the epineurium, the primary repair must be abandoned. Forcing a tight repair leads to immediate ischemia, massive scar proliferation, and guaranteed failure. In such cases, the gap must be bridged using an autologous nerve graft (e.g., sural nerve) or, for very small non-critical sensory gaps (< 3 cm), a biological or synthetic nerve conduit. Fibrin glue is frequently utilized as an adjunct to suture repair, providing immediate tensile strength, sealing the coaptation site, and potentially reducing the number of required sutures, thereby minimizing foreign body reaction.
Complications, Incidence Rates, and Salvage Management
Despite flawless microsurgical technique, peripheral nerve repair is fraught with potential complications, largely due to the unforgiving biological nature of axonal regeneration. The surgeon must proactively counsel the patient regarding these risks and maintain a high index of suspicion during the prolonged postoperative recovery phase. Failure of regeneration is the most profound complication, often resulting from an unrecognized tension at the repair site, inadequate resection of the zone of injury, or severe scar tissue formation within the surrounding soft tissue bed.
Neuroma-in-continuity or terminal neuroma formation occurs when regenerating axons escape the coaptation site or fail to penetrate the distal stump, respectively. These present as exquisitely tender, palpable masses that elicit severe radiating pain upon percussion (a hyperactive Tinel sign that fails to advance). Joint stiffness and debilitating contractures are common secondary complications resulting from prolonged immobilization and the loss of dynamic muscle balance following motor denervation. Complex Regional Pain Syndrome (CRPS) is a devastating, sympathetically mediated complication characterized by burning pain, allodynia, sudomotor changes, and severe stiffness, which can completely derail functional recovery even if the nerve repair itself is biologically successful.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage Management / Treatment |
|---|---|---|---|
| Failure of Regeneration | 10% - 30% (varies by age/nerve) | Tension, inadequate debridement, severe crush, advanced age. | Re-exploration, neurolysis, resection and nerve grafting, or nerve/tendon transfers. |
| Symptomatic Neuroma | 5% - 15% | Axonal escape, poor epineurial closure, gap formation. | Excision and grafting, targeted muscle reinnervation (TMR), burying in muscle/bone. |
| Joint Contracture | 20% - 40% | Prolonged immobilization, lack of therapy, muscle imbalance. | Aggressive hand therapy, dynamic splinting, eventual surgical capsulotomy/release. |
| CRPS (Type II) | 2% - 5% | High-energy trauma, severe pain cycle, psychological factors. | Multidisciplinary pain management, sympathetic blocks, gabapentinoids, aggressive desensitization. |
| Cold Intolerance | > 50% | Sympathetic denervation, abnormal reinnervation of vasculature. | Conservative management, thermal protection, biofeedback; often permanent but improves slowly. |
When a primary repair fails, salvage management requires a strategic paradigm shift. If serial clinical exams and electrodiagnostic studies at 3 to 6 months demonstrate an absolute lack of motor or sensory progression, surgical re-exploration is mandated. If a dense neuroma-in-continuity is found with no distal conduction, the scarred segment must be resected and reconstructed with autologous nerve grafts. However, in cases of delayed presentation or high proximal injuries where the time required for axons to reach distal targets exceeds the viability window of the motor endplates (typically 12 to 18 months), nerve transfers (neurotization) have revolutionized salvage surgery. For example, transferring the expendable anterior interosseous nerve (AIN) to the deep motor branch of the ulnar nerve bypasses the long regeneration distance from the elbow, providing a reliable source of motor axons close to the target intrinsic muscles. When motor endplates are irreversibly lost, classic tendon transfers remain the definitive reconstructive option to restore functional biomechanics.
Phased Post-Operative Rehabilitation Protocols
The surgical repair is only the first step in the arduous journey of nerve recovery; meticulous, phased postoperative rehabilitation, guided by a specialized certified hand therapist (CHT), is absolutely essential to optimize functional outcomes. The rehabilitation protocol is strictly divided into distinct phases, balancing the initial need to protect the fragile microscopic repair with the eventual necessity of preventing adhesions and retraining the brain.
The initial phase (Weeks 0 to 3) is focused entirely on protection. The extremity is immobilized in a custom orthosis designed to remove all tension from the repair site. For a volar wrist median or ulnar nerve repair, this typically involves a dorsal blocking splint with the wrist in 20 to 30 degrees of flexion. The patient is strictly instructed to avoid any active or passive stretching of the repaired nerve. However, isolated joint movements that do not tension the nerve (e.g., passive digital flexion and extension while the wrist remains flexed) are initiated immediately to prevent tendon adhesions and joint contractures.
The early mobilization phase (Weeks 3 to 6) marks the gradual introduction of nerve gliding and tensioning. The protective splint is slowly adjusted toward neutral (e.g., extending the wrist by 10 degrees per week). Active range of motion exercises are initiated, focusing on differential tendon gliding to prevent the nerve from becoming tethered in the surrounding scar bed. The therapist closely monitors for any signs of sharp, radiating pain that might indicate excessive tension on the healing coaptation.
The functional recovery phase (Weeks 6 and beyond) shifts focus to sensory re-education and motor strengthening as regeneration progresses. Sensory re-education is a critical cognitive process; as axons reach the skin, the brain receives novel, often distorted signals. Patients undergo structured protocols involving the identification of different textures, shapes, and temperatures, forcing the somatosensory cortex to remap and interpret the new afferent input. Once motor function begins to return (initially presenting as a flicker of contraction), gravity-eliminated exercises are started, progressing to progressive resistance training. Biofeedback and electrical stimulation may be utilized as adjuncts to maximize motor unit recruitment.
Summary of Landmark Literature and Clinical Guidelines
The modern principles of peripheral nerve repair are built upon decades of foundational research and evolving clinical paradigms. Sir Sydney Sunderland’s classic 1951 classification of nerve injuries remains the definitive framework for understanding the internal architecture of nerve trauma, expanding upon Seddon’s earlier concepts of neurapraxia, axonotmesis, and neurotmesis. Sunderland’s work underscored the critical importance of the perineurium and endoneurium in predicting spontaneous recovery versus the need for surgical intervention.
Landmark studies by Millesi in the 1970s fundamentally altered the surgical approach to nerve gaps. Millesi demonstrated that repairs performed under tension resulted in massive intraneural fibrosis and failure of regeneration. His advocacy for the use of interfascicular nerve grafting for any gap that could not be closed without tension remains a cornerstone of modern microsurgery. Subsequent research has continually reinforced that tension is the absolute enemy of nerve healing.
Contemporary clinical guidelines strongly advocate for the acute primary repair of sharp transections within the first 72 hours, as early repair minimizes stump retraction and capitalizes on the optimal biological environment before severe fibrotic changes occur. A seminal meta-analysis by Ruijs et al. (2005) reviewing median and ulnar nerve repairs confirmed that patient age is the single most significant prognostic factor for recovery, with children achieving vastly superior outcomes compared to adults over 40. Furthermore, the literature consistently demonstrates that the median nerve generally achieves better functional recovery than the ulnar nerve, likely due to the ulnar nerve's heavy reliance on fine motor intrinsic function, which is notoriously difficult to restore. Recent advancements detailed in the literature focus on the use of biological conduits, the application of tacrolimus (FK506) to enhance regeneration rates, and the expanding indications for distal nerve transfers as primary or early salvage procedures.
