Mastering Carpal Tunnel Release: An Intraoperative Guide to Open and Endoscopic Techniques
Key Takeaway
This masterclass provides an exhaustive, real-time walkthrough of Carpal Tunnel Release, covering both open and endoscopic techniques. Fellows will gain deep insights into surgical anatomy, meticulous intraoperative steps, critical decision-making, and advanced strategies for managing potential complications. Learn precise instrument handling and anatomical landmarks to ensure successful median nerve decompression and optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
Carpal Tunnel Syndrome (CTS) represents the most ubiquitous compressive neuropathy in the human body, serving as a foundational pathology that every orthopedic surgeon must master intimately. It is an entrapment neuropathy of the median nerve as it traverses the fibro-osseous carpal canal at the wrist. Carpal Tunnel Release (CTR), the definitive surgical intervention for this condition, remains one of the most frequently performed orthopedic procedures globally. Mastery of this procedure—encompassing both the classic open approach and the modern endoscopic techniques—is absolutely paramount for ensuring optimal patient outcomes, minimizing iatrogenic morbidity, and facilitating a rapid return to function. The goal of this definitive chapter is to provide an immersive, highly technical, and scrubbed-in perspective, illuminating every critical anatomical nuance, procedural step, and potential intraoperative pitfall.
To understand the surgical imperative, one must first deconstruct the underlying pathogenesis and epidemiology of CTS. The vast majority of cases are classified as idiopathic, characterized by a chronic, progressive increase in interstitial pressure within the rigid confines of the carpal tunnel. Normal resting pressure within the carpal canal ranges from 2 to 10 mm Hg. In patients with CTS, baseline pressures frequently exceed 30 mm Hg, and dynamic positioning—such as extreme wrist flexion or extension—can drive these pressures well above 90 mm Hg. This sustained mechanical compression initiates a predictable microvascular ischemic cascade. Initial venular congestion leads to breakdown of the blood-nerve barrier, resulting in protein-rich endoneurial edema. If the pressure is not relieved, this edema incites intraneural fibrosis, localized demyelination, and eventually, irreversible Wallerian degeneration of the axonal architecture.
Systemic conditions profoundly influence this microvascular environment. Endocrinopathies such as diabetes mellitus and hypothyroidism compromise peripheral nerve resilience, making the median nerve highly susceptible to even minor mechanical compression—a phenomenon often described within the context of the "double crush" hypothesis. Inflammatory arthritides, most notably rheumatoid arthritis, can induce massive tenosynovial proliferation that mechanically obliterates the available space within the canal. Other established risk factors include chronic renal failure (associated with amyloid deposition), sarcoidosis, and the fluid retention characteristic of the third trimester of pregnancy. Furthermore, anomalous anatomical structures, such as a persistent median artery, proximally extending lumbrical muscle bellies, or occult ganglion cysts, can act as space-occupying lesions directly impinging upon the median nerve.
Understanding the natural history of this compressive neuropathy is critical for surgical decision-making. In its early stages, the pathophysiology is dominated by transient ischemia and localized neuropraxia, which are often fully reversible with conservative management or prompt surgical decompression. However, in advanced stages characterized by chronic axonal degeneration, the prognosis shifts dramatically. Patients presenting with profound sensory loss, fixed motor deficits, and severe thenar atrophy may experience incomplete neurological recovery even after a technically flawless surgical release. This stark clinical reality underscores the absolute necessity for timely, accurate diagnosis and decisive surgical intervention before irreversible end-organ damage occurs.
Detailed Surgical Anatomy and Biomechanics
Before a scalpel touches the skin, an exhaustive, three-dimensional understanding of the carpal tunnel anatomy is non-negotiable. The carpal tunnel is an unyielding fibro-osseous conduit situated at the volar aspect of the wrist. It functions not merely as a passive anatomical space, but as a dynamic biomechanical pulley system that facilitates the efficient transmission of digital flexor tendon forces.
Osteoligamentous Boundaries of the Carpal Canal
The architecture of the carpal canal is defined by rigid boundaries that leave virtually no margin for volumetric expansion. The dorsal floor is formed by the concave arch of the carpal bones: proximally by the scaphoid, lunate, triquetrum, and pisiform; and distally by the trapezium, trapezoid, capitate, and hamate. The radial border is anchored by the prominent tubercle of the scaphoid and the crest of the trapezium. Conversely, the ulnar border is defined by the pisiform proximally and the robust hook of the hamate distally.
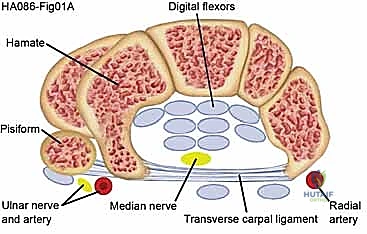
The palmar roof—and the primary target of our surgical intervention—is the transverse carpal ligament (TCL), also known as the flexor retinaculum. The TCL is a dense, thick, inelastic fibrous band that spans the carpal arch. It is contiguous proximally with the deep antebrachial fascia of the distal forearm and blends distally with the palmar aponeurosis. The TCL plays a crucial biomechanical role in preventing volar bowstringing of the flexor tendons during forceful wrist and digital flexion. Surgical division of this ligament predictably alters carpal arch kinematics, slightly increasing the cross-sectional area of the canal while occasionally leading to transient postoperative "pillar pain" due to micro-instability of the carpal bones.
Contents and Topography of the Carpal Tunnel
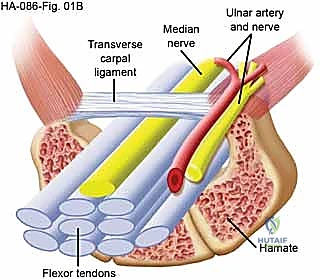
Within this highly constrained space, ten critical structures must navigate the wrist. These include the median nerve and nine digital flexor tendons: four flexor digitorum superficialis (FDS) tendons, four flexor digitorum profundus (FDP) tendons, and the single flexor pollicis longus (FPL) tendon. The FDS tendons to the middle and ring fingers typically lie volar to those of the index and small fingers. The FPL occupies the most radial aspect of the canal, intimately associated with the radial bursa. All nine tendons are enveloped by complex tenosynovial sheaths that facilitate frictionless gliding. Pathological hypertrophy of this tenosynovium is a primary driver of increased intracanalicular pressure.
The median nerve is the most superficial and vulnerable structure within the tunnel, typically positioned directly beneath the TCL and slightly radial to the midline. It provides critical sensory innervation to the palmar aspect of the thumb, index, middle, and the radial half of the ring finger. Motor innervation is supplied to the intrinsic thenar musculature, specifically the abductor pollicis brevis, the opponens pollicis, the superficial head of the flexor pollicis brevis, and the radial two lumbricals. The fascicular topography of the median nerve at the wrist is highly organized, with the motor fascicles consistently located in the radial-volar quadrant of the nerve proper.
Critical Neurovascular Variants and Surgical Hazards
The surgical release of the TCL is fraught with potential hazards, primarily due to the highly variable branching patterns of the median nerve. The recurrent motor branch is of paramount concern. According to the classic Lanz classification, the motor branch exhibits significant anatomical variability. The most common configuration (approximately 46-90% of cases) is extraligamentous, where the branch arises distal to the TCL and recurves radially to innervate the thenar muscles. However, subligamentous variants (arising within the tunnel and running beneath the TCL before exiting) and transligamentous variants (piercing directly through the substance of the TCL) pose a severe risk of iatrogenic transection during surgical release.
Adjacent neurovascular structures also demand meticulous respect. The ulnar artery and nerve traverse Guyon's canal, located superficial to the TCL and immediately ulnar to the hook of the hamate. While an appropriately placed radial-sided incision protects these structures, aggressive or errant ulnar dissection can result in catastrophic neurovascular injury. Distally, the superficial palmar arch crosses the palm just distal to the distal margin of the TCL. Incomplete visualization during the distal extent of the release, particularly in endoscopic procedures, can risk laceration of this vital arterial arcade.
Furthermore, the palmar cutaneous branch of the median nerve (PCBMN) must be protected. This sensory branch typically arises 5 to 8 cm proximal to the wrist crease, traveling distally between the palmaris longus and flexor carpi radialis tendons before crossing superficial to the TCL to innervate the base of the thenar eminence. An incision placed too radially or excessively proximal dissection can easily transect the PCBMN, resulting in a debilitating, painful neuroma that often overshadows the benefits of the carpal tunnel release.
Exhaustive Indications and Contraindications
The decision to proceed with surgical intervention must be rooted in a rigorous clinical evaluation, supported by objective diagnostic data, and tailored to the individual patient's anatomical and physiological profile. While Carpal Tunnel Release is highly successful, inappropriate patient selection is the leading cause of persistent postoperative symptoms.
The primary indication for surgical release is a confirmed diagnosis of Carpal Tunnel Syndrome that has proven refractory to a comprehensive trial of conservative management. This typically involves 3 to 6 months of nocturnal splinting, activity modification, nonsteroidal anti-inflammatory drugs, and frequently, a diagnostic and therapeutic corticosteroid injection into the carpal canal. A positive, albeit temporary, response to a corticosteroid injection is highly predictive of a successful surgical outcome.
Absolute indications for expedited surgical intervention include the presence of progressive motor weakness, visible thenar atrophy, or profound sensory loss indicating advanced axonal degeneration. In these scenarios, prolonged conservative management is detrimental, as it permits further irreversible neural damage. Electrodiagnostic studies (EMG/NCS) demonstrating severe median neuropathy—characterized by absent sensory responses, significantly prolonged motor latencies, or fibrillations in the thenar musculature—strongly support early surgical decompression. Furthermore, acute Carpal Tunnel Syndrome, often precipitated by high-energy distal radius fractures, carpal dislocations, or acute hemorrhage, constitutes a surgical emergency requiring immediate open release to prevent catastrophic nerve ischemia.
Table: Indications and Contraindications for Carpal Tunnel Release
| Parameter | Open Carpal Tunnel Release (OCTR) | Endoscopic Carpal Tunnel Release (ECTR) |
|---|---|---|
| Primary Indications | Refractory primary CTS; Severe CTS with thenar atrophy; Acute CTS (trauma/fracture); Revision surgery for recurrent CTS; Space-occupying lesions (tumors, severe tenosynovitis). | Refractory primary CTS; Bilateral CTS (allows concurrent release with faster rehab); High demand for early return to work/activities. |
| Absolute Contraindications | Active local infection at the surgical site; Medically unstable patient unable to tolerate local anesthesia. | Acute CTS secondary to trauma/fracture; Suspected anatomical anomalies or space-occupying lesions; Revision CTS surgery; Severe joint contracture preventing positioning. |
| Relative Contraindications | Severe bleeding diathesis (requires optimization prior to surgery); Unrealistic patient expectations regarding nerve recovery in end-stage disease. | Small wrist anatomy limiting cannula insertion; Severe proliferative tenosynovitis limiting endoscopic visualization; Previous wrist surgery altering local anatomy. |
When selecting between open and endoscopic techniques, the surgeon must carefully weigh the anatomical constraints and the specific pathology. Endoscopic techniques offer the distinct advantages of a smaller incision, reduced postoperative pain, and a statistically faster return to work and activities of daily living in the early postoperative period. However, endoscopic release is contraindicated in cases of recurrent CTS, where scar tissue obscures visualization, or when space-occupying lesions such as ganglion cysts, lipomas, or amyloid deposits must be excised concurrently. Open release remains the gold standard, providing unparalleled, direct visualization of the median nerve, its branches, and the entire contents of the carpal canal.
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the cornerstone of a flawless surgical execution. This phase begins in the clinic with a comprehensive physical examination and a thorough review of all diagnostic modalities. While clinical diagnosis is paramount, adjunctive imaging and electrodiagnostic studies refine the surgical strategy.
Electrodiagnostic testing (EMG/NCS) serves as the objective gold standard for confirming the diagnosis, localizing the focal point of compression, and quantifying the severity of nerve impairment. It is crucial for differentiating CTS from proximal compressive neuropathies, such as cervical radiculopathy or pronator syndrome. High-resolution ultrasound is increasingly utilized as a rapid, non-invasive tool to assess the cross-sectional area of the median nerve, evaluate for tenosynovitis, and identify anatomical anomalies such as a persistent median artery or a bifid median nerve. Magnetic Resonance Imaging (MRI), while not routinely indicated for primary CTS, is invaluable in the workup of recurrent cases, suspected tumors, or complex traumatic presentations.
Surgical preparation involves precise anatomical marking and optimal patient positioning. The patient is placed supine on the operating table with the operative arm extended on a radiolucent hand table. A pneumatic tourniquet is typically applied to the proximal arm, although the rising popularity of the WALANT (Wide Awake Local Anesthesia No Tourniquet) technique has revolutionized this paradigm. WALANT utilizes a combination of lidocaine for anesthesia and epinephrine for hemostasis, eliminating the need for a tourniquet, reducing patient discomfort, and allowing for intraoperative assessment of active digital motion.
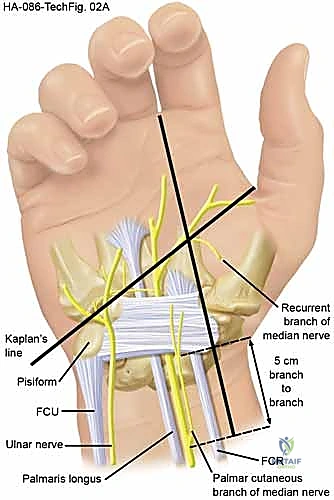
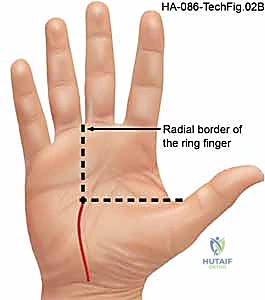
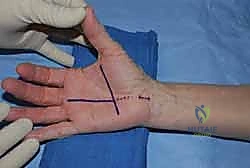
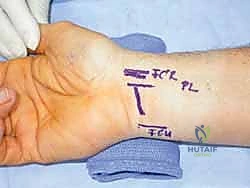
Accurate surface landmarking is absolutely critical to avoid iatrogenic injury. Kaplan's cardinal line is drawn from the apex of the interdigital fold between the thumb and index finger, extending toward the ulnar border of the hand parallel to the proximal palmar crease. The intersection of this line with a longitudinal line drawn along the radial border of the ring finger marks the approximate location of the hook of the hamate and the distal extent of the TCL. The incision for a standard open release is planned longitudinally, just ulnar to the thenar crease, remaining in line with the radial border of the ring finger to avoid the palmar cutaneous branch of the median nerve.
For endoscopic procedures, the surface markings are equally critical. The proximal wrist crease, the palmaris longus tendon, and the flexor carpi radialis tendon are carefully delineated. The entry portal is typically marked just proximal to the distal wrist crease, ulnar to the palmaris longus tendon, ensuring a safe trajectory into the carpal canal that avoids the median nerve radially and the ulnar neurovascular bundle ulnarly.
Step-by-Step Surgical Approach and Fixation Technique
The execution of a Carpal Tunnel Release demands absolute precision, a deep respect for soft tissue envelopes, and an unwavering commitment to complete decompression. We will detail both the standard Open Carpal Tunnel Release (OCTR) and the single-portal Endoscopic Carpal Tunnel Release (ECTR).
Open Carpal Tunnel Release (OCTR)
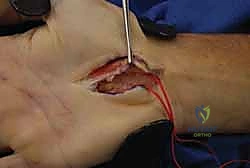
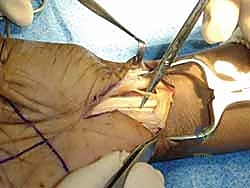
The open approach provides definitive visualization and is the benchmark against which all other techniques are measured. Following exsanguination and tourniquet inflation (or under WALANT conditions), a 3 to 4 cm longitudinal incision is made in the palm. The incision begins at the intersection of Kaplan's cardinal line and the radial border of the ring finger, extending proximally toward, but not crossing, the wrist crease. Crossing the wrist crease perpendicularly can lead to restrictive, hypertrophic scar contractures.
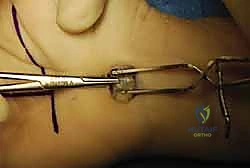
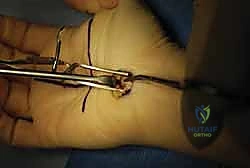
Sharp dissection proceeds through the skin and subcutaneous adipose tissue. Small bridging vessels are meticulously coagulated using bipolar electrocautery to maintain a bloodless field. The palmar aponeurosis is identified; its longitudinally oriented fibers are sharply divided in line with the incision. Immediately deep to the aponeurosis lies the palmaris brevis muscle. Its transversely oriented muscle fibers are carefully separated or incised to expose the dense, white, transversely oriented fibers of the underlying Transverse Carpal Ligament (TCL).
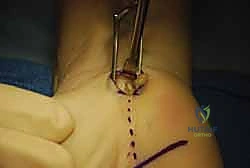
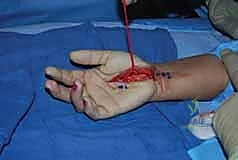
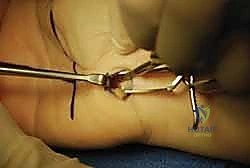
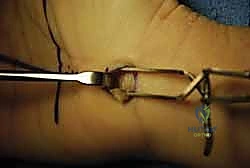
The release of the TCL is the critical juncture of the procedure. Using a scalpel or dissecting scissors, the ligament is carefully incised longitudinally, slightly ulnar to the midline to maximize distance from the median nerve and its recurrent motor branch. The release is carried distally until the palmar fat pad is encountered, signifying the complete division of the distal edge of the ligament. The superficial palmar arch lies just distal to this point and must be rigorously protected.
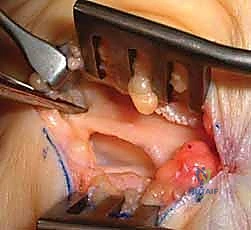
Attention is then directed proximally. A Ragnell or Senn retractor is placed at the proximal pole of the incision. Under direct vision, utilizing tenotomy scissors or a specialized ligamentotome, the proximal extent of the TCL and the contiguous distal antebrachial fascia are divided for a distance of 2 to 3 cm proximal to the wrist crease. Complete release is confirmed by passing a blunt probe or elevator proximally and distally within the canal; there should be absolutely no residual fibrous bands tethering the nerve. The median nerve is inspected for hour-glassing, hyperemia, or anatomical variants. Following copious irrigation and release of the tourniquet, meticulous hemostasis is achieved, and the skin is closed with non-absorbable monofilament sutures.
Endoscopic Carpal Tunnel Release (ECTR)
Endoscopic techniques, such as the single-portal Agee method, offer a minimally invasive alternative that preserves the overlying palmar skin and subcutaneous tissues, thereby accelerating early functional recovery.
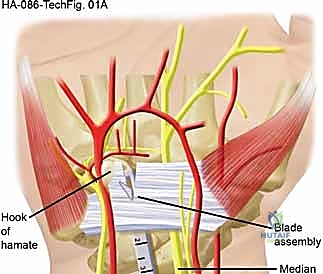
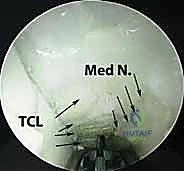
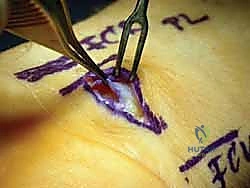
A 1 to 1.5 cm transverse incision is made in the proximal wrist crease, positioned ulnar to the palmaris longus tendon. Dissection is carried down through the antebrachial fascia. A U-shaped flap of fascia is often elevated to create a secure entry portal. Serial dilation of the carpal canal is performed using blunt synovial elevators to clear the undersurface of the TCL and displace the median nerve radially and volarly, creating a safe optical cavity.

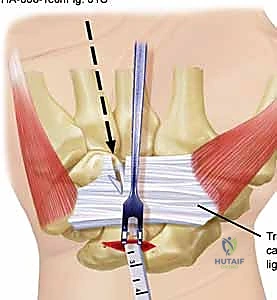
The endoscopic blade assembly is introduced into the canal, hugging the undersurface of the TCL. The surgeon must maintain strict spatial orientation, keeping the device aligned with the ring finger. The transverse fibers of the TCL must be clearly visualized across the entire width of the endoscopic monitor. If visualization is obscured by tenosynovium or if the anatomy is unclear, the procedure must be immediately converted to an open approach.

Once the distal edge of the ligament and the intervening fat pad are positively identified, the retrograde blade is deployed. The device is smoothly withdrawn proximally, sharply dividing the TCL under continuous direct visualization. A second pass with the endoscope is mandatory to confirm complete division of all ligamentous fibers. The portal is then closed with simple skin sutures.

Complications, Incidence Rates, and Salvage Management
While Carpal Tunnel Release is generally considered a safe and highly effective procedure, complications can and do occur. A profound understanding of
Clinical & Radiographic Imaging Archive