Masterclass: Surgical Management of Femoral Metastatic Bone Disease – Proximal and Diaphyseal Lesions
Key Takeaway
This masterclass guides fellows through the surgical management of metastatic bone disease in the proximal and diaphyseal femur. We meticulously cover comprehensive anatomy, detailed preoperative planning, and granular intraoperative execution, including curettage, cement augmentation, and internal fixation with intramedullary nails or endoprostheses. Critical pearls, potential pitfalls, and essential postoperative care are emphasized to ensure durable reconstruction and optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
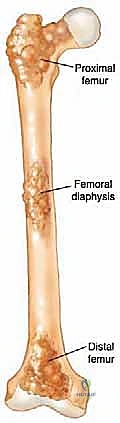
Welcome, esteemed colleagues and fellows, to the operating theater. Today, we are undertaking a rigorous examination of a critical, multifaceted challenge in orthopedic oncology: the surgical management of metastatic bone disease (MBD) localizing to the femur. This endeavor transcends the fundamental principles of fracture fixation; it is a highly specialized discipline aimed at restoring mechanical integrity, alleviating debilitating oncologic pain, and providing a durable, immediate reconstruction for patients who, despite a limited life expectancy, are profoundly deserving of optimal functional independence and quality of life. The femur represents the most frequent site of appendicular skeletal metastasis requiring surgical intervention. Because it is a primary weight-bearing pillar of the human skeleton, our margin for error is infinitesimally small, demanding meticulous preoperative templating and flawless intraoperative execution.
The pathophysiology of metastatic bone disease in the femur is driven by a complex, vicious cycle of cellular signaling between tumor cells and the bone microenvironment. Carcinomas of the breast, prostate, lung, kidney, and thyroid exhibit a profound osteotropism. Once tumor cells arrest in the highly vascularized red marrow of the proximal femur or diaphysis, they secrete factors such as Parathyroid Hormone-related Protein (PTHrP). This stimulates osteoblasts to upregulate Receptor Activator of Nuclear Factor Kappa-B Ligand (RANKL), which in turn drives massive osteoclastogenesis and subsequent osteolysis. This uncoupled bone remodeling rapidly degrades both trabecular microarchitecture and cortical integrity, significantly reducing the load-bearing capacity of the femur and predisposing the patient to catastrophic pathologic fractures.

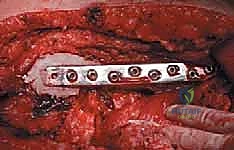
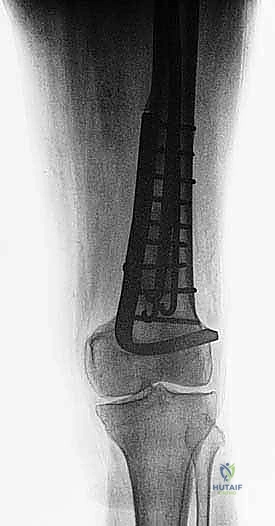
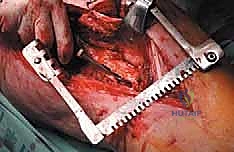
Historically, the orthopedic approach to these lesions was largely reactive, focusing on the salvage of completed fractures. However, contemporary orthopedic oncology mandates a proactive, prophylactic paradigm. The philosophical shift dictates that our reconstructive constructs must outlive the patient. We are not merely aiming for bone union—which is frequently impaired by local tumor burden, systemic chemotherapy, and adjuvant radiotherapy—but rather for immediate mechanical stability. This requires a transition from traditional osteosynthesis techniques, which rely on host bone healing, to load-sharing or load-bearing constructs such as cephalomedullary nails augmented with polymethylmethacrylate (PMMA) or massive endoprosthetic replacements.
A successful intervention relies heavily on a multidisciplinary tumor board approach. The orthopedic surgeon must collaborate seamlessly with medical oncologists, radiation oncologists, and interventional radiologists. The decision to operate, and the choice of operation, is intimately tied to the patient's systemic disease status, anticipated survival, and the specific histologic subtype of the primary tumor. For instance, highly vascular metastases from renal cell carcinoma or thyroid carcinoma necessitate preoperative embolization to mitigate the risk of catastrophic intraoperative hemorrhage. Ultimately, our goal is to execute a masterfully planned procedure that allows for immediate weight-bearing, thereby preventing the myriad complications associated with prolonged recumbency in the oncologic patient.
Detailed Surgical Anatomy and Biomechanics
Before we even consider making a scalpel incision, a profound, three-dimensional understanding of the regional anatomy and biomechanics is paramount. Each segment of the femur presents unique anatomical considerations, dictating our surgical approach, our choice of implant, and our navigation of potential iatrogenic risks.
Proximal Femur Considerations
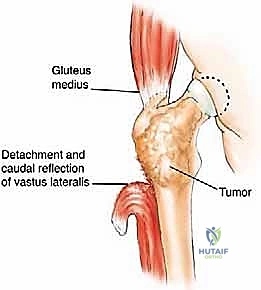
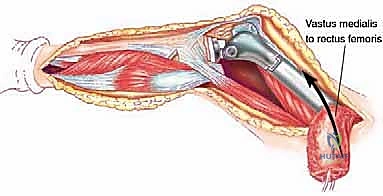
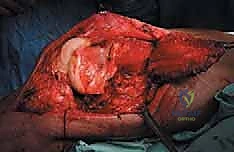
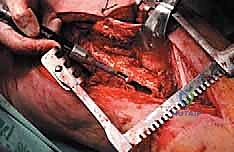
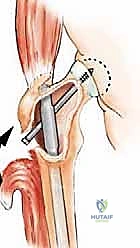
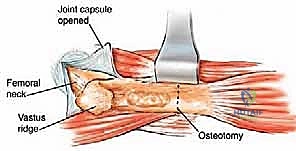
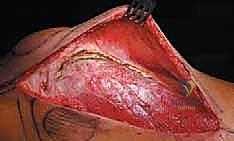
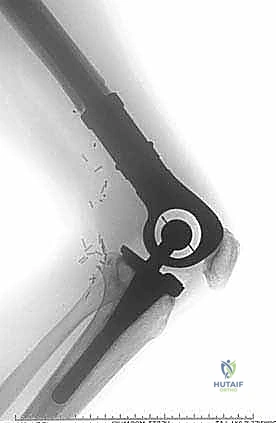
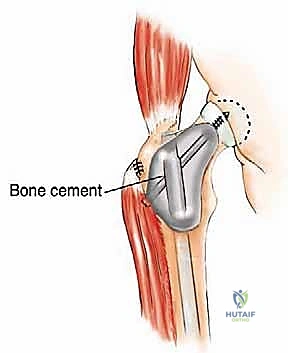
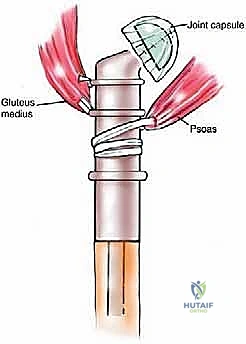
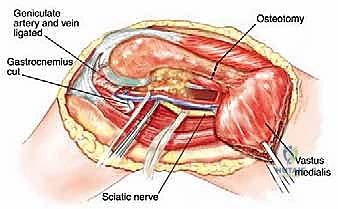
The proximal femur is a biomechanical marvel, subjected to immense joint reaction forces that can exceed several times body weight during normal ambulation. The anatomy here is complex and critical for maintaining pelvic stability and hip kinematics. The robust joint capsule, attaching anteriorly to the intertrochanteric line and posteriorly to the femoral neck, provides essential static stability. When performing a proximal femoral replacement for extensive metastatic destruction, meticulous capsulotomy and subsequent capsulorrhaphy (when possible) are vital steps. Extracapsular resection is often preferred if the tumor breaches the cortex of the femoral neck, ensuring adequate oncologic margins even in a palliative setting.
The lateral aspect of the proximal femur is dominated by the greater trochanter, the indispensable insertion site for the abductor complex, primarily the gluteus medius and minimus. These muscles act as the primary lateral stabilizers of the pelvis. Compromise of this insertion, either by tumor destruction or surgical excision, results in a debilitating Trendelenburg gait and profound functional impairment. During endoprosthetic reconstruction, reattaching the abductor mechanism to the prosthesis using heavy non-absorbable sutures, synthetic meshes, or specialized trochanteric claw grips is one of the most technically demanding yet critical steps of the procedure. Furthermore, deep lateral dissection places the superior gluteal neurovascular bundle at risk; meticulous hemostasis and careful retraction are mandatory.
Medially, the lesser trochanter serves as the confluence and insertion point for the iliopsoas tendon, the primary flexor of the hip. While isolated loss of the lesser trochanter is better tolerated than loss of the greater trochanter, preserving or reattaching the iliopsoas optimizes postoperative hip flexion strength, crucial for stair climbing and rising from a seated position. More importantly, the medial approach to the proximal femur is fraught with danger due to the proximity of the femoral neurovascular bundle (femoral artery, vein, and nerve) residing within the femoral triangle. Plunging instruments or overzealous medial retraction during tumor curettage or resection can lead to devastating vascular injury.
Femoral Diaphysis Considerations
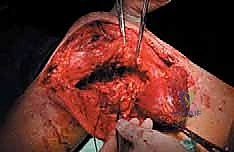
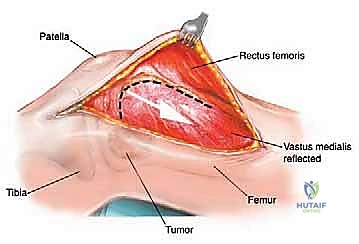
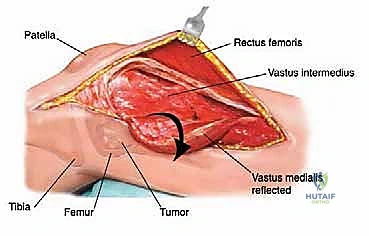
The femoral diaphysis is a dense, thick-walled tubular bone designed to withstand massive torsional and bending moments. Unlike primary bone sarcomas, which frequently present with large, aggressive soft tissue extensions, metastatic carcinomas to the diaphysis typically remain confined within the medullary canal or present with relatively small extraosseous soft tissue components, even in the presence of extensive cortical osteolysis. This is a critical clinical distinction. It often allows the orthopedic oncologist to spare vital extracortical structures, such as the enveloping muscle layers, which is invaluable for preserving postoperative motor function and providing soft tissue coverage for our implants.
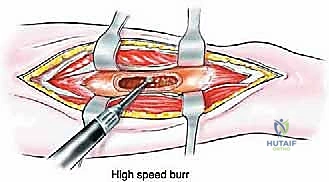
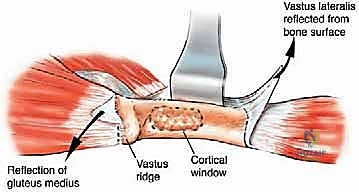
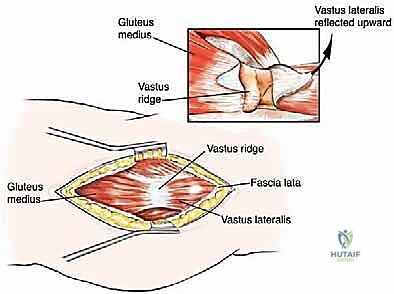
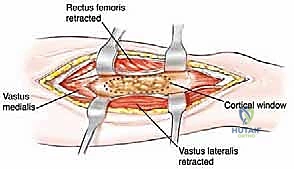
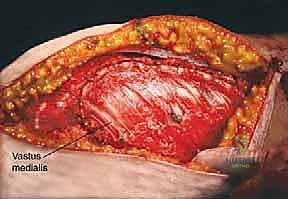
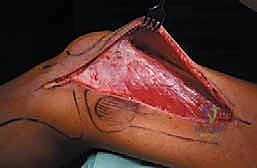
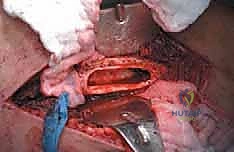
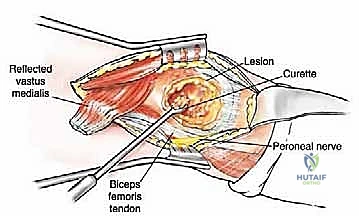
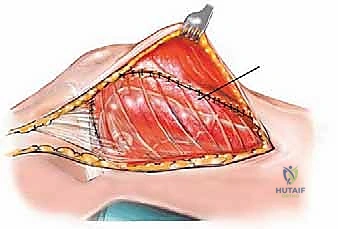
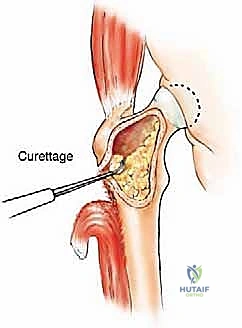
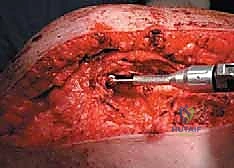
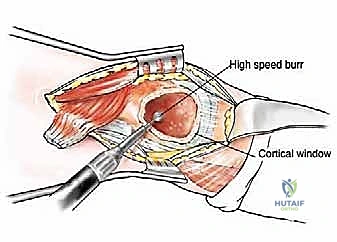
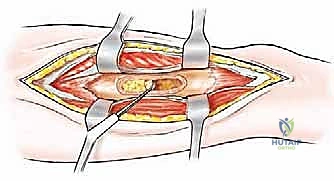
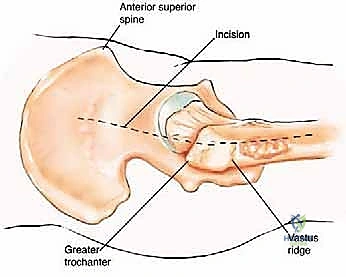
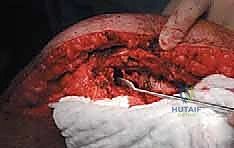
The diaphysis is encased by multiple, robust muscle compartments. The first layer, the vastus intermedius, directly envelops the anterior and lateral periosteum. Superficial to this, the rectus femoris and vastus lateralis intersect anterolaterally. Standard surgical approaches, such as the lateral approach to the femur, exploit the internervous plane between the vastus lateralis (femoral nerve) and the lateral intermuscular septum, though practically, splitting the vastus lateralis fibers is often required for direct lateral access. When performing open curettage and cemented plating or nailing, meticulous handling of these muscle envelopes ensures adequate blood supply for soft tissue healing and minimizes the risk of deep surgical site infections.
Neurovascular risks along the diaphysis are significant and must be respected. Deep to the vastus medialis and running intimately with the adductor magnus are the profunda femoris artery and its perforating branches. These perforators tether the vastus lateralis to the linea aspera and must be systematically identified and ligated during extensive lateral exposures to prevent massive hematoma formation. Posteriorly, the sciatic nerve descends through the posterior compartment. In cases of posterior cortical blowout from metastatic disease, extruded tumor or cement during reconstruction can compress the sciatic nerve, leading to foot drop and severe neuropathic pain. Careful digital exploration and protection of the posterior cortex during cement pressurization are therefore mandatory.
Exhaustive Indications and Contraindications
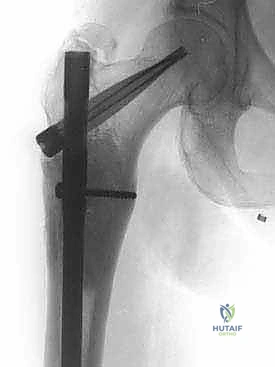
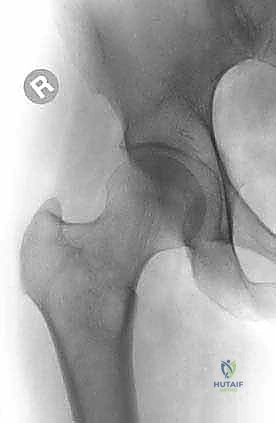
The decision to proceed with surgical intervention in the setting of femoral metastatic disease requires a nuanced synthesis of mechanical principles and oncologic prognosis. The absolute indication for surgery is a completed pathologic fracture in a patient who is medically fit for anesthesia. However, the true art of orthopedic oncology lies in the prophylactic management of impending fractures. We utilize the Mirels' scoring system, which evaluates four parameters: site of the lesion (upper limb, lower limb, trochanteric), nature of the lesion (blastic, mixed, lytic), size of the lesion (<1/3, 1/3-2/3, >2/3 of cortical diameter), and the presence of pain (mild, moderate, functional). A Mirels' score of 8 or higher strongly dictates the need for prophylactic surgical stabilization to avert the massive morbidity associated with a sudden mechanical failure.
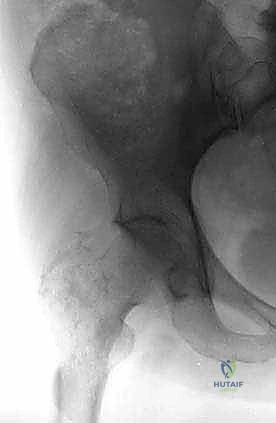
Beyond mechanical instability, intractable oncologic pain that is refractory to narcotic analgesia, bisphosphonates, and radiation therapy serves as a primary indication for surgical palliation. Intramedullary nailing or resection dramatically alters the local mechanical environment, eliminating micro-motion at the site of osteolysis and providing immediate, profound pain relief. Furthermore, in highly selected patients presenting with a solitary bone metastasis from favorable histologies (such as breast carcinoma or renal cell carcinoma) and a long anticipated life expectancy, wide en bloc resection followed by endoprosthetic reconstruction may be indicated with curative or long-term disease-free intent.
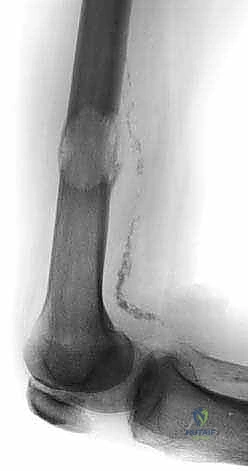
Contraindications must be evaluated with absolute rigor. Absolute contraindications include patients who are medically unfit for anesthesia due to severe cardiopulmonary compromise or profound coagulopathy that cannot be corrected. Additionally, if the patient's overall oncologic trajectory suggests a life expectancy of less than 4 to 6 weeks, major surgical intervention is generally contraindicated, as the surgical recovery period would consume their remaining lifespan without conferring a meaningful improvement in quality of life. In such terminal scenarios, compassionate pain management and supportive care are the appropriate clinical pathways.
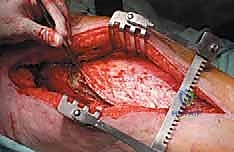
Relative contraindications include local active infection or massive, fungating tumor masses that preclude primary soft tissue closure. In cases of highly vascular tumors (e.g., renal cell, thyroid, hepatocellular carcinoma), proceeding without preoperative selective arterial embolization is a severe relative contraindication due to the high probability of exsanguinating intraoperative hemorrhage.
| Parameter | Indications for Surgical Intervention | Contraindications (Absolute & Relative) |
|---|---|---|
| Mechanical Status | Completed pathologic fracture; Mirels' score $\ge$ 8 (Impending fracture); >50% cortical destruction. | Stable lesion (Mirels' $\le$ 7) responsive to radiation; Non-weight bearing bone (less relevant for femur). |
| Pain Profile | Intractable, mechanical pain with weight-bearing; Failure of conservative/radiation therapy. | Pain well-controlled with oral analgesics; Pain purely neuropathic in nature. |
| Oncologic Status | Solitary metastasis (potential for wide resection); Life expectancy > 6-12 weeks. | Life expectancy < 4-6 weeks (Absolute); Multiorgan failure; Uncontrolled systemic sepsis. |
| Patient Physiology | Medically optimized for general or regional anesthesia; Hemodynamically stable. | Severe, uncorrectable coagulopathy (Absolute); Unembolized highly vascular primary (Relative). |
Pre-Operative Planning, Templating, and Patient Positioning
Fellows, I cannot overstate this: a meticulously planned surgery is a surgery half-won. The operating room is not the environment for improvisation when dealing with metastatic bone disease. Our objective is to execute a definitive, durable reconstruction that minimizes complications and maximizes immediate functional recovery. This necessitates a rigorous, protocol-driven approach to imaging, templating, and patient positioning.
Advanced Imaging and Staging Protocols
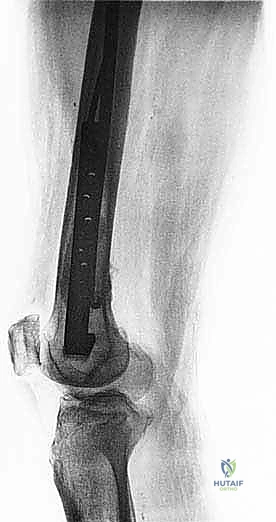
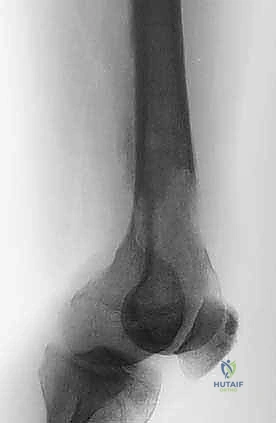
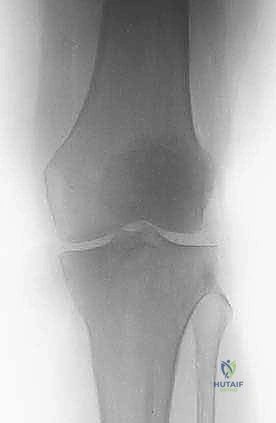
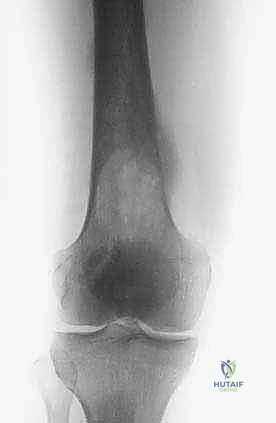
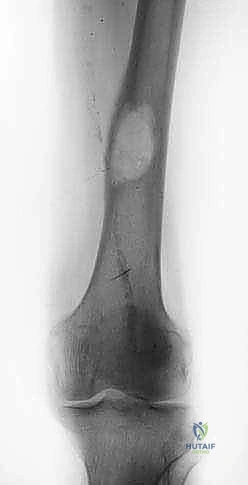
The foundation of our preoperative plan rests on comprehensive imaging. Standard orthogonal plain radiographs of the entire femur are absolutely mandatory. A localized view of the painful hip or knee is insufficient; we must rule out synchronous, asymptomatic skip metastases within the same bone that could dictate a different reconstructive strategy (e.g., opting for a full-length cephalomedullary nail instead of a short reconstruction nail). Radiographs provide an initial assessment of cortical thinning, deformity, and the overall biomechanical axis of the limb.
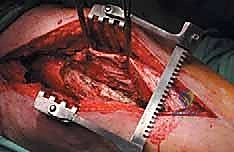
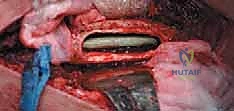
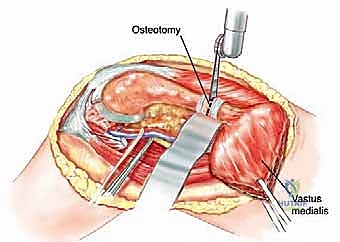
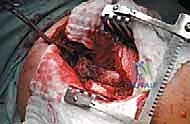
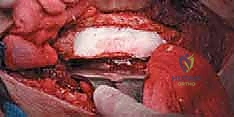
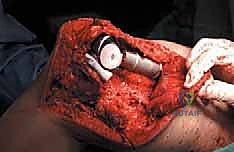
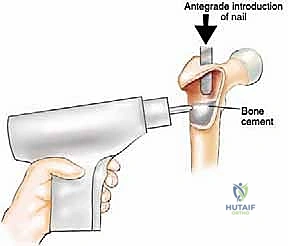
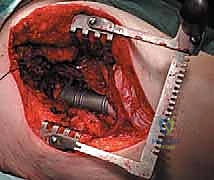
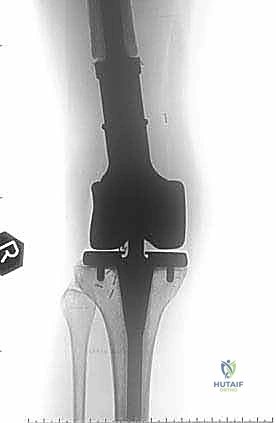
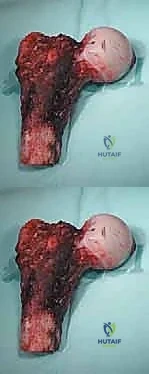
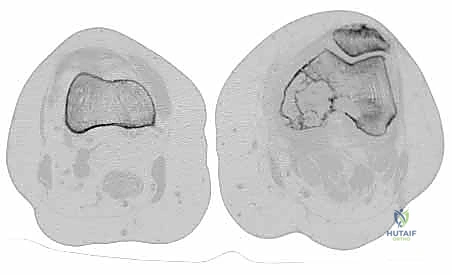
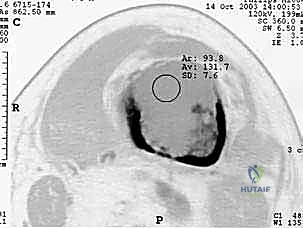
Computed Tomography (CT) is the gold standard for defining the precise geometry of bone destruction. Thin-slice CT with multiplanar reconstructions allows us to quantify the remaining cortical stock. This is the critical decision point: if sufficient circumferential cortex remains to contain a fixation device and cement, we proceed with intralesional curettage and cemented osteosynthesis (nail or plate). If the CT reveals massive, uncontained cortical loss, particularly in the proximal femur, the bone is deemed mechanically unsalvageable, and we must pivot our plan to segmental resection and endoprosthetic reconstruction.
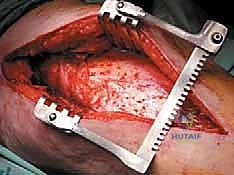
Magnetic Resonance Imaging (MRI) of the entire femur is utilized to delineate the intramedullary extent of the tumor, which is frequently far more extensive than plain radiographs suggest. MRI is critical for identifying soft tissue extension, neurovascular involvement, and skip lesions within the marrow cavity. Concurrently, systemic staging via Total Body Bone Scintigraphy (Technetium-99m) or, increasingly, PET-CT, is essential to identify the global skeletal tumor burden. Finally, if the patient presents with a destructive femoral lesion of unknown primary origin, a core needle biopsy is strictly mandated prior to definitive fixation. Nailing an undiagnosed primary bone sarcoma (like osteosarcoma) leads to catastrophic intramedullary dissemination and turns a potentially curable limb-salvage scenario into an absolute indication for hip disarticulation.
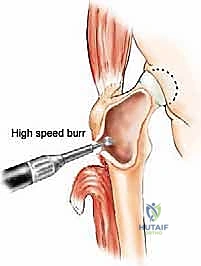
Biomechanical Templating and Positioning
Digital templating is a non-negotiable step. Using calibrated radiographs, we template the anticipated implant. For intramedullary nailing, we determine the optimal nail diameter, length, and the required radius of curvature to match the patient's native femoral bow. We must also plan the trajectory of the cephalomedullary screws to ensure they bypass the tumor defect and achieve purchase in the dense trabecular bone of the femoral head. For endoprosthetic reconstructions, templating determines the level of the femoral osteotomy, the required length of the prosthetic body to restore leg length, and the diameter of the intramedullary stem.
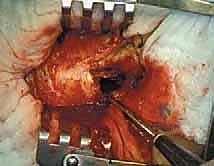
Patient positioning dictates our surgical access and intraoperative fluoroscopic capabilities. For the vast majority of femoral lesions, the patient is positioned supine. While a fracture table allows for excellent traction in traumatic fractures, it is frequently avoided in metastatic disease. The fixed boots and rigid positioning can easily induce iatrogenic fractures in osteoporotic, tumor-laden bone during setup. Therefore, a radiolucent flat Jackson table or a standard operating table is preferred.