Masterclass: Resection of Extracompartmental Sarcomas of the Lower Extremity
Key Takeaway
This masterclass provides an exhaustive, real-time walkthrough of lower extremity extracompartmental sarcoma resection. Fellows will learn intricate surgical anatomy, advanced preoperative planning, and granular intraoperative techniques for Type 1, 2, and 3 tumors. We cover precise instrument use, neurovascular protection, graft reconstruction, and managing critical complications, ensuring wide margins and optimal limb function.
Comprehensive Introduction and Patho-Epidemiology
The surgical management of soft tissue sarcomas arising within the extracompartmental spaces of the lower extremity represents one of the most formidable challenges in musculoskeletal oncology. Unlike typical intracompartmental lesions—where the tumor is anatomically constrained by robust fascial boundaries, allowing for straightforward en bloc resection with the muscle of origin—extracompartmental tumors defy these physical limitations. These "space tumors" inhabit the loose, areolar, and adipose-filled intervals between distinct muscular compartments. Because these spaces serve as the primary conduits for the extremity’s major neurovascular bundles, tumors arising here are intimately associated with critical limb-salvaging structures from their inception. Navigating this treacherous anatomy requires not only a masterful understanding of regional topography but also a nuanced appreciation of tumor biology and surgical margins.
Historically, the conceptualization and surgical approach to these extracompartmental lesions were poorly elucidated, leading to high rates of both local recurrence and unnecessary amputations. The seminal work of Enneking, which introduced the concept of anatomical compartmentalization, provided a crucial framework for surgical oncology. However, while Enneking’s principles perfectly dictated the management of intracompartmental disease, they often fell short in providing a pragmatic guide for the delicate, margin-compromising resections mandated by extracompartmental lesions. Today, while the American Joint Committee on Cancer (AJCC) classification has shifted its primary staging focus toward tumor grade, size, and depth, the fundamental understanding of anatomical spaces remains the absolute cornerstone for planning an oncologically sound resection while maximizing postoperative limb function.
The patho-epidemiology of these tumors dictates a highly individualized surgical strategy. Extracompartmental spaces are devoid of tight fascial restraints; they are filled with fat, loose fibrous connective tissue, and critical neurovascular structures. Consequently, soft tissue sarcomas in these regions—frequently undifferentiated pleomorphic sarcomas, liposarcomas, and synovial sarcomas—can extend longitudinally with minimal anatomical resistance. They often grow to massive proportions before clinical detection and frequently arise in direct, intimate proximity to major arteries, veins, and nerves. This biological reality means that standard wide margins (traditionally defined by a thick cuff of normal muscle) are often impossible to achieve without sacrificing the main blood supply or innervation to the distal extremity.
Our overarching goals in the modern era of limb-salvage surgery remain steadfast: achieving wide, negative surgical margins to prevent local recurrence, and preserving satisfactory, durable extremity function. However, the path to these goals is exceptionally delicate when dealing with space tumors. We must routinely navigate around, dissect free from, or entirely resect and reconstruct vital neurovascular structures. This task demands an extraordinary degree of precision, preoperative foresight, and a comprehensive understanding of both the regional anatomy and the reconstructive options available, often necessitating a multidisciplinary approach involving orthopedic oncologists, vascular surgeons, and plastic microsurgeons.
Detailed Surgical Anatomy and Biomechanics
Understanding the intricate, three-dimensional anatomy of the lower extremity’s extracompartmental spaces is paramount for safe and effective oncologic resection. The lower extremity contains three primary extracompartmental spaces that serve as critical transit routes for neurovascular structures: the femoral triangle, the sartorial (Hunter’s) canal, and the popliteal space. Each of these regions presents unique anatomical boundaries, distinct contents, and specific biomechanical implications following surgical extirpation.
The Femoral Triangle
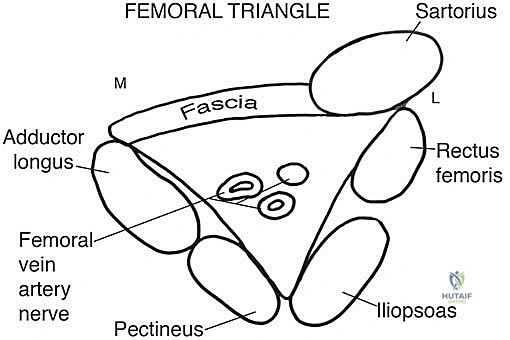
The femoral triangle is a three-dimensional pyramid located at the proximal anterior thigh, serving as the critical gateway for neurovascular structures transitioning from the pelvis into the lower limb. Its base is formed superiorly by the inguinal ligament. The lateral border is defined by the medial edge of the sartorius muscle, while the medial border is formed by the medial edge of the adductor longus muscle (or occasionally the anterior border of the gracilis). The floor of this space is muscular, composed of the iliopsoas laterally and the pectineus and adductor longus medially. The apex points distally, marking the point where the sartorius crosses over the adductors to form the roof of the sartorial canal.
The contents of the femoral triangle are arranged in a highly predictable medial-to-lateral fashion, classically remembered by the mnemonic NAVEL (Nerve, Artery, Vein, Empty space, Lymphatics). The femoral vein is the most medial structure, the femoral artery lies centrally, and the femoral nerve is situated most laterally. These structures emerge from beneath the inguinal ligament and traverse distally. The surgical risks in this region are profound; the close proximity of these structures means that even minor iatrogenic trauma can result in catastrophic hemorrhage, limb ischemia, or debilitating neurological deficits. Resection of the femoral nerve, for instance, results in profound quadriceps weakness, necessitating extensive postoperative bracing and rehabilitation to restore ambulatory capacity.

The Sartorial Canal (Hunter's Canal)
Distal to the apex of the femoral triangle, the neurovascular bundle enters the sartorial canal, a robust muscular tunnel connecting the anterior thigh to the popliteal fossa. In cross-section, this canal resembles an inverted triangle. The roof is formed by the sartorius muscle and the dense subsartorial fascia. The floor consists of the adductor longus proximally and the adductor magnus distally. The lateral boundary is formed by the thick fascial envelope of the vastus medialis muscle.
Deep within this heavily constrained canal lie the superficial femoral artery, the superficial femoral vein (positioned deep and slightly lateral to the artery), and the saphenous nerve. These structures are enveloped in a very thick, unyielding fascial sheath. They exit the canal distally through the adductor hiatus—a tendinous arch in the adductor magnus—to transition into the popliteal vessels. Dissection within the sartorial canal is notoriously challenging due to its depth and the dense fascial adhesions surrounding the vessels. Tumors here frequently compress or directly invade the superficial femoral vessels, making the decision between adventitial dissection (epifascial) and en bloc vascular resection with bypass grafting a frequent intraoperative dilemma.
The Popliteal Space
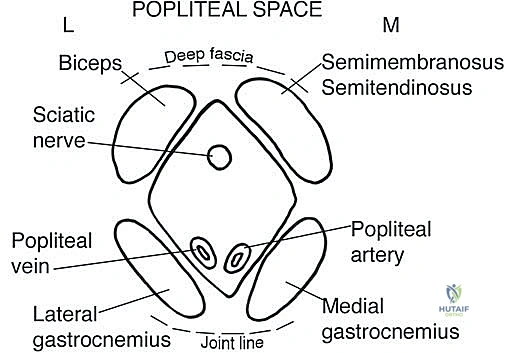
The popliteal space is a complex, diamond-shaped fossa located posterior to the knee joint, serving as the primary distribution center for the leg and foot. Its proximal boundaries are defined laterally by the biceps femoris and medially by the semitendinosus and semimembranosus muscles. Distally, it is bounded by the lateral and medial heads of the gastrocnemius muscle. The anterior boundary (floor) consists of the popliteal surface of the femur, the posterior capsule of the knee joint, and the popliteus muscle. Posteriorly, the space is roofed by the thick, unyielding popliteal fascia.
The spatial arrangement of the neurovascular structures within the popliteal fossa is critical for surgical planning. The popliteal artery is the deepest and most anterior structure, lying directly against the joint capsule. The popliteal vein is situated superficial and slightly lateral to the artery. The sciatic nerve enters the proximal apex of the space and typically bifurcates into the tibial nerve (which continues vertically down the midline) and the common peroneal nerve (which tracks laterally along the medial border of the biceps femoris to wrap around the fibular neck). Tumors in this space frequently encase the tibial nerve and popliteal vessels. Resection of the tibial nerve results in a devastating loss of plantarflexion and intrinsic foot function, while injury to the common peroneal nerve causes a classic foot drop.
Exhaustive Indications and Contraindications
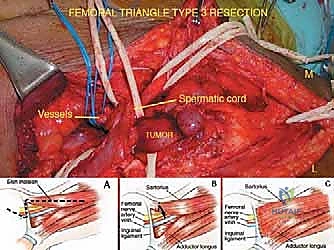
The decision-making process for resecting extracompartmental sarcomas of the lower extremity is complex, requiring a delicate balance between achieving local oncologic control and preserving a functional limb. The primary indication for surgical intervention is a biopsy-proven soft tissue sarcoma (regardless of grade) that is localized to the extracompartmental space without evidence of widespread, uncontrolled systemic metastasis. The goal in over 90% of these cases is a limb-sparing wide local excision.
Limb-salvage surgery is indicated when the tumor can be resected with negative margins while preserving or reconstructing the major neurovascular structures necessary for a viable, functional extremity. Advances in vascular grafting and microsurgical flap coverage have significantly expanded the indications for limb salvage. Even in cases where the tumor completely encases the superficial femoral or popliteal artery and vein (Type 3 tumors), en bloc resection of the vessels followed by immediate saphenous vein or synthetic interposition grafting is highly indicated, provided the patient has adequate distal runoff and lacks severe peripheral vascular disease.
Conversely, amputation remains a necessary, albeit less frequent, intervention. Absolute contraindications for limb-salvage surgery include tumors that extensively involve multiple major nerve trunks (e.g., complete encasement of the sciatic nerve extending into the lumbosacral plexus) where resection would leave a flail, insensate, and painful limb that is functionally inferior to a modern prosthesis. Additionally, massive fungating tumors complicated by severe infection, or tumors in patients with severe medical comorbidities precluding prolonged reconstructive surgery, are better served by amputation.
Relative contraindications must be evaluated on a case-by-case basis. These include extensive distant metastatic disease where the anticipated lifespan is short; in such scenarios, massive limb-salvage surgery may subject the patient to prolonged recovery without meaningful quality of life improvement, making palliative resection or radiation more appropriate. Neoadjuvant radiation therapy is frequently utilized to sterilize the reactive zone, effectively shrinking the required surgical margin and shifting a borderline unresectable tumor into the realm of limb salvage.
| Parameter | Indications for Limb-Sparing Resection | Contraindications for Limb-Sparing Resection (Favor Amputation) |
|---|---|---|
| Neurovascular Status | Major vessels/nerves free of tumor, or vessels resectable with viable bypass options. | Extensive involvement of major nerves (e.g., entire sciatic) resulting in a functionless, insensate limb if resected. |
| Tumor Extent | Localized disease, margins achievable via wide excision or planned marginal excision post-radiation. | Massive, fungating tumors with extensive skin/soft tissue loss precluding adequate flap coverage. |
| Systemic Disease | No metastases, or stable/resectable oligometastatic disease. | Widespread, uncontrolled metastatic disease with poor short-term prognosis (relative contraindication). |
| Patient Factors | Medically fit for prolonged surgery, compliant with extensive postoperative rehabilitation. | Severe peripheral vascular disease precluding bypass, profound medical comorbidities. |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the absolute blueprint for success in the resection of extracompartmental sarcomas. Before an incision is even considered, the surgical team must anticipate every potential anatomical challenge, formulate a primary resection strategy, and establish backup plans for unexpected intraoperative findings.
Patient Assessment and Advanced Imaging
Clinical evaluation begins with a meticulous history and physical examination. The duration of symptoms, presence of rest pain, paresthesias, or motor weakness are critical clues indicating potential nerve encasement. A detailed neurological assessment is mandatory; a dense motor deficit preoperatively suggests that the nerve is directly invaded and will likely require sacrifice. Plain radiography is the initial imaging step, utilized primarily to rule out secondary cortical erosion or periosteal reaction of the adjacent femur or tibia, which would necessitate a composite bone and soft tissue resection.
Advanced cross-sectional imaging is the workhorse of preoperative templating. Magnetic Resonance Imaging (MRI) with and without gadolinium contrast is the gold standard. It provides exquisite detail of the tumor’s size, its relationship to the muscular walls of the space, and its proximity to the neurovascular bundles. T1-weighted images are excellent for defining the fat planes, while T2-weighted STIR sequences highlight the peritumoral edema (the reactive zone), which often harbors microscopic satellite lesions. Computed Tomography Angiography (CTA) or MR Angiography (MRA) with three-dimensional reconstruction is indispensable for space tumors. These modalities map the arterial supply, demonstrate displacement or encasement of major vessels, and are critical for planning vascular bypass procedures.
Biopsy Strategy and Surgical Classification
The biopsy is the definitive diagnostic maneuver, but it must be executed with extreme caution. Because space tumors reside in loose tissue adjacent to major vessels, an open biopsy carries a high risk of iatrogenic hemorrhage and subsequent hematoma, which can widely seed the entire extracompartmental space with tumor cells. Therefore, image-guided Core Needle Biopsy (CNB) is the preferred modality. The biopsy tract must be meticulously planned so that it lies directly within the planned surgical incision, allowing the entire tract to be excised en bloc with the tumor.
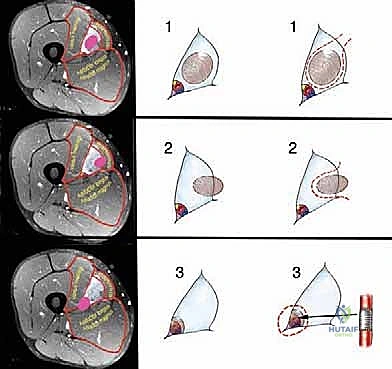
Based on the preoperative MRI and anticipated intraoperative findings, we categorize these extracompartmental lesions into three distinct groups to dictate the surgical plane of resection:
* Type 1 (Intraluminal) Tumors: These arise from the fat or fibrous tissue within the lumen of the space. They approximate but do not directly adhere to the muscular walls or neurovascular structures. The resection plane is through the normal fat surrounding the tumor.
* Type 2 (Wall) Tumors: These originate from the muscular boundaries or fascia bordering the space and grow inward. Resection requires taking the involved muscle en bloc with the tumor.
* Type 3 (Vessel) Tumors: The most treacherous lesions, originating from or directly invading the adventitia of arteries, veins, or the epineurium of nerves. These require subadventitial dissection or en bloc segmental resection and reconstruction of the involved structures.
Patient Positioning and Exposure
Proper patient positioning is critical for achieving adequate exposure, securing proximal and distal vascular control, and accessing potential autologous graft harvest sites. For tumors in the femoral triangle and sartorial canal, the patient is placed in the supine position. The entire abdomen, ipsilateral hip, and lower extremity down to the toes must be prepped and draped in a sterile fashion. This extensive field is required because proximal vascular control may necessitate an incision above the inguinal ligament to access the external iliac vessels, and the contralateral leg may need to be prepped if a great saphenous vein harvest is anticipated for bypass grafting.
For tumors located in the popliteal space, the patient is typically positioned prone. The limb is prepped from the gluteal fold down to the toes. A sterile tourniquet may be placed high on the thigh, though it is often left uninflated unless catastrophic bleeding occurs, as an inflated tourniquet can distort the venous anatomy and hinder the identification of subtle tumor margins. In all cases, the surgical table must be radiolucent to allow for intraoperative fluoroscopy if bone resection or orthopedic hardware placement becomes necessary.
Step-by-Step Surgical Approach and Fixation Technique
The surgical extirpation of extracompartmental sarcomas requires a meticulous, systematic approach predicated on the "no-touch" technique. The tumor pseudocapsule must never be breached, as this leads to microscopic spillage and a dramatically increased risk of local recurrence. The fundamental principle is to dissect through normal, healthy tissue planes, isolating the tumor along with a cuff of normal surrounding tissue.
Approach to the Femoral Triangle and Sartorial Canal
For anterior lesions, a longitudinal incision is made over the anterior thigh, carefully incorporating the previous biopsy tract within an elliptical island of skin. Skin flaps are elevated at the subfascial level to ensure adequate thickness and viability. The first and most critical step is obtaining proximal and distal vascular control. For femoral triangle tumors, the common femoral artery and vein are identified and looped with vessel loops just distal to the inguinal ligament, well away from the tumor margin. The femoral nerve is identified laterally and traced distally.
Once control is established, the dissection proceeds circumferentially. If the tumor is a Type 2 lesion involving the sartorius or adductor longus, these muscles are transected at their origins and insertions and mobilized en bloc with the mass. For Type 3 lesions abutting the vessels, a delicate epifascial or subadventitial dissection is performed. If the tumor directly invades the vessel wall, the vessel is clamped, the involved segment is resected en bloc with the tumor, and immediate vascular reconstruction is performed, typically utilizing a reversed great saphenous vein graft or a synthetic PTFE graft.
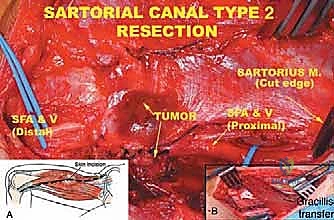
When transitioning into the sartorial canal, the dense subsartorial fascia is incised longitudinally. The sartorius muscle is retracted laterally or resected if involved. The superficial femoral artery and vein are meticulously separated from the tumor mass. The saphenous nerve is frequently sacrificed to achieve adequate margins in this tight space, resulting in expected numbness over the medial aspect of the lower leg, which is generally well-tolerated by the patient.
Approach to the Popliteal Space
Resection within the popliteal fossa is performed with the patient prone. An S-shaped or lazy-Z incision is utilized across the popliteal crease; straight longitudinal incisions across the joint line are strictly avoided to prevent severe postoperative flexion contractures. The deep fascia is incised, and the proximal boundaries of the space (the hamstrings) are identified and retracted.
The initial deep dissection focuses on identifying the sciatic nerve proximally before it plunges into the tumor bed. The nerve is traced distally to its bifurcation into the tibial and common peroneal nerves. Vessel loops are utilized for gentle retraction. Next, the popliteal artery and vein are identified. The popliteal vein is often splayed over the tumor and can be extremely thin-walled and friable. Ligation of the multiple genicular branches is frequently required to mobilize the popliteal vessels away from the tumor capsule.
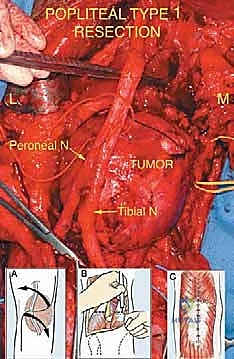
If the tumor arises from the posterior joint capsule (a frequent occurrence), an extra-articular resection may be required, taking a portion of the posterior femoral condyles or the entire posterior capsule, which subsequently necessitates complex soft tissue reconstruction or even a distal femoral replacement. Following tumor removal, meticulous hemostasis is achieved. Dead space management is critical in the popliteal fossa; local muscle flaps, such as a medial gastrocnemius rotational flap, are frequently employed to cover exposed vessels and nerves, providing a robust, vascularized bed that promotes healing and resists infection, particularly in the setting of preoperative radiotherapy.
Complications, Incidence Rates, and Salvage Management
The aggressive nature of extracompartmental sarcoma resections, combined with the frequent use of neoadjuvant radiation therapy, predisposes these patients to a high rate of postoperative complications. A thorough understanding of these risks and their respective salvage pathways is essential for the operating surgeon.
Wound complications are the most frequently encountered adverse events, occurring in approximately 25% to 35% of patients who undergo preoperative radiation followed by wide resection. These range from minor delayed healing and seroma formation to catastrophic major wound dehiscence with exposure of underlying vascular grafts or nerves. Immediate management involves aggressive surgical debridement, negative pressure wound therapy (NPWT), and often requires the mobilization of regional or free vascularized tissue transfers (e.g., Anterolateral Thigh or Latissimus Dorsi free flaps) by the plastic surgery team to achieve stable coverage.
Vascular and neurological complications are less frequent but carry devastating functional consequences. Thrombosis of a vascular bypass graft occurs in 5% to 10% of cases, presenting as acute limb ischemia. This requires emergent return to the operating room for thrombectomy or graft revision; failure to intervene promptly inevitably leads to limb loss. Neurological deficits are often expected (e.g., planned sacrifice of the common peroneal nerve), but iatrogenic traction injuries can cause unexpected neuropraxia. For permanent nerve sacrifices, secondary reconstructive procedures such as posterior tibial tendon transfers for foot drop, or the use of custom Ankle-Foot Orthoses (AFOs), are critical for restoring ambulatory function.
Local recurrence is the ultimate oncologic failure, occurring in 10% to 15% of high-grade space tumors despite aggressive local therapy. Recurrence in an extracompartmental space is particularly problematic due to the scarring from previous surgery and radiation, which obliterates normal anatomical planes. Salvage management for local recurrence often mandates amputation, as a second limb-sparing resection rarely achieves negative margins without causing unacceptable morbidity.
| Complication | Estimated Incidence | Salvage Management / Intervention |
|---|---|---|
| Major Wound Dehiscence / Necrosis | 25% - 35% (higher with radiation) | Serial debridement, NPWT, regional rotational flaps (gastrocnemius, sartorius) or free tissue transfer. |
| Vascular Graft Thrombosis / Ischemia | 5% - 10% | Emergent vascular surgery consultation, thrombectomy, revision of bypass graft, potential fasciotomies. |
| Deep Surgical Site Infection (SSI) | 10% - 15% | Intravenous antibiotics, operative irrigation and debridement, removal of infected synthetic material if present. |
| Permanent Neurological Deficit | Variable (depends on tumor location) | Orthotic bracing (AFO), targeted physical therapy, delayed tendon transfer surgery (e.g., for foot drop). |
| Local Oncologic Recurrence | 10% - 15% | Restaging imaging, multidisciplinary tumor board review; highly likely to require salvage amputation. |
Phased Post-Operative Rehabilitation Protocols
Postoperative rehabilitation following the resection of an extracompartmental sarcoma is a rigorous, protracted process that must be carefully tailored to the specific anatomical deficits created during surgery. The rehabilitation protocol is generally divided into distinct, progressive phases, with constant communication required between the orthopedic oncologist, the physical therapy team, and the patient.
Phase I: Immediate Post-Operative Period (Days 1-14)
The primary focus during the first two weeks is the protection of the surgical reconstruction, wound healing, and the prevention of systemic complications such as deep vein thrombosis (DVT) and pneumonia. If a vascular bypass or a complex microvascular free flap was performed, the limb is typically immobilized in a custom splint to prevent tension on the anastomoses. Strict elevation is maintained to combat the inevitable dependent edema. Passive range of motion (PROM) of adjacent, uninvolved joints may be initiated carefully, but active muscle contraction of the resected compartments is strictly prohibited. Aggressive DVT prophylaxis (chemical and mechanical) is mandatory, given the high-risk oncologic and surgical profile of these patients.
Phase II: Early Mobilization and Tissue Healing (Weeks 2-6)
As the surgical incisions heal and the risk of acute graft failure diminishes, the focus shifts to early mobilization. Active-assisted range of motion (AAROM) is gradually introduced. If a major nerve was sacrificed, appropriate orthotics are fitted at this stage; for example, a rigid AFO is provided for a patient with a common peroneal nerve resection to prevent equinus contracture and facilitate safe transfers. Weight-bearing status is highly variable; if no bone was resected, patients may progress to weight-bearing as tolerated with assistive devices. Lymphedema management, including compression garments and manual lymphatic drainage, is often initiated during this phase, as the resection of lymphatic channels within the femoral triangle or popliteal space frequently results in chronic limb swelling.
Phase III: Strengthening and Functional Restoration (Weeks 6-12 and Beyond)
Once soft tissue healing is secure, the rehabilitation protocol becomes significantly more aggressive. The goal is to maximize the strength of the remaining, intact musculature to compensate for the resected tissues. For instance, if the quadriceps mechanism was partially resected in the anterior thigh, intensive hamstring and gluteal strengthening is required to stabilize the knee during the stance phase of gait. Gait training is paramount, focusing on eliminating compensatory limps and improving proprioception.
Long-term functional outcomes are routinely monitored using standardized metrics such as the Musculoskeletal Tumor Society (MSTS) scoring system. While patients rarely return to high-impact, competitive athletics following massive extracompartmental resections, the majority achieve a stable, pain-free limb that allows for independent activities of daily living and a return to the workforce. Ongoing psychological support is also a critical component of
