Masterclass: Surgical Management of Benign Bone Cysts – UBC and ABC
Key Takeaway
Join this immersive surgical masterclass on managing unicameral (UBC) and aneurysmal bone cysts (ABC). We delve into precise patient positioning, detailed surgical anatomy, and real-time, step-by-step intraoperative execution. Learn critical techniques for percutaneous decompression, open curettage, and grafting, including pearls, pitfalls, and comprehensive postoperative care for optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
Welcome to this definitive masterclass on the surgical management of benign bone cysts. Within the realm of orthopedic oncology and pediatric orthopedics, the Unicameral Bone Cyst (UBC) and the Aneurysmal Bone Cyst (ABC) represent two of the most frequently encountered, yet fundamentally distinct, cystic lesions of the developing skeleton. While both entities present as radiolucent, fluid-filled cavities within bone, their underlying histopathology, biological behavior, and, most critically, their requisite surgical management diverge significantly. The overarching objective of this chapter is to transcend basic procedural descriptions, providing the orthopedic surgeon with an exhaustive understanding of the nuances, anticipated intraoperative challenges, and precise execution strategies required to optimize outcomes and minimize recurrence in our young patient demographic.
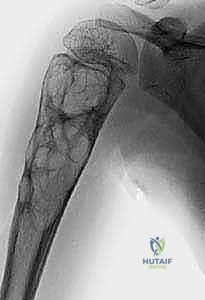
To master the surgical management of these lesions, one must first possess a granular understanding of their patho-epidemiology. The Unicameral Bone Cyst, frequently termed a simple bone cyst, is a benign, solitary, fluid-filled cavity that predominantly manifests in the metaphysis of long bones. Epidemiologically, the proximal humerus and proximal femur are the most ubiquitous sites, accounting for an estimated 40% to 80% of all documented cases. The demographic distribution is heavily skewed toward the pediatric and adolescent populations, with approximately 90% of patients presenting before the age of 20. Pathophysiologically, UBCs have historically been attributed to localized venous obstruction leading to increased interstitial fluid pressure and subsequent bone resorption. However, contemporary cytogenetic analyses have revolutionized our understanding, revealing translocations such as t(16;20)(p11.2;q13) and TP53 mutations in recurrent cases. This suggests that a subset of UBCs may harbor a true neoplastic etiology rather than being purely reactive phenomena. Macroscopically, these cysts are filled with a characteristic yellowish or serosanguineous fluid and are lined by a tenuous, semi-permeable fibrous membrane.
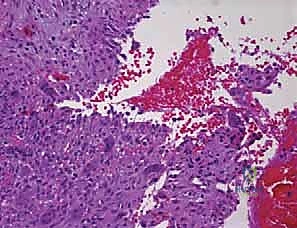
In stark contrast, the Aneurysmal Bone Cyst is a benign but highly active, expansile, and locally aggressive osteolytic lesion. ABCs are classically eccentric in their location and exhibit a predilection for the metaphyses of long bones and the posterior elements of the axial skeleton, particularly during the first two decades of life. The pathogenesis of ABCs is now definitively understood to be neoplastic in the majority of primary cases. This is driven by the recurrent chromosomal translocation t(16;17)(q22;p13), which results in the promoter swapping and subsequent upregulation of the USP6 (ubiquitin-specific protease 6) oncogene. This genetic aberration triggers a cascade of matrix metalloproteinase production and osteoclastogenesis, driving the rapid, destructive expansion characteristic of the lesion. Furthermore, ABCs can present as secondary lesions, arising in tandem with other primary bone tumors such as giant cell tumors, osteoblastomas, or chondroblastomas, necessitating a high index of suspicion during histopathological evaluation.
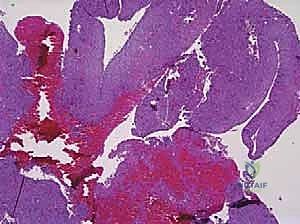
Histologically, the differentiation between UBCs and ABCs is paramount and dictates the aggressiveness of the surgical approach. A UBC is characterized by a paucicellular fibrous lining with occasional giant cells and hemosiderin-laden macrophages, lacking any endothelial lining. Conversely, the microscopic architecture of an ABC is defined by blood-filled cystic spaces separated by robust fibrous septa. Crucially, these blood-filled spaces are not lined by vascular endothelium, distinguishing them from true vascular malformations or hemangiomas. The fibrous septa are highly cellular, populated by fibroblasts, multinucleated osteoclast-like giant cells, and reactive woven bone. This hypercellular, aggressive stromal component is the target of surgical eradication; failure to adequately address these septa during curettage is the primary driver of the unacceptably high recurrence rates historically associated with ABCs.
Histological and Genetic Differentiation
The precise histological diagnosis often dictates the necessity for adjuvant therapies intraoperatively. When examining biopsy specimens, the presence of the USP6 rearrangement via fluorescence in situ hybridization (FISH) has become the gold standard for confirming a primary ABC, particularly in anatomically complex regions like the spine or pelvis where complete resection is fraught with morbidity. Understanding these cellular mechanisms empowers the surgeon to transition from mere mechanical curettage to a biologically targeted approach, recognizing that the fibrous septa of the ABC act as a proliferating neoplastic engine that must be systematically dismantled.
Detailed Surgical Anatomy and Biomechanics
The surgical management of benign bone cysts is inextricably linked to the intricate anatomy of the pediatric metaphysis and the biomechanical forces exerted upon the weakened host bone. UBCs are classically categorized based on their proximity to the physis. "Active" cysts are situated immediately adjacent to the epiphyseal plate. These lesions pose a dual threat: they possess a higher propensity for continued growth and expansion, and surgical intervention carries a significant risk of iatrogenic physeal injury, potentially culminating in premature growth arrest and subsequent angular deformity or limb length discrepancy. "Latent" cysts, conversely, have migrated diaphyseally away from the physis as a result of normal longitudinal bone growth. While the risk of physeal damage during surgery is mitigated in latent cysts, their diaphyseal location alters the biomechanical stress profile, often predisposing the bone to unique fracture patterns under torsional loads.
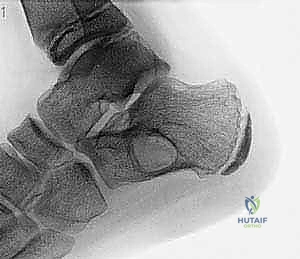
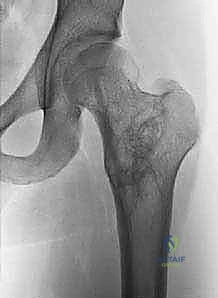
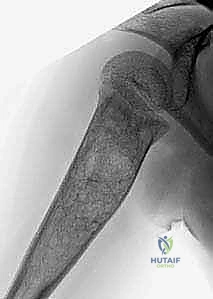
Biomechanically, a cystic lesion acts as a significant stress riser within the tubular structure of a long bone. The structural integrity of the bone is compromised proportionally to the cross-sectional area occupied by the cyst and the degree of cortical thinning. In the proximal femur, a common site for both UBCs and ABCs, the biomechanical implications are particularly dire. The proximal femur is subjected to massive compressive forces medially and tensile forces laterally during normal ambulation. A cyst occupying the femoral neck or intertrochanteric region drastically reduces the bone's load-bearing capacity, making prophylactic internal fixation a frequent necessity to prevent catastrophic displacement and avascular necrosis of the femoral head. The classic "fallen fragment" sign, pathognomonic for a UBC, occurs when a cortical fragment fractures and descends through the fluid-filled cavity, resting dependently. This sign not only confirms the fluid nature of the cyst but also highlights the extreme fragility of the thinned cortical shell.
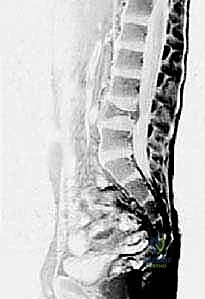
When evaluating Aneurysmal Bone Cysts, the anatomical considerations become even more complex due to their expansile and locally destructive nature. ABCs frequently violate the cortical boundaries, expanding into the surrounding soft tissues, contained only by a thin, reactive periosteal shell. In the appendicular skeleton, this eccentric expansion can distort normal muscle attachments and neurovascular routing. However, it is in the axial skeleton that the anatomy of an ABC presents the most formidable surgical challenge. Spinal ABCs exhibit a strong predilection for the posterior elements—the lamina, pedicles, and spinous processes.

As a spinal ABC expands, it frequently encroaches upon the spinal canal or neural foramina, leading to radiculopathy or frank myelopathy. The destruction of the pedicles and facet joints rapidly compromises the structural stability of the spinal column, often resulting in asymmetric vertebral body collapse and progressive kyphoscoliosis. Surgical planning in these regions requires meticulous 3D visualization to navigate the distorted anatomy, decompress neural elements, and reconstruct the destabilized spinal segment. The highly vascular nature of ABCs further complicates the anatomical approach, as massive intraoperative hemorrhage is a well-documented risk, particularly in pelvic and spinal lesions where proximal vascular control may be challenging to achieve.
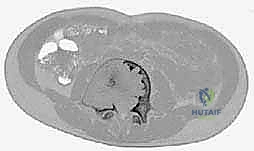
Proximal Femoral Vascularity
A critical anatomical consideration when approaching proximal femoral cysts is the preservation of the medial circumflex femoral artery (MCFA) and its retinacular branches. Surgical approaches, particularly those involving extensive curettage or the placement of pediatric dynamic hip screws (DHS), must be meticulously planned to avoid compromising this precarious blood supply. Iatrogenic avascular necrosis in a pediatric patient is a devastating complication that overshadows the successful treatment of the underlying cyst. Therefore, fluoroscopic guidance and a thorough understanding of the extracapsular and intracapsular vascular anatomy are mandatory prerequisites for any intervention in this region.
Exhaustive Indications and Contraindications
The decision-making algorithm for intervening in benign bone cysts requires a delicate balance between the natural history of the lesion, the biomechanical risk of fracture, and the potential morbidity of the surgical procedure. The philosophy of intervention has evolved significantly; while observation remains a valid strategy for select, asymptomatic lesions, proactive management is increasingly favored to prevent the profound functional impairment associated with pathologic fractures, particularly in weight-bearing extremities.
For Unicameral Bone Cysts, the indications for surgery are highly stratified based on location and size. Small, asymptomatic UBCs located in non-weight-bearing bones (such as the distal radius or ulna) or those occupying less than one-third of the transverse diameter of the bone can often be managed with vigilant radiographic observation. However, surgical intervention is strongly indicated for lesions that are painful, progressively enlarging, or located in high-stress weight-bearing areas such as the proximal femur or proximal tibia. Furthermore, if a UBC occupies greater than 50% of the bone's cross-sectional area or exhibits a cortical thickness of less than 2 millimeters, the risk of pathologic fracture is deemed unacceptably high, warranting prophylactic stabilization and cyst management. While a pathologic fracture through a UBC can occasionally stimulate spontaneous healing of the cyst (occurring in approximately 10-15% of cases), waiting for a fracture to occur in a critical anatomical zone is not a recommended primary strategy.
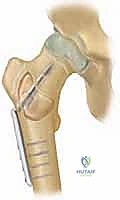
The indications for treating Aneurysmal Bone Cysts are markedly more aggressive, reflecting the lesion's destructive biological behavior. Conservative management or observation has virtually no role in the treatment of a confirmed ABC. Surgical intervention is indicated upon diagnosis to halt local bone destruction, alleviate pain, and prevent impending fractures. In the spine, the indications are emergent if there is evidence of progressive neurological deficit or impending spinal instability due to pedicle or vertebral body destruction. The rapid expansion of an ABC can lead to severe, irreversible functional deficits if not addressed decisively. Furthermore, the presence of an ABC often necessitates a biopsy to definitively rule out telangiectatic osteosarcoma, a malignant entity that can mimic an ABC both radiographically and clinically.
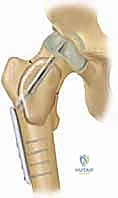
Contraindications to surgical intervention are relatively few but absolute when present. Active, uncontrolled local or systemic infection precludes elective cyst curettage and bone grafting due to the high risk of catastrophic deep space infection. Severe medical comorbidities that render the patient unfit for anesthesia are also contraindications, though these are rare in the typical pediatric demographic. A relative contraindication exists for active UBCs directly abutting the physis in very young children; in these scenarios, less invasive modalities such as percutaneous steroid or bone marrow aspirate injections may be prioritized over aggressive curettage to minimize the risk of physeal arrest, delaying definitive mechanical treatment until the cyst has migrated to a latent position.
Summary Table of Indications and Contraindications
| Lesion Type | Primary Surgical Indications | Relative/Absolute Contraindications |
|---|---|---|
| Unicameral Bone Cyst (UBC) | - Lesion >50% of bone diameter - Cortical thickness <2mm - Weight-bearing bone (e.g., prox femur) - Recurrent pathologic fractures - Failure of conservative/injection therapy |
- Small, asymptomatic lesions in upper extremity - Active infection - Cyst immediately adjacent to physis (relative - favor injection over curettage) |
| Aneurysmal Bone Cyst (ABC) | - Progressive expansile growth - Pain and functional impairment - Impending or actual pathologic fracture - Spinal involvement with instability - Neurological compromise (emergent) |
- Active local infection - Medically unstable patient - Inability to obtain clear margins in expendable bones (may favor en bloc resection instead) |
Pre-Operative Planning, Templating, and Patient Positioning
The hallmark of a master surgeon is the exhaustive nature of their preoperative planning. For benign bone cysts, this phase is not merely administrative; it is the blueprint for success that dictates the trajectory of the entire surgical intervention. The process begins with high-resolution, multi-planar imaging. While plain orthogonal radiographs provide the baseline assessment of the lesion's size, location, and the presence of a "fallen fragment" or cortical breach, advanced imaging is non-negotiable for comprehensive templating. Computed Tomography (CT) is invaluable for assessing the precise cortical thickness, identifying subtle micro-fractures, and mapping the 3D geometry of the cyst, which is particularly critical in anatomically complex regions like the pelvis, calcaneus, or spine.
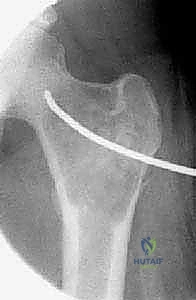
Magnetic Resonance Imaging (MRI) serves a dual purpose: it aids in the differential diagnosis and delineates the extent of soft tissue involvement or perilesional edema. For ABCs, MRI is critical for identifying the characteristic, though not pathognomonic, fluid-fluid levels caused by the settling of blood products of varying ages within the cystic cavities. Furthermore, MRI provides the most accurate assessment of the lesion's proximity to the unossified cartilaginous physis, a vital metric for planning the trajectory of curettes or hardware to avoid growth arrest. Once the imaging is secured, digital templating is performed to select the appropriate implants. If internal fixation is required—such as flexible intramedullary nails for a humeral UBC or a pediatric dynamic hip screw for a femoral neck lesion—the exact length, diameter, and trajectory of the hardware must be mapped preoperatively.
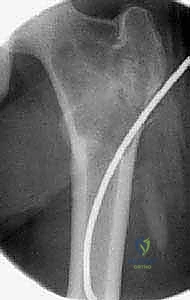
Biopsy planning is a critical sub-component of the preoperative phase. The surgeon must decide between a core needle biopsy and an open incisional biopsy. For suspected UBCs, aspiration of the characteristic serosanguineous fluid combined with a core biopsy of the cyst wall is often sufficient. However, for ABCs, a needle biopsy is frequently fraught with sampling error, yielding only non-diagnostic blood and failing to capture the diagnostic fibrous septa. Therefore, an open incisional biopsy, often performed with frozen section analysis, is frequently required. The biopsy tract must be meticulously planned so that it can be excised en bloc during the definitive surgical procedure, a principle borrowed from orthopedic oncology to prevent potential seeding if the lesion surprisingly proves to be malignant (e.g., telangiectatic osteosarcoma).
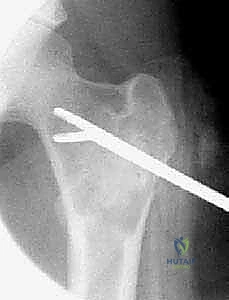
Patient positioning and operating room setup must be optimized for both surgical access and fluoroscopic visualization. The patient is typically positioned on a radiolucent table to allow for unimpeded orthogonal fluoroscopy. For proximal femoral lesions, a fracture table or a flat Jackson table with a bump under the ipsilateral hip may be utilized. Anesthesia considerations are paramount, particularly for large ABCs. Given their highly vascular nature, massive, rapid intraoperative blood loss is a distinct possibility. The anesthesia team must be briefed on this risk, and large-bore intravenous access, continuous arterial blood pressure monitoring, and the availability of cross-matched blood products or cell-saver technology are mandatory prerequisites before the first incision is made.
Fluoroscopic Integration
The integration of the C-arm fluoroscope must be seamless. The surgeon should verify that perfect anteroposterior and lateral views of the target anatomy can be obtained prior to prepping and draping. In cases involving the continuous decompression technique using flexible nails for UBCs, the ability to visualize the entire length of the long bone is necessary to ensure proper nail advancement and cortical purchase without breaching the joint space or the physis.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution for benign bone cysts demands a meticulous, systematic approach, tailored specifically to the biological behavior of the lesion. The approach begins with a carefully planned incision that respects the local neurovascular anatomy and allows for extensile exposure if necessary. Soft tissue handling must be atraumatic; the periosteum should be sharply incised and elevated as a continuous flap, preserving its osteogenic potential, which is critical for postoperative bone remodeling and graft incorporation.
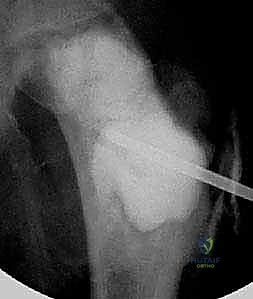
For Unicameral Bone Cysts, the surgical armamentarium ranges from minimally invasive injections to formal curettage and grafting. If an injection technique is selected (utilizing corticosteroids or concentrated bone marrow aspirate), two large-bore trocars are introduced into the cyst under fluoroscopic guidance. The cyst is thoroughly aspirated, and radiopaque contrast is injected to confirm that the lesion is truly unicameral and to map its extent. The cyst is then aggressively irrigated with saline to disrupt the fibrous lining before the therapeutic agent is instilled. For more recalcitrant UBCs, the continuous decompression technique utilizing flexible intramedullary nails has proven highly effective. Nails are introduced in a retrograde or antegrade fashion, passing through the cyst to provide immediate biomechanical stability while simultaneously creating a permanent channel for the egress of cyst fluid, thereby neutralizing the localized venous hypertension that drives cyst expansion.
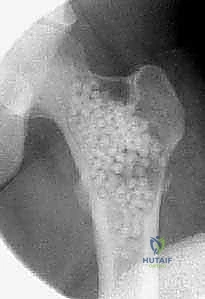
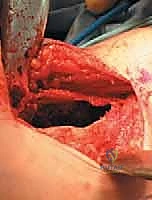
The surgical management of Aneurysmal Bone Cysts requires a significantly more aggressive strategy: intralesional extended curettage. A large cortical window is created to gain unrestricted access to the entire cystic cavity. The surgeon must systematically and aggressively curette the lesion, removing all fibrous septa and the semi-solid, hypervascular tissue. This is not a gentle procedure; the goal is to reach healthy, bleeding, punctate cortical bone in all dimensions. Because ABCs possess a high propensity for recurrence from microscopic cellular remnants, mechanical curettage alone is insufficient.
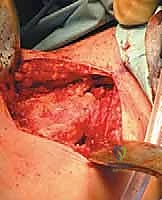
Following mechanical debridement, the application of local adjuvant therapy is mandatory to achieve extended margins. The high-speed burr is utilized to aggressively decorticate the inner walls of the cavity, breaking down the microscopic ridges where neoplastic cells hide. Subsequently, chemical or thermal adjuvants are employed. Phenol (followed by alcohol neutralization), hydrogen peroxide, or the argon beam coagulator are systematically applied to the cavity walls to induce a zone of cellular necrosis extending 1-2 millimeters beyond the mechanical margin. The argon beam coagulator is particularly favored for its ability to achieve excellent hemostasis while delivering controlled, uniform thermal necrosis.
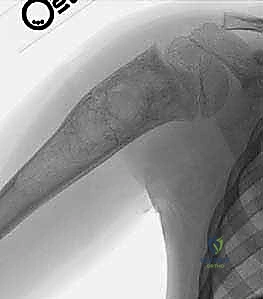
Once the cavity is sterilized of neoplastic tissue, the resulting defect must be managed. For small defects, the cavity may be left to fill with hematoma or packed with autologous cancellous bone graft harvested from the iliac crest. For larger, structurally significant defects, a combination of allograft (cancellous chips or structural struts) and synthetic bone void fillers (such as calcium phosphate or calcium sulfate) is utilized. If the mechanical integrity of the bone is critically compromised, prophylactic internal fixation must be applied. In the proximal femur, this typically involves the placement of a pediatric dynamic hip screw or cannulated screws, carefully bypassing the physis while providing rigid stabilization to allow the graft to incorporate and the bone to remodel.
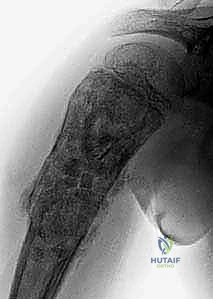

Hemostasis in ABC Resection
Managing the profound hemorrhage associated with ABC curettage is a critical intraoperative challenge. The bleeding is often diffuse and pulsatile from the bone margins. Systematic packing with laparotomy sponges, the use of topical hemostatic agents (e.g., bone wax, thrombin-soaked Gelfoam), and the rapid, sequential application of the argon beam coagulator are essential techniques. The surgeon must work expediently yet methodically, achieving hemostasis in one quadrant before moving to the next, ensuring that blood loss is minimized without compromising the thoroughness of the oncologic curettage.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, the management of benign bone cysts is fraught with potential complications, primarily driven by the biological persistence of the lesions and the vulnerability of the pediatric skeleton. Recurrence remains the most frustrating complication. For UBCs managed with simple aspiration and injection, recurrence rates can approach 20% to 40%. The use of flexible intramedullary nailing has reduced this significantly, but recurrence is still a documented risk, particularly in younger patients with active cysts near the physis. For ABCs, historical recurrence rates following simple curettage were unacceptably high, ranging from 30% to 50%. However, the modern implementation of extended intralesional curettage with high-speed burring and local adjuvants (phenol or argon beam) has drastically reduced the recurrence rate to approximately 10% to 15%.
Phy
