Masterclass: ORIF of Capitellum and Capitellar–Trochlear Shear Fractures
Key Takeaway
Welcome, fellows, to an immersive intraoperative masterclass on open reduction and internal fixation (ORIF) of capitellum and capitellar–trochlear shear fractures. We'll navigate complex elbow anatomy, meticulous surgical techniques, and critical decision-making. From precise patient positioning to advanced fixation strategies, this session will equip you with the expertise to achieve optimal outcomes for these challenging distal humerus injuries.
Comprehensive Introduction and Patho-Epidemiology
Welcome to the operating theater, colleagues. Today, we are undertaking a comprehensive masterclass on a fascinating, technically demanding, and often unforgiving injury: fractures of the capitellum and the more complex capitellar–trochlear coronal shear fractures. While statistically uncommon—accounting for less than 1% of all elbow fractures and roughly 6% of distal humerus fractures—their management is absolutely critical for the restoration of radiocapitellar and ulnohumeral kinematics. These injuries frequently present in conjunction with radial head fractures, lateral ulnar collateral ligament (LUCL) avulsions, and posterior elbow dislocations, demanding a highly sophisticated diagnostic and operative approach to prevent catastrophic joint failure.
The mechanism of injury typically involves a high-energy axial load transmitted through an outstretched hand. As the patient falls, the radial head acts as a dense bony battering ram, impacting the capitellum. In capitellar–trochlear shear fractures, this impaction occurs with the elbow in a semi-extended or slightly flexed position, leading to a profound coronal shearing force across the articular surface of the distal humerus. The fracture fragments, devoid of substantial soft-tissue tethers, commonly displace proximally and anteriorly into the radial and coronoid fossae, causing an immediate mechanical block to elbow flexion.
Demographically, isolated capitellar fractures are almost exclusively observed in the adult population, with a pronounced preponderance in females. This gender discrepancy is traditionally attributed to the female anatomical tendency toward a greater carrying angle (cubitus valgus), which inherently directs more axial force through the lateral column of the elbow during a fall. Furthermore, the elderly population is highly susceptible due to underlying osteopenia and osteoporosis, transforming low-energy ground-level falls into complex articular shear injuries.
The natural history of untreated or poorly managed capitellar and coronal shear fractures is universally dismal. Nonoperative management of displaced fractures invariably leads to progressive loss of motion, chronic pain, mechanical crepitus, and rapid-onset post-traumatic arthrosis. The intra-articular nature of these fractures, combined with the precise tolerances of the radiocapitellar joint, dictates that anything short of absolute anatomic reduction and rigid internal fixation is unacceptable. As orthopedic surgeons, we must recognize that these are not simply "lateral condyle" fractures; they are complex articular shear injuries that require meticulous preoperative planning and masterful intraoperative execution.
Detailed Surgical Anatomy and Biomechanics
To successfully navigate the surgical management of capitellar fractures, one must possess an intimate understanding of the intricate osseous, vascular, and neurologic anatomy of the distal humerus. The distal humerus flares into two distinct condyles, forming the lateral and medial columns that act as a structural arch supporting the central trochlea. The lateral column is uniquely designed to withstand the compressive forces transmitted from the wrist through the radius.
Osseous and Articular Architecture
The capitellum represents the anterior aspect of the lateral column. It is a hemispherical structure entirely covered by hyaline articular cartilage anteriorly and inferiorly, but notably devoid of cartilage posteriorly. It is directed distally and anteriorly at an angle of approximately 30 degrees relative to the longitudinal axis of the humeral shaft. The radial head articulates with the anterior surface of the capitellum during elbow flexion and glides to its inferior surface during full extension. The center of rotation of the capitellum is collinear with the center of the trochlea, a biomechanical absolute that must be restored to preserve the natural flexion-extension arc of the elbow.
Vascular Supply and the Risk of Osteonecrosis
The vascular anatomy of the capitellum is a critical consideration during surgical exposure. The blood supply is derived predominantly from posterior perforating vessels originating from the lateral arcade—an intricate anastomosis between the radial collateral arteries of the profunda brachii and the radial recurrent artery. Because the capitellum lacks significant soft-tissue attachments anteriorly, its viability relies heavily on this posterior intraosseous perfusion. Extensive posterior surgical dissection, aggressive soft-tissue stripping, or severe posterior comminution can obliterate this vascular network, significantly increasing the risk of postoperative osteonecrosis and subsequent articular collapse.
Critical Neurovascular Structures
Neurologic considerations dictate our surgical approaches. Proximally, the radial nerve travels between the brachialis and brachioradialis muscles before crossing the elbow joint. Meticulous dissection is required to protect it, especially when extending the lateral approach proximally along the supracondylar ridge. Distally, the posterior interosseous nerve (PIN)—the deep motor branch of the radial nerve—is highly vulnerable as it dives into the supinator muscle. When reflecting the common extensor origin or developing the interval between the anconeus and the extensor carpi ulnaris (ECU), the PIN is at risk. Maintaining the forearm in full pronation intraoperatively is a vital maneuver, as it dynamically shifts the PIN anteriorly and medially, safely away from the lateral retractor blades. The ulnar nerve, located in the cubital tunnel behind the medial epicondyle, must also be respected, particularly in Type 4 coronal shear fractures that require medial column exposure or when placing trans-condylar posterior-to-anterior screws.
Ligamentous and Muscular Intervals
The lateral collateral ligament (LCL) complex, specifically the lateral ulnar collateral ligament (LUCL), inserts adjacent to the lateral margin of the capitellum at the isometric point of the lateral epicondyle. Traumatic avulsion of this complex is ubiquitous with coronal shear fractures. Recognizing and anatomically repairing the LUCL at the conclusion of the case is mandatory to prevent chronic posterolateral rotatory instability (PLRI). Muscularly, the Köcher approach utilizes the internervous plane between the anconeus (radial nerve) posteriorly and the ECU (PIN) anteriorly. Alternatively, the Kaplan approach exploits the interval between the extensor digitorum communis (EDC) and the extensor carpi radialis brevis/longus (ECRB/ECRL), though it places the PIN at slightly higher risk if extended too far distally.
Exhaustive Indications and Contraindications
The philosophy governing the treatment of capitellar and coronal shear fractures is firmly rooted in the principles of rigid internal fixation and early mobilization. Because the elbow joint is notoriously prone to post-traumatic stiffness, prolonged immobilization is the enemy of a functional outcome. Therefore, the primary goal of surgery is to achieve a construct stable enough to permit immediate, active range of motion.
Indications for Open Reduction and Internal Fixation (ORIF) encompass nearly all capitellar fractures. Any fracture exhibiting displacement greater than 2 millimeters, any fracture causing a mechanical block to flexion or extension, and all capitellar-trochlear shear fractures (Bryan and Morrey Type 4) mandate surgical intervention. Even seemingly "nondisplaced" fractures on initial plain radiographs often reveal subtle articular step-offs or instability on advanced imaging, pushing the pendulum heavily toward operative management. The presence of concomitant injuries, such as a radial head fracture or an LUCL tear, further solidifies the absolute need for surgical exploration and stabilization.
Nonoperative management is an extreme rarity in modern orthopedic practice and is reserved for a very narrow clinical subset. Truly nondisplaced, isolated Type 1 capitellar fractures in highly compliant patients may theoretically be managed with a brief period of splinting (1-2 weeks) followed by heavily supervised, protected motion. However, the risk of secondary displacement is high, and weekly radiographic surveillance is mandatory. We strongly caution against nonoperative management for any multi-fragmentary or shear-type pattern due to the inherent instability of the joint surface.
Contraindications to surgery are generally limited to absolute medical unfitness for anesthesia. Patients with severe, advanced dementia who cannot participate in postoperative rehabilitation, or non-ambulatory patients with a painless, functional pseudarthrosis of a chronic fracture, may be managed nonoperatively. Active local infection over the surgical site is an absolute contraindication until the soft-tissue envelope is optimized.
| Parameter | Indications for ORIF | Contraindications / Non-Op Management |
|---|---|---|
| Fracture Displacement | > 2 mm displacement; Articular step-off | Strictly < 2 mm (Requires CT confirmation) |
| Fracture Pattern | Bryan & Morrey Types 2, 3, 4; Coronal shears | Truly isolated, non-displaced Type 1 (Rare) |
| Mechanical Status | Block to flexion/extension; Joint incongruity | Full, painless ROM with no mechanical block |
| Associated Injuries | Radial head fracture; LUCL tear; Dislocation | None (Isolated injury only) |
| Patient Factors | Active, compliant, medically optimized | Medically unstable; Severe dementia; Local infection |
Pre-Operative Planning, Templating, and Patient Positioning
Before a scalpel ever touches the skin, a meticulous, exhaustive preoperative plan must be formulated. The complexity of coronal shear fractures demands a three-dimensional understanding of the injury pattern, as intraoperative surprises in the elbow often lead to suboptimal outcomes.
Advanced Imaging and Classification
Standard anteroposterior (AP) and lateral radiographs are the starting point but are notoriously insufficient for definitive planning. The AP view is often unrevealing because the thin, sheared articular fragment overlies the intact posterior columns, obscuring the fracture lines. The lateral radiograph is far more diagnostic. Look specifically for the characteristic "double arc" sign, pathognomonic for a Type 4 coronal shear fracture involving both the capitellum and the lateral ridge of the trochlea.

To fully delineate the injury, a Computed Tomography (CT) scan with 1- to 2-mm fine cuts is absolutely mandatory in all cases. CT imaging identifies the exact size of the articular fragments, the presence of posterior column comminution, and the involvement of the medial trochlea. Three-dimensional (3D) CT reconstructions provide the ultimate roadmap, allowing the surgeon to virtually rotate the distal humerus and appreciate the precise anatomical orientation of the fracture lines.
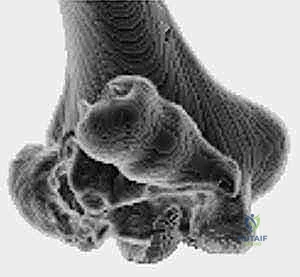
We utilize the Bryan and Morrey classification, modified by McKee, to categorize these injuries. Type 1 (Hahn-Steinthal) involves a large osseous fragment of the capitellum. Type 2 (Kocher-Lorenz) is a superficial articular shear with minimal subchondral bone. Type 3 (Broberg-Morrey) is highly comminuted. Type 4, the McKee modification, is a severe coronal shear including the capitellum and a significant portion of the trochlea.
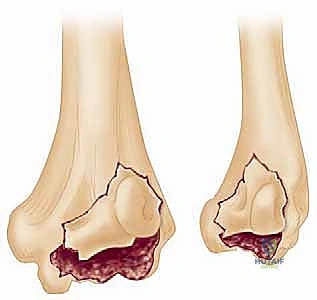
Furthermore, we heavily rely on the Dubberley classification, which categorizes fractures based on the presence (Type B) or absence (Type A) of posterior condylar comminution. This is critical: if posterior comminution is present, anterior-to-posterior screws alone will fail due to lack of a stable posterior buttress, mandating the use of posterior anti-glide plating.
Logistics and Patient Positioning
Timing is critical; surgery should ideally be performed within 5 to 10 days of injury. Operating too early through massively swollen tissues invites wound breakdown, while delaying beyond two weeks allows early osseous consolidation and severe soft-tissue contractures, making anatomic reduction nearly impossible. Ensure the operating room is stocked with headless compression screws (e.g., 2.4mm and 3.0mm Herbert or Acutrak screws), 0.045-inch K-wires, and a comprehensive mini-fragment (2.0mm/2.4mm) plating set.
General anesthesia is recommended to ensure complete muscle paralysis, facilitating joint distraction and reduction. The patient is typically positioned supine with the operative arm extended onto a radiolucent hand table. A sterile tourniquet is applied high on the brachium. This position allows for an unencumbered lateral approach and excellent fluoroscopic visualization. For highly complex Type 4 fractures with severe posterior comminution (Dubberley Type B) that may require an olecranon osteotomy for exposure, the patient may be positioned in the lateral decubitus or prone position, with the arm draped over a well-padded bolster.
Step-by-Step Surgical Approach and Fixation Technique
Let us proceed to the surgical execution. The goal is anatomic reduction of the articular surface, rigid fixation to allow early motion, and meticulous repair of the lateral ligamentous complex.
Exposure: The Extensile Lateral Approach
We begin with a lateral incision, starting 3 to 4 cm proximal to the lateral epicondyle and extending distally toward the radial neck. Dissect through the subcutaneous tissues, achieving meticulous hemostasis. Frequently, the traumatic energy has already violated the lateral capsule and avulsed the LUCL from the epicondyle. We exploit this traumatic window.
If the capsule is intact, we develop the Köcher interval between the anconeus and the ECU. To achieve adequate exposure of a coronal shear fracture, we must sharply elevate the common extensor origin (ECU, EDC, ECRL) off the anterior aspect of the lateral epicondyle. Reflecting this muscle mass anteriorly exposes the entire anterior radiocapitellar joint. Remember to keep the forearm fully pronated to protect the PIN during this anterior retraction. By applying a gentle varus stress to the elbow, the joint hinges open on the intact medial collateral ligament (MCL), providing a panoramic view of the articular surface.
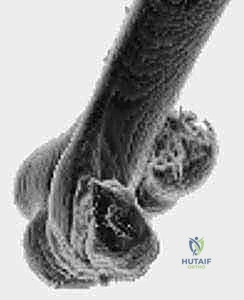
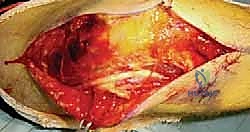
Debridement and Anatomic Reduction
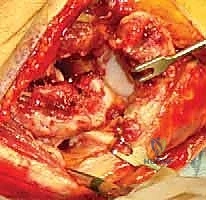
The joint is invariably filled with fracture hematoma and small, loose chondral fragments. Thoroughly irrigate the joint. The main capitellar fragment is usually found displaced proximally and rotated within the radial fossa.
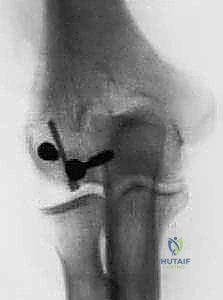
Using a fine dental pick or a Freer elevator, gently tease the fragment out of the fossa and manipulate it back into its anatomic bed. Reduction must be perfect; even a 1-mm step-off will lead to rapid joint destruction. Once reduced, provisionally secure the fragment using smooth 0.045-inch K-wires. Insert these wires strategically so they do not interfere with the planned trajectory of your definitive screws.
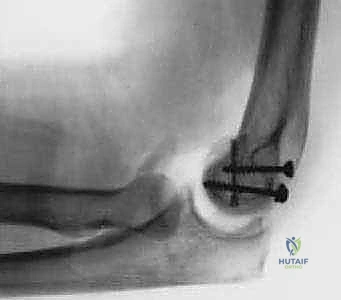
Definitive Internal Fixation
For Type 1 and simple Type 4 fractures without posterior comminution (Dubberley Type A), anterior-to-posterior fixation with headless compression screws is the gold standard.
Guide wires for the headless screws are placed perpendicular to the fracture plane, passing from the anterior articular surface of the capitellum into the intact posterior lateral column.
The screw lengths are measured, and the near cortex is overdrilled to allow the trailing head of the screw to bury completely beneath the hyaline cartilage. It is imperative that the screws are countersunk by at least 1 to 2 millimeters to prevent catastrophic abrasion against the radial head during postoperative motion.
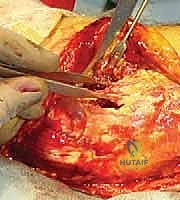
Managing Complex Coronal Shears and Posterior Comminution
When faced with a Dubberley Type B fracture featuring posterior condylar comminution, anterior-to-posterior screws alone are biomechanically insufficient. The anterior screws will simply pull the capitellum into the posterior void, resulting in malreduction and joint incongruity.
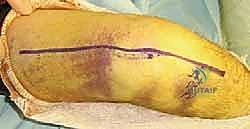
In these complex scenarios, the fixation strategy must be reversed or augmented. We must transition to a posterior-to-anterior fixation construct. This often requires extending the exposure posteriorly.
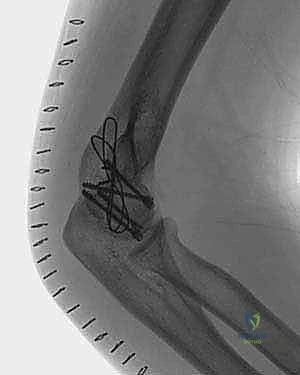
A mini-fragment plate (e.g., a 2.0mm or 2.4mm T-plate or straight plate) is applied to the posterior aspect of the lateral column, acting as an anti-glide buttress. Screws are then passed through the plate, from posterior to anterior, capturing the capitellar articular fragment and pulling it securely against the reconstructed posterior column.
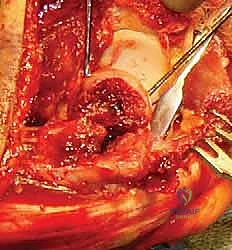
Once fixation is complete, the tourniquet is deflated, and hemostasis is achieved. The elbow is taken through a full range of motion under direct visualization and fluoroscopy to confirm absolute stability, absence of hardware impingement, and concentric reduction of the joint. Finally, the LUCL is rigorously repaired back to its isometric footprint on the lateral epicondyle using suture anchors, and the extensor origin is repaired.
Complications, Incidence Rates, and Salvage Management
Despite masterful surgical technique, complications following ORIF of capitellar shear fractures remain a significant clinical challenge due to the unforgiving nature of the elbow joint.
Post-traumatic stiffness is the most ubiquitous complication, affecting up to 40% of patients to some degree. The elbow capsule is highly reactive to trauma and surgical dissection, rapidly forming dense fibrotic adhesions. While a mild loss of terminal extension (10 to 15 degrees) is common and functionally well-tolerated, severe stiffness restricting the functional arc of motion (30 to 130 degrees) may require intervention. If aggressive physical therapy and static progressive splinting fail after 6 months, an open or arthroscopic capsular release (arthrolysis) may be indicated as a salvage procedure.
Avascular necrosis (AVN) of the capitellum occurs in approximately 10-15% of cases, primarily due to the disruption of the posterior intraosseous blood supply during the initial trauma or iatrogenic stripping. Interestingly, radiographic AVN does not always correlate with clinical failure. Many patients with fragmented, sclerotic capitella remain surprisingly asymptomatic. However, if AVN leads to structural collapse, severe pain, and arthrosis, salvage options include radial head excision, radiocapitellar arthroplasty, or total elbow arthroplasty in older, lower-demand patients.
Hardware failure and nonunion are less common (under 5%) but catastrophic when they occur. Backing out of headless compression screws can cause severe third-body wear on the radial head. This is usually the result of failing to adequately countersink the screws or failing to recognize posterior comminution, leading to construct failure. Revision ORIF with bone grafting is required for nonunions, often utilizing posterior plating constructs.
| Complication | Estimated Incidence | Primary Etiology | Salvage / Management Strategy |
|---|---|---|---|
| Post-Traumatic Stiffness | 30% - 40% | Capsular fibrosis; Prolonged immobilization | Static progressive splinting; Operative arthrolysis |
| Avascular Necrosis (AVN) | 10% - 15% | Disruption of posterior blood supply | Observation if painless; Arthroplasty if collapsed |
| Hardware Impingement | 5% - 10% | Inadequate screw countersinking | Immediate hardware removal; Chondroplasty |
| Nonunion / Failure | < 5% | Missed posterior comminution (Dubberley B) | Revision ORIF with structural bone graft and plating |
| Nerve Palsy (PIN or Ulnar) | 2% - 5% | Iatrogenic traction; Retractor placement | Observation (most neurapraxias resolve); Nerve exploration |
Phased Post-Operative Rehabilitation Protocols
The success of capitellar ORIF is equally dependent on the surgical construct and the postoperative rehabilitation protocol. The paradigm has shifted entirely away from prolonged casting toward immediate, controlled mobilization.
Phase 1: Tissue Healing and Early Motion (Days 0 to 14)
Postoperatively, the patient is placed in a bulky, well-padded posterior splint with the elbow at 90 degrees of flexion and the forearm in neutral rotation. This splint is removed within 3 to 5 days. Provided rigid internal fixation was achieved and the LUCL repair is secure, early active and active-assisted range of motion (ROM) is initiated immediately. Gravity-assisted extension and active flexion are encouraged. If the LUCL was repaired, varus stress and combined extension/supination must be strictly avoided to protect the ligamentous healing. An anterior extension splint is often fabricated for nighttime use to combat the inevitable flexion contracture.
Phase 2: Restoring the Functional Arc (Weeks 2 to 6)
Sutures are removed at 14 days. The focus shifts to aggressively restoring the functional arc of motion (30 to 130 degrees of flexion, 50 degrees of pronation/supination). Supervised physical therapy is critical. Edema control, scar massage, and continuous passive motion (CPM) machines may be utilized as adjuncts, though active motion remains the cornerstone. Lifting is restricted to less than 1 pound. Radiographs are obtained at 2 and 6 weeks to ensure maintenance of reduction and monitor for early signs of hardware migration.
Phase 3: Strengthening and Maturation (Weeks 6 to 12+)
By 6 weeks, early radiographic union is typically evident. We begin to wean the patient off any protective splinting. Isometric strengthening of the biceps, triceps, and forearm musculature is initiated, gradually progressing to isotonic resistance exercises. Dynamic or static progressive splinting may be introduced at 8 weeks if the patient has plateaued and significant stiffness remains. Patients must be counseled that maximal medical improvement (MMI)
