Ulnar Nerve Decompression at Guyon's Canal: An Intraoperative Masterclass
Key Takeaway
This masterclass details ulnar nerve decompression at Guyon's Canal, from intricate anatomy to real-time surgical execution. Fellows learn patient positioning, precise incision, meticulous neurovascular bundle dissection, and management of various compressive pathologies. We cover critical pearls, potential pitfalls, and comprehensive postoperative rehabilitation, ensuring optimal patient outcomes and surgical proficiency.
Comprehensive Introduction and Patho-Epidemiology
Ulnar nerve decompression at Guyon's canal represents a highly specialized surgical intervention that requires an intimate understanding of the complex neurovascular architecture of the volar-ulnar wrist. As orthopedic surgeons and hand specialists, we recognize that the ulnar nerve is the cornerstone of intrinsic hand function, governing the fine motor dexterity, powerful pinch, and grip strength that define human manipulation. When patients present with progressive ulnar nerve dysfunction, localizing the precise epicenter of compression is our foremost diagnostic imperative. While the cubital tunnel at the elbow remains the most ubiquitous site of entrapment, Guyon's canal at the wrist stands as the second most frequent anatomic bottleneck, presenting a unique set of diagnostic and surgical challenges that demand meticulous attention.
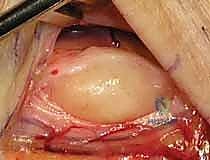
The pathogenesis of ulnar tunnel syndrome is remarkably diverse, distinguishing it from the more homogeneous etiology of carpal tunnel syndrome. Unlike the median nerve, which is most often compressed by idiopathic tenosynovitis or elevated compartmental pressures, the ulnar nerve within Guyon's canal is frequently compromised by discrete structural anomalies or space-occupying lesions. Soft tissue masses, particularly ganglion cysts originating from the pisotriquetral joint, lipomas, and benign peripheral nerve sheath tumors, account for a substantial 32% to 48% of all documented cases.
Another critical etiologic category involves vascular pathology, most notably hypothenar hammer syndrome. This condition arises from repeated blunt trauma to the hypothenar eminence—often seen in industrial workers who use the heel of their hand as a percussive tool—leading to thrombosis, intimal hyperplasia, or pseudoaneurysm of the ulnar artery. Because the ulnar artery shares the unyielding confines of Guyon's canal with the nerve, any vascular expansion or perivascular inflammation translates directly into compressive neuropathy.

Beyond structural masses and vascular insults, the operating surgeon must remain vigilant for anomalous muscular anatomy. Accessory muscle bellies, such as an aberrant abductor digiti minimi or a hypertrophied palmaris brevis, are implicated in approximately 16% of ulnar tunnel syndromes. Furthermore, osteological trauma, including fractures of the hook of the hamate or malunions of the distal radius, can distort the canal's architecture. Inflammatory diatheses like rheumatoid synovitis, gouty tophi, and iatrogenic causes following tendon transfers (e.g., Huber opponensplasty) further populate the differential diagnosis. Understanding this extensive patho-epidemiology is not merely an academic exercise; it dictates our preoperative imaging strategy and fundamentally alters our intraoperative approach, moving us from a generic "release" to a targeted extirpation of the offending pathology.
Detailed Surgical Anatomy and Biomechanics
The Architecture of Guyon's Canal
Guyon's canal, originally described by the French surgeon Jean Casimir Félix Guyon in 1861, is a complex, triangular, fibro-osseous conduit situated at the proximal, ulnar aspect of the palm. Measuring approximately 4 centimeters in length, it serves as the critical transition zone for the ulnar neurovascular bundle as it exits the distal forearm and arborizes into the hand. The canal begins proximally at the proximal edge of the palmar carpal ligament and extends distally to the fibrous aponeurotic edge of the hypothenar musculature. Understanding the rigid borders of this physiologic tunnel is paramount, as any volume increase within this unyielding space precipitously elevates interstitial pressure, precipitating ischemic neuropathy.
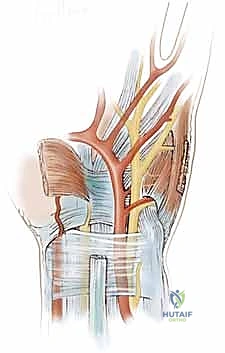
The boundaries of Guyon's canal are defined by robust osteoligamentous structures. The lateral (radial) wall is formed by the unyielding hook of the hamate and the dense transverse carpal ligament. The medial (ulnar) wall is composed of the pisiform bone and the tendinous insertions of the flexor carpi ulnaris, along with the pisohamate ligament. The floor of the canal is primarily constituted by the transverse carpal ligament and the pisohamate ligament, which provide a firm bed over which the nerve glides. Finally, the roof is formed by the palmar carpal ligament—a superficial thickening of the deep antebrachial fascia—and distally by the palmaris brevis muscle. It is crucial to note that the ulnar nerve is accompanied by the ulnar artery and its venae comitantes throughout this passage, situated just radial to the nerve.
Before the ulnar nerve even enters this confined space, it gives off a critical branch: the dorsal ulnar sensory branch. Arising approximately 5 to 8 centimeters proximal to the pisiform, this branch courses dorsally deep to the flexor carpi ulnaris to provide sensation to the dorsoulnar aspect of the hand and the dorsal surfaces of the small and ulnar half of the ring fingers. Because this bifurcation occurs proximal to Guyon's canal, a pure lesion within the canal will classically spare dorsal ulnar sensation. This is a vital clinical pearl; sensory deficits encompassing the dorsal hand strongly suggest a more proximal lesion, such as cubital tunnel syndrome or a cervical radiculopathy, rather than isolated ulnar tunnel syndrome.
The Zone System of the Ulnar Nerve
To systematically correlate clinical symptomatology with precise anatomic pathology, Guyon's canal is universally divided into three distinct zones, originally popularized by Shea and McClain. Zone 1 represents the most proximal segment of the canal, measuring approximately 3 centimeters in length. It encompasses the area proximal to the bifurcation of the main ulnar nerve trunk into its terminal motor and sensory branches. Compression within Zone 1 typically results in a combined motor and sensory deficit, as the nerve fibers are still bundled together. Pathologies frequently encountered in this zone include ganglion cysts dissecting from the pisotriquetral articulation, fractures of the hook of the hamate, and idiopathic thickening of the palmar carpal ligament at the canal's inlet.
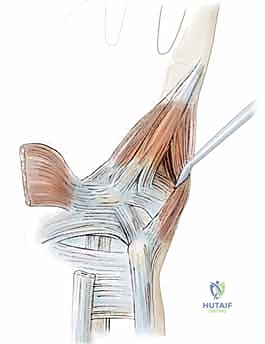
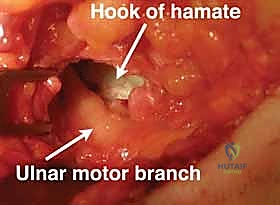
Zone 2 is the most intricate and clinically precarious region, encompassing the deep motor branch of the ulnar nerve. After bifurcating, this motor branch, accompanied by the deep palmar branch of the ulnar artery, dives dorsoradially. It courses through a tight fibrous hiatus between the abductor digiti quinti and the flexor digiti quinti brevis, eventually perforating the opponens digiti quinti. It then tracks along the deep volar arch to innervate the hypothenar muscles, all interossei, the third and fourth lumbricals, the adductor pollicis, and the deep head of the flexor pollicis brevis. Compression in Zone 2 manifests as pure motor weakness and intrinsic wasting, with complete sparing of palmar sensation.

Zone 3 is situated ulnar to Zone 2 and contains the superficial sensory branch of the ulnar nerve. This branch supplies motor innervation to the small palmaris brevis muscle before continuing distally to provide critical tactile sensation to the volar aspect of the hypothenar eminence, the entire small finger, and the ulnar half of the ring finger. Compression isolated to Zone 3 yields pure sensory deficits in this specific distribution, without intrinsic muscle atrophy or weakness. The most common etiologies for Zone 3 compression include ulnar artery thrombosis, pseudoaneurysms, and localized synovial inflammation. Understanding this zonal anatomy allows the surgeon to perform a targeted, symptom-directed exploration.
Nerve Anomalies and Diagnostic Confounders
The peripheral nervous system is notorious for its anatomic variability, and the upper extremity is no exception. When evaluating a patient for ulnar nerve decompression, the surgeon must be acutely aware of neural anastomoses that can confound both clinical examination and electrodiagnostic studies. The most prominent of these is the Martin-Gruber anastomosis, present in approximately 15% to 20% of the population. In this anomaly, motor fibers destined for the ulnar-innervated intrinsic hand muscles travel initially within the median nerve through the arm and proximal forearm, before crossing over to join the ulnar nerve in the mid-to-distal forearm. Consequently, a severe ulnar nerve injury proximal to this anastomosis (e.g., at the elbow) may present with paradoxically preserved intrinsic hand function, masking the severity of the proximal lesion.
Similarly, the Riche-Cannieu anastomosis occurs distally within the palm itself. This represents a neural communication between the deep motor branch of the ulnar nerve and the recurrent motor branch of the median nerve. When present, this anastomosis can result in the median nerve providing motor innervation to muscles traditionally supplied by the ulnar nerve (such as the first dorsal interosseous or adductor pollicis), or vice versa. In the setting of severe ulnar nerve compression at Guyon's canal, a Riche-Cannieu anastomosis might preserve significant pinch strength and intrinsic function, leading the unwary clinician to underestimate the degree of entrapment.
These anomalies underscore the necessity of a comprehensive, proximal-to-distal physical examination and the judicious use of electromyography and nerve conduction studies (EMG/NCS). The electromyographer must specifically test for these crossover anomalies to ensure accurate localization of the compressive lesion. Failure to recognize a Martin-Gruber or Riche-Cannieu anastomosis can lead to misdiagnosis, inappropriate surgical targeting, and ultimately, surgical failure. The master surgeon anticipates these variations and incorporates them into the preoperative planning and intraoperative decision-making process.
Exhaustive Indications and Contraindications
The decision to proceed with surgical decompression of the ulnar nerve at Guyon's canal must be predicated on a rigorous synthesis of clinical history, physical examination findings, and advanced diagnostic modalities. The primary indication for operative intervention is the failure of a comprehensive trial of conservative management. This trial typically includes activity modification, rigid wrist splinting in a neutral position to maximize canal volume, non-steroidal anti-inflammatory drugs (NSAIDs), and occupational therapy. When patients experience persistent, intractable pain, or refractory paresthesias despite these measures for a period of 6 to 12 weeks, surgical decompression is strongly warranted.
More urgent indications for surgery revolve around the presence of objective neurological deficits or identifiable space-occupying lesions. The onset of progressive motor weakness, manifested by a positive Froment's sign, Wartenberg's sign, or visible atrophy of the first dorsal interosseous and hypothenar musculature, dictates prompt surgical exploration to prevent irreversible motor endplate degradation. Similarly, if preoperative imaging (MRI or high-resolution ultrasound) reveals a discrete mass—such as a ganglion cyst, lipoma, or anomalous muscle belly—surgical excision combined with nerve decompression is the definitive standard of care. Furthermore, vascular etiologies like ulnar artery thrombosis or pseudoaneurysm require immediate surgical attention to restore perfusion and relieve secondary neural compression.
Conversely, there are strict contraindications that the surgeon must respect to avoid catastrophic outcomes or failure to relieve symptoms. The most absolute contraindication is an incorrect diagnosis, specifically when symptoms are entirely attributable to a more proximal pathology, such as cervical radiculopathy (C8-T1), thoracic outlet syndrome, or severe cubital tunnel syndrome, without a concomitant distal compression. Operating on the wrist when the primary lesion is at the cervical root will yield no clinical benefit. Relative contraindications include severe, unoptimized medical comorbidities that preclude safe anesthesia, or advanced, long-standing neuropathy where the intrinsic musculature has undergone complete fatty infiltration and fibrosis, rendering the potential for motor recovery negligible.
| Category | Indications for Surgical Decompression | Contraindications (Absolute and Relative) | Clinical Rationale |
|---|---|---|---|
| Neurological | Progressive intrinsic muscle weakness/atrophy; Refractory sensory deficits despite 3 months of conservative care. | Purely proximal lesions (Cervical radiculopathy, Thoracic Outlet) without distal involvement. | Decompressing the wrist in the presence of a purely proximal lesion will not alleviate symptoms. Early intervention for motor weakness prevents irreversible muscle fibrosis. |
| Structural | MRI/Ultrasound confirmed space-occupying lesions (ganglion, lipoma, tumor); Anomalous musculature causing mechanical block. | Advanced, irreversible fatty atrophy of intrinsic muscles (Relative contraindication). | Structural lesions require physical extirpation. If muscles are completely fibrosed, motor recovery is impossible, though surgery may still be offered for pain relief. |
| Vascular | Ulnar artery thrombosis; Pseudoaneurysm (Hypothenar Hammer Syndrome) causing secondary nerve compression. | Active, untreated systemic vasculitis or severe coagulopathy precluding safe dissection. | Vascular lesions in the canal cause rapid, severe ischemic neuropathy and require immediate exploration, potential arterial reconstruction, and nerve release. |
| Traumatic | Acute fracture of the hook of the hamate with acute ulnar neuropathy; Compartment syndrome of the hand/wrist. | Active, uncontrolled local infection at the surgical site. | Traumatic impingement requires immediate skeletal stabilization/excision and nerve decompression to prevent permanent axonotmesis. |
Pre-Operative Planning, Templating, and Patient Positioning
Clinical Evaluation and Diagnostic Modalities
A masterful surgical outcome begins long before the first incision, rooted in a fastidious preoperative evaluation. The physical examination must follow a strict proximal-to-distal algorithm to rule out the "double-crush" phenomenon, where a proximal nerve irritation renders the distal nerve more susceptible to compression. The surgeon must evaluate the cervical spine for range of motion and radicular signs, palpate the supraclavicular fossa for thoracic outlet involvement, and perform Tinel's testing at the cubital tunnel. At the wrist, a positive Tinel's sign over Guyon's canal is highly suggestive. Motor testing must isolate the ulnar-innervated intrinsics: assessing pinch strength (Froment's sign), small finger abduction (Wartenberg's sign), and the ability to cross the index and middle fingers.
Diagnostic imaging is non-negotiable in the workup of ulnar tunnel syndrome, given the high prevalence of structural etiologies. High-resolution diagnostic ultrasound is an excellent, cost-effective first-line modality to assess nerve cross-sectional area, identify ganglion cysts, and evaluate ulnar artery patency dynamically. However, Magnetic Resonance Imaging (MRI) without and with intravenous contrast remains the gold standard. MRI provides unparalleled soft-tissue resolution, allowing the surgeon to precisely delineate the size, origin, and extent of tumors, lipomas, or anomalous muscles, and to evaluate the intrinsic muscles for edema (an early sign of denervation) or fatty infiltration (a late sign).
Electromyography and Nerve Conduction Studies (EMG/NCS) are critical adjuncts. They not only confirm the diagnosis and localize the lesion to the wrist (distinguishing it from cubital tunnel syndrome) but also quantify the severity of axonal loss and demyelination. The electrophysiologist must specifically evaluate the dorsal ulnar sensory branch; normal conduction in this branch coupled with delayed conduction across the wrist confirms a lesion within Guyon's canal. Finally, if vascular pathology is suspected (e.g., hypothenar hammer syndrome), an Allen test followed by a dedicated MR Angiography or Doppler ultrasound is mandatory to map the palmar arch collateral circulation before any surgical manipulation of the ulnar artery.
Operative Setup and Anesthesia
The operative setup for ulnar nerve decompression requires a meticulously organized environment to facilitate delicate neurovascular dissection. The procedure is typically performed on an outpatient basis. While general anesthesia or a regional brachial plexus block (supraclavicular or axillary) can be utilized, many master surgeons prefer wide-awake local anesthesia no tourniquet (WALANT) or a regional block with a well-padded proximal pneumatic tourniquet. If a tourniquet is used, it should be inflated to 250 mm Hg after exsanguination with an Esmarch bandage to provide a bloodless surgical field, which is absolutely critical for identifying microvascular structures and small nerve fascicles.
The patient is positioned supine on the operating table with the operative extremity extended onto a radiolucent hand table. The shoulder is abducted to 90 degrees, and the forearm is supinated. A rolled towel may be placed under the dorsal wrist to provide mild extension, thereby bringing the volar structures into greater relief. The surgeon sits seated in the axilla, with the surgical assistant positioned opposite. High-quality surgical loupes (minimum 3.5x magnification, though 4.5x is preferred by many for the deep motor branch dissection) and a fiber-optic headlight are indispensable tools for this procedure.
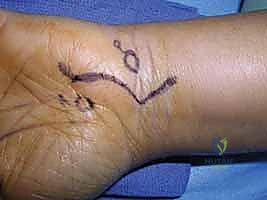
Preoperative templating involves marking the surgical landmarks with a sterile surgical pen before exsanguination. The pisiform, the hook of the hamate, and the distal wrist crease are palpated and marked. The planned incision is drawn, ensuring it crosses the wrist flexion creases at an oblique angle to prevent postoperative scar contracture. The surgeon must mentally rehearse the three-dimensional anatomy of the canal, anticipating the location of the bifurcation and the deep motor branch based on the preoperative MRI findings. This mental templating minimizes intraoperative hesitation and ensures a fluid, precise surgical execution.
Step-by-Step Surgical Approach and Fixation Technique
Incision Design and Superficial Dissection
The surgical approach to Guyon's canal demands precision to prevent iatrogenic injury to superficial sensory nerves and to ensure adequate exposure of all three zones. The incision typically begins 2 to 3 centimeters proximal to the distal wrist flexion crease, positioned directly over the flexor carpi ulnaris (FCU) tendon. It extends distally in a curvilinear or zig-zag fashion, crossing the wrist crease obliquely to avoid a longitudinal scar that could lead to a restrictive flexion contracture. The incision then curves radially around the base of the hypothenar eminence, terminating near the mid-palmar crease, aligning with the longitudinal axis of the fourth ray.
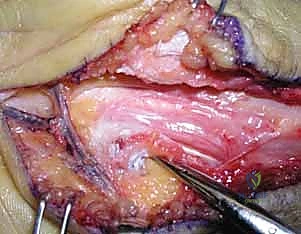
Upon incising the epidermis and dermis, the surgeon encounters the superficial palmar fascia and the subcutaneous fat. Dissection here must proceed with extreme caution using tenotomy scissors or a fine hemostat. The palmar cutaneous branch of the ulnar nerve, which supplies sensation to the proximal ulnar palm, is highly variable and can be easily transected if the dissection is overly aggressive. Furthermore, the surgeon must be cognizant of the dorsal ulnar sensory branch, which, although typically branching proximal to the wrist, can occasionally have an anomalous distal takeoff. Meticulous hemostasis is maintained using bipolar electrocautery to keep the field pristine.
The superficial dissection concludes with the identification of the palmaris brevis muscle distally and the superficial fascia of the distal forearm proximally. The FCU tendon is identified and retracted ulnarly. Just radial and deep to the FCU, the ulnar neurovascular bundle is located. The ulnar artery, usually situated radial to the nerve, serves as an excellent landmark. At this stage, the surgeon has exposed the proximal entrance to Guyon's canal, defined by the proximal edge of the palmar carpal ligament, setting the stage for the deep decompression.
Deep Dissection and Canal Release
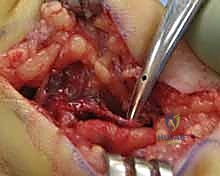
The deep dissection begins with the systematic release of the roof of Guyon's canal. Using a combination of blunt and sharp dissection, the palmar carpal ligament is incised longitudinally from proximal to distal. It is imperative to keep the tips of the scissors directly superficial to the nerve to avoid inadvertent injury to the underlying epineurium or the accompanying ulnar artery. As the ligament is divided, the ulnar nerve and artery are visualized traversing Zone 1. The nerve is gently mobilized using vessel loops, taking care not to apply excessive traction which could cause a neuropraxic injury.
The dissection then proceeds distally into the palm. The palmaris brevis muscle is carefully split longitudinally or retracted ulnarly to expose the bifurcation of the ulnar nerve into its superficial sensory (Zone 3) and deep motor (Zone 2) branches. The superficial sensory branch is traced distally to ensure no fascial bands are compressing it against the hypothenar fascia. The most critical and technically demanding portion of the procedure is the decompression of the deep motor branch in Zone 2. This branch dives deeply between the abductor digiti minimi and the flexor digiti minimi.
The surgeon must identify the fibrous leading edge of the hypothenar muscles (the pisohamate hiatus) that arches over the deep motor branch. This fibrous arch must be completely released. Using fine tenotomy scissors, the fascia is divided while directly visualizing the motor branch as it courses around the hook of the hamate. The proximity of the motor branch to the hamate hook makes it highly vulnerable; the surgeon must maintain a clear visual field and avoid blind cutting. The release is considered complete only when the motor branch can be seen diving freely into the deep palmar space without any fascial tethering.
Management of Specific Pathologies
Once the canal is fully unroofed and the nerve branches are mobilized, the surgeon must address the specific underlying pathology identified during the preoperative workup. If a ganglion cyst is present, typically arising from the pisotriquetral joint, it must be meticulously dissected free from the nerve. The stalk of the ganglion must be traced down to the joint capsule and excised completely, often requiring a small capsulotomy to prevent recurrence. Lipomas or anomalous muscle bellies (e.g., accessory abductor digiti minimi) are similarly excised, ensuring that no residual mass effect remains within the canal.
In cases of hypothenar hammer syndrome or ulnar artery thrombosis, the management of the vascular bundle dictates the flow of the operation. If a thrombosed segment or pseudoaneurysm is identified, vascular control is obtained proximally and distally. Depending on the extent of the lesion and the adequacy of the collateral circulation (as determined by preoperative angiography and intraoperative assessment), the diseased arterial segment may simply be ligated and resected, relieving the pressure on the adjacent nerve.
If collateral flow is insufficient, an arterial reconstruction using a reverse saphenous vein graft or primary end-to-end anastomosis (if tension-free) must be performed under operating microscope magnification. Regardless of the specific pathology, the final step before closure involves a thorough inspection of the nerve to ensure complete hemostasis and to verify that all compressive elements across Zones 1, 2, and 3 have been eradicated. The wound is then irrigated, and the skin is closed with non-absorbable monofilament sutures; the deep ligaments are intentionally left open to prevent recurrent compression.
Complications, Incidence Rates, and Salvage Management
Despite the high success rates of ulnar nerve decompression at Guyon's canal, complications can and do occur, often stemming from technical errors or anatomic misjudgments. The most common cause of surgical failure is an incomplete release, specifically the failure to adequately decompress the deep motor branch in Zone 2. The fibrous arch of the hypothenar muscles can be dense and difficult to visualize; if not completely divided, the patient will continue to experience intrinsic weakness and cramping. Revision surgery in these cases is fraught with difficulty due to scar tissue, requiring meticulous neurolysis and a high degree of microsurgical skill.
Iatrogenic nerve injury is a catastrophic complication that demands immediate recognition. The deep motor branch is particularly susceptible to transection or crush injury as it rounds the hook of the hamate, especially during the excision of hamate hook fractures or deep ganglion cysts. Similarly, the palmar cutaneous branch or the dorsal ulnar sensory branch can be injured during the superficial exposure, leading to the formation of a painful neuroma. Neuromas in this region are notoriously difficult to treat and often result in chronic, debilitating pain that overshadows the original compressive symptoms.
Vascular complications, including uncontrolled hemorrhage or secondary thrombosis of the ulnar artery, can occur if the vessel is manipulated roughly during dissection. Complex Regional Pain Syndrome (CRPS) is another potential postoperative sequela, characterized by disproportionate pain, swelling, and autonomic dysfunction. Prevention of CRPS relies on meticulous surgical technique, adequate postoperative pain control, and early, supervised mobilization. Should complications arise, salvage management requires a multidisciplinary approach, often involving revision neurolysis, nerve grafting for transections, or targeted neuroma management (e.g., targeted muscle reinnervation or relocation into bone/muscle).
| Complication | Estimated Incidence Rate | Prevention Strategy | Salvage Management |
|---|---|---|---|
| Incomplete Release (Zone 2) | 3% - 5% | Direct visualization of the deep motor branch diving into the deep palmar space; complete division of the pisohamate hiatus. | Revision exploration with neurolysis under microscopic magnification; ensuring all fascial bands are resected. |
| Iatrogenic Nerve Injury (Motor Branch) | < 1% | Avoid blind cutting near the hook of the hamate; use 3.5x to 4.5x loupe magnification; meticulous hemostasis. | Immediate primary microsurgical repair. If discovered late, nerve grafting or tendon transfers (e.g., for pinch/clawing) are required. |
| Painful Neuroma (Sensory Branches) | 1% - 2% | Careful superficial dissection; avoid aggressive retraction of skin flaps; identify and protect palmar cutaneous branches. | Excision of the neuroma and relocation of the proximal nerve stump into deep muscle (e.g., pronator quadratus) or bone. |
| Complex Regional Pain Syndrome (CRPS) | 2% - 4% | Gentle tissue handling; avoid excessive tourniquet times; early postoperative digit mobilization; adequate analgesia. | Aggressive hand therapy; sympathetic nerve blocks |
