Comprehensive Management of Charcot Arthropathy: Operative and Nonoperative Strategies
Key Takeaway
Charcot arthropathy management requires a multidisciplinary approach. Nonoperative treatment centers on total contact casting and protected weight-bearing to achieve consolidation. Operative intervention, indicated in approximately 25% of cases, focuses on deformity correction, stabilization, and ulcer prevention. Surgical strategies range from exostectomy and Achilles tendon lengthening to complex midfoot arthrodesis and tibiotalocalcaneal fusion, aiming to create a stable, plantigrade, and braceable foot while avoiding amputation.
Comprehensive Introduction and Patho-Epidemiology
The management of Charcot neuroarthropathy (CN) represents one of the most formidable, complex, and resource-intensive challenges in orthopedic foot and ankle surgery. Characterized by progressive, relentless joint destruction, subluxation, fragmentation, and eventual profound deformity in a neuropathic patient, the condition demands a rigorous, stage-specific therapeutic approach. The overarching goal of treatment—whether nonoperative or operative—is to achieve a stable, plantigrade, and braceable foot, thereby preventing recalcitrant ulceration, deep soft-tissue infection, osteomyelitis, and the ultimate endpoint of major lower extremity amputation. Understanding this pathology requires a deep appreciation of its etiology, which is intimately tied to profound peripheral neuropathy, most commonly secondary to long-standing diabetes mellitus, though neurosyphilis, syringomyelia, and chronic alcoholism remain well-documented causes.
The pathophysiology of Charcot arthropathy is classically described by two competing but ultimately synergistic theories: the neurotraumatic (German) theory and the neurovascular (French) theory. The neurotraumatic theory posits that a loss of protective sensation (LOPS) and proprioception allows for repetitive, unrecognized microtrauma to the foot and ankle. Without the protective pain feedback loop, the patient continues to bear weight on microfractures and ligamentous sprains, leading to progressive joint subluxation and catastrophic osseous collapse. Conversely, the neurovascular theory suggests that an autonomic neuropathy leads to a loss of sympathetic tone in the lower extremity. This autonomic failure induces profound arteriovenous shunting and localized hyperemia. The resultant hyperdynamic blood flow washes out bone mineral density, leading to localized osteopenia and rendering the osseous structures highly susceptible to fracture under normal physiological loads.
Modern molecular medicine has synthesized these historical theories into a unified inflammatory pathway centered on the Receptor Activator of Nuclear Factor Kappa-B (RANK), its ligand (RANKL), and Osteoprotegerin (OPG). In the acute phase of Charcot arthropathy, repetitive trauma in the setting of neuropathy triggers a massive release of pro-inflammatory cytokines, including Interleukin-1 (IL-1), Interleukin-6 (IL-6), and Tumor Necrosis Factor-alpha (TNF-α). These cytokines upregulate the expression of RANKL, which binds to RANK receptors on osteoclast precursors, driving aggressive osteoclastic differentiation and monumental bone resorption. The localized hyperemia further delivers a continuous supply of osteoclast precursors to the site of injury. Consequently, the bone becomes structurally incompetent, leading to the dramatic fragmentation and periarticular debris characteristic of Eichenholtz Stage I disease.
Epidemiologically, the incidence of Charcot arthropathy in the general diabetic population is estimated between 0.1% and 0.9%, but this figure skyrockets to nearly 13% in high-risk diabetic clinics treating patients with established peripheral neuropathy. The condition is bilateral in up to 30% of cases, either sequentially or simultaneously, necessitating vigilant longitudinal surveillance of the contralateral limb. The mortality rate associated with Charcot arthropathy is alarmingly high, primarily driven by the systemic cardiovascular complications of long-standing diabetes and the devastating physiologic toll of deep infection and amputation. Patients who develop a Charcot-related ulceration face a 5-year mortality rate that rivals or exceeds many common malignancies, underscoring the absolute necessity for aggressive, limb-salvaging interventions by the orthopedic surgeon.

Detailed Surgical Anatomy and Biomechanics
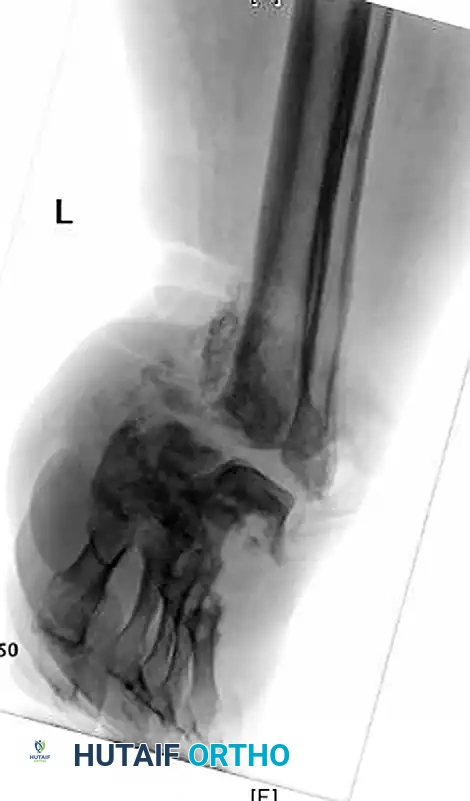
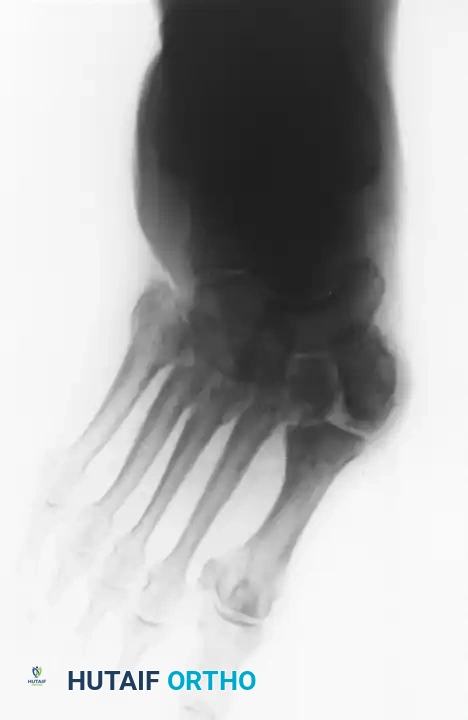
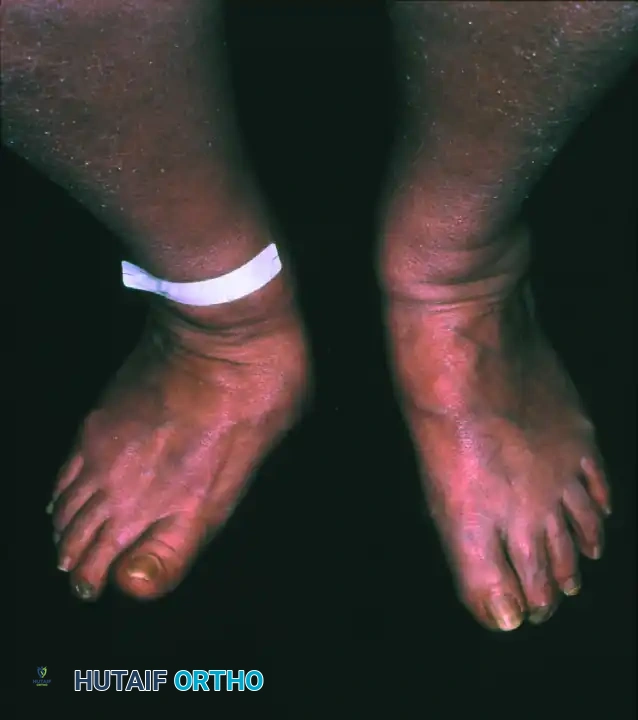
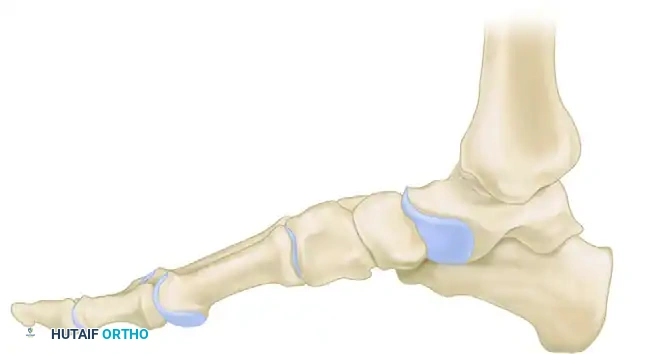
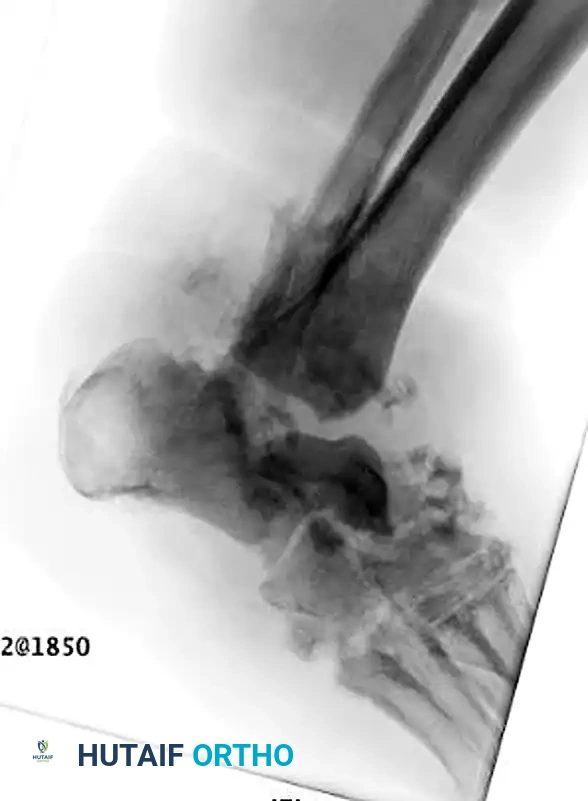
A profound understanding of the surgical anatomy and the biomechanical forces driving deformity is an absolute prerequisite for successfully reconstructing the Charcot foot. The anatomic location of the deformity heavily dictates the surgical approach, the choice of fixation, and the ultimate prognosis. The Brodsky classification, later modified by Trepman, remains the most clinically relevant anatomic staging system. Type 1 involves the tarsometatarsal (Lisfranc) and naviculocuneiform joints, representing the most common presentation (approximately 60% of cases). Type 2 involves the hindfoot joints (subtalar, talonavicular, and calcaneocuboid). Type 3 involves the tibiotalar (ankle) joint and carries the highest risk of rapid, unbraceable collapse. Type 4 involves multiple regions, and Type 5 involves the forefoot (metatarsophalangeal joints).
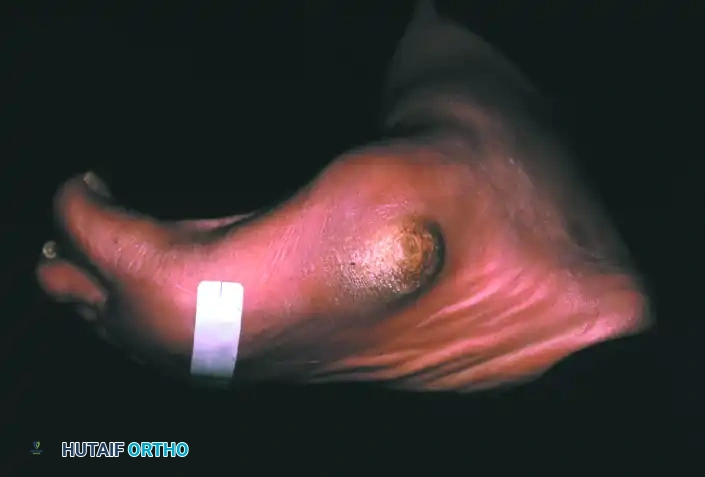
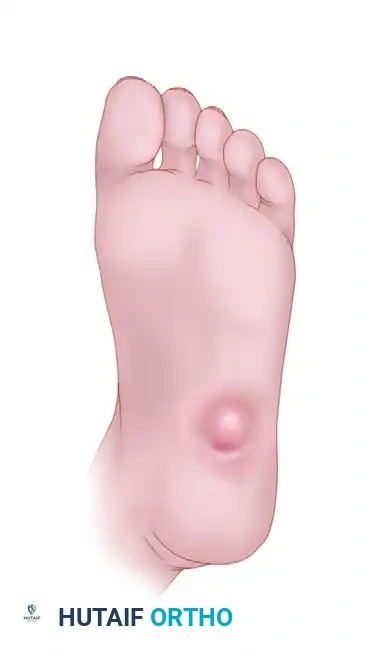
The biomechanical collapse in a Type 1 midfoot Charcot event is almost universally driven by an underlying equinus contracture. Autonomic neuropathy leads to glycosylation of the Achilles tendon and gastrocnemius-soleus complex, rendering these structures rigid and inelastic. During the terminal stance phase of the gait cycle, the inability of the ankle to dorsiflex forces the tremendous load of the body weight anteriorly onto the midfoot joints. In a healthy foot, the plantar fascia, spring ligament, and interlocking keystone anatomy of the midfoot bones resist these forces. However, in the hyperemic, osteopenic Charcot foot, these stabilizing structures fail. The midfoot collapses into abduction and plantarflexion, creating the classic "rocker-bottom" deformity. The cuboid or medial cuneiform is driven plantarly, becoming a massive bony prominence that directly compresses the overlying skin from the inside out, leading to highly recalcitrant ulcerations.
Surgical anatomy in the neuropathic foot is further complicated by the critical concept of angiosomes. The foot is supplied by distinct three-dimensional vascular territories fed by specific source arteries (the posterior tibial, anterior tibial, and peroneal arteries). In a patient with microvascular disease, the choke vessels connecting these angiosomes are often non-functional. Therefore, surgical incisions must be meticulously planned to respect these vascular boundaries. For example, an extensile medial utility incision utilizes the territory of the medial plantar artery. If a concomitant lateral incision is required, an adequate skin bridge must be maintained to prevent catastrophic flap necrosis. The surgeon must operate under the assumption that the soft tissue envelope is profoundly ischemic at the microvascular level, even in the presence of palpable pedal pulses.
Biomechanical principles of internal fixation in the Charcot foot differ radically from standard orthopedic trauma. The bone is severely osteopenic, and the healing potential is drastically reduced. Standard plates and screws applied in a conventional manner will inevitably fail. The surgeon must apply fixation based on tension band principles. The plantar aspect of the foot represents the tension side during weight-bearing, while the dorsal aspect represents the compression side. Therefore, whenever anatomically feasible, hardware should be applied to the plantar surface to resist the massive deforming forces of the Achilles tendon and body weight. When plantar plating is not possible, intramedullary "beaming" techniques must be utilized to create an internal load-sharing splint that spans across multiple joints, bypassing the focal area of osteopenic collapse and anchoring into healthy, dense bone proximally and distally.
Exhaustive Indications and Contraindications
The decision-making algorithm for intervening in Charcot arthropathy is complex, requiring a delicate balance between the risks of surgical reconstruction and the natural history of the disease. Nonoperative management remains the gold standard for acute Eichenholtz Stage I disease and is successful in the majority of patients when initiated promptly. Total contact casting (TCC) with protected weight-bearing aims to arrest the inflammatory cycle, prevent further mechanical destruction, and protect the soft-tissue envelope. However, when conservative measures fail or the deformity becomes fundamentally unbraceable, operative intervention transitions from an elective procedure to mandatory limb salvage surgery.
Historically, surgery was strictly deferred until the coalescence or consolidation phases (Eichenholtz II or III) due to the profound hyperemia, massive edema, and severely osteopenic bone of the acute phase that provides poor purchase for internal fixation. However, a paradigm shift has occurred in modern orthopedic practice. With the advent of stronger fixation techniques ("superconstructs") and advanced external fixation, a more aggressive approach is now justified. Operating during the fragmentation phase may be necessary for severe, unbraceable instability, acute dislocation with soft-tissue compromise, or concurrent deep infection. The primary indications for surgery include severe instability that cannot be controlled with immobilization, recurrent ulceration caused by a rigid deformity in the consolidated phase, deep infection or osteomyelitis requiring surgical debridement, and acute, highly unstable fracture-dislocations.
Contraindications to surgical reconstruction are equally critical to recognize, as operating on an inappropriate candidate can rapidly accelerate the path to amputation. Absolute contraindications include profound, uncorrectable peripheral arterial ischemia, active systemic sepsis, and severe medical comorbidities that preclude safe anesthesia. Relative contraindications include profound patient non-compliance, active smoking, and severe malnutrition. In patients with inadequate vascular runoff, a formal vascular surgery consultation is mandatory, and revascularization (endovascular or open bypass) must precede any orthopedic reconstruction. If the limb cannot be revascularized, major amputation is often the only viable option.
Summary of Indications and Contraindications
| Procedure Category | Primary Indications | Absolute Contraindications | Relative Contraindications |
|---|---|---|---|
| Exostectomy | Consolidated (Stage III) deformity; Recurrent focal ulceration; Stable midfoot architecture. | Acute fragmentation (Stage I); Gross clinical instability; Active osteomyelitis in adjacent joints. | Poor soft-tissue envelope over the prominence; Inadequate vascular perfusion. |
| Internal Arthrodesis (Superconstructs) | Severe, unbraceable instability; Failed conservative management; Impending ulceration from progressive collapse. | Active deep infection/osteomyelitis; Severe peripheral arterial disease; Inadequate bone stock for fixation. | Active smoking; HbA1c > 10%; Documented profound patient non-compliance. |
| External Fixation (Ilizarov/TSF) | Active osteomyelitis requiring resection; Massive soft-tissue defects; Poor bone quality precluding internal fixation. | Inability to tolerate frame care; Severe proximal joint disease; Uncorrectable ischemia. | Severe psychiatric illness; Lack of social support for pin care. |
| Major Amputation (BKA/AKA) | Ascending necrotizing infection; Unsalvageable ischemia; Massive bone loss precluding reconstruction; Failed multiple limb salvage attempts. | Medically unstable for surgery (requires optimization); Salvageable limb with functional potential. | Patient refusal (requires extensive counseling and shared decision making). |

Pre-Operative Planning, Templating, and Patient Positioning
Pre-operative planning for Charcot reconstruction is a meticulous process that begins long before the patient enters the operating theater. Comprehensive medical optimization is mandatory to mitigate the exceptionally high risk of postoperative complications. Glycemic control is paramount; an HbA1c level below 8% is highly desirable, as hyperglycemia impairs leukocyte function and drastically increases the risk of surgical site infection. Nutritional status must be evaluated via serum albumin (target > 3.5 g/dL) and total lymphocyte count (target > 1500/mm³). A formal vascular assessment is non-negotiable. Non-invasive arterial studies, including Ankle-Brachial Indices (ABI), Toe-Brachial Indices (TBI), and Doppler waveforms, must confirm adequate perfusion. A TBI greater than 0.6 or a transcutaneous oxygen pressure (TcPO2) greater than 40 mmHg is generally required for predictable wound healing.
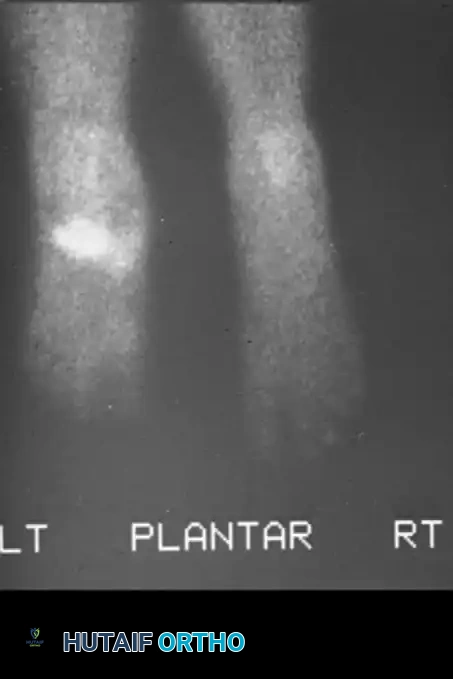
Advanced imaging is the cornerstone of surgical templating. While initial weight-bearing radiographs provide a baseline assessment of the deformity, a multi-planar Computed Tomography (CT) scan is essential for evaluating bone stock, identifying areas of precise fragmentation, and planning the trajectory of large-diameter beaming screws. Magnetic Resonance Imaging (MRI) with and without intravenous contrast is heavily utilized to differentiate between acute Charcot neuroarthropathy (bone marrow edema, periarticular fluid) and concomitant osteomyelitis (sinus tracts, cortical destruction, rim-enhancing fluid collections). When planning for a "superconstruct," the surgeon must template the fixation to extend well beyond the zone of injury. For a midfoot collapse, this often means planning a medial column beam that originates in the posterior talus, traverses the navicular and medial cuneiform, and terminates in the diaphyseal bone of the first metatarsal.
Patient positioning is dictated by the specific anatomic location of the deformity and the planned surgical approach. For midfoot reconstructions, the patient is typically positioned supine with a bump under the ipsilateral hip to internally rotate the leg, bringing the medial column into a neutral, accessible position. A radiolucent table is absolutely critical, as intraoperative fluoroscopy is used extensively to guide deformity correction and hardware placement. For hindfoot or ankle reconstructions requiring a posterior approach or an Achilles tendon lengthening, the patient may be positioned prone or in a floppy lateral decubitus position.
The use of a pneumatic tourniquet in the diabetic neuropathic patient is highly controversial. While a tourniquet provides a bloodless field and facilitates complex dissection, it poses a significant risk of inducing ischemic injury to already compromised microvasculature. Furthermore, tourniquet inflation can cause severe calcified vessels to rupture or thrombose. Many experienced limb salvage surgeons advocate for a tourniquet-less approach, utilizing meticulous hemostasis and bipolar electrocautery to minimize tissue trauma. If a tourniquet is used, the pressure should be set to the lowest effective level (typically 100 mmHg above systolic pressure) and inflation time strictly limited to less than 90 minutes.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution of Charcot reconstruction demands strict adherence to the principles of "superconstructs," a term popularized by Sammarco to describe fixation techniques robust enough to withstand the unique biomechanical demands of the neuropathic foot. These principles mandate that fusion must extend beyond the zone of injury to include normal, healthy bone; bone resection must be adequate to correct the deformity without placing the soft-tissue envelope under tension; the strongest possible fixation devices must be utilized; and hardware should be applied to the tension side of the deformity.
Exostectomy (Ostectomy) Procedures
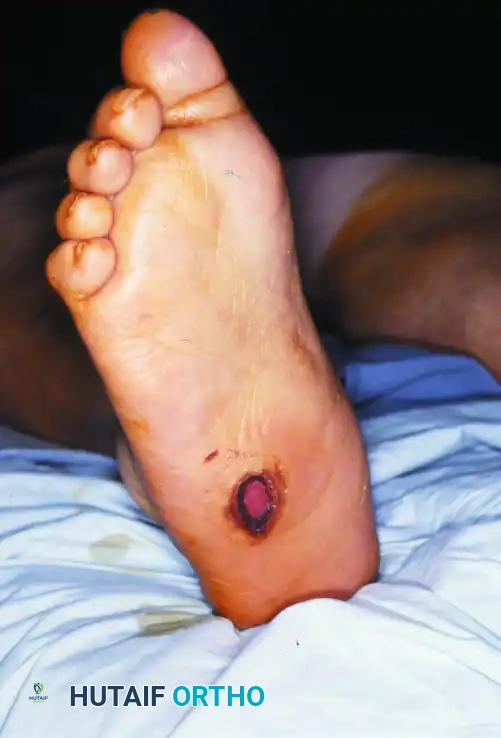
Exostectomy is reserved for stable, consolidated deformities (Eichenholtz Stage III) where a focal bony prominence causes recurrent ulceration. The surgical approach must be meticulously planned. Incisions should ideally be placed away from the ulcer bed, utilizing healthy, non-irradiated skin. The incision is made full-thickness straight down to the bone; undermining of the superficial soft tissue must be strictly avoided to preserve the fragile subdermal vascular plexus. The bony prominence, typically the plantar aspect of the medial cuneiform or cuboid, is exposed circumferentially. Using a broad osteotome or an oscillating saw, the prominence is resected flush with the surrounding contour of the foot. It is imperative to protect or reattach important tendinous insertions, such as the tibialis anterior or peroneus longus, using suture anchors if their footprint is compromised. An Achilles tendon lengthening (ATL) or gastrocnemius recession is almost universally performed concomitantly to address the underlying equinus contracture and permanently reduce forefoot pressures.
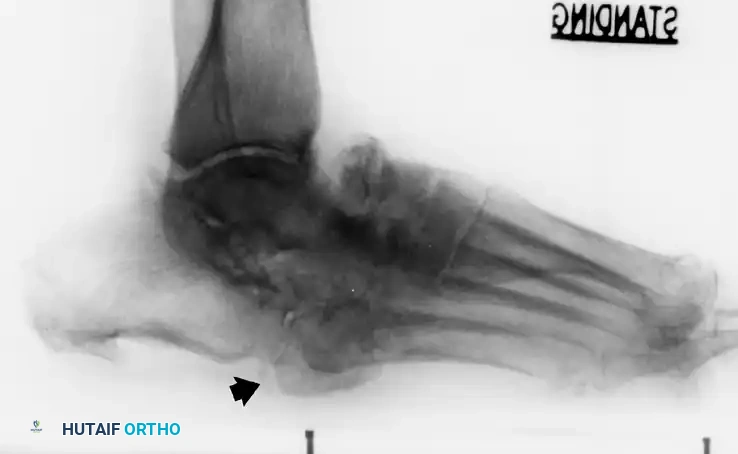
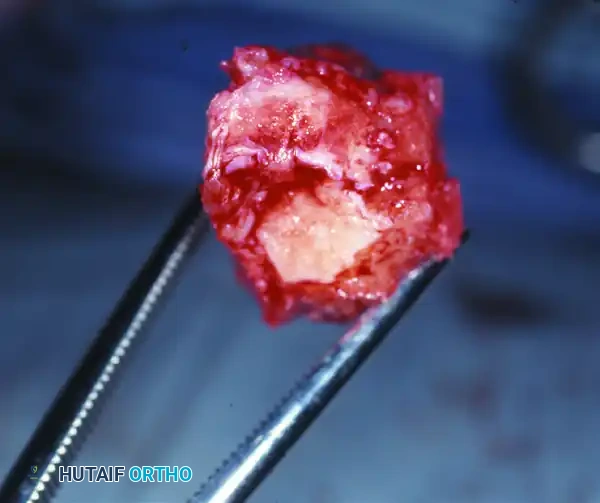
Midfoot Arthrodesis and Beaming
For unstable Type 1 midfoot Charcot, complex arthrodesis is required. A dual-incision approach is commonly utilized: an extensile medial utility incision to access the medial column (talonavicular, naviculocuneiform, and first tarsometatarsal joints) and a dorsolateral incision to access the lateral column (calcaneocuboid and 4th/5th tarsometatarsal joints). The joints are aggressively debrided of all cartilage, fibrous tissue, and sclerotic bone down to bleeding subchondral bone. Biplanar wedges of bone are often resected to restore a plantigrade alignment.
Fixation frequently relies on intramedullary beaming. For the medial column, a guidewire is driven from the posterior aspect of the talus, across the navicular and medial cuneiform, and into the medullary canal of the first metatarsal. After sequential over-reaming, a large-diameter (7.0 mm to 8.0 mm) solid or cannulated intramedullary screw is inserted. This beam acts as an internal splint, sharing the load across the entire medial column. The lateral column is similarly beamed, typically from the posterior calcaneus through the cuboid and into the fourth metatarsal base. To augment rotational stability and apply tension band principles, robust plantar plating may be added. The fusion sites are densely packed with autograft, allograft, or bone marrow aspirate concentrate (BMAC) to stimulate osteogenesis in the hostile neuropathic environment.
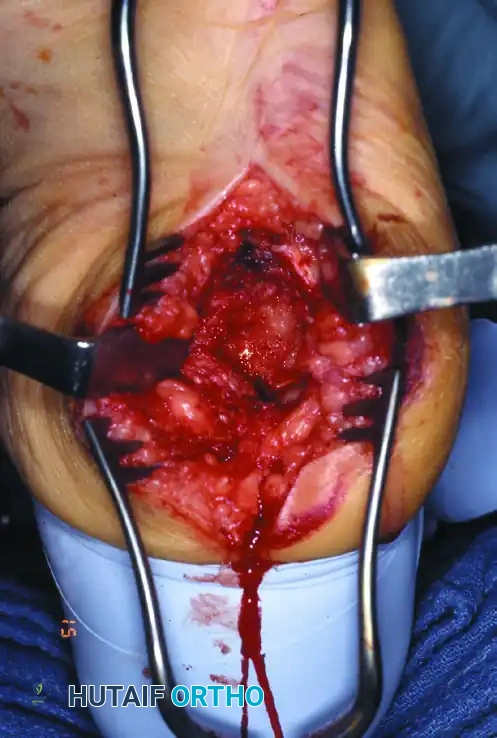
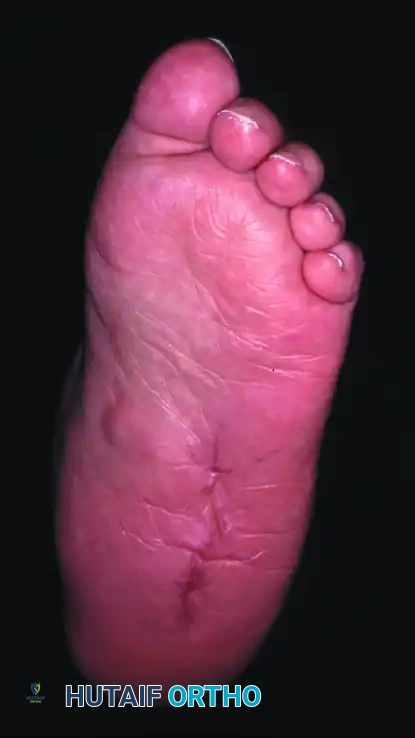
Hindfoot and Ankle Reconstruction
Type 2 and Type 3 Charcot deformities involving the hindfoot and ankle are highly unstable and prone to rapid varus or valgus collapse. Tibiotalocalcaneal (TTC) arthrodesis utilizing a retrograde intramedullary nail is the workhorse procedure. The ankle and subtalar joints are approached laterally via a transfibular approach or anteriorly. After aggressive joint preparation and deformity correction, a heavy guidewire is passed from the plantar aspect of the calcaneus, through the talus, and into the tibial diaphysis. The canal is reamed, and a robust, rigid TTC nail is inserted. In cases of severe talar avascular necrosis or massive fragmentation, a primary talectomy may be required, converting the procedure to a tibiocalcaneal fusion. This drastically alters the biomechanics and shortens the limb, but it is often necessary to achieve a solid bony apposition and eradicate necrotic bone.
External Fixation Techniques
When internal fixation is contraindicated due to active osteomyelitis, massive soft-tissue defects, or profoundly poor bone quality, thin-wire circular external fixation (e.g., Ilizarov frames or Taylor Spatial Frames) becomes the ultimate limb salvage tool. Infected and nonviable tissue is aggressively debrided. The joints are prepared for arthrodesis, and the frame is applied to compress the fusion sites and stabilize the limb. Smooth and olive wires are tensioned across the tibia, hindfoot, and forefoot, providing immense biomechanical stability while allowing full access to the soft tissue envelope. Open wounds can be managed concurrently with negative pressure wound therapy (NPWT) or advanced biological dressings through the frame, often avoiding the need for complex free flap coverage.
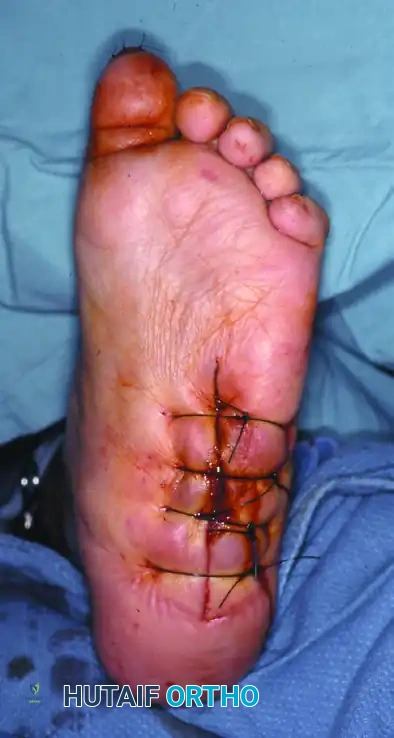

Complications, Incidence Rates, and Salvage Management
Surgical intervention in the Charcot foot is fraught with complications, and the operating surgeon must be intimately familiar with their diagnosis and management. The complication profile in this patient population is significantly higher than in standard orthopedic trauma or elective foot and ankle surgery, primarily due to the systemic manifestations of diabetes, profound neuropathy, and compromised microvascular perfusion. Patients must be extensively counseled preoperatively that limb salvage is a marathon, not a sprint, and that multiple trips to the operating room may be required.
Hardware failure is a pervasive risk, driven by the delayed healing potential of osteopenic bone and the sheer biomechanical forces exerted during weight-bearing. Screw breakage, plate pullout, and intramedullary nail migration can occur even with the most robust superconstructs. Nonunion and pseudoarthrosis are exceedingly common, with literature reporting rates between 10% and 30% for complex midfoot and hindfoot fusions in Charcot patients. However, a critical distinction must be made between a symptomatic nonunion and a stable, asymptomatic fibrous union. Because of the underlying sensory neuropathy, many nonunions are completely painless. If clinical stability is maintained, the hardware remains largely intact, and the foot remains plantigrade and braceable, a stable fibrous nonunion is considered a clinical success and generally does not require revision surgery.
Surgical site infections (SSI) and deep wound dehiscence are the most devastating complications, often precipitating a rapid cascade toward amputation. The incidence of SSI in complex Charcot reconstruction can approach 15% to 20%. Superficial dehiscence can often be managed with local wound care, oral antibiotics, and negative pressure wound therapy. However, deep infections involving the hardware or the fusion mass require aggressive surgical debridement, hardware removal (if the fusion is stable or the hardware is loose), and prolonged intravenous antibiotic therapy guided by deep intraoperative cultures. In cases where the hardware is providing critical stability but is colonized, a suppressive antibiotic regimen may be utilized until bony consolidation is achieved, at which point the hardware can be safely explanted.
Complication Profile and Salvage Strategies
| Complication | Estimated Incidence | Clinical Presentation | Salvage Management Strategy |
|---|---|---|---|
| Stable Fibrous Nonunion | 15% - 30% | Asymptomatic; Radiographic lucency at fusion site; Hardware intact. | Continued bracing (CROW/PTB); Observation; External bone stimulator. |
| Hardware Failure with Instability | 10% - 20% | Progressive deformity; Hardware breakage on XR; Recurrent ulceration. | Revision ORIF with larger diameter hardware; Conversion to external fixation. |
| Deep Surgical Site Infection | 10% - 15% | Erythema, purulent drainage, systemic leukocytosis, probing to bone. | Aggressive I&D; Hardware removal if loose; IV antibiotics; Antibiotic cement spacers. |
| Recurrent Ulceration | 15% - 25% | Breakdown over new bony prominences or residual deformity. | Exostectomy; Revision deformity correction; Modification of custom orthotics. |
| Catastrophic Failure / Sepsis | 5% - 10% | Ascending gas gangrene; Unsalvageable vascular collapse; Massive bone loss. | Major lower extremity amputation (BKA/AKA) to preserve life. |
Phased Post-Operative Rehabilitation Protocols
The postoperative management of the Charcot patient is as critical as the surgical execution itself. A flawlessly performed surgical reconstruction will rapidly fail if the postoperative rehabilitation protocol is not strictly enforced. The timeline for healing in a neuropathic foot is substantially protracted compared to a sensate foot. The overarching principle of postoperative care is prolonged, absolute immobilization and strict adherence to non-weight-bearing (NWB) protocols.
Phase I encompasses the immediate postoperative period (Weeks 0 to 6). The patient is placed in a well-padded, short-leg splint or a bivalved total contact cast
