Surgical Management of Rheumatoid Arthritis: A Comprehensive Orthopaedic Guide
Key Takeaway
Rheumatoid arthritis is a systemic inflammatory disease characterized by progressive synovitis and joint destruction. Orthopaedic management requires a multidisciplinary approach, balancing aggressive medical therapies like biologic agents with timely surgical interventions. Procedures range from arthroscopic synovectomy in early stages to complex total joint arthroplasty for severe articular collapse. Perioperative optimization, particularly regarding immunosuppressive medications and corticosteroid stress dosing, is critical to minimizing complications and maximizing functional outcomes.
Comprehensive Introduction and Patho-Epidemiology
Rheumatoid arthritis (RA) represents a chronic, systemic, inflammatory autoimmune disorder characterized by a predilection for the synovial lining of diarthrodial joints. While it most frequently manifests in the small articular articulations of the hands, wrists, and feet, its insidious progression spares virtually no synovial joint, ultimately culminating in severe biomechanical derangement if left unchecked. The disease exhibits a global prevalence ranging from 1% to 2%, with a distinct female-to-male predilection of approximately 2.5:1. Adult-onset RA is characteristically polyarticular and symmetric, whereas systemic extra-articular involvement affecting visceral organs, the pulmonary interstitium, or the ocular structures is relatively rare compared to its juvenile counterpart. The modern orthopaedic surgeon must recognize that RA is not merely a localized articular pathology but a systemic inflammatory cascade that profoundly alters the patient’s physiologic reserve, bone density, and soft-tissue integrity.
The precise etiology of rheumatoid arthritis remains elusive, though it is universally accepted within the rheumatologic and orthopaedic communities to be a complex, multifactorial interplay of genetic susceptibility and environmental triggers. A strong association exists with the human leukocyte antigen (HLA) class II genotypes, specifically the HLA-DR4 and HLA-DR1 alleles, which share a common amino acid sequence known as the "shared epitope." Environmental factors, most notably cigarette smoking and potentially infectious agents (such as Porphyromonas gingivalis in periodontitis), are thought to trigger epigenetic modifications, leading to the citrullination of host proteins. This loss of immunologic tolerance results in the production of anti-citrullinated protein antibodies (ACPAs), which predate clinical symptoms by years and serve as highly specific diagnostic markers.
The clinical and pathophysiologic hallmark of rheumatoid arthritis is the transformation of the normally delicate synovial membrane into a hyperplastic, aggressive, and locally invasive tissue known as the "pannus." This hypertrophic synovium is densely infiltrated by CD4+ T cells, B cells, and activated macrophages, which orchestrate a relentless cytokine storm. Tumor necrosis factor-alpha (TNF-α), interleukin-1 (IL-1), and interleukin-6 (IL-6) are the primary biochemical culprits, driving the production of matrix metalloproteinases (MMPs) that degrade articular cartilage. Furthermore, these cytokines potently upregulate the receptor activator of nuclear factor kappa-B ligand (RANKL), which binds to RANK on osteoclast precursors, driving massive osteoclastogenesis and localized bone resorption. This molecular cascade directly translates to the classic radiographic triad of RA: periarticular osteopenia, marginal erosions at the bare areas of the joint, and symmetric joint space narrowing.
Adult RA diagnosis has historically relied on the 1987 American Rheumatism Association (ARA) criteria, which required four of seven specific clinical and radiographic findings (such as morning stiffness lasting over an hour, symmetric arthritis, and rheumatoid nodules) present for at least six weeks. However, the paradigm has shifted toward the 2010 ACR/EULAR classification criteria, which prioritize early diagnosis before the onset of irreversible joint destruction. The 2010 criteria utilize a scoring system based on joint involvement, serology (Rheumatoid Factor and ACPAs), acute-phase reactants (CRP and ESR), and symptom duration. The ultimate goal of both medical and surgical management is the preservation of functional capacity, historically stratified into four ARA classes ranging from Class I (unrestricted activity) to Class IV (complete incapacitation). Because structural joint destruction typically commences within the first one to two years of disease onset, early aggressive pharmacological intervention is paramount to altering the natural history of the disease and preventing catastrophic articular collapse.
Detailed Surgical Anatomy and Biomechanics
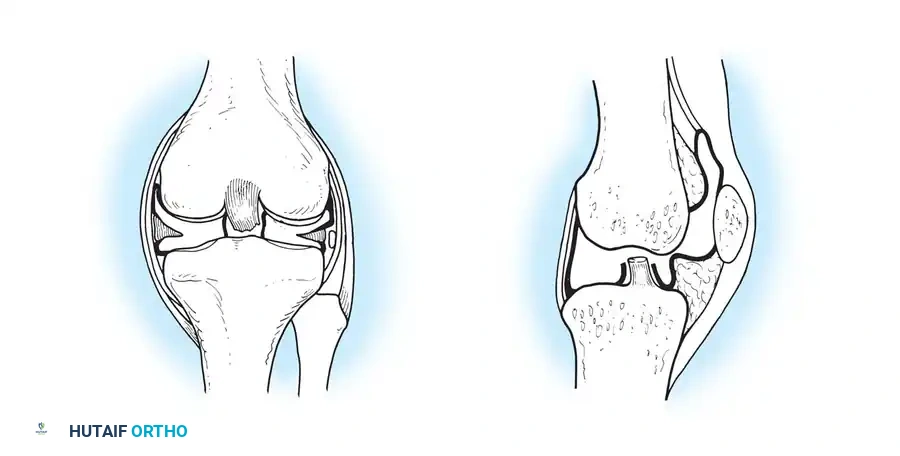
The biomechanical consequences of rheumatoid arthritis are profound, stemming directly from the dual assault of enzymatic cartilage degradation and mechanical distension of capsuloligamentous restraints by the hypertrophic pannus. Unlike primary osteoarthritis, which is fundamentally a disease of mechanical wear and asymmetric cartilage loss leading to osteophyte formation and subchondral sclerosis, RA is characterized by symmetric cartilage loss, severe periarticular osteopenia, and a distinct lack of osteophytosis. The inflammatory exudate and pannus physically stretch the joint capsule and collateral ligaments, rendering the joint grossly unstable. This ligamentous laxity, combined with the normal vector forces of muscle pull across the joint, leads to predictable, severe, and often fixed deformities that challenge even the most experienced reconstructive surgeon.
In the upper extremity, the hand and wrist are particularly vulnerable to these deforming forces. At the wrist, the destruction of the radioulnar ligaments leads to dorsal subluxation of the distal ulna (caput ulnae syndrome) and supination of the carpus. This creates a biomechanical environment where the extensor tendons are subjected to attrition over the prominent distal ulna, frequently resulting in sequential extensor tendon ruptures (Vaughan-Jackson syndrome). At the metacarpophalangeal (MCP) joints, the attenuation of the radial sagittal bands allows the extensor tendons to subluxate ulnarly into the valleys between the metacarpal heads. The intrinsic muscles, now acting with an altered line of pull, exacerbate volar subluxation and ulnar drift of the proximal phalanges. Proximal interphalangeal (PIP) joint involvement manifests as either a swan-neck deformity (PIP hyperextension with distal interphalangeal flexion due to volar plate laxity and intrinsic tightness) or a boutonniere deformity (PIP flexion with DIP hyperextension secondary to central slip rupture).
The rheumatoid knee presents a unique biomechanical challenge, typically manifesting as a severe valgus deformity combined with a fixed flexion contracture. The valgus alignment is driven by the preferential destruction of the lateral compartment cartilage and bone, coupled with the contracture of the iliotibial band, lateral collateral ligament, and popliteus tendon. Simultaneously, the medial collateral ligament becomes chronically attenuated and incompetent. The flexion contracture is exacerbated by hamstring spasm, posterior capsular scarring, and the patient's tendency to hold the knee in flexion to maximize joint volume and minimize capsular distension from chronic effusions. Reconstructing such a knee requires extensive lateral soft-tissue releases, meticulous gap balancing, and frequently the use of constrained or semi-constrained prosthetic designs to substitute for the deficient medial collateral ligament.
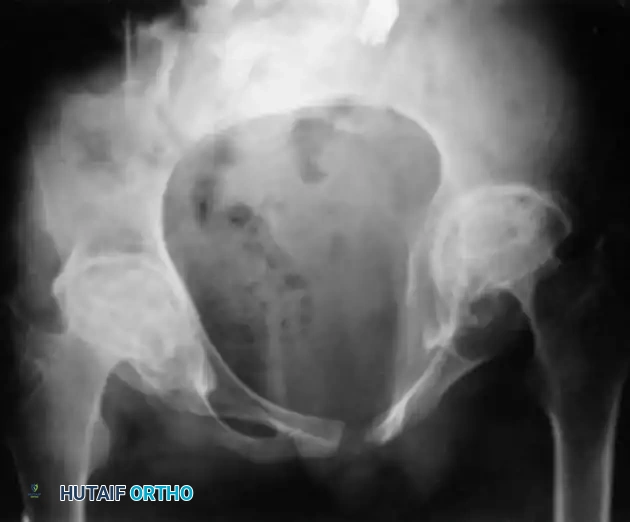
In the hip, the biomechanical hallmark of rheumatoid arthritis is protrusio acetabuli, defined radiographically by the medial migration of the femoral head past the ilioischial (Kohler's) line. The severe osteopenia of the medial acetabular wall, combined with the constant medially directed vector force of the hip abductors and iliopsoas, drives the femoral head deep into the pelvis. This medialization significantly alters the center of rotation, shortening the abductor moment arm and leading to profound abductor weakness and a Trendelenburg gait. Interestingly, protrusio acetabuli in juvenile rheumatoid arthritis (JRA) tends to migrate more cephalad (superiorly) compared to the strictly medial migration typically observed in adult RA. Surgical reconstruction must focus on restoring the anatomic center of rotation laterally and inferiorly, often requiring structural or impaction bone grafting of the medial wall defect.
Exhaustive Indications and Contraindications
The indications for surgical intervention in the rheumatoid patient have evolved significantly with the advent of disease-modifying antirheumatic drugs (DMARDs) and biologic therapies. Historically, orthopaedic surgeons frequently performed early synovectomies to halt disease progression. Today, the primary indications for surgery are the relief of intractable, mechanically driven pain, the prevention of imminent tendon rupture or neurologic compromise, and the restoration of functional independence in the setting of end-stage joint destruction. Surgery is generally considered only after the patient has failed a comprehensive, maximally optimized regimen of systemic pharmacotherapy. The orthopaedic surgeon must distinguish between pain caused by active inflammatory synovitis (which may respond to medical adjustment or intra-articular corticosteroid injection) and pain resulting from structural, bone-on-bone mechanical destruction (which necessitates surgical reconstruction).
Contraindications to elective orthopaedic surgery in the rheumatoid population are predominantly related to the patient's systemic medical optimization and infectious risk profile. Absolute contraindications include active local or systemic infection, severe uncorrected medical comorbidities (such as unstable coronary artery disease or decompensated heart failure), and the presence of an active, severe polyarticular disease flare. Operating during an acute flare not only increases the risk of postoperative stiffness and poor rehabilitation but also complicates the perioperative management of immunosuppressive medications. Relative contraindications include profound malnutrition, severe unmanaged osteoporosis, and active cutaneous ulcerations or vasculitic lesions near the proposed surgical site, which exponentially increase the risk of delayed wound healing and surgical site infection (SSI).
The perioperative management of the rheumatoid patient's pharmacological armamentarium is a critical component of surgical decision-making. Biologic agents, particularly tumor necrosis factor (TNF) antagonists (e.g., etanercept, infliximab, adalimumab), profoundly inhibit the host’s immune response and significantly elevate the risk of perioperative SSIs. Current guidelines dictate that surgery should be scheduled at the end of the dosing cycle for these agents, and they should be withheld for at least one dosing interval prior to moderate or high-risk procedures. Conversely, traditional DMARDs like methotrexate are generally continued perioperatively, as the risk of a severe systemic flare from cessation outweighs the theoretical risk of infection or wound healing complications, provided the patient does not have severe renal or hepatic impairment.
Table: Surgical Indications and Contraindications in Rheumatoid Arthritis
| Category | Specific Parameters | Clinical Rationale |
|---|---|---|
| Absolute Indications | Cervical myelopathy (AADI > 9mm, SAC < 14mm); Impending tendon rupture; End-stage structural joint collapse with intractable pain. | Neurologic preservation is paramount; structural failure cannot be salvaged medically; mechanical pain requires mechanical solutions (arthroplasty/arthrodesis). |
| Relative Indications | Chronic refractory synovitis (Larsen grade 0-1); Progressive flexible deformities impairing ADLs. | Synovectomy or soft-tissue rebalancing may delay joint destruction if performed before significant cartilage loss occurs. |
| Absolute Contraindications | Active systemic or localized infection; Unstable cardiopulmonary disease; Active vasculitic ulcers at the surgical site. | Prohibitive risk of catastrophic sepsis, perioperative mortality, or complete wound failure. |
| Relative Contraindications | Active polyarticular disease flare; Severe unmanaged osteoporosis; Poor psychosocial support or inability to participate in rehab. | High risk of implant subsidence, periprosthetic fracture, and severe postoperative stiffness or contracture. |
Pre-Operative Planning, Templating, and Patient Positioning
Pre-operative planning for the rheumatoid patient requires a meticulous, multidisciplinary approach that extends far beyond standard orthopaedic templating. The most critical, life-saving step in the pre-operative evaluation of any RA patient—regardless of the planned surgical procedure—is the assessment of the cervical spine. Rheumatoid involvement of the cervical spine occurs in up to 80% of patients, leading to atlantoaxial subluxation (AAS), basilar invagination (cranial settling), and subaxial subluxation. The destruction of the transverse ligament allows the odontoid process to migrate posteriorly during neck flexion, compressing the spinal cord. Therefore, dynamic flexion-extension cervical spine radiographs are absolutely mandatory prior to any surgical intervention requiring intubation. An Anterior Atlantodental Interval (AADI) greater than 3.5 mm indicates instability, while a Space Available for the Cord (SAC) of less than 14 mm is highly predictive of impending neurologic compromise.

Radiographic templating for total joint arthroplasty in RA must account for severe periarticular osteopenia, bone cysts, and complex deformities. In the knee, full-length standing alignment films are essential to quantify the magnitude of the valgus deformity and assess extra-articular bowing. The surgeon must anticipate the need for stemmed tibial and femoral components to bypass metaphyseal osteopenia and achieve secure diaphyseal fixation. Furthermore, metal augments or trabecular metal cones should be available to address contained and uncontained bone defects. In the hip, templating for protrusio acetabuli requires careful assessment of the medial wall thickness and the location of the anatomic center of rotation. The surgeon must plan for the use of impaction bone grafting, reinforcement cages, or highly porous multi-hole titanium shells to achieve stable fixation in compromised host bone.
Patient positioning and intraoperative handling demand extreme caution. The rheumatoid patient's osteopenic bone is highly susceptible to iatrogenic fracture during routine surgical maneuvers, such as dislocation of the hip or eversion of the patella. Limbs must be manipulated with broad, even pressure rather than focal force. Furthermore, the anesthesiologist must be acutely aware of the cervical spine status; if instability is present, fiberoptic awake intubation or the use of a video laryngoscope (GlideScope) with manual in-line stabilization is required to prevent catastrophic spinal cord injury during airway management. Positioning pads must be liberally applied to protect fragile, paper-thin rheumatoid skin and to prevent compression neuropathies, as these patients often have pre-existing peripheral nerve vulnerabilities.
Finally, pre-operative medical optimization must address the hypothalamic-pituitary-adrenal (HPA) axis. Patients on chronic corticosteroid therapy (typically defined as >5-10 mg of prednisone equivalent daily for more than 3 weeks) suffer from HPA axis suppression and cannot mount an appropriate endogenous cortisol response to surgical stress. Preoperative "stress" dosages of intravenous hydrocortisone are mandatory to prevent acute, potentially fatal adrenal crisis (Addisonian crisis), characterized by refractory hypotension, hypoglycemia, and cardiovascular collapse. For moderate stress procedures (e.g., total hip/knee arthroplasty), 50 to 75 mg of hydrocortisone is administered on the day of the procedure, with an expeditious taper over 1 to 2 days back to the patient's baseline preoperative dose.
Step-by-Step Surgical Approach and Fixation Technique
The surgical approach to the rheumatoid joint must be meticulously executed, respecting the compromised soft-tissue envelope and the fragile osseous structures. In the setting of a Total Knee Arthroplasty (TKA) for a severe valgus rheumatoid knee, a standard medial parapatellar arthrotomy is typically utilized, though a lateral parapatellar approach (Keblish) may be considered for fixed, severe valgus deformities to directly address the contracted lateral structures. Soft-tissue balancing is the crux of the procedure. The surgeon must systematically release the contracted lateral structures—often requiring a "pie-crusting" technique of the iliotibial band, followed by release of the lateral collateral ligament (LCL) and popliteus tendon off the lateral femoral epicondyle. Because the medial collateral ligament (MCL) is chronically attenuated, achieving symmetric flexion and extension gaps is exceedingly difficult. Consequently, the surgeon must have a low threshold to utilize a constrained condylar knee (CCK) prosthesis, which provides varus-valgus stability through a large, reinforced central post and cam mechanism.
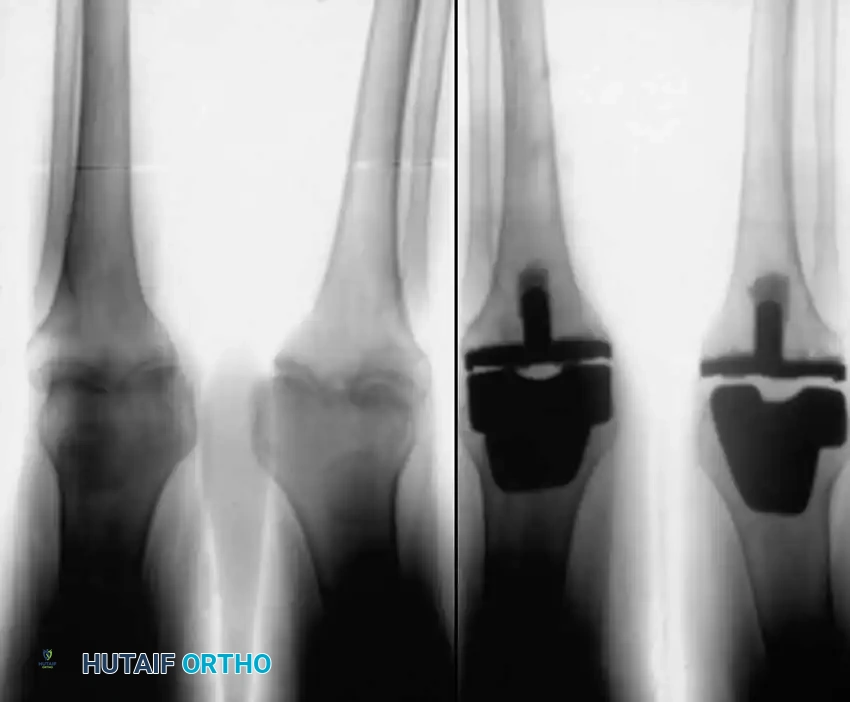
Fixation in the rheumatoid knee frequently necessitates the use of diaphyseal engaging stems. The metaphyseal bone is often characterized by cystic degeneration and profound osteopenia, rendering standard primary components susceptible to early subsidence and aseptic loosening. By utilizing cemented or press-fit diaphyseal stems, the surgeon effectively bypasses the compromised metaphysis, transferring the load to the more robust cortical bone of the diaphysis. Care must be taken during the preparation of the canals, utilizing sequential reaming without excessive force to avoid catastrophic periprosthetic fractures. The patella in RA is often thin and osteopenic; while resurfacing is generally recommended to prevent anterior knee pain from residual rheumatoid synovitis, the surgeon must ensure a minimum of 12-15 mm of residual composite patellar thickness to prevent postoperative patellar fracture.
In the surgical management of the rheumatoid hip, particularly in the presence of severe protrusio acetabuli, the primary objective is the lateralization and inferiorization of the center of rotation to its anatomic location. The standard posterior or anterolateral approach may be utilized. Upon exposure, dislocation of the hip must be performed with extreme care; in cases of severe protrusio, it is often safer to perform an in situ femoral neck osteotomy and extract the femoral head with a corkscrew to prevent iatrogenic femoral or acetabular fracture. Once the acetabulum is exposed, aggressive reaming is contraindicated, as it will further compromise the already deficient medial wall. Instead, the peripheral rim is gently prepared to bleeding bone.
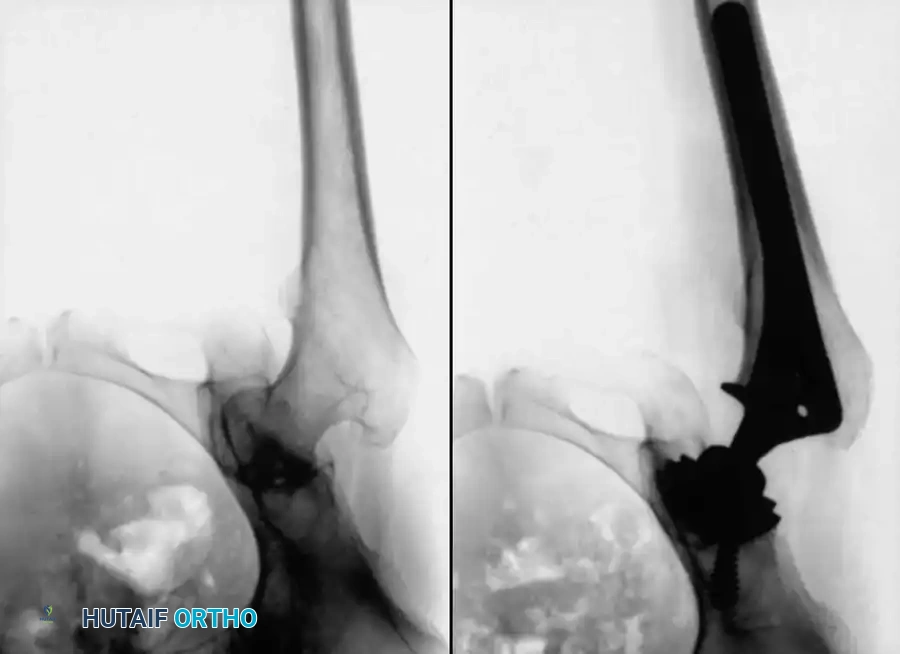
Reconstruction of the medial wall defect is most effectively achieved using the impaction bone grafting technique (Slooff-Ling technique). Morselized cancellous allograft or autograft (from the excised femoral head) is densely packed into the medial defect using specialized tamps. A highly porous, multi-hole hemispherical titanium shell is then impacted, achieving a "rim fit" on the intact peripheral acetabular bone, thereby bridging the medial defect and compressing the bone graft. If the peripheral rim is incompetent or the defect is massive, an antiprotrusio cage (e.g., Burch-Schneider cage) may be required. This device spans the acetabulum, fixing to the ilium superiorly and the ischium inferiorly, protecting the medial wall graft and providing a stable platform into which a polyethylene liner can be cemented.
Complications, Incidence Rates, and Salvage Management
The rheumatoid patient is uniquely predisposed to a myriad of perioperative and postoperative complications, driven by systemic immunosuppression, poor bone stock, and compromised soft tissues. The most devastating complication is Periprosthetic Joint Infection (PJI). The incidence of PJI in RA patients undergoing total joint arthroplasty is significantly higher than in the osteoarthritic population, ranging from 2% to 4% for primary procedures. This elevated risk is multifactorial, stemming from the use of biologic agents, chronic corticosteroid therapy, inherently altered host immunity, and fragile skin that is prone to delayed wound healing. Salvage management for chronic PJI typically requires a two-stage revision arthroplasty: explantation of the components, aggressive thorough debridement, placement of an antibiotic-eluting cement spacer, and a prolonged course of targeted intravenous antibiotics, followed by reimplantation once infection eradication is confirmed.
Intraoperative and postoperative periprosthetic fractures represent another major complication, directly attributable to severe rheumatoid osteopenia and the stress risers created by prosthetic implants. The incidence of intraoperative femur fractures during THA in RA patients can approach 3% to 5%, particularly when utilizing cementless, press-fit stems in "stovepipe" (Dorr Type C) femoral canals. Surgeons must maintain a low threshold for utilizing cemented femoral stems in the elderly rheumatoid population to mitigate this risk. If an intraoperative fracture occurs, it must be recognized immediately and bypassed with a longer stem, supplemented with cerclage wiring or strut allografts. Postoperative periprosthetic fractures require complex revision surgery, often utilizing long, fluted, tapered modular stems or distal femoral replacement (megaprosthesis) in cases of catastrophic distal femur bone loss.
Instability and dislocation following total joint arthroplasty are also more prevalent in the rheumatoid cohort. In THA, the incidence of dislocation is elevated due to the attenuation of the abductor musculature, altered joint biomechanics from pre-existing protrusio, and generalized capsular laxity. Salvage management for recurrent instability may involve the use of dual-mobility articulations, which provide a significantly greater jump distance and enhanced stability, or constrained acetabular liners in cases of profound abductor deficiency. In TKA, late instability often results from progressive attenuation of the collateral ligaments or failure of the extensor mechanism. Extensor mechanism rupture (patellar tendon or quadriceps tendon) is particularly disastrous in the RA patient; primary repair frequently fails due to poor tissue quality, necessitating complex salvage with synthetic mesh augmentation or whole extensor mechanism allograft reconstruction.
Table: Complications, Incidence Rates, and Salvage Management
| Complication | Estimated Incidence in RA | Primary Etiology | Salvage Management Strategy |
|---|---|---|---|
| Periprosthetic Joint Infection (PJI) | 2.0% - 4.0% | Immunosuppression (Biologics/Steroids); Poor wound healing; Altered host immunity. | Two-stage revision with antibiotic spacer; Chronic suppressive antibiotics if medically unfit for surgery. |
| Intraoperative Periprosthetic Fracture | 3.0% - 5.0% | Severe osteopenia; Cortical thinning; Excessive force during press-fit or dislocation. | Immediate recognition; Bypass fracture with long-stem components; Cerclage wiring; Strut grafting. |
| THA Dislocation / TKA Instability | 3.0% - 6.0% | Abductor/ligamentous attenuation; Capsular laxity; Failure to restore offset. | Dual-mobility bearing surfaces (THA); Constrained condylar or hinged prostheses (TKA). |
| Extensor Mechanism Rupture | 1.0% - 2.0% | Chronic steroid use; Attritional wear; Poor soft-tissue vascularity. | Extensor mechanism allograft; Synthetic mesh reconstruction (e.g., Marlex mesh); Arthrodesis (rare). |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation of the rheumatoid patient requires a delicate, highly individualized balance. The orthopaedic surgeon, physiatrist, and physical therapist must coordinate early mobilization to prevent debilitating stiffness—a pervasive and rapid complication in RA—while simultaneously protecting osteopenic bone, tenuous soft-tissue repairs, and unhealed osteotomies. Unlike osteoarthritic patients, who typically have a single symptomatic joint, RA patients suffer from polyarticular disease. This fundamental difference drastically alters rehabilitation mechanics. For instance, a patient recovering from a total knee arthroplasty cannot simply utilize standard axillary crutches or a standard walker if they suffer from severe rheumatoid involvement of the wrists, elbows, or shoulders. The rehabilitation team must proactively provide platform walkers or custom-molded forearm crutches to distribute weight-bearing forces away from the fragile small joints of the upper extremity.
Phase I of rehabilitation (Postoperative Days 0 to 14) focuses on immediate joint mobilization, edema control, and the prevention of venous thromboembolism. Continuous Passive Motion (CPM) machines, while controversial in standard osteoarthritis pathways, remain highly beneficial in the rheumatoid knee to prevent the rapid formation of intra-articular adhesions. Pain management must be multimodal, utilizing regional nerve blocks, acetaminophen, and judicious opioids, while carefully managing the reintroduction of the patient's baseline NSAIDs and DMARDs in coordination with their rheumatologist. Weight-bearing status is generally advanced as tolerated for cemented arthroplasties, but may be restricted for cementless diaphyseal stems or complex bone-grafting procedures (such as impaction grafting for protrusio acetabuli) to allow for biological incorporation.
Phase II (Weeks 2 to 6) emphasizes the restoration of active range of motion, isometric strengthening, and the improvement of proprioception. In the rheumatoid population, aggressive passive stretching by the physical therapist must be strictly avoided, as the osteopenic bone is highly susceptible to iatrogenic fracture, and attenuated tendons are prone to rupture under high tensile loads. Aquatic therapy is exceptionally beneficial during this phase, providing buoyancy that unloads the polyarticular joints while offering gentle, uniform resistance for global muscle strengthening. Occupational therapy is equally critical during this period, particularly following upper extremity procedures or for patients with severe hand deformities. The occupational therapist provides custom dynamic or static splinting to protect surgical repairs (e.g., post-MCP arthroplasty) and introduces adaptive devices (e.g., long-handled reachers, modified utensils) to compensate for permanent functional deficits and promote independence in Activities of Daily Living (ADLs).
Phase III (Weeks 6 to 12 and beyond) focuses on functional integration, endurance training, and long-term joint protection strategies. The rheumatoid patient must be educated that while their surgically reconstructed joint is mechanically sound, the surrounding musculature and adjacent joints remain vulnerable to the systemic disease process. High-impact activities and heavy lifting are permanently restricted to prevent accelerated polyethylene wear and implant loosening. The successful surgical management of rheumatoid arthritis does not end at discharge; it relies on a seamless, lifelong multidisciplinary team approach encompassing rheumatologists, orthopaedic surgeons, and rehabilitation specialists to dynamically manage the patient's evolving functional needs and systemic disease progression.
Summary of Landmark Literature and Clinical Guidelines
The surgical management of rheumatoid arthritis is heavily guided by evolving literature, particularly concerning the perioperative management of immunomodulatory medications and the long-term efficacy of specific surgical interventions. A landmark consensus document is the American College of Rheumatology (ACR) and American Association of Hip and Knee Surgeons (AAHKS) Guideline for the Perioperative Management of Antirheumatic Medication in Patients with Rheumatic Diseases Undergoing Elective Total Hip or Total Knee Arthroplasty. This comprehensive guideline established the modern standard of care, definitively recommending the continuation of traditional synthetic DMARDs (like methotrexate) through the perioperative period, while mandating the withholding of biologic agents (such as TNF inhibitors) prior to surgery. The guidelines dictate scheduling surgery at the end of the biologic's dosing cycle and restarting the medication roughly 14 days postoperatively, strictly contingent upon the presence of a clean, dry, and healing surgical incision without clinical signs of infection.
In the realm of Juvenile Idiopathic Arthritis (JIA), the literature surrounding the utility of synovectomy has undergone a dramatic paradigm shift. Historically championed as a joint-preserving procedure, longitudinal studies have severely tempered early enthusiasm. The seminal work by
