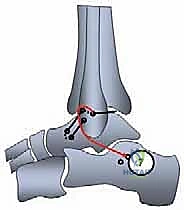
Anatomic Lateral Ankle Ligament Reconstruction: A Masterclass in Plantaris Longus Autograft Technique
Key Takeaway
Join us in the OR for an immersive masterclass on anatomic lateral ankle ligament reconstruction. We'll meticulously detail the plantaris longus autograft technique, from comprehensive anatomical considerations and precise patient positioning to step-by-step intraoperative execution. Learn critical pearls, pitfalls, and postoperative protocols for optimal outcomes in chronic ankle instability. This guide ensures a thorough understanding of this complex procedure.
Welcome, colleagues and fellows, to the operating theater. Today, we are addressing a common yet uniquely challenging orthopedic pathology: chronic lateral ankle instability (CLAI). This condition is far more insidious than a simple, isolated ligamentous sprain; it represents a complex cascade of biomechanical failures leading to persistent functional impairment, altered joint kinematics, and, if left surgically untreated in the symptomatic patient, inevitable degenerative osteoarthritic changes. Our definitive goal is the precise restoration of the native ankle biomechanics, providing robust, enduring stability while meticulously preserving physiologic motion. We will achieve this through an anatomic lateral ligament reconstruction utilizing a plantaris longus autograft—a masterclass technique that has demonstrated superior long-term outcomes in restoring both mechanical and functional stability.
Comprehensive Introduction and Patho-Epidemiology
Chronic lateral ankle instability typically arises from an initial, often severe, inversion ankle sprain that fails to progress through the normal phases of tissue healing. This failure initiates a vicious cycle of recurrent macro-trauma and micro-trauma. The pathogenesis of CLAI is multifactorial, encompassing mechanical laxity, proprioceptive deficits, and neuromuscular dysregulation. When the anterior talofibular ligament (ATFL) and calcaneofibular ligament (CFL) are disrupted, the talus is permitted to translate anteriorly and tilt into varus, fundamentally altering the contact mechanics of the tibiotalar joint.
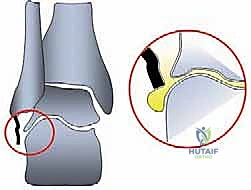
The etiology of this chronic state can be attributed to several compounding factors. Inadequate primary conservative management is a frequent culprit; initial sprains are routinely underestimated by primary care providers, leading to premature return to weight-bearing and athletic activities before the collagen matrix has adequately remodeled. Furthermore, the intra-articular environment itself is hostile to ligamentous repair. Synovial fluid interposition between the torn ligament ends significantly impedes fibrin clot formation and subsequent fibroblastic bridging, resulting in a structurally inferior, elongated scar tissue formation.
Repetitive trauma exacerbates this attenuation. Each subsequent inversion event further degrades the tensile strength of the healing tissue, creating a self-perpetuating cycle of instability. Crucially, this mechanical damage is intrinsically linked to compromised proprioception. The lateral ligamentous complex is densely populated with mechanoreceptors (Ruffini endings, Pacinian corpuscles, and Golgi tendon organs). Disruption of these ligaments severs the afferent feedback loop to the central nervous system, drastically delaying the reflex activation of the dynamic peroneal stabilizers. The epidemiological data is staggering: the risk of an ankle sprain increases exponentially from approximately 1 in 1,000,000 steps in an uninjured individual to 1 in 1,000 steps following a severe, poorly rehabilitated sprain.
Historically, the orthopedic community relied heavily on non-anatomic tenodesis procedures, such as the Evans or Watson-Jones techniques, utilizing the peroneus brevis tendon. While these procedures successfully eliminated gross laxity, they achieved this at a severe biomechanical cost. Non-anatomic routing inherently restricts subtalar and tibiotalar motion, leading to altered hindfoot kinematics, persistent lateral column pain, and accelerated degenerative joint disease. Consequently, the modern gold standard has shifted definitively toward anatomic reconstruction. By precisely recreating the native origins and insertions of the ATFL and CFL using a free tendon graft, we restore the isometric properties of the lateral complex, yielding superior functional outcomes and longevity, particularly in the high-demand athletic population.
Detailed Surgical Anatomy and Biomechanics
Before initiating any surgical incision, an exhaustive, three-dimensional understanding of the regional osteology, ligamentous footprints, and neurovascular topography is absolutely paramount. The lateral ankle is an unforgiving anatomic zone where millimeter-level deviations in tunnel placement can drastically alter joint kinematics.
Lateral Ligamentous Complex
The primary static stabilizers of the lateral ankle comprise the lateral collateral ligamentous complex. The Anterior Talofibular Ligament (ATFL) is a flat, quadrilateral band, approximately 1.5 to 2 cm in length and 6 to 10 mm in width. It is the weakest of the complex and the first to fail under inversion stress. It originates from the anterior border of the distal fibula—specifically, the inferior segment of the anterior colliculus—and inserts onto the lateral articular facet of the talar neck. Biomechanically, it is the primary restraint to anterior translation of the talus relative to the tibia, particularly when the ankle is in plantarflexion.
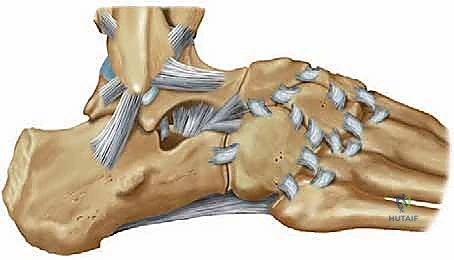
The Calcaneofibular Ligament (CFL) is a robust, cord-like structure that spans two joints (the talocrural and subtalar joints). Originating from the anterior aspect of the lateral malleolar tip, it courses obliquely downwards and backwards to insert on a small tubercle on the lateral surface of the calcaneus. The CFL is extra-articular and intimately related to the peroneal tendon sheath. It serves as the primary restraint to inversion when the ankle is in a neutral or dorsiflexed position. The Posterior Talofibular Ligament (PTFL) is the strongest component, originating from the malleolar fossa of the fibula and inserting onto the posterior talar process. It is rarely torn except in severe, massive ankle dislocations and is generally not addressed in standard lateral reconstructions.
Neurovascular Topography
The surgical approach places two critical sensory nerves at risk. The Superficial Peroneal Nerve (SPN) courses anterior to the fibula in the lateral compartment before piercing the crural fascia approximately 10 to 12 cm proximal to the lateral malleolus. It then divides into the medial and intermediate dorsal cutaneous nerves. The SPN is highly variable and extremely susceptible to iatrogenic injury during the anterior extent of the lateral incision or via aggressive retractor placement. Injury results in painful neuromas and sensory deficits over the dorsum of the foot.
The Sural Nerve courses posteriorly, running alongside the small saphenous vein posterior to the lateral malleolus and lateral to the Achilles tendon. It provides sensation to the lateral hindfoot and the fifth digit. While less commonly injured than the SPN during ATFL/CFL reconstruction, it remains at risk during posterior dissection, particularly when preparing the calcaneal footprint for the CFL graft or during overly aggressive retraction of the peroneal tendons.
Plantaris Longus Tendon Anatomy
The plantaris muscle is a small, fusiform muscle with a short belly and a tremendously long, slender tendon. It originates from the lateral supracondylar line of the femur, just superior to the lateral head of the gastrocnemius. The tendon courses distally and medially between the gastrocnemius and soleus muscles. In the distal third of the leg, it emerges on the medial aspect of the Achilles tendon to insert into the calcaneus.
The plantaris tendon is an ideal autograft for lateral ankle reconstruction due to its exceptional tensile strength, adequate length (often exceeding 30 cm), and minimal donor site morbidity. It is considered a vestigial structure, and its harvest does not result in any clinically significant loss of plantarflexion power. However, surgeons must be aware that the plantaris is absent in approximately 7% to 20% of the population, necessitating a contingency plan (such as a gracilis autograft or an allograft) during preoperative planning.
Exhaustive Indications and Contraindications
The decision to proceed with an anatomic reconstruction utilizing a tendon graft, rather than a primary anatomic repair (e.g., the modified Broström-Gould procedure), requires careful clinical judgment. The Broström procedure remains the workhorse for standard instability; however, reconstruction is strictly indicated when local tissue quality is insufficient to withstand physiologic loads.
| Clinical Parameter | Modified Broström Repair | Anatomic Tendon Reconstruction |
|---|---|---|
| Tissue Quality | Robust, identifiable ligament remnants | Attenuated, friable, or absent remnants |
| Prior Surgery | Primary intervention | Failed prior Broström repair |
| Patient Phenotype | Normal connective tissue | Generalized ligamentous laxity (e.g., Ehlers-Danlos) |
| BMI / Demand | Low to moderate BMI, recreational | High BMI, elite/professional athletes |
| Chronicity | Acute-on-chronic, moderate duration | Long-standing chronic instability (> 10 years) |
Indications for plantaris longus anatomic reconstruction include patients with a clearly documented history of recurrent lateral ankle sprains who have failed a minimum of 3 to 6 months of dedicated physical therapy (focusing on peroneal strengthening and proprioceptive training). It is the procedure of choice for revision scenarios where a previous primary repair has stretched out or failed. Furthermore, patients with profound generalized hypermobility often fail primary repairs because their native collagen is inherently flawed; augmenting the lateral complex with a robust tendon graft provides the necessary structural rigidity.
Contraindications must be rigorously respected. Absolute contraindications include active local or systemic infection, severe peripheral vascular disease precluding wound healing, and advanced degenerative osteoarthritis of the tibiotalar or subtalar joints (where a fusion or arthroplasty would be more appropriate). Relative contraindications include profound, uncorrectable hindfoot varus alignment. If a patient presents with a cavovarus foot type, reconstructing the lateral ligaments in isolation is a recipe for catastrophic graft failure. The deforming bony forces will inevitably stretch out the graft. In such cases, a concomitant lateralizing calcaneal osteotomy or first metatarsal dorsiflexion osteotomy must be performed to correct the mechanical axis prior to, or concurrently with, the ligament reconstruction.
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the foundation of a successful surgical outcome. The clinical assessment must be exhaustive. The patient history should quantify the frequency of "giving way" episodes, the severity of pain, and the specific functional limitations. Physical examination must include an assessment of the overall lower extremity alignment, specifically evaluating for hindfoot varus. Range of motion is documented, and the Silfverskiöld test is performed to rule out an isolated gastrocnemius contracture, which, if present, increases strain on the lateral ligaments and may necessitate a concurrent gastrocnemius recession.
Stability testing—specifically the anterior drawer test (evaluating ATFL integrity) and the talar tilt test (evaluating CFL integrity)—should be performed and compared to the contralateral, asymptomatic limb. These tests are often best appreciated under anesthesia immediately prior to the incision, eliminating involuntary muscle guarding. Imaging is mandatory. Weight-bearing AP, lateral, and mortise radiographs assess for osteochondral lesions, loose bodies, and degenerative changes. Stress radiographs, while somewhat operator-dependent, can quantify the degree of anterior translation and varus tilt. Magnetic Resonance Imaging (MRI) is the gold standard for evaluating the soft tissue envelope. It details the extent of ligamentous scarring, identifies concomitant peroneal tendon pathology (tears or subluxation), and highlights osteochondral lesions of the talar dome, which are present in a significant percentage of chronic instability cases.
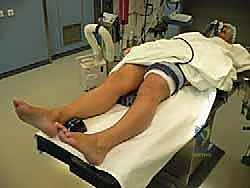
Patient positioning is critical for optimal surgical ergonomics. The patient is placed supine on the operating table. A large sandbag or a specialized positioning bump is placed under the ipsilateral hip. This maneuver internally rotates the lower extremity, bringing the lateral malleolus and the lateral aspect of the foot into a prominent, easily accessible position for the primary surgeon. A well-padded pneumatic tourniquet is applied to the proximal thigh.
Following standard induction of anesthesia, a prophylactic intravenous antibiotic is administered. The limb is exsanguinated with an Esmarch bandage, and the tourniquet is inflated to a standard pressure (typically 250-300 mmHg) to ensure a bloodless surgical field. The entire lower extremity, from the tourniquet down to the toes, is prepped with a chlorhexidine or iodine-based antiseptic solution and draped in a sterile fashion. This extensive draping is necessary to allow simultaneous access to the medial calf for the plantaris graft harvest and the lateral ankle for the reconstructive phase.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution is divided into two distinct phases: the harvest of the autograft and the anatomic reconstruction of the lateral complex. Precision in both phases is non-negotiable.
Phase I: Harvesting the Plantaris Longus Autograft
We begin on the medial aspect of the calf. The plantaris tendon is most reliably identified at the musculotendinous junction of the medial gastrocnemius.
A 3-centimeter longitudinal incision is made over the medial calf, approximately three fingerbreadths medial to the tibial crest, at the level of the maximal girth of the gastrocnemius muscle belly. Dissection is carried sharply through the skin and subcutaneous fat. Hemostasis is achieved meticulously using bipolar electrocautery to avoid thermal injury to the surrounding superficial nerves.
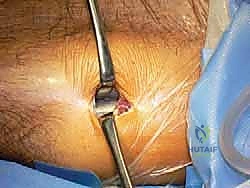
The deep crural fascia is identified and incised longitudinally. Using blunt dissection—often a combination of a surgical sweep with the index finger and a blunt elevator—the interval between the medial head of the gastrocnemius and the underlying soleus muscle is developed. This is an avascular plane that opens easily.
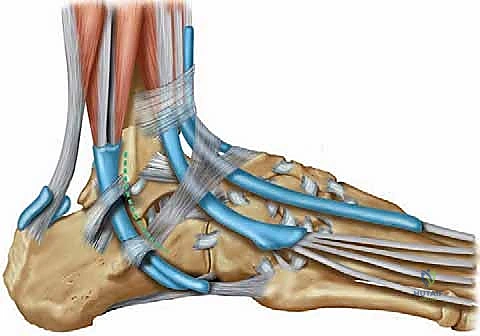
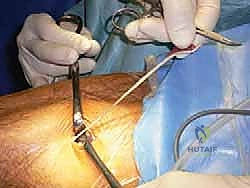
Deep within this interval, the plantaris longus tendon is visualized. It presents as a distinct, pearlescent, ribbon-like structure running obliquely from proximal-lateral to distal-medial. It is crucial to definitively differentiate it from the medial margin of the Achilles tendon or fascial bands of the soleus. Once isolated, a right-angle clamp is passed beneath the tendon.
The tendon is sharply transected as proximally as possible within the incision. The free distal end is secured with a heavy non-absorbable traction suture (e.g., #2 FiberWire). The tendon is then threaded into a closed-end tendon stripper. While maintaining firm, inline traction on the suture, the stripper is steadily advanced distally toward the calcaneus. The knee should be slightly flexed and the ankle plantarflexed to reduce tension on the posterior compartment structures. The stripper is advanced until it reaches the calcaneal insertion, at which point a firm push cuts the tendon free. The graft is delivered onto the back table, cleared of residual muscle tissue, and prepared with whipstitches at both ends.
Phase II: Lateral Approach and Footprint Preparation
Attention is now turned to the lateral ankle.

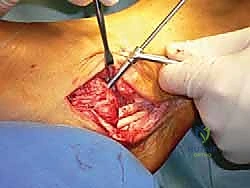
A curvilinear incision is made, starting approximately 2 cm proximal to the tip of the lateral malleolus, curving distally and anteriorly along the course of the ATFL, and extending toward the base of the fourth metatarsal. This "J-shaped" incision provides excellent exposure of both the fibula and the talar/calcaneal footprints.
Dissection proceeds carefully through the subcutaneous tissues. The superficial peroneal nerve branches must be identified and gently retracted anteriorly. The inferior extensor retinaculum is identified, incised, and tagged for later repair.
The anterior capsule of the ankle joint is exposed. The attenuated remnants of the ATFL and CFL are identified and sharply excised from their fibular origins to expose pristine cortical bone. A limited anterolateral arthrotomy is performed to inspect the joint for loose bodies and to visualize the talar footprint of the ATFL.
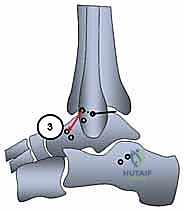
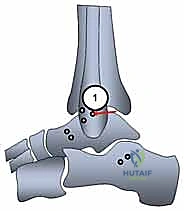
We now prepare the osseous tunnels. The anatomic footprint of the ATFL on the talar neck is identified. A guide pin is placed at this isometric point, directed medially and slightly plantarward to avoid penetrating the subtalar joint. A 4.5 mm or 5.0 mm cannulated reamer is used to create a blind tunnel approximately 15-20 mm deep.
Next, the calcaneal footprint of the CFL is localized. This requires retracting the peroneal tendons superiorly and anteriorly. The insertion site is typically a small tubercle on the lateral calcaneal wall. A guide pin is inserted here, directed medially and slightly superiorly, and over-reamed to create the calcaneal tunnel.
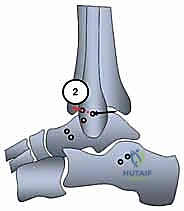
Attention returns to the fibula. Two convergent drill holes are required to recreate the origins of the ATFL and CFL. The ATFL origin is located at the anterior inferior border of the fibula, while the CFL origin is at the very tip of the malleolus.
Guide pins are placed at these precise anatomic origins, ensuring an adequate bone bridge (at least 1 cm) remains between them to prevent cortical blowout.
These pins are over-reamed, and the tunnels are connected internally using a curved curette or a specialized passing instrument, creating a trans-fibular tunnel.
Phase III: Graft Routing and Fixation
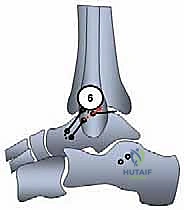
The prepared plantaris graft is brought to the field.
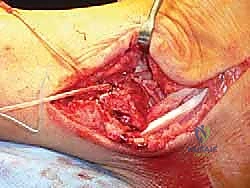
One end of the graft is passed into the talar tunnel. It is secured using a bio-tenodesis or PEEK interference screw. The screw must be inserted parallel to the graft to ensure maximum pull-out strength, adhering to the "deadman's angle" principles.
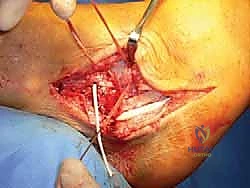
The free end of the graft is then shuttled beneath the extensor retinaculum, passed into the anterior fibular hole (ATFL origin), routed through the fibula, and brought out through the inferior hole (CFL origin).
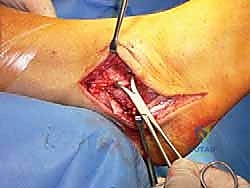
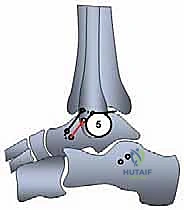
The graft is then directed deep to the peroneal tendons and introduced into the calcaneal tunnel. Tensioning is the most critical step of the procedure. The ankle is held in neutral dorsiflexion and slight eversion to restore appropriate physiological tension to the reconstructed complex. While maintaining this precise position, the graft is tensioned and secured within the calcaneal tunnel using a second interference screw.
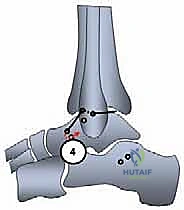
The final construct is rigorously tested. An anterior drawer and varus stress test are performed manually to confirm the elimination of pathologic laxity. The graft should be taut, and the ankle should demonstrate full, unimpeded range of motion without impingement.
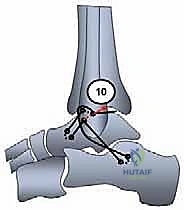
Closure is performed in a layered fashion. The extensor retinaculum is mobilized and sutured over the fibular bone bridge to provide an additional layer of biologic reinforcement (a modified Gould augmentation). The subcutaneous tissues are closed with absorbable sutures.
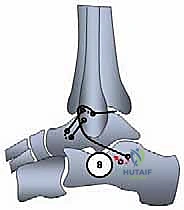
The skin is closed meticulously with non-absorbable nylon or a subcuticular Monocryl stitch, depending on surgeon preference and skin tension.

A sterile, non-adherent dressing is applied over the incisions.