Limb-Sparing Gluteus Maximus Resection for Sarcoma: An Intraoperative Masterclass
Key Takeaway
This masterclass details the limb-sparing gluteus maximus resection for soft tissue sarcomas. We cover essential preoperative planning, precise patient positioning, and a granular, real-time walkthrough of the surgical technique. Fellows will gain insights into critical anatomical landmarks, neurovascular preservation, meticulous tumor excision, and advanced strategies for seroma prevention. Postoperative care and potential complications are also thoroughly discussed, ensuring comprehensive understanding of this complex oncological procedure.
Comprehensive Introduction and Patho-Epidemiology
The evolution of orthopedic oncology over the past four decades has been defined by a paradigm shift from radical, morbid amputations to sophisticated, function-preserving limb salvage surgeries. Today, we are focusing on a quintessential limb-sparing procedure: the gluteus maximus resection for high-grade soft tissue sarcoma (STS). Historically, large, deep-seated sarcomas of the posterior pelvis and buttock were managed with radical hemipelvectomies or hindquarter amputations, procedures carrying profound physical and psychological morbidity. However, driven by advancements in high-resolution magnetic resonance imaging (MRI), targeted neoadjuvant chemoradiation protocols, and refined microvascular and macroscopic surgical techniques, limb-sparing resections are now the standard of care for approximately 90% of patients. This approach prioritizes the triad of oncologic safety, functional preservation, and maximization of the patient's postoperative quality of life.
The gluteus maximus, a massive, powerful hip extensor and external rotator, represents a classic "quiet area" for sarcomagenesis. Because the posterior gluteal compartment is highly expansile and lacks rigid fascial constraints compared to the distal extremities, tumors arising in this location frequently grow to substantial, sometimes massive, dimensions before producing clinical symptoms. Patients typically present with a painless, progressively enlarging mass, and by the time of clinical detection, these lesions have often encroached upon critical neurovascular structures or adjacent pelvic anatomy. The most common histologic subtypes encountered in this region include undifferentiated pleomorphic sarcoma (UPS), myxofibrosarcoma, and dedifferentiated liposarcoma. Understanding the specific tumor biology is critical, as it dictates the propensity for microscopic local invasion and distant metastasis, thereby influencing our surgical margin strategy.
From a pathophysiological standpoint, soft tissue sarcomas expand centripetally, compressing surrounding normal tissue to form a pseudocapsule. This pseudocapsule is a deceptive anatomical boundary; it is invariably permeated by microscopic satellite tumor cells. Therefore, a simple "shelling out" or marginal excision is oncologically unacceptable and guarantees local recurrence. Our fundamental surgical objective is an en bloc wide local excision. This necessitates resecting the tumor along with its pseudocapsule and a continuous cuff of normal, healthy tissue—in this case, the entirety or a significant functional unit of the gluteus maximus muscle—to achieve R0 (microscopically negative) margins. The deep fascia, periosteum of the ilium, and the epineurium of the sciatic nerve frequently serve as our deep anatomic barriers.
The success of a limb-sparing gluteus maximus resection hinges entirely on rigorous multidisciplinary collaboration. Preoperative presentation at a sarcoma tumor board is mandatory. Neoadjuvant radiation therapy is frequently employed for high-grade, deep-seated lesions >5 cm to sterilize the reactive zone, induce tumor necrosis, and potentially downstage the tumor, thereby facilitating a more conservative resection. Neoadjuvant chemotherapy may be indicated for specific chemosensitive histologies or high-risk systemic profiles. The integration of these modalities requires the orthopedic oncologist to meticulously plan the surgical approach, anticipating radiation-induced tissue fibrosis and compromised wound healing, which heavily influence our flap design and closure strategies.
Detailed Surgical Anatomy and Biomechanics
Mastery of the gluteal regional anatomy is the foundation upon which safe and oncologically sound resections are built. The gluteus maximus is the largest and most superficial muscle of the gluteal complex, characterized by its coarse fascicular architecture and robust vascularity. It possesses a broad, multi-focal origin, arising from the posterior gluteal line of the ilium, the posterior surface of the lower sacrum and the coccyx, the aponeurosis of the erector spinae, and the robust sacrotuberous ligament. The muscle fibers course obliquely in an inferolateral direction. The superior and larger portion of the muscle inserts into the iliotibial (IT) tract of the fascia lata, while the deeper, inferior fibers insert directly onto the gluteal tuberosity of the proximal femur. This extensive footprint requires meticulous, systematic detachment during en bloc resection to avoid inadvertent tumor violation.
Deep to the gluteus maximus lies the critical retrogluteal space, a complex anatomical compartment that houses the primary neurovascular conduits of the lower extremity and the deep external rotators of the hip. Upon elevating the gluteus maximus, one encounters a fascial plane covering the gluteus medius superiorly and the piriformis muscle centrally. Inferior to the piriformis, the superior gemellus, obturator internus, inferior gemellus, and quadratus femoris are sequentially aligned. The interval between the piriformis and the superior gemellus is the most critical anatomical landmark in this region, as it serves as the primary exit point for the sciatic nerve from the greater sciatic foramen.
The sciatic nerve is the paramount neurovascular structure requiring identification and protection during this procedure. Exiting the pelvis typically inferior to the piriformis (though anatomical variants, such as the nerve piercing the piriformis, exist and must be anticipated), it descends vertically between the greater trochanter of the femur and the ischial tuberosity. It lies deep to the gluteus maximus and superficial to the short external rotators and the adductor magnus. While massive gluteal sarcomas frequently displace the sciatic nerve, direct epineurial invasion is relatively uncommon unless the tumor originates from the nerve sheath itself (e.g., Malignant Peripheral Nerve Sheath Tumor). The surgical strategy relies on early, proximal identification of the nerve at the sciatic notch and continuous visual and tactile protection throughout the resection.
The vascular supply to the gluteus maximus is robust, derived primarily from the superior and inferior gluteal arteries, which are terminal branches of the internal iliac artery. The superior gluteal artery exits the pelvis superior to the piriformis, supplying the upper portion of the muscle, while the inferior gluteal artery exits inferior to the piriformis, accompanying the sciatic nerve, to supply the lower portion. These vessels, along with their accompanying veins and the inferior gluteal nerve (which provides exclusive motor innervation to the gluteus maximus), must be meticulously identified, isolated, and ligated. Biomechanically, the loss of the gluteus maximus results in a predictable deficit in forceful hip extension and external rotation. While routine level-ground walking is often surprisingly well-compensated by the hamstrings and adductor magnus, patients will exhibit a characteristic "gluteus maximus lurch" (posterior trunk lean during the stance phase) when ascending stairs or rising from a seated position.
Exhaustive Indications and Contraindications
The decision to proceed with a limb-sparing gluteus maximus resection versus a radical amputation requires a nuanced assessment of tumor characteristics, anatomical involvement, and patient-specific factors. The primary indication for this procedure is a biopsy-proven, intermediate- to high-grade soft tissue sarcoma anatomically confined to the gluteus maximus muscle or its immediate fascial envelopes, where a wide en bloc excision can be achieved without compromising the major neurovascular supply to the distal extremity. Furthermore, it is indicated for recurrent low-grade sarcomas or massive benign aggressive lesions (e.g., extra-abdominal desmoid tumors) that have failed conservative management and require extensive muscular resection for local control.
Contraindications to limb-sparing gluteal resection are primarily dictated by the inability to achieve negative oncologic margins without rendering the limb flail, ischemic, or insensate. Absolute contraindications include extensive tumor extension through the greater sciatic notch into the true pelvis, which precludes adequate posterior clearance and necessitates a combined abdominopelvic and posterior approach, often culminating in a hemipelvectomy. Direct, irreparable encasement of the sciatic nerve by a high-grade sarcoma historically mandated amputation; however, in highly selected cases at tertiary centers, en bloc sciatic nerve resection with subsequent nerve grafting or tendon transfers may be considered, though this remains controversial and highly morbid.
Relative contraindications revolve around soft tissue integrity and patient comorbidities. Severe, fungating tumors with massive skin involvement or concurrent polymicrobial infection of the tumor bed significantly elevate the risk of catastrophic postoperative wound failure and sepsis, potentially making a clean amputation a safer, more definitive oncologic and palliative choice. Furthermore, direct osseous invasion into the sacrum or the ilium requires composite bone and soft tissue resections (e.g., partial sacrectomy or partial iliectomy). While these are technically limb-sparing, they escalate the complexity, morbidity, and complication rates exponentially compared to an isolated gluteus maximus resection.
The management of the prior biopsy tract is a critical determinant of surgical eligibility. The biopsy tract is universally considered contaminated with malignant cells and must be excised en bloc with the definitive tumor specimen. If an outside institution performed a poorly planned transverse incision or a biopsy that widely contaminated the gluteal fasciocutaneous flaps or the ischiorectal fossa, achieving a wide margin may require sacrificing so much skin that primary closure or local flap coverage becomes impossible, thereby pushing the surgeon toward a more radical procedure.
| Category | Limb-Sparing Gluteus Maximus Resection | Radical Hemipelvectomy / Amputation |
|---|---|---|
| Primary Indication | Sarcoma confined to gluteus maximus with clear fascial boundaries. | Massive pelvic extension; intractable fungating tumors. |
| Neurovascular Status | Sciatic nerve displaceable/free of direct tumor encasement. | Irreparable sciatic nerve encasement (relative) or iliac vessel involvement. |
| Bone Involvement | No direct invasion of ilium, sacrum, or proximal femur. | Massive destruction of the pelvic ring or sacral neuroforamina. |
| Biopsy Tract | Strategically placed, easily excisable en bloc with the specimen. | Widely contaminated, precluding safe flap elevation and closure. |
| Patient Status | Medically optimized for prolonged surgery and complex rehab. | Palliative scenario where rapid local control is prioritized. |
Pre-Operative Planning, Templating, and Patient Positioning
Preoperative planning for a gluteal sarcoma resection demands a meticulous, multimodal imaging review. High-resolution, contrast-enhanced MRI of the pelvis and entire affected femur is the gold standard. The surgeon must scrutinize the T1-weighted images for precise anatomical delineation of the tumor within the muscle bellies and the T2-weighted/STIR images to assess peritumoral edema, which may harbor microscopic disease and dictate the extent of the wide margin. The relationship of the tumor to the sciatic nerve, the greater sciatic notch, and the sacrospinous/sacrotuberous ligaments must be mapped millimeter by millimeter. A non-contrast CT scan of the chest is mandatory for pulmonary staging, while a PET-CT or bone scintigraphy is utilized to rule out osseous metastases or direct bony invasion not readily apparent on MRI.
The architectural blueprint of the surgery begins with the biopsy. As a cardinal rule of orthopedic oncology, the surgeon who will perform the definitive resection should ideally perform or directly supervise the core needle biopsy. The biopsy tract must be placed longitudinally, in line with the planned definitive posterior incision, and strictly within the anatomical compartment of the tumor. Trans-compartmental biopsies or those placed too medially near the anal sphincter or ischiorectal fossa can disastrously compromise the ability to perform a limb-sparing procedure. The tract is tattooed or meticulously documented to ensure its inclusion in the final en bloc resection.

Patient positioning is a critical intraoperative variable that directly impacts surgical exposure and patient safety. Following the induction of general endotracheal anesthesia and the placement of a Foley catheter and arterial line, the patient is transitioned to the lateral decubitus position. The affected side is oriented superiorly. We utilize a rigid beanbag positioner and kidney rests at the level of the ASIS and the sacrum to ensure absolute stability, as the table will frequently be tilted during the procedure to optimize visualization of the deep pelvic structures. An axillary roll is placed to protect the dependent brachial plexus, and all bony prominences (fibular head, lateral malleolus) are meticulously padded.
The prepping and draping protocol must allow for maximum intraoperative flexibility. The entire affected lower extremity, from the costal margin and midline of the sacrum down to the toes, is prepped with a chlorhexidine-based solution and draped free. A sterile tourniquet is not applicable in this region. Draping the leg free is indispensable; it allows the surgical assistant to manipulate the limb—flexing, extending, and rotating the hip—which dynamically alters the tension on the gluteus maximus and the sciatic nerve, vastly improving exposure deep within the retrogluteal space. Intraoperative neuromonitoring, including somatosensory evoked potentials (SSEPs) and transcranial motor evoked potentials (TcMEPs) of the sciatic nerve distributions, is highly recommended to provide real-time feedback during tumor dissection adjacent to the nerve.
Step-by-Step Surgical Approach and Fixation Technique
Incision Design and Fasciocutaneous Flap Elevation
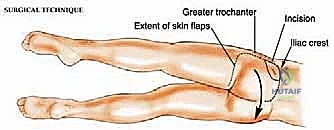
The procedure commences with the design of a generous, curvilinear incision tailored to encompass the tumor, the biopsy tract, and provide access to the expansive gluteal origins and insertions. The incision begins at the posterior third of the iliac crest, curving inferiorly and laterally along the anterior border of the gluteus maximus. It encompasses the prior biopsy site with a minimum 2-3 cm elliptical margin. The incision then traverses over the greater trochanter, extending 5-8 cm distally along the lateral thigh, before sweeping posteriorly and medially to parallel the gluteal fold. This extensive "question mark" or "hockey stick" configuration is non-negotiable; inadequate incisions lead to forceful retraction, tissue ischemia, and compromised oncologic margins.
Using electrocautery, the dissection proceeds through the skin and subcutaneous adipose tissue. The objective is to elevate a massive, robust posterior fasciocutaneous flap. The surgeon must maintain a precise depth, dissecting just superficial to the deep epimysial fascia of the gluteus maximus. If the preoperative MRI demonstrates tumor proximity to the superficial fascia, the dissection plane must be moved more superficially into the subcutaneous fat to ensure a healthy cuff of normal tissue remains on the tumor specimen. Gentle, broad traction utilizing rakes or penetrating towel clips is essential to elevate the flap medially toward the sacrum, exposing the entire posterior envelope of the gluteus maximus muscle while preserving the subdermal vascular plexus to prevent postoperative flap necrosis.
Distal Mobilization and Retrogluteal Space Exposure
Once the superficial aspect of the gluteus maximus is entirely exposed, the mobilization phase begins distally. The vast insertion of the gluteus maximus into the iliotibial (IT) tract is identified. Using a combination of electrocautery and Mayo scissors, the surgeon incises the IT band longitudinally, effectively releasing the lateral and superior fibers of the muscle. Attention is then directed to the deeper, inferior fibers that insert directly onto the gluteal tuberosity of the proximal femur. This tendinous insertion is sharply detached, taking care to identify and coagulate the robust perforating branches of the profunda femoris artery that frequently arborize in this region.
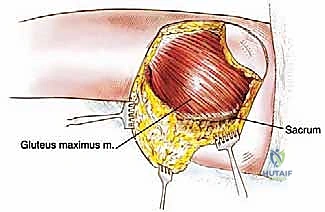
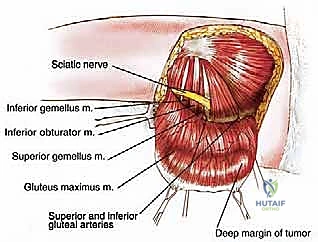
With the distal insertions completely released, the surgeon can now place a hand deep to the inferior border of the gluteus maximus, gently elevating the muscle belly superiorly and medially. This maneuver opens the retrogluteal space. At this juncture, initial blunt palpation is performed to assess the tumor's mobility and its spatial relationship to the underlying structures. The deep fascial plane separating the gluteus maximus from the short external rotators (piriformis, gemelli, obturator internus, quadratus femoris) is developed using blunt finger dissection and judicious use of electrocautery.
Neurovascular Control and Sciatic Nerve Protection
As the gluteus maximus is reflected medially, the critical neurovascular pedicles are encountered. The inferior gluteal artery, vein, and nerve reliably emerge from the greater sciatic foramen, inferior to the piriformis, and dive directly into the deep, central surface of the gluteus maximus. These structures are meticulously skeletonized using a right-angle clamp and blunt dissecting peanuts. The vessels are doubly ligated with heavy non-absorbable silk sutures (e.g., 0 or 2-0 silk) and divided. The inferior gluteal nerve is similarly transected. If the tumor involves the superior aspect of the muscle, the superior gluteal vessels, exiting above the piriformis, must also be identified, ligated, and divided in a similar fashion.
The overriding priority now shifts to the absolute protection of the sciatic nerve. Having been identified at its exit point below the piriformis, the nerve is traced distally throughout the extent of the operative field. It is gently neurolysed from surrounding areolar tissue using blunt dissection. Vascular loops or a soft Penrose drain can be passed around the nerve to allow for gentle, atraumatic retraction. Under no circumstances should sharp dissection or aggressive electrocautery be used in direct proximity to the nerve. A broad, malleable retractor is placed to shield the nerve anteriorly and medially, creating a safe corridor for the final, aggressive detachment of the tumor-bearing muscle from its medial origins.
Proximal Detachment and En Bloc Resection
With the sciatic nerve visually secured and protected, the final and most physically demanding phase of the resection occurs: detaching the gluteus maximus from its robust medial origins. The muscle is retracted laterally, placing the medial attachments under tension. Using high-setting electrocautery, the surgeon systematically releases the muscle from the sacrotuberous and sacrospinous ligaments. Extreme caution is required here; penetrating too deeply through these ligaments risks entering the ischiorectal fossa or the true pelvis, potentially injuring the pudendal neurovascular bundle or causing catastrophic pelvic bleeding.
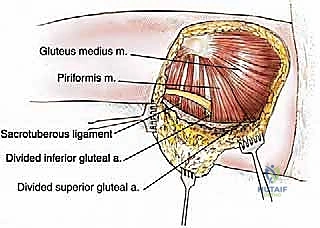
The dissection continues superiorly, detaching the aponeurotic origins from the posterior surface of the sacrum and the coccyx. The periosteum of the sacrum may be taken en bloc with the specimen if the tumor abuts the bone. Finally, the broad origin along the posterior gluteal line of the iliac crest is sharply incised. Throughout this process, the surgeon must continuously verify that the deep margin remains intact and that the tumor pseudocapsule is not inadvertently breached. Once the final attachments to the ilium and the thoracolumbar fascia are released, the entire gluteus maximus, containing the sarcoma and the isolated biopsy tract, is delivered en bloc from the surgical field.
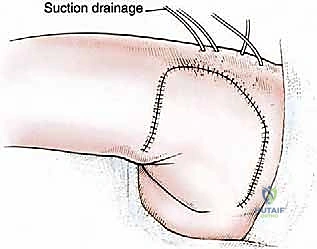
Defect Reconstruction and Wound Closure
Following specimen removal, the massive surgical defect is rigorously irrigated with sterile saline or an oncologic irrigant (e.g., sterile water to induce hypotonic lysis of stray cells, though evidence is mixed). Meticulous hemostasis is achieved using argon beam coagulation and bipolar electrocautery, paying special attention to the venous plexuses near the sacrum and the muscular branches of the external rotators. The surgical margins are oriented with sutures or clips for the pathologist, and frozen sections may be sent from any suspicious areas of the tumor bed, particularly adjacent to the sciatic nerve or the sacral periosteum.
Closure of a gluteus maximus resection defect is notoriously challenging due to the immense dead space created. Two to three large-bore (e.g., 15 French or 19 French) closed-suction fluted drains are placed deep within the retrogluteal space and brought out through separate stab incisions distally and laterally. The massive fasciocutaneous flap is then advanced over the defect. In cases of significant skin resection, local advancement flaps or even pedicled myocutaneous flaps (e.g., V-Y advancement, or utilizing the tensor fasciae latae) may be required, necessitating plastic surgery collaboration. The deep dermal layers are closed with interrupted, heavy absorbable sutures (e.g., 2-0 Vicryl) to alleviate tension on the skin edges, followed by a meticulous skin closure using surgical staples or non-absorbable monofilament. A bulky, compressive posterior dressing is applied to minimize seroma formation.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, limb-sparing gluteus maximus resections carry a substantial complication profile, heavily influenced by the massive creation of dead space and the frequent use of neoadjuvant radiation therapy. Wound complications are the most pervasive issue. Radiation-induced endarteritis compromises the microvascular supply to the massive fasciocutaneous flap, leading to delayed healing, marginal necrosis, or frank wound dehiscence. When dehiscence occurs, it exposes the deep retrogluteal space and potentially the sciatic nerve to desiccation and polymicrobial infection. Aggressive early intervention is required, including operative debridement, negative pressure wound therapy (NPWT), and frequently, the mobilization of rotational flaps for definitive coverage.
Seroma formation is a near-universal phenomenon following this procedure, given the extensive lymphatic disruption and the massive potential space left by the excised muscle. While closed-suction drains mitigate this initially, prolonged drainage is common. Premature drain removal inevitably leads to a tense, symptomatic seroma that can compromise flap perfusion or become secondarily infected. Management involves maintaining drains until output is minimal (typically <30 cc per 24 hours), which may take weeks. Persistent, sterile seromas post-drain removal may require serial ultrasound-guided aspirations or, rarely, operative sclerosis or marsupialization.
Neurologic complications, specifically sciatic nerve dysfunction, represent a devastating functional setback. While direct transection is rare in experienced hands, traction neurapraxia due to prolonged retraction or thermal injury from adjacent electrocautery is a recognized risk. Patients may present with foot drop (peroneal division is more susceptible) or sensory deficits in the distal extremity. Most neurapraxic injuries recover over 3 to 6 months with supportive care, including ankle-foot orthoses (AFOs) and intensive physical therapy. However, permanent deficits require long-term orthotic management or consideration of tendon transfers (e.g., posterior tibial tendon transfer for foot drop).
Oncologic failure, primarily local recurrence, remains the most dreaded complication. Despite attempting wide margins, the complex three-dimensional anatomy of the pelvis and the microscopic infiltrative nature of high-grade sarcomas result in a local recurrence rate of approximately 10-15%. Recurrences in the irradiated, fibrotic tumor bed are notoriously difficult to manage and frequently necessitate radical salvage procedures, including hemipelvectomy, to achieve definitive local control, highlighting the absolute critical nature of the index operation.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage Management / Mitigation |
|---|---|---|---|
| Wound Dehiscence / Necrosis | 15% - 30% | Neoadjuvant radiation, smoking, excessive flap tension, obesity. | NPWT, serial debridement, rotational/pedicled flap coverage. |
| Prolonged Seroma | 40% - 60% | Massive dead space, lymphatic disruption, early drain removal. | Prolonged drain retention, US-guided aspiration, compressive garments. |
| Sciatic Nerve Neurapraxia | 5% - 10% | Traction injury, thermal spread, hematoma compression. | AFO bracing, physical therapy, gabapentinoids; usually self-resolving. |
| Local Tumor Recurrence | 10% - 15% | R1/R2 margins, high histologic grade, lack of adjuvant radiation. | Re-resection, salvage hemipelvectomy, systemic targeted therapies. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following a total gluteus maximus resection is a protracted, multi-phased process designed to protect the fragile soft tissue reconstruction while progressively restoring ambulatory function. The immediate postoperative phase (Days 0-7) is strictly focused on wound protection and hematoma/seroma prevention. The patient is typically managed on an air-fluidized or pressure-relieving mattress. Direct pressure on the posterior gluteal flap is strictly minimized; patients are nursed in the lateral decubitus or prone positions. Hip flexion is restricted to less than 45-60 degrees to prevent excessive tension on the posterior skin closure. Ambulation is initiated early, usually on postoperative day 1 or 2, but requires a walker or crutches with weight-bearing as tolerated, emphasizing a stiff-legged gait to avoid stretching the posterior wound.
Phase I of rehabilitation (Weeks 1-4) continues to prioritize wound healing. As the drains are removed and the incision consolidates, physical therapy focuses on gentle, passive, and active-assisted range of motion (ROM) of the hip, knee, and ankle. Active hip extension is strictly prohibited to avoid disrupting the healing deep tissues, although the primary extensor has been removed. Gait training focuses on safety and the use of assistive devices. Patients are educated on the biomechanical reality of their resection: they will experience a "gluteus maximus lurch," a compensatory mechanism where the patient thrusts the trunk posteriorly immediately after heel strike to maintain the center of gravity behind the hip axis, preventing the hip from collapsing into flexion.
Phase II (Weeks 4-8) marks the transition to active strengthening and functional compensation. With the wound fully healed, the focus shifts to hypertrophying the remaining hip musculature to compensate for the absent gluteus maximus. Intensive strengthening of the hamstrings (the primary secondary hip extensors) and the adductor magnus is initiated. Core stabilization exercises are critical, as a strong lumbar and abdominal core helps control the compensatory trunk movements during gait. Closed kinetic chain exercises, such as modified shallow squats and leg presses, are gradually introduced. Patients are transitioned from a walker to a cane, with the goal of independent, level-ground ambulation.
Phase III (Weeks 8 and beyond) represents the advanced functional restoration phase. Patients engage in advanced gait training, specifically addressing the challenges of ascending stairs, rising from low chairs, and walking on inclines—activities that heavily rely on forceful hip extension. Proprioceptive training and balance exercises are incorporated. While patients will permanently lack the explosive hip extension required for sprinting or heavy lifting, a dedicated adherence to this phased protocol allows the vast majority of patients to return to their baseline activities of daily living and recreational walking with minimal to no assistive devices, demonstrating the remarkable compensatory capacity of the human musculoskeletal system.
Summary of Landmark Literature and Clinical Guidelines
The conceptual framework for limb-sparing surgery in orthopedic oncology is deeply rooted in the pioneering work of William Enneking in the 1980s. Enneking's surgical staging system for musculoskeletal sarcomas established the fundamental definitions of surgical margins—intralesional, marginal, wide, and radical. His seminal papers demonstrated that a wide en bloc resection, achieving a continuous cuff of normal tissue around the tumor, yielded local control rates comparable to radical amputation for extremity sarcomas. This work provided the oncologic justification for procedures
