Limb-Sparing Adductor Compartment Resection: An Intraoperative Masterclass
Key Takeaway
Join us in the OR for a masterclass on adductor compartment resection. We'll navigate complex anatomy, meticulously dissect around neurovascular structures, and perform en bloc tumor removal for sarcomas. Learn critical techniques for vascular reconstruction, ensuring optimal limb preservation and function, while proactively managing potential complications from incision to rehabilitation.
Comprehensive Introduction and Patho-Epidemiology
Welcome, fellows and colleagues. Today, we embark on a profound exploration of one of the most demanding procedures in orthopedic oncology: the limb-sparing resection of a high-grade soft tissue sarcoma originating within the adductor compartment of the thigh. This procedure transcends the basic mechanics of surgical excision; it represents a meticulous, high-stakes dissection, a precise anatomical dance around vital neurovascular structures, and a testament to our unwavering commitment to preserving maximum limb function while achieving definitive oncologic control. The adductor compartment represents the second most common anatomic site for soft tissue tumors in the lower extremity, surpassed only by the anterior quadriceps compartment. Because of the deep, expansive nature of this fascial space, these tumors often present insidiously, growing to surprisingly massive proportions before clinical detection.
Understanding the patho-epidemiology of these lesions is the cornerstone of our surgical rationale. Soft tissue sarcomas (STS) are a heterogeneous group of rare mesenchymal malignancies. In the adductor compartment, we frequently encounter undifferentiated pleomorphic sarcomas (UPS), myxoid and dedifferentiated liposarcomas, and synovial sarcomas. The biological behavior of these tumors dictates our surgical margins. Unlike carcinomas, sarcomas expand by compressing surrounding normal tissue, forming a "pseudocapsule." This pseudocapsule is a deceptive boundary; it is invariably infiltrated with microscopic satellite tumor cells. Therefore, a simple "shelling out" or marginal excision is oncologically unacceptable and guarantees local recurrence. Our goal is a wide, en bloc resection, taking a cuff of normal, healthy tissue in all dimensions to achieve R0 (microscopically negative) margins.
The modern paradigm of sarcoma management is inherently multidisciplinary. As orthopedic oncologists, we do not operate in a vacuum. The decision to proceed with a limb-sparing adductor resection is made in concert with our colleagues in musculoskeletal radiology, pathology, medical oncology, and radiation oncology. The advent of neoadjuvant radiation therapy has revolutionized our approach, sterilizing the reactive zone and allowing for closer surgical margins adjacent to critical, un-resectable structures like the superficial femoral artery or the sciatic nerve. However, this preoperative radiation significantly alters the surgical landscape, rendering tissues fibrotic, increasing bleeding propensity, and exponentially raising the risk of postoperative wound complications. We must approach the irradiated adductor compartment with profound respect and technical precision.
Detailed Surgical Anatomy and Biomechanics
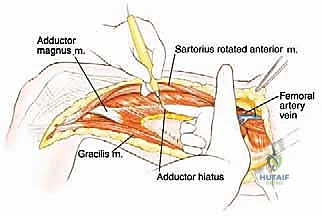
Before we even contemplate the initial skin incision, we must comprehensively re-familiarize ourselves with the intricate, three-dimensional landscape of the medial thigh. The adductor compartment is a complex, robust fascial space, classically conceptualized as an inverted, oblique funnel. Mastery of this anatomy is non-negotiable; it is the map that guides our oncologic resection and prevents catastrophic iatrogenic injury.
The proximal base of this anatomical funnel originates directly from the osseous structures of the pelvic floor and the medial aspect of the ipsilateral pelvic ring. Specifically, the muscular origins span the symphysis pubis, the inferior pubic ramus, the ischial tuberosity, and the dense obturator fascia. The lateral border of the compartment is rigidly defined by the femur, specifically the rough, longitudinal ridge of the linea aspera, where the adductor aponeuroses insert. Distally, the conical space tapers rapidly, terminating at the adductor hiatus—a critical tendinous arch in the adductor magnus through which the superficial femoral vessels transition into the popliteal fossa.

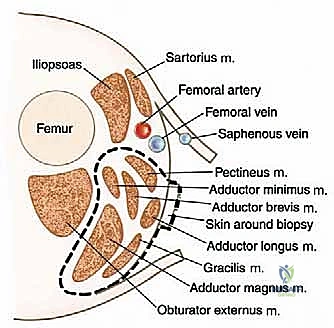
The primary muscular constituents of this compartment are the adductor magnus, adductor brevis, adductor longus, gracilis, and pectineus. The adductor magnus is the largest and most posterior, often described as having both an adductor and a hamstring portion. These muscles are primarily innervated by the obturator nerve (L2-L4), which exits the pelvis through the obturator canal and immediately bifurcates into anterior and posterior divisions, straddling the adductor brevis. In a comprehensive extra-compartmental resection, these muscles, along with their innervating nerve branches, are often sacrificed en bloc.
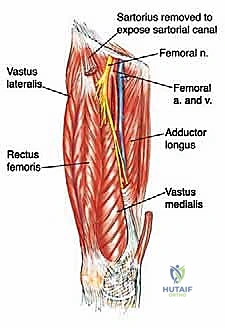
The neurovascular considerations within and adjacent to this compartment are paramount and dictate the feasibility of limb salvage. The superficial femoral artery (SFA) and vein (SFV) course along the anterior and lateral margins of the compartment within the sartorial (Hunter's) canal, essentially forming the anterolateral boundary of our resection. While massive tumors frequently displace these vessels medially or laterally, direct adventitial invasion is less common but must be anticipated. Conversely, the profundus femoris artery (deep femoral artery) is intimately associated with the adductor musculature, frequently diving directly posterior to the adductor longus and passing through or adjacent to the adductor brevis. In high-grade sarcomas of the central adductor compartment, ligation and en bloc resection of the profundus femoris is routinely necessary to achieve adequate deep margins.
From a biomechanical standpoint, the adductor muscles are primary stabilizers of the pelvis and femur during the stance phase of gait and assist in hip flexion and internal rotation during the swing phase. While the loss of the entire adductor compartment seems biomechanically devastating, clinical experience demonstrates that patients compensate remarkably well. The remaining intact muscles—specifically the gluteus maximus, the medial hamstrings, and the iliopsoas—hypertrophy over time to stabilize the pelvis. The primary functional deficit observed post-resection is a mild, persistent weakness in active hip adduction against resistance, but normal, unassisted ambulation is almost universally recovered.
Exhaustive Indications and Contraindications
The decision to proceed with a limb-sparing adductor resection requires a nuanced balance between achieving local oncologic cure and preserving a functional, viable extremity. Not every tumor is amenable to this approach, and recognizing the absolute and relative contraindications is a hallmark of an experienced orthopedic oncologist.
Indications for Resection
The primary indication for a radical adductor compartment resection is the presence of a primary soft tissue sarcoma (e.g., undifferentiated pleomorphic sarcoma, liposarcoma, synovial sarcoma) localized within the medial thigh. The goal is complete extirpation with negative microscopic margins. A secondary indication includes locally aggressive but histologically benign lesions, such as desmoid-type fibromatosis, particularly when they are symptomatic, rapidly enlarging, or threatening critical neurovascular structures. Additionally, massive, symptomatic benign lipomas or isolated, solitary metastatic lesions (e.g., from renal cell carcinoma or melanoma) may warrant resection for palliative pain relief and functional restoration, provided the patient's overall oncologic prognosis justifies the surgical morbidity.
Contraindications to Limb-Sparing Surgery
While limb preservation is our primary objective, amputation (such as a hip disarticulation or high transfemoral amputation) remains a necessary and life-saving alternative in specific, advanced scenarios. Absolute contraindications to limb salvage include complete, circumferential encasement of the major neurovascular bundle (superficial femoral artery, vein, and sciatic nerve) where vascular reconstruction is either anatomically impossible or would result in a functionally useless, insensate limb.
Relative contraindications involve massive tumors with extensive extracompartmental extension into the deep pelvic floor, the ischiorectal fossa, or direct invasion into the osseous structures of the hip joint or ischium, where achieving an R0 margin would require a highly morbid hemipelvectomy. Furthermore, the presence of severe, untreated peripheral vascular disease where the superficial femoral artery is occluded, and the limb relies entirely on the profundus femoris (which must be resected with the tumor), precludes limb salvage without complex, high-risk extra-anatomic bypass grafting.
| Category | Specific Clinical Scenario | Surgical Implication |
|---|---|---|
| Absolute Indication | High-grade STS confined to adductor compartment | Wide local excision / Limb-sparing resection |
| Absolute Indication | Symptomatic, growing desmoid fibromatosis | Marginal to wide excision (depending on morbidity) |
| Relative Contraindication | SFA/SFV encasement > 180 degrees | Consider neoadjuvant therapy; prepare for vascular bypass/reconstruction |
| Relative Contraindication | Extension into medial hamstrings/sciatic nerve | Requires epineural dissection or partial nerve resection with resulting deficit |
| Absolute Contraindication | Complete neurovascular encasement with insensate foot | High transfemoral amputation or Hip disarticulation |
| Absolute Contraindication | Inability to achieve R0 margins despite radical resection | Amputation to prevent fungating local recurrence |
Pre-Operative Planning, Templating, and Patient Positioning
The success of a complex adductor resection is entirely predicated on exhaustive, meticulous preoperative planning. The operation itself should merely be the physical execution of a well-rehearsed mental blueprint.
Imaging and Staging Studies
Our preoperative imaging protocol is comprehensive. Plain radiographs of the femur and pelvis are obtained to rule out subtle periosteal reactions or direct osseous invasion. The cornerstone of our planning, however, is a high-resolution, multi-planar Magnetic Resonance Imaging (MRI) scan of the entire thigh, ipsilateral hip, and hemipelvis, utilizing both T1-weighted and T2-weighted fat-suppressed sequences, with and without intravenous gadolinium contrast. This study delineates the exact dimensions of the tumor, the presence of peritumoral edema (which often harbors microscopic disease), and the precise relationship of the mass to the sartorial canal, the profundus femoris origin, and the ischial tuberosity.
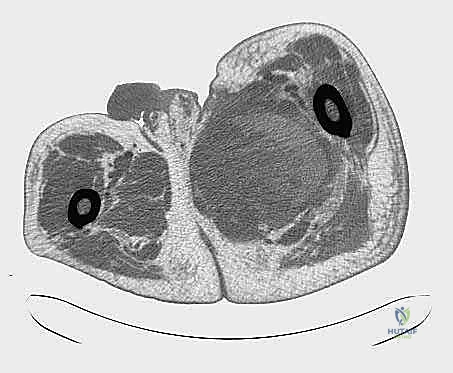
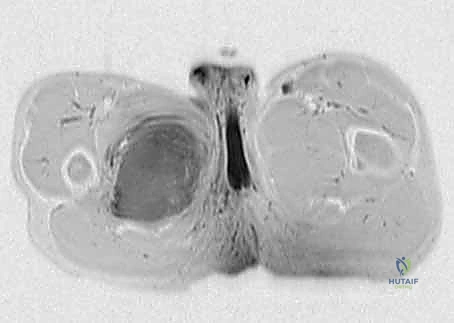
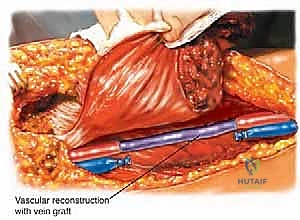
In cases where the tumor abuts or displaces the major vessels, CT Angiography or MR Angiography is mandatory. We must map the vascular supply, specifically evaluating the patency of the superficial femoral artery. If the profundus femoris must be sacrificed, the viability of the limb relies entirely on the SFA. We routinely obtain Ankle-Brachial Indices (ABIs) and duplex ultrasounds to confirm distal perfusion and to map the contralateral greater saphenous vein, ensuring we have a suitable autologous conduit should an interpositional vascular graft become necessary. Furthermore, a staging CT of the chest is non-negotiable to rule out pulmonary metastases, which would fundamentally alter our surgical intent from curative to palliative.
Biopsy Principles and Patient Positioning
The diagnostic biopsy must be performed with surgical precision, ideally by the operating orthopedic oncologist or a highly experienced interventional radiologist under direct consultation. We utilize a core needle biopsy, placing the tract meticulously within the planned surgical incision line. The tract must not traverse uncontaminated fascial planes or the sartorial canal. During the definitive resection, this entire biopsy tract, including the skin entry point, is excised en bloc with the tumor specimen.
For the resection, the patient is positioned supine on a standard radiolucent operating table. The affected lower extremity is placed in a "frog-leg" position—flexed at the hip and knee, abducted, and externally rotated. This posture maximally exposes the medial thigh, the femoral triangle, and the perineal region. A soft, gel bump is placed under the ipsilateral hemipelvis to facilitate this rotation. The surgical prep must be extensive, extending from the costal margin down to the toes, circumferentially around the entire limb, and including the ipsilateral perineum. The contralateral leg is also prepped and draped into the sterile field to allow for immediate access to the contralateral greater saphenous vein if vascular reconstruction is mandated.
Step-by-Step Surgical Approach and Fixation Technique
We are now ready to execute the operative plan. Scalpel, please. The room must be quiet; this dissection requires absolute focus.
Incision and Initial Exposure
We begin with a generous, longitudinal incision to ensure unhindered access to the entire medial thigh and the femoral triangle. The incision originates proximally near the pubic tubercle, just inferior to the inguinal ligament, and extends distally, paralleling the medial border of the sartorius muscle, curving gently toward the posteromedial aspect of the knee. Crucially, the incision is designed as an ellipse around the previous biopsy site, ensuring the contaminated tract remains firmly attached to the underlying tumor mass.
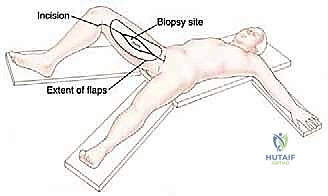
Using a #10 blade, we incise through the skin and subcutaneous fat down to the superficial investing fascia of the thigh. We then elevate massive anterior and posterior fasciocutaneous flaps using electrocautery. The thickness of these flaps is dictated by the tumor's depth; if the tumor is deep within the muscle belly, subfascial flaps are elevated. If the tumor abuts the fascia, the fascia is left attached to the tumor, and suprafascial flaps are developed. The anterior flap is retracted laterally to expose the sartorius muscle and the vastus medialis, while the posterior flap is retracted medially to expose the gracilis and the medial hamstrings.
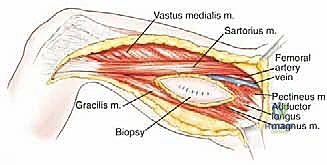
Vascular Dissection and Protection
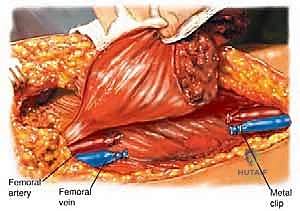
The most critical initial phase of the deep dissection is the identification, isolation, and protection of the superficial femoral neurovascular bundle. We identify the medial border of the sartorius muscle and retract it laterally. This opens the roof of the sartorial (Hunter's) canal. Using meticulous sharp and blunt dissection, we identify the superficial femoral artery and vein. Vessel loops are passed around these structures proximally and distally to maintain control.
We trace the vessels proximally to the bifurcation of the common femoral artery. Here, we identify the origin of the profundus femoris artery. If our preoperative MRI indicates that the tumor involves the adductor brevis or longus near this origin, the profundus must be sacrificed. We carefully dissect the profundus, ligate it securely with heavy silk ties and surgical clips, and transect it. This maneuver immediately devascularizes a significant portion of the adductor compartment and the tumor itself, reducing intraoperative blood loss. We must then carefully dissect the SFA and SFV away from the tumor pseudocapsule, ligating all small, medial-directed muscular branches with surgical clips to prevent avulsion and catastrophic bleeding.
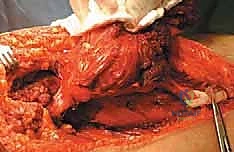

Muscular Mobilization and En Bloc Resection
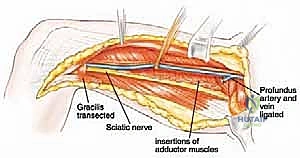
With the major lateral vascular structures protected, we turn our attention to the muscular margins. Proximally, we trace the adductor longus, brevis, and gracilis to their origins on the pubis and ischium. Using electrocautery, we detach these muscles directly off the bone, taking the periosteum if necessary to ensure a wide margin. During this proximal dissection, we encounter the obturator nerve and artery exiting the obturator foramen. These are clamped, ligated, and transected, as they plunge directly into the tumor-bearing compartment.

Posteriorly, we develop the plane between the adductor magnus and the medial hamstrings (semimembranosus and semitendinosus). If the tumor is confined to the adductor compartment, this areolar plane is usually avascular and easily developed. However, if there is extracompartmental extension, a portion of the hamstrings must be taken en bloc. Distally, we detach the adductor insertions from the linea aspera of the femur. We use a combination of electrocautery and a periosteal elevator to strip the muscular aponeurosis off the bone. Finally, we address the adductor hiatus, carefully protecting the popliteal vessels as we release the distal-most extent of the adductor magnus.
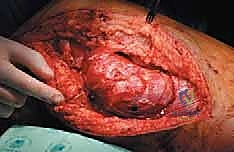

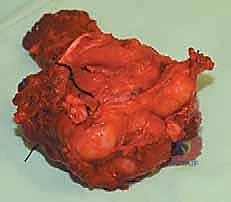
The entire adductor compartment, containing the tumor enveloped in normal muscle, is now free. It is lifted from the surgical bed as a single, contiguous specimen and passed off the field for immediate pathologic orientation and margin assessment. The massive surgical void is then irrigated copiously with sterile saline. Hemostasis is achieved meticulously using electrocautery, argon beam coagulation, and topical hemostatic agents. Closed suction drains are placed deep within the resection bed, and the wound is closed in multiple layers, ensuring a tension-free skin closure to mitigate the risk of necrosis.
Complications, Incidence Rates, and Salvage Management
Despite flawless surgical technique, the radical resection of the adductor compartment—especially in the setting of neoadjuvant radiation—carries a significant risk of postoperative complications. Recognizing these early and implementing aggressive salvage management is vital to preventing limb loss and systemic morbidity.
Wound complications are by far the most frequent adverse event. The combination of large fasciocutaneous flaps, an extensive dead space, and radiation-induced microvascular fibrosis creates a hostile environment for healing. Dehiscence, major skin flap necrosis, and deep surgical site infections occur in 20% to 35% of irradiated patients. Management requires a proactive approach: early recognition of ischemia, aggressive surgical debridement of necrotic tissue, and the liberal use of negative pressure wound therapy (NPWT). Frequently, we must collaborate with our plastic surgery colleagues for pedicled or free flap coverage (e.g., a vertical rectus abdominis myocutaneous flap) to obliterate the dead space and provide healthy, vascularized tissue over exposed vessels.
Vascular and neurologic complications, while less frequent, are infinitely more devastating. Iatrogenic injury to the superficial femoral artery or profound venous thrombosis can lead to acute limb ischemia. If the SFA is injured during dissection, immediate primary repair or interposition bypass grafting with the reversed saphenous vein is mandatory. Postoperative deep vein thrombosis (DVT) is a constant threat due to altered venous hemodynamics and the hypercoagulable state of malignancy; thus, aggressive chemical and mechanical prophylaxis is standard. Neurologically, the intentional sacrifice of the obturator nerve results in permanent numbness over the medial thigh and profound weakness in active hip adduction, though, as previously discussed, functional ambulation is preserved.
| Complication | Estimated Incidence | Prevention & Salvage Management |
|---|---|---|
| Wound Dehiscence / Necrosis | 20% - 35% (higher with XRT) | Tension-free closure, thick flaps, NPWT, Plastic surgery flap coverage |
| Deep Surgical Site Infection | 10% - 15% | Meticulous hemostasis, prolonged deep drains, aggressive I&D, targeted IV antibiotics |
| Deep Vein Thrombosis (DVT) | 5% - 10% | Intraoperative heparin (if vascular repair), post-op LMWH, early mobilization |
| Local Tumor Recurrence | 10% - 15% (at 5 years) | Strict R0 margins, adjuvant XRT. Salvage: Re-resection or Amputation |
| SFA/SFV Injury or Thrombosis | < 5% | Careful adventitial dissection. Salvage: Immediate saphenous vein bypass |
Phased Post-Operative Rehabilitation Protocols
The surgical resection is only the first half of the patient's journey; rigorous, phased postoperative rehabilitation is essential to maximize functional recovery and integrate the biomechanical compensations required after the loss of the adductor compartment.
Phase I: Immediate Post-Operative Period (Weeks 0-2)
The primary goals in the immediate postoperative phase are the promotion of wound healing, the prevention of deep vein thrombosis, and the management of acute pain. Patients are typically kept on bed rest or limited to chair transfers for the first 24 to 48 hours to prevent hematoma formation in the massive surgical dead space. Closed suction drains remain in place until output is minimal (typically less than 30cc per 24 hours). Gentle, passive range of motion (ROM) of the hip and knee is initiated early to prevent joint contractures, but active adduction is strictly prohibited to avoid placing tension on the deep fascial closures. Weight-bearing status is generally dictated by the extent of the resection; if the femur was not compromised, patients are usually allowed touchdown weight-bearing with a walker.
Phase II: Intermediate Mobilization (Weeks 2-6)
Once the surgical incisions have healed and the sutures or staples are removed, the focus shifts to progressive mobilization and gait training. Weight-bearing is advanced to tolerance. Patients will demonstrate a noticeable functional deficit—specifically, a tendency for the hip to abduct during the swing phase of gait due to the unopposed pull of the gluteus medius and minimus. Physical therapy focuses heavily on training the compensatory musculature. The medial hamstrings, the pectineus (if spared), and the lower fibers of the gluteus maximus are targeted with isometric and progressive resistive exercises to stabilize the pelvis and assist in bringing the leg back to the midline during ambulation.
Phase III: Advanced Strengthening and Return to Function (Weeks 6-12+)
In the final phase of rehabilitation, the emphasis is on maximizing strength, endurance, and proprioception. Patients engage in closed-kinetic-chain exercises, balance training, and advanced gait mechanics. While active, forceful adduction against resistance (e.g., squeezing the knees together against a heavy band) will remain permanently weakened, patients are trained to utilize core stability and pelvic tilt to compensate. By 12 weeks, the vast majority of patients are ambulating independently without assistive devices and can return to their baseline activities of daily living, bearing witness to the remarkable adaptability of the human musculoskeletal system following limb-sparing oncologic surgery.
Summary of Landmark Literature and Clinical Guidelines
Our current surgical protocols are not born of anecdote; they are deeply rooted in decades of rigorous oncologic research and landmark clinical trials. Understanding this literature is vital for any practicing orthopedic oncologist.
The foundational shift from routine amputation to limb-sparing surgery for extremity soft tissue sarcomas was solidified by the landmark prospective randomized trial conducted by Rosenberg et al. at the National Cancer Institute in the early 1980s. This study definitively proved that wide local excision paired with adjuvant radiation therapy yielded overall survival rates identical to those of radical amputation, forever altering the standard of care. This principle remains the bedrock of the National Comprehensive Cancer Network (NCCN) guidelines, which strongly advocate for limb-sparing surgery whenever R0 margins can be achieved without rendering the limb non-functional.
Furthermore, the timing of radiation therapy—a critical factor in our adductor resections—was elegantly addressed by the Canadian Sarcoma Group trial led by O'Sullivan et al. This randomized study compared preoperative versus postoperative radiation for extremity sarcomas. The data clearly demonstrated that while preoperative radiation significantly increased the risk of acute wound complications (a reality we face constantly in medial thigh resections), it provided superior long-term functional outcomes by reducing the required radiation field size and minimizing late tissue fibrosis and joint contracture. Consequently, preoperative neoadjuvant radiation followed by wide surgical resection has become the preferred treatment paradigm for high-grade, deep-seated adductor sarcomas at major academic centers worldwide.
