Elbow Contracture Release: A Masterclass in Lateral Columnar Technique
Key Takeaway
Step into the operating theater for a comprehensive masterclass on lateral columnar release for extrinsic elbow contracture. We'll meticulously detail preoperative planning, precise surgical anatomy, and granular intraoperative execution from incision to closure. Learn to navigate neurovascular risks, manage heterotopic ossification, and ensure optimal outcomes. This guide covers pearls, pitfalls, and postoperative rehabilitation, providing a complete understanding of this challenging procedure.
Comprehensive Introduction and Patho-Epidemiology
My fellows, welcome to the operating theater. Today, we are undertaking a technically demanding yet profoundly rewarding procedure: a Lateral Columnar Release for Extrinsic Elbow Contracture. This patient presents with significant post-traumatic elbow stiffness, a ubiquitous and debilitating sequela of elbow trauma that can severely impact a patient's independence and quality of life. Our definitive surgical goal is to restore a functional arc of motion. As defined by the seminal biomechanical studies of Morrey and colleagues, the functional arc typically requires 30 to 130 degrees of flexion-extension and 50 degrees of both pronation and supination. Restoring this specific arc is paramount, as it accommodates the vast majority of activities of daily living, from personal hygiene to feeding.
To effectively treat this pathology, we must first deeply understand the pathophysiology driving it. Elbow stiffness, particularly extrinsic contracture, is invariably initiated by a cascade of soft tissue trauma, intra-articular hemorrhage, and a hyperactive inflammatory response. Imagine this biochemical cascade: a mechanical injury causes tearing and contusion of the highly vascular periarticular tissues, leading to immediate hematoma formation within the joint space and surrounding fascial planes. The patient instinctively splints the elbow in a flexed, guarded position—typically around 70 to 80 degrees—to maximize intra-articular volume and alleviate capsular distension pain. This protective posture, while initially beneficial for pain control, catalyzes a vicious cycle of immobility and fibrosis.
Within that organizing hematoma and traumatized muscle bed, a robust fibroblastic and myofibroblastic tissue response ensues. Driven by inflammatory cytokines such as Transforming Growth Factor-beta (TGF-β) and Platelet-Derived Growth Factor (PDGF), this cellular infiltrate rapidly synthesizes disorganized Type III collagen, which later remodels into dense, unyielding Type I collagen. Over time, this fibrous tissue profoundly thickens the joint capsule, rendering it non-compliant. In severe cases, this reactive stroma can ossify, forming heterotopic bone that physically blocks the joint's mechanical excursion. It is critical to note that overly aggressive early physical therapy, characterized by painful passive stretching, can actually exacerbate this inflammatory process, inciting micro-tearing of the nascent scar tissue and further potentiating the cycle of pain, swelling, and frank contracture.
Extrinsic contracture specifically refers to stiffness primarily resulting from this fibrosis, thickening, and ossification of the elbow capsule and the surrounding periarticular soft tissues. Crucially, in a pure extrinsic contracture, the articular cartilage itself remains either minimally involved or entirely pristine. This delineates it from intrinsic contracture, wherein intra-articular adhesions, osteochondral defects, or frank cartilage destruction are the primary drivers of stiffness. While we make this rigid distinction for conceptual clarity and preoperative planning, it is imperative to recognize that these entities frequently overlap in clinical practice, particularly in cases of complex intra-articular fracture-dislocations where both capsular scarring and articular incongruity coexist.
Detailed Surgical Anatomy and Biomechanics
The elbow is a marvel of biomechanical engineering, functioning as a compound uniaxial synovial joint comprising three highly congruous articulations enveloped within a single, continuous capsule. Understanding the intricate interplay of these articulations is non-negotiable for safe and effective surgical release. The ulnohumeral joint is our primary ginglymus, or hinge joint, dictating the majority of flexion and extension. Its inherent osseous stability is derived from the highly constrained articulation between the spool-shaped trochlea of the humerus and the greater sigmoid notch of the ulna. The radiocapitellar joint functions as a gliding joint between the concave radial head and the convex capitellum, contributing to both flexion/extension and the complex mechanics of forearm rotation. Finally, the proximal radioulnar joint (PRUJ) allows for the pivotal pronation and supination of the forearm.
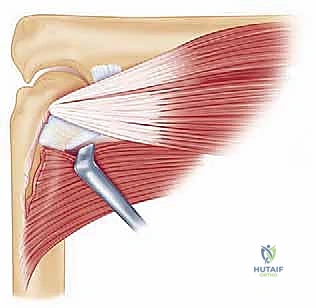
These osseous articulations are dynamically and statically stabilized by an intricate network of intracapsular ligaments and overlying extracapsular musculature. The collateral ligament complexes are the primary static stabilizers. The medial ulnar collateral ligament (MUCL), specifically its anterior bundle, is the primary restraint to valgus stress. Conversely, the lateral collateral ligament (LCL) complex, and most critically the lateral ulnar collateral ligament (LUCL), restrains varus and posterolateral rotatory forces. Injury to these ligaments, or secondary fibrosis resulting from prolonged immobilization, contributes significantly to capsular contracture. During our lateral columnar approach, we must be exceptionally vigilant to preserve the origin and insertion of the LUCL; iatrogenic transection during capsulectomy will invariably result in posterolateral rotatory instability (PLRI), a devastating complication that converts a stiff elbow into an unstable, painful one.
The periarticular musculature and the joint capsule itself are the primary anatomical structures we must manipulate and release. Significant injury to the anterior joint capsule and the overlying brachialis muscle leads to profound capsular hypertrophy and fibrotic tethering, a primary cause of extension loss. The brachialis muscle can undergo myostatic contracture, acting as a formidable tether. Posteriorly, the triceps muscle and its tendinous insertion can become densely adherent to the posterior humerus and the olecranon fossa, restricting terminal flexion. The common extensor origin on the lateral epicondyle—consisting of the extensor carpi radialis longus (ECRL), extensor carpi radialis brevis (ECRB), extensor digitorum communis (EDC), and extensor carpi ulnaris (ECU)—serves as our anatomical roadmap. Our lateral approach will meticulously navigate these intervals, specifically utilizing the internervous plane between the anconeus (radial nerve) and ECU (posterior interosseous nerve), as well as the interval between the ECRL and ECRB/EDC.
Navigating the neurovascular topography is the most perilous aspect of this procedure. The ulnar nerve, residing in the cubital tunnel posterior to the medial epicondyle, is highly susceptible to traction injury. As we release the posterior and anterior structures and rapidly restore flexion, the ulnar nerve can experience acute stretch. Preoperative ulnar neuropathy or a severe flexion contracture (less than 100 degrees of preoperative flexion) mandates a prophylactic ulnar nerve neurolysis and anterior transposition. Laterally, the radial nerve bifurcates into its superficial sensory and deep motor (Posterior Interosseous Nerve - PIN) branches anterior to the radiocapitellar joint. The PIN wraps around the radial neck within the supinator muscle. During distal and anterior extracapsular dissection, the PIN is at extreme risk. Retractors must be placed with precision, avoiding blind plunging distal to the annular ligament. Anteriorly, the median nerve and brachial artery are typically shielded by the robust belly of the brachialis muscle; maintaining dissection strictly between the anterior capsule and the brachialis muscle guarantees their safety.
Exhaustive Indications and Contraindications
Patient selection is the cornerstone of successful elbow contracture release. The decision to proceed to the operating room must be predicated on a comprehensive evaluation of the patient's functional deficit, their response to conservative measures, and their psychological readiness for the grueling postoperative rehabilitation.
Indications for Lateral Columnar Release
The primary indication for surgical intervention is a persistent, functionally limiting elbow contracture that has proven refractory to a dedicated, protracted course of nonoperative management. Typically, we require patients to have undergone at least 3 to 6 months of rigorous conservative therapy, including static progressive splinting, active-assisted range of motion exercises, and judicious use of NSAIDs. Functionally, patients generally seek surgical intervention when their extension loss exceeds 30 to 40 degrees, or when their maximum flexion is restricted to less than 115 to 120 degrees, as these deficits severely impede reaching the face, head, and perineum. The lateral columnar approach is specifically indicated for extrinsic contractures characterized by anterior and posterior capsular fibrosis, mild to moderate heterotopic ossification localized to the lateral or posterior compartments, and post-traumatic stiffness where the articular surfaces remain largely congruous and viable.
Contraindications to Surgical Release
Contraindications must be rigorously respected to avoid catastrophic outcomes. Absolute contraindications include active local or systemic infection, which must be eradicated prior to any elective reconstructive procedure. The presence of acute, fulminant Complex Regional Pain Syndrome (CRPS) is an absolute contraindication; operating during the hyperalgesic, inflammatory phase of CRPS will invariably result in a massive flare of the disease, leading to profound, irreversible stiffness and pain. Severe intrinsic joint destruction, such as end-stage post-traumatic osteoarthritis or advanced inflammatory arthropathy with complete loss of the articular cartilage space, renders a soft-tissue release futile. In these scenarios, the patient is better served by an interposition arthroplasty or a total elbow arthroplasty, depending on their age and functional demands.
Relative Contraindications and Patient Factors
Relative contraindications require nuanced clinical judgment. A non-compliant patient, or one lacking the psychological resilience to endure the painful, daily postoperative rehabilitation protocol, is a poor candidate. The success of an elbow release is arguably 50% surgery and 50% postoperative therapy; without the latter, the former is doomed to fail. Furthermore, extensive, bridging heterotopic ossification that completely encases the joint (ankylosis) may require a more extensile, combined medial and lateral approach, or a posterior universal approach, rather than an isolated lateral columnar release. Severe, unaddressed ulnar neuropathy should also prompt a combined approach to allow for simultaneous nerve transposition.
| Category | Specific Factors | Clinical Implications |
|---|---|---|
| Primary Indications | Failure of 3-6 months conservative Tx | Splinting/PT plateaued; no further ROM gains. |
| Extension loss > 40° | Impedes reaching, lifting, and spatial hand positioning. | |
| Flexion < 120° | Impedes feeding, hygiene, and grooming. | |
| Extrinsic capsular fibrosis | Ideal pathology for lateral columnar capsulectomy. | |
| Absolute Contraindications | Active joint space infection | High risk of systemic sepsis and hardware/tissue failure. |
| Acute phase CRPS | Surgical trauma exacerbates sympathetic dystrophy. | |
| End-stage articular destruction | Requires arthroplasty (TEA or interposition), not release. | |
| Relative Contraindications | Non-compliant patient | Post-op rehab is grueling; failure to comply guarantees recurrent stiffness. |
| Massive bridging HO | May require extensile posterior/combined approaches. | |
| Severe untreated ulnar neuropathy | Requires simultaneous medial approach for transposition. |
Pre-Operative Planning, Templating, and Patient Positioning
Before a scalpel ever touches the skin, meticulous and exhaustive preoperative planning is paramount. This begins with a granular patient history, elucidating the specific etiology of the contracture—be it a simple radial head fracture, a complex terrible triad injury, a burn, or a closed head injury (which carries a notoriously high risk of aggressive heterotopic ossification). We must document the exact duration and progression of symptoms, all prior therapeutic modalities, and the presence of any retained orthopedic hardware that may require concurrent removal.
The physical examination is rigorous. Goniometric measurement of both active and passive motion is recorded to establish a baseline. We meticulously check for changes in ulnohumeral motion with varying degrees of forearm pronation and supination; increased extension in supination compared to pronation may suggest subtle posterolateral rotatory instability—a critical finding that alters our surgical plan. A comprehensive neurologic examination is mandatory. We assess for a positive Tinel's sign over the cubital tunnel, perform the elbow flexion test, and evaluate intrinsic hand strength to rule out subclinical ulnar neuropathy.
Imaging serves as our architectural blueprint. Routine Anteroposterior (AP) and Lateral radiographs are the foundational workhorses, providing a gross assessment of joint congruity, joint space narrowing, and the presence of prominent osteophytes or heterotopic bone.
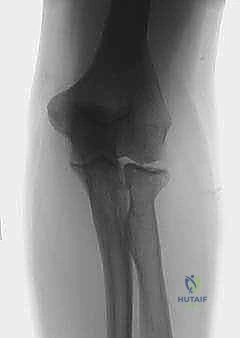

However, plain films are often insufficient for complex contractures. A 3D Computed Tomography (CT) scan is invaluable and routinely obtained in my practice. It provides exquisite cross-sectional detail, allowing us to precisely map the topography of heterotopic ossification, identify hidden osteophytes impinging within the coronoid or olecranon fossae, and assess the true integrity of the articular surfaces. If the patient reports severe pain at rest or pain throughout the remaining arc of motion—symptoms atypical for a pure extrinsic contracture—a serologic workup including a Complete Blood Count (CBC), Erythrocyte Sedimentation Rate (ESR), and C-Reactive Protein (CRP) is strictly indicated to rule out indolent infection or inflammatory arthropathy.
Patient positioning and anesthesia are critical logistical steps. The procedure can be performed under general anesthesia, but I strongly advocate for the addition of an indwelling continuous regional nerve block (e.g., supraclavicular or infraclavicular catheter). This provides profound intraoperative muscle relaxation and, more importantly, facilitates aggressive, pain-free continuous passive motion (CPM) in the immediate postoperative period. The patient is positioned supine on the operating table. The involved arm is placed on a radiolucent hand table. Crucially, the patient's torso must be brought to the absolute edge of the operating table. This specific positioning allows the arm to drop into maximal extension without being blocked by the table mattress, and it provides unimpeded access for the C-arm fluoroscope to obtain true lateral views during the procedure. A sterile tourniquet is applied high on the brachium to ensure a bloodless field, which is essential for identifying the delicate neurovascular structures and fascial intervals.
Step-by-Step Surgical Approach and Fixation Technique
We utilize the extended lateral columnar approach, a highly versatile exposure that provides sequential access to the posterior, anterior, and lateral compartments of the elbow without disrupting the primary stabilizing ligaments or the flexor-pronator mass.
Skin Incision and Superficial Dissection
The skin incision is curvilinear, measuring approximately 10 to 12 centimeters. It originates 5 centimeters proximal to the lateral epicondyle, following the lateral supracondylar ridge of the humerus. It passes slightly anterior to the lateral epicondyle and curves distally, aligning with the anatomic interval between the anconeus muscle posteriorly and the extensor carpi ulnaris (ECU) anteriorly.
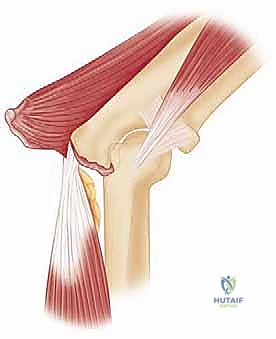
Using a #15 blade, the skin and subcutaneous tissues are incised. Meticulous hemostasis is achieved immediately with bipolar electrocautery to maintain pristine visualization. Full-thickness fasciocutaneous flaps are raised anteriorly and posteriorly. It is critical to maintain the integrity of these flaps to prevent postoperative skin necrosis, a disastrous complication over the lateral epicondyle.
The Posterior Release
We systematically address the posterior compartment first, as gaining extension is often the primary goal and posterior structures act as significant tethers. We identify the Köcher interval between the anconeus and the ECU.
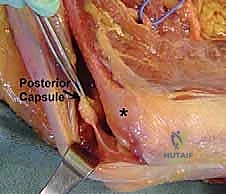
Using blunt Metzenbaum scissors and precise sharp dissection, we split the deep fascia along this interval. The anconeus is elevated subperiosteally off the lateral ulna and reflected posteriorly, remaining in continuity with the lateral head of the triceps. This maneuver dramatically exposes the posterior joint capsule and the radiocapitellar joint.
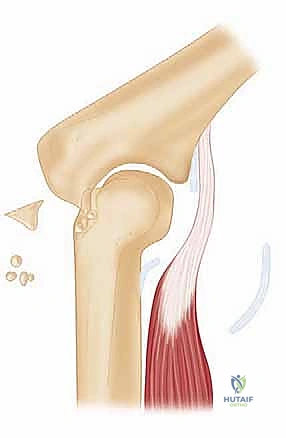
We incise the posterior capsule, which is often thickened to the consistency of shoe leather. Using a combination of rongeurs and a high-speed burr, we meticulously clear the olecranon fossa of all fibrotic scar tissue, loose bodies, and impinging osteophytes.
The tip of the olecranon is frequently hypertrophied; we routinely perform a partial olecranon ostectomy (removing the proximal 3-5 mm) to ensure it seats deeply into the cleared fossa without bony impingement. The posterior band of the medial collateral ligament, which is often contracted and contributes to extension loss, can be carefully released from within the joint capsule, taking immense care not to plunge medially and injure the ulnar nerve.
The Anterior Release
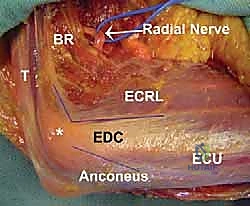
Once maximal posterior release is achieved, we shift our focus anteriorly. We develop the interval between the extensor carpi radialis longus (ECRL) and the extensor digitorum communis (EDC). The ECRL and brachioradialis are elevated anteriorly, exposing the anterior joint capsule.
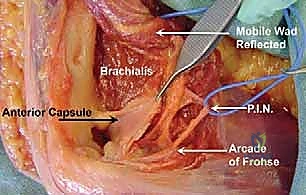
This is the most treacherous part of the procedure. The brachialis muscle is often densely adherent to the anterior capsule. Using a Cobb elevator and sharp dissection, we must meticulously elevate the brachialis muscle belly completely off the anterior capsule, sweeping from lateral to medial. This creates a safe "buffer zone"; as long as our instruments remain posterior to the brachialis, the median nerve and brachial artery are safe.
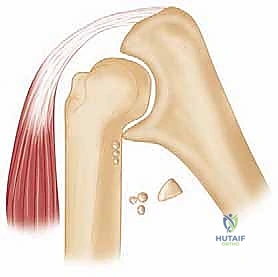
With the brachialis safely retracted anteriorly, we perform a radical anterior capsulectomy. We excise a wide swath of the thickened anterior capsule, extending from the lateral epicondyle across the joint to the medial epicondyle.
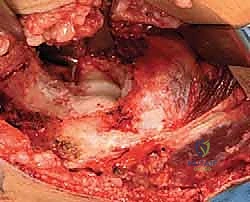
During this lateral dissection, the origin of the lateral ulnar collateral ligament (LUCL) on the lateral epicondyle must be strictly identified and preserved. We then inspect the radiocapitellar joint.
If there is significant radiocapitellar arthritis or a malunited radial head fracture blocking rotation, a radial head resection or arthroplasty may be performed concurrently. Finally, we assess the coronoid fossa; any osteophytes or heterotopic bone blocking full flexion are removed with an osteotome or burr.

After copious irrigation, the tourniquet is deflated, and meticulous hemostasis is achieved. A deep closed-suction drain is placed to prevent hematoma formation, which is a potent stimulus for recurrent heterotopic ossification. The intervals are closed loosely, and the skin is reapproximated.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, elbow contracture release carries a high complication profile, necessitating rigorous informed consent and vigilant postoperative management. The most frequent and concerning complications are neurologic.
Ulnar neuropathy is the most common complication, with incidence rates reported between 5% and 15% in the literature if prophylactic decompression is not performed. This occurs due to acute traction on the nerve as the elbow's flexion arc is rapidly restored, or secondary to postoperative hematoma and swelling within the cubital tunnel. If a patient develops new-onset postoperative ulnar neuropathy, conservative management with splinting in extension is initially trialed. However, if motor deficits arise or sensory symptoms persist beyond 6 to 8 weeks, a secondary surgical decompression and anterior transposition is mandated. Injury to the Posterior Interosseous Nerve (PIN) is less common (1-3%) but catastrophic, resulting in the inability to extend the fingers and thumb. This is almost exclusively an iatrogenic injury caused by aggressive retractor placement distal to the annular ligament or over-zealous lateral dissection.
Iatrogenic instability is another major pitfall. Posterolateral rotatory instability (PLRI) occurs if the LUCL is inadvertently transected during the lateral capsulectomy. If recognized intraoperatively, it must be repaired immediately with suture anchors to the lateral epicondyle. If it presents postoperatively, the patient will complain of painful clicking and a sensation of the elbow "giving out" when pushing off a chair. Delayed presentation requires formal LUCL reconstruction using a palmaris longus or allograft tendon.
Recurrent stiffness and heterotopic ossification (HO) are the banes of elbow release surgery. Hematoma formation in the large dead space created by the capsulectomy serves as a scaffold for recurrent fibrosis and HO. Incidence of recurrent stiffness requiring re-operation ranges from 10% to 20%. To mitigate this, we employ meticulous hemostasis, indwelling drains, and routine prophylaxis against HO.
| Complication | Estimated Incidence | Prevention & Salvage Management |
|---|---|---|
| Ulnar Neuropathy | 5% - 15% | Prevention: Prophylactic transposition if pre-op flexion <100° or positive Tinel's. Salvage: Delayed anterior transposition if symptoms persist >6 weeks. |
| PIN Palsy | 1% - 3% | Prevention: Avoid retractor placement distal to radial neck; respect supinator anatomy. Salvage: Tendon transfers if no recovery at 6-12 months. |
| Iatrogenic PLRI | 2% - 5% | Prevention: Meticulous identification and preservation of LUCL footprint during capsulectomy. Salvage: Acute anchor repair; delayed allograft/autograft reconstruction. |
| Recurrent Stiffness / HO | 10% - 20% | Prevention: Meticulous hemostasis, closed suction drains, Indomethacin prophylaxis, early CPM. Salvage: Revision release; hinged external fixator; Total Elbow Arthroplasty (TEA) in low-demand patients. |
| Deep Infection | < 2% | Prevention: Strict sterile technique, perioperative antibiotics, careful soft-tissue handling. Salvage: Aggressive I&D, targeted IV antibiotics, possible hardware removal. |
Phased Post-Operative Rehabilitation Protocols
The surgical release is merely the first step; the postoperative rehabilitation protocol dictates the ultimate success or failure of the procedure. The rehabilitation is phased, intensive, and requires immense patient dedication.
Phase I: Immediate Postoperative Period (Days 0-7)
The primary goal in the first week is the immediate initiation of motion to prevent the nascent hematoma from organizing into restrictive scar tissue. This is where the continuous regional nerve block proves invaluable. While the block is active, the patient is placed in a Continuous Passive Motion (CPM) machine immediately in the recovery room, aiming to cycle through the full intraoperatively achieved arc of motion. If a CPM is not utilized, active-assisted range of motion (AAROM) guided by a specialized hand therapist begins on postoperative day one. Gravity-assisted extension exercises are emphasized. Edema control is critical; the arm is kept elevated, and compressive dressings are utilized.
Phase II: Intermediate Phase (Weeks 1-6)
As the regional block wears off, pain management transitions to oral analgesics. The focus shifts to maintaining the ROM gained in the OR. This phase introduces static progressive splinting or dynamic bracing. Based on the patient's primary deficit, the therapist will fabricate custom splints. If extension is the primary issue, the patient will wear an extension splint at night and perform frequent, short-duration stretching sessions during the day. We employ a "tissue stress" model—prolonged, low-load stretching is far superior and less inflammatory than short, aggressive, painful stretching. The patient must understand that pain during stretching is counterproductive, as it incites an inflammatory cascade that promotes further fibrosis.
Phase III: Late Phase (Weeks 6-12 and Beyond)
By week six, the soft tissues have begun to remodel. The focus shifts from pure ROM to incorporating strengthening and functional use. Daytime splinting is gradually weaned, though nighttime splinting may continue for up to 6 months if morning stiffness is prominent. Patients are counseled that they may experience intermittent flare-ups of inflammation and stiffness; these are managed with short courses of NSAIDs and temporary increases in splinting time. We must manage patient expectations: while we strive for the functional arc of 30-130 degrees, a residual terminal extension loss of 1
Clinical & Radiographic Imaging Archive

