Arthroscopic Contracture Release of the Elbow: A Masterclass in Restoring Motion
Key Takeaway
This masterclass guides fellows through arthroscopic elbow contracture release. We meticulously cover surgical anatomy, patient positioning, and real-time intraoperative execution. Learn precise portal establishment, capsular release, and osteophyte resection techniques. Critical pearls for neurovascular protection, complication management, and postoperative rehabilitation are emphasized, ensuring comprehensive understanding for restoring elbow motion.
Comprehensive Introduction and Patho-Epidemiology
Welcome, esteemed colleagues and surgeons in training, to this definitive masterclass focusing on a uniquely challenging yet profoundly rewarding orthopedic procedure: the arthroscopic release of the elbow contracture. The overarching objective of this intervention is the restoration of a functional arc of motion, thereby alleviating the severe functional impairment that this condition invariably imposes upon our patients. The stiff elbow is not merely a mechanical hindrance; it is a complex pathophysiological entity that drastically reduces a patient's ability to perform essential activities of daily living (ADLs). This procedure transcends the basic mechanical act of excising fibrotic tissue; it demands an intimate, three-dimensional understanding of the elbow’s intricate biomechanics, its unforgiving neurovascular topography, and the underlying biological drivers of capsular fibrosis.
Historically, the management of the stiff elbow was relegated to extensive open surgical releases, which, while effective, carried significant morbidity, including massive surgical trauma, prolonged rehabilitation, and a high risk of recurrent stiffness secondary to the surgical insult itself. The evolution of elbow arthroscopy has revolutionized our approach, offering a minimally invasive corridor to achieve a comprehensive 360-degree capsular release. However, this minimally invasive nature belies the technical complexity of the procedure. The arthroscopic management of elbow contractures is widely considered an advanced, tertiary-level skill, fraught with potential neurovascular pitfalls. It requires meticulous preoperative planning, precise portal placement, and an unwavering respect for the proximity of critical neurovascular structures that intimately surround the joint capsule.
To truly master this technique, the surgeon must first comprehend the etiology and pathogenesis of elbow stiffness. Elbow contractures are most frequently posttraumatic in origin, arising as a sequela of fractures, dislocations, or severe soft-tissue trauma. However, they can also manifest secondary to degenerative osteoarthritis, inflammatory arthritides (such as rheumatoid arthritis), systemic conditions like traumatic brain injury or severe burns, and various neurologic disorders. Patients who present with a combination of head trauma and local elbow trauma, as well as those with severe burns, are at an exceptionally high risk for developing heterotopic ossification (HO) concurrently with severe capsular thickening.
The biological mechanisms driving altered capsular properties in the stiff elbow are multifactorial and heavily driven by chronic inflammation and altered cellular mechanics. Histological analysis of contracted elbow capsules consistently reveals a marked increase in the population of myofibroblasts, particularly within the anterior capsule. These specialized cells, rich in alpha-smooth muscle actin, actively contribute to collagen contraction and tissue retraction. Concomitantly, there is a documented increase in pathological collagen cross-linking, overall capsular hypertrophy, a profound decrease in tissue water content, and a reduction in proteoglycan concentration. This biochemical cascade culminates in a rigid, non-compliant capsular envelope that mechanically tethers the joint, severely restricting both the flexion-extension arc and the pronation-supination axis.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of the elbow's unique surgical anatomy is the absolute prerequisite for performing a safe and effective arthroscopic contracture release. Unlike the shoulder complex, which benefits from the compensatory mobility of the scapulothoracic articulation to mask glenohumeral motion loss, the elbow operates as an isolated mechanical linkage in the upper extremity. It lacks any proximal or distal compensatory mechanisms, meaning that even a minor degree of stiffness—particularly in the functional arc of 30 to 130 degrees—is poorly tolerated and immediately translates to functional disability.
Osteology and Articular Architecture
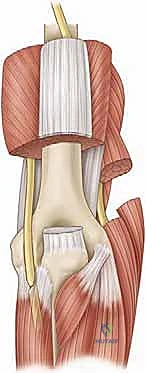
The elbow joint is a highly congruent, complex structural marvel comprising three distinct articulations housed within a single, continuous synovial-lined cavity. The ulnohumeral articulation functions as a highly constrained hinge (ginglymus) joint, serving as the primary osseous stabilizer and dictating the flexion and extension arc. The radiohumeral articulation operates as a rotatory joint, contributing to both the flexion/extension arc and the complex mechanics of forearm rotation. Finally, the proximal radioulnar joint acts as a trochoid (pivot) joint, working in concert with the distal radioulnar joint to facilitate pronation and supination.
The inherent osseous stability of the elbow is derived from the deep articulation of the trochlear notch of the ulna with the spool-shaped trochlea of the humerus. This high degree of congruency, while providing excellent stability, renders the joint highly susceptible to mechanical blocks from relatively small osteophytes or loose bodies. The radial head articulates with the capitellum, and its smooth rotation is essential for unrestricted forearm mechanics. Any incongruity in these articular surfaces, whether from post-traumatic malunion or degenerative osteophyte formation, will result in intrinsic stiffness that must be addressed concurrently with any soft-tissue release.

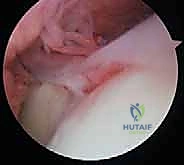
The close anatomical relationship of the joint capsule to the surrounding collateral ligaments and the crossing musculotendinous units dictates that any intra-articular inflammatory process, hemarthrosis, or surgical trauma will rapidly induce widespread, global stiffness. The anterior and posterior compartments of the elbow must be viewed as distinct yet interconnected spaces, both of which require meticulous evaluation and potential decompression during an arthroscopic release.
Capsuloligamentous Complex and Pathophysiological Alterations
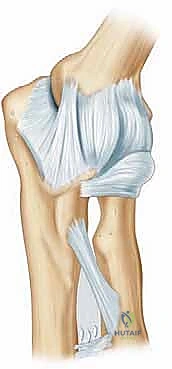
The capsuloligamentous envelope of the elbow is the primary pathological focus in extrinsic, static contractures. The anterior capsule is of paramount importance; proximally, it originates just superior to the coronoid and radial fossae on the anterior distal humerus. Distally, it inserts onto the anterior margin of the coronoid process medially and blends intimately with the annular ligament laterally. The mechanical strength of the anterior capsule is derived from its robust, cruciate-oriented collagen fibers, which become maximally taut in full extension and lax during flexion.
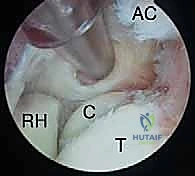
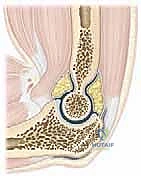
The posterior capsule originates proximally just superior to the olecranon fossa and inserts distally along the articular margin of the sigmoid notch and the posterior aspect of the annular ligament. In a healthy, non-pathological elbow, the maximal capsular capacity is approximately 25 to 30 mL. However, in the setting of a severe posttraumatic contracture, this volume can be drastically attenuated to as little as 5 to 6 mL. Furthermore, the capsule itself hypertrophies significantly, often thickening from a normal physiological width of 1-2 mm to a dense, fibrotic band measuring 5 mm or more. This profound loss of joint volume and the presence of thickened, rigid capsular tissue—most notably in the anterior compartment—are the absolute hallmarks of posttraumatic elbow contractures.

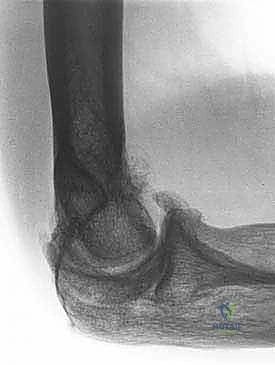
The medial and lateral collateral ligament complexes must be carefully evaluated. While the capsule is routinely resected, the collateral ligaments must be preserved to maintain joint stability. The anterior bundle of the medial collateral ligament (AMCL) is the primary restraint to valgus stress, while the lateral ulnar collateral ligament (LUCL) prevents posterolateral rotatory instability. Inadvertent resection of these structures during aggressive capsectomy will convert a stiff elbow into an unstable one, a disastrous complication that is exceedingly difficult to salvage.
Neurovascular Topography and Hilton's Law
The neurovascular anatomy surrounding the elbow is notoriously unforgiving, and a thorough understanding of these relationships is non-negotiable. Hilton's Law dictates that the nerves supplying a joint also supply the muscles moving that joint and the skin covering their respective insertions. The elbow capsule receives rich sensory innervation from branches of the musculocutaneous, median, ulnar, and radial nerves. This dense, overlapping innervation profile explains the severe, often debilitating pain associated with a stiff, inflamed elbow joint, and underscores the necessity of adequate postoperative analgesia.


The ulnar nerve is a particularly critical and vulnerable structure in the context of elbow contractures. It courses posteriorly through the cubital tunnel, a fibro-osseous canal bordered by the medial epicondyle anteriorly, the olecranon laterally, and the arcuate ligament (Osborne's fascia) superficially. As the elbow flexes, the attachment sites of this retinaculum diverge, rendering the cubital tunnel increasingly taut and decreasing the cross-sectional area of the canal. This anatomical reality means that as surgical release restores elbow flexion, the ulnar nerve may become acutely compressed.
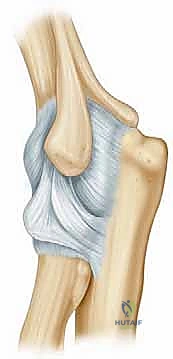
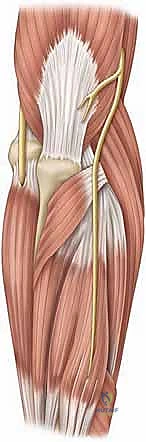
Pre-existing flexion contractures can also directly tether and compress the ulnar nerve, leading to subclinical or overt neuropathy. Consequently, meticulous preoperative clinical assessment of ulnar nerve function is mandatory. In many instances, particularly when significant flexion gains are anticipated or when preoperative neuropathy is present, a prophylactic open decompression or transposition of the ulnar nerve must be performed prior to or concurrently with the arthroscopic capsular release to prevent catastrophic postoperative palsy.
Exhaustive Indications and Contraindications
The clinical decision to proceed with operative intervention for a stiff elbow is nuanced and must never be predicated solely on absolute goniometric measurements of motion loss. Instead, the decision-making process must be entirely patient-centric, focusing heavily on the individual's functional impairment, occupational demands, and inability to perform ADLs. A functional arc of approximately 100 degrees (ranging from 30 degrees of extension to 130 degrees of flexion), combined with 50 degrees of both pronation and supination, is generally accepted as the minimum requirement for the vast majority of daily tasks.
Clinical Decision Making and Nonoperative Modalities
Nonoperative management remains the mandatory first line of treatment for nearly all elbow contractures and should be aggressively pursued for a minimum of 4 to 6 months following the onset of stiffness or the inciting trauma. The cornerstone of conservative therapy is a highly structured, patient-directed physical therapy program emphasizing edema control, gentle active and active-assisted range of motion, and the rigorous application of static progressive splinting.
Dynamic splinting, which applies a constant, elastic tension, often incites a reactive inflammatory response and muscle guarding, ultimately proving counterproductive. Conversely, static progressive splinting utilizes the principle of stress relaxation, allowing the fibrotic capsular tissue to gradually elongate over time without triggering an acute inflammatory flare. Patients must be extensively counseled regarding the protracted nature of conservative treatment and the necessity of strict compliance. Surgical intervention is only entertained when a documented plateau in motion has been reached despite exhaustive nonoperative efforts, and the residual stiffness continues to cause unacceptable functional limitation or pain.
Patient Selection Criteria
Proper patient selection is the ultimate determinant of surgical success. Morrey's classification of elbow stiffness remains highly relevant; he characterized stiffness as either static (the most common variant, involving the capsule, ligaments, and heterotopic ossification) or dynamic (less common, involving muscle spasticity, nerve injuries, or poor muscle excursion). Arthroscopic release is exquisitely suited for addressing static, extrinsic contractures primarily driven by capsular fibrosis and accessible osteophyte formation.
| Category | Specific Parameters and Considerations |
|---|---|
| Primary Indications | - Loss of functional ROM precluding ADLs or occupational duties. - Significant, debilitating pain at the terminal arcs of motion. - Documented failure of ≥ 6 months of comprehensive nonoperative therapy (including static progressive splinting). - Contracture etiology primarily driven by anterior/posterior capsular thickening or intra-articular osteophytes/loose bodies. |
| Absolute Contraindications | - Active, untreated intra-articular or periarticular infection. - Severe architectural distortion of the joint secondary to malunion or massive trauma, precluding safe arthroscopic navigation. - Prior surgical incisions or trauma that have severely distorted normal neurovascular anatomy, making standard portal placement unsafe. - Bridging, extra-articular Heterotopic Ossification (HO) that is the primary mechanical block to motion (requires open excision). |
| Relative Contraindications | - Prior ulnar nerve transposition (requires extreme caution; often necessitates open exploration of the nerve prior to medial portal placement). - Severe, advanced osteoarthritis with near-complete loss of joint space and massive deformity. - Poor patient compliance or inability to participate in aggressive postoperative rehabilitation. - Active inflammatory arthropathy in a flare state. |

Heterotopic ossification (HO) warrants special consideration. While small, isolated intra-articular osteophytes can be readily excised arthroscopically, massive, bridging extra-articular HO is a strict contraindication to an exclusively arthroscopic approach. The presence of significant HO often signifies a complex, multifactorial extrinsic contracture that is simply not amenable to arthroscopic resection and requires a formal, extensile open approach to safely identify and protect neurovascular structures while excising the ectopic bone.
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the bedrock upon which a safe and successful arthroscopic elbow release is built. The surgeon must meticulously map the patient's specific pathoanatomy, anticipating the location of critical neurovascular structures and planning the sequence of the arthroscopic release to maximize efficiency and safety.
Clinical Assessment and Diagnostic Imaging
The physical examination must be exhaustive, beginning proximally at the cervical spine to rule out any concurrent radiculopathy that might confound the clinical picture. The shoulder and wrist must also be assessed to ensure no concomitant pathology is contributing to the upper extremity dysfunction. At the elbow, precise goniometric measurements of active and passive flexion, extension, pronation, and supination are recorded. The quality of the endpoint is a critical diagnostic clue; a firm, abrupt endpoint typically suggests a bony block (osteophyte or HO), whereas a softer, spongy endpoint is indicative of capsular fibrosis, which is highly amenable to arthroscopic capsectomy.


Ligamentous stability must be rigorously tested by applying varus and valgus stress at both 0 and 30 degrees of flexion. Pre-existing instability is a major red flag; aggressive capsular release in an unstable elbow will exacerbate the problem. A comprehensive neurological examination, with a specific focus on the ulnar nerve, is mandatory. The cubital tunnel is palpated for tenderness or a positive Tinel's sign. Two-point discrimination (normal being < 6mm) and a Froment sign are assessed to evaluate intrinsic hand muscle function. Any evidence of ulnar neuropathy dictates that the nerve must be addressed surgically.
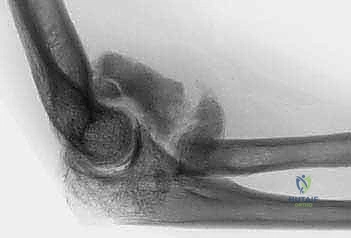
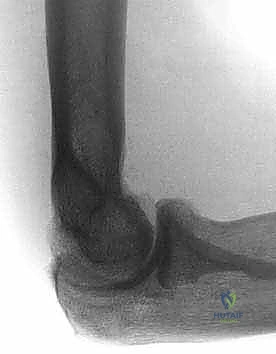
Standard radiographic evaluation includes high-quality anteroposterior (AP) and lateral views. The lateral radiograph is invaluable for identifying osteophytes impinging on the olecranon or coronoid fossae, as well as assessing the congruency of the ulnohumeral joint. The AP view helps evaluate the joint line and subchondral bone architecture. However, if the elbow has a flexion contracture exceeding 45 degrees, the AP projection will be distorted, limiting its utility.

In cases of complex posttraumatic stiffness, articular incongruity, or suspected heterotopic ossification, a non-contrast Computed Tomography (CT) scan with 3D reconstructions is highly recommended. The CT scan provides an unparalleled, three-dimensional roadmap of the osseous anatomy, allowing the surgeon to precisely localize osteophytes, loose bodies, and areas of malunion, thereby facilitating accurate preoperative templating for bony resection.
Anesthesia, Setup, and Patient Positioning
The procedure is typically performed under general anesthesia, often supplemented with a regional block (such as an interscalene or supraclavicular nerve block) to provide profound postoperative analgesia, which is critical for facilitating early, aggressive rehabilitation. However, the use of regional anesthesia must be carefully weighed against the need to perform an immediate postoperative neurological examination, particularly if there are concerns regarding the ulnar or radial nerves.

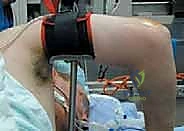
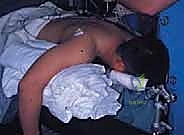
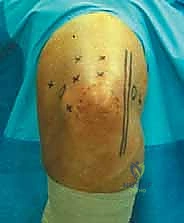
Patient positioning is a matter of surgeon preference, with the lateral decubitus, prone, and supine suspended positions all being viable options. The lateral decubitus position, with the arm draped over a specialized padded post, is highly favored by many advanced arthroscopists. This position allows for excellent access to both the anterior and posterior compartments, facilitates airway management by the anesthesia team, and permits the elbow to hang freely at 90 degrees, which naturally distends the joint and drops the neurovascular structures away from the anterior capsule. A sterile tourniquet is routinely applied high on the brachium to ensure a bloodless field, though its inflation time must be strictly monitored.
Step-by-Step Surgical Approach and Release Technique
The arthroscopic release of a stiff elbow is a highly orchestrated, sequential procedure. The overarching philosophy is to perform a complete, 360-degree capsectomy while meticulously protecting the crossing neurovascular structures. The surgeon must maintain excellent spatial awareness, constantly visualizing the anatomical landmarks and understanding the depth and trajectory of their instruments.
Portal Placement and Initial Diagnostic Arthroscopy
Precise portal placement is the most critical step in preventing iatrogenic neurovascular injury. The joint is first maximally distended by injecting 20-30 mL of sterile saline through a lateral soft spot approach (through the anconeus triangle). This distension pushes the anterior capsule and the closely applied neurovascular structures (brachial artery, median nerve, radial nerve) anteriorly, creating a safer working space.

The anteromedial portal is typically established first. To minimize risk to the ulnar nerve and medial antebrachial cutaneous nerve, the proximal anteromedial (PAM) portal is utilized. This is located approximately 2 cm proximal to the medial epicondyle, just anterior to the medial intermuscular septum. An incision is made in the skin only, and a blunt trocar is used to carefully dissect down to the anterior capsule, maintaining contact with the anterior aspect of the humerus.
Once the arthroscope is introduced medially, the proximal anterolateral (PAL) portal is established under direct intra-articular visualization. The PAL portal is located 2 cm proximal and 1 cm anterior to the lateral epicondyle. A spinal needle is used to localize the ideal trajectory, ensuring it enters the joint safely above the radial head and capitellum. The radial nerve is at the highest risk during the establishment of the anterolateral portal; keeping the portal proximal and utilizing a strictly blunt dissection technique through the brachialis muscle minimizes this risk.
Anterior Compartment Capsectomy and Debridement
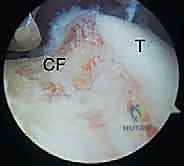
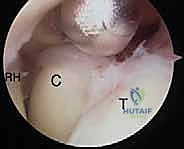
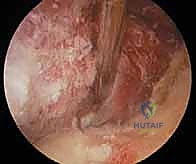
With both anterior portals established, a thorough diagnostic sweep of the anterior compartment is performed. The coronoid fossa, radial fossa, and the articular surfaces of the radiocapitellar and ulnohumeral joints are inspected. In a contracted elbow, the anterior capsule will appear as a dense, opaque, fibrotic sheet, often obscuring the underlying musculature.
The anterior capsectomy begins by utilizing a motorized shaver and an arthroscopic radiofrequency wand. Working from the PAL portal while viewing from the PAM portal, the surgeon systematically resects the anterior capsule. The release must begin proximally, exposing the fibers of the brachialis muscle, which serves as a critical anatomical barrier protecting the brachial artery and median nerve. The resection proceeds distally towards the radiocapitellar joint. Extreme caution must be exercised laterally to avoid injuring the radial nerve, which lies in close proximity to the capsule anterior to the radiocapitellar joint.
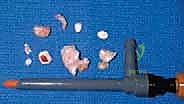
Once the lateral half of the capsule is resected, the viewing and working portals are switched. Viewing from the PAL portal, the surgeon resects the medial half of the anterior capsule using instruments in the PAM portal. The medial resection must be meticulous to avoid injury to the medial collateral ligament and the median nerve. Any osteophytes impinging within the coronoid or radial fossae are subsequently resected using an arthroscopic burr, ensuring the fossae are deep enough to accommodate the coronoid and radial head during terminal flexion.
Posterior Compartment Decompression and Osteophyte Resection
Following the anterior release, attention is turned to the posterior compartment. The posterior portals include the direct posterior portal (located 2-3 cm proximal to the olecranon tip, directly in the midline) and the posterolateral portal (located just lateral to the triceps tendon).

Viewing from the posterolateral portal, the direct posterior portal is established. The posterior compartment is often filled with dense fibrotic tissue and adhesions. A thorough posterior capsectomy is performed, exposing the olecranon fossa. In many posttraumatic contractures, the olecranon fossa is filled with osteophytes or fibrotic debris, preventing terminal extension. An arthroscopic burr is utilized to clear the fossa and resect any impinging osteophytes from the tip of the olecranon.
Throughout the posterior release, the surgeon must remain acutely aware of the ulnar nerve medially. If a prophylactic ulnar nerve decompression was not performed, the medial aspect of the posterior capsule must be approached with extreme trepidation. The release is considered complete when the olecranon seats deeply and congruently into the olecranon fossa during full passive extension, and the coronoid process clears the anterior humerus during full passive flexion.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, arthroscopic elbow contracture release carries a distinct and unforgiving complication profile. The dense concentration of vital neurovascular structures surrounding the joint capsule mandates absolute precision; even minor deviations in portal placement or aggressive soft-tissue resection can result in catastrophic, life-altering morbidity for the patient.
Neurologic Injury and Heterotopic Ossification
Neurologic injury remains the most feared complication, with transient nerve palsies reported in 2% to 5% of cases, and permanent nerve damage occurring in less than 1% in the hands of highly experienced surgeons. The ulnar, radial, median, and medial antebrachial cutaneous (MABC) nerves are all at risk.
| Complication | Incidence | Etiology / Risk Factors | Prevention and Management Strategies |
|---|---|---|---|
| Ulnar Nerve Neuropathy | 2% - 5% | Traction during flexion restoration; direct injury during medial portal placement or posterior capsectomy. | Prophylactic open decompression/transposition if preoperative symptoms exist or >30° flexion gain anticipated. Avoid blind instrumentation medially. |
| Radial Nerve Injury | 1% - 3% | Direct laceration during anterolateral portal placement; thermal injury from RF wand laterally. | Use proximal anterolateral portal. Blunt dissection to capsule. Maintain brachialis muscle as a protective barrier. Avoid aggressive lateral capsectomy near radiocapitellar joint. |
| Median Nerve / Brachial Artery Injury | < 1% | Penetration of anterior capsule during capsectomy; aggressive use of motorized shavers anteriorly. | Maintain visualization of brachialis muscle fibers. Do not resect tissue anterior to the brachialis. Use RF wand with extreme caution anteriorly. |
| Heterotopic Ossification (HO) Recurrence | 5% - 15% | Surgical trauma in predisposed individuals (head trauma, burns); inadequate hemostasis. | Meticulous hemostasis. Consider postoperative prophylaxis (Indomethacin or localized radiation) in high-risk patients. |
| Recurrent Stiffness | 10% - 20% | Inadequate initial release; poor postoperative pain control |
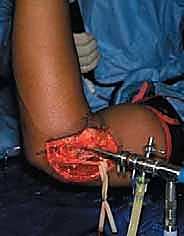
Clinical & Radiographic Imaging Archive