Masterclass: Flexor Carpi Ulnaris Transfer for Spastic Wrist Flexion Deformity
Key Takeaway
This masterclass details the Flexor Carpi Ulnaris (FCU) transfer for spastic wrist flexion deformity in cerebral palsy. We meticulously cover patient selection, comprehensive anatomy, step-by-step intraoperative execution including FCU mobilization and ECRB transfer using the Pulvertaft weave, and critical pearls for optimal tensioning. Postoperative rehabilitation and complication management are also thoroughly discussed for maximal functional outcomes.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues and fellows, to the operating theater. Today, we are addressing a common yet functionally debilitating manifestation of cerebral palsy in the upper extremity: the spastic wrist flexion deformity. Our procedure today will be a Flexor Carpi Ulnaris (FCU) transfer to the Extensor Carpi Radialis Brevis (ECRB). This operation remains a cornerstone in improving upper limb function in these challenging patients, aiming to convert a primary deforming force into a corrective, functional vector. To master this procedure, one must first possess a deep understanding of the underlying pathophysiology and the natural history of the spastic upper extremity.
Cerebral palsy represents a heterogeneous group of non-progressive, primary central nervous system dysfunctions occurring in the developing fetal or infant brain. However, while the neurologic lesion is static, its secondary peripheral manifestations in the musculoskeletal system are fiercely dynamic and progressive. In spastic hemiplegia—the predominant subtype of cerebral palsy for which upper extremity surgical intervention is indicated—we observe a profound loss of normal descending cortical inhibition. This upper motor neuron syndrome manifests as a triad of spasticity, weakness of antagonist muscles, and severely impaired selective motor control. The resulting muscle imbalance across the radiocarpal and midcarpal joints initiates a cascade of debilitating peripheral changes.
Over time, this relentless muscle imbalance leads to structural alterations within the muscle–tendon unit itself. The Flexor Carpi Ulnaris (FCU) is almost universally the primary culprit in this patho-anatomic cascade. Its increased spastic tone overpowers the relatively paretic Extensor Carpi Radialis Longus (ECRL) and ECRB. Initially, this presents as a dynamic deformity, where the wrist assumes a characteristic flexed and ulnar-deviated posture during active use or ambulation, yet remains passively correctable. However, persistent overpull induces rheologic changes within the FCU muscle belly, including sarcomere loss, increased intramuscular collagen deposition, and ultimately, a fixed myostatic contracture.
If left unaddressed, this myostatic contracture inevitably progresses to a fixed skeletal deformity by the time of skeletal maturity, characterized by volar subluxation of the carpus and adaptive changes in the articular cartilage. This flexed posture significantly interferes with the tenodesis effect critical for normal hand function, devastating the patient's capacity for grasp and release. Our goal with tendon transfer surgery is to intervene precisely during the dynamic phase, before fixed joint contractures develop, thereby preserving joint mobility, rebalancing the biomechanical forces, and maximizing the patient's functional potential. If fixed articular contractures are already present, isolated tendon transfers are destined to fail, and salvage procedures such as proximal row carpectomy or total wrist arthrodesis become necessary.
Detailed Surgical Anatomy and Biomechanics
Before we make our initial incision, a rigorous review of the critical anatomy and wrist biomechanics is mandatory. The wrist joint is a complex intercalated segment, and its spatial position is primarily controlled by five key dedicated motors, heavily influenced by the secondary contributions of the extrinsic digital flexors and extensors.
Wrist Extensors and Flexors
The extensor mechanism of the wrist is dominated by the Extensor Carpi Radialis Brevis (ECRB), the Extensor Carpi Radialis Longus (ECRL), and the Extensor Carpi Ulnaris (ECU). The ECRB is the primary and most powerful wrist extensor. Inserting into the base of the third metacarpal, it possesses a central line of pull, making it the ideal recipient tendon for our transfer to achieve pure wrist extension without exacerbating radial or ulnar deviation. The ECRL, inserting into the base of the second metacarpal, acts as a strong wrist extensor but carries a significant radial deviation vector. The ECU extends and ulnar deviates the wrist. On the volar aspect, the Flexor Carpi Radialis (FCR) flexes and radially deviates the wrist. The FCU, our donor tendon, inserts distally onto the pisiform, with robust structural extensions to the hook of the hamate and the base of the fifth metacarpal via the pisohamate and pisometacarpal ligaments. Biomechanically, the FCU commands the greatest flexion and ulnar deviation moment arm at the wrist; hence, its overactivity is the primary deforming force we must neutralize.
Extrinsic Digital Motors Crossing the Wrist
The extrinsic finger and thumb musculature significantly impacts radiocarpal positioning. The Flexor Digitorum Profundus (FDP), Flexor Digitorum Superficialis (FDS), and Flexor Pollicis Longus (FPL) cross the volar aspect of the wrist joint, exerting a substantial secondary wrist flexion force. Spasticity or contracture within these units can severely exacerbate the primary wrist flexion deformity. Conversely, the Extensor Pollicis Longus (EPL), Extensor Indicis Proprius (EIP), and Extensor Digitorum Communis (EDC) exert a secondary wrist extension force. The interplay between these extrinsic digital motors and the dedicated wrist motors dictates the success of any tendon transfer, necessitating careful preoperative evaluation of the Volkmann angle to ensure digital flexor tightness will not negate the transferred FCU's extension vector.

Critical Neurovascular Topography
The surgical anatomy of the volar-ulnar forearm is unforgiving, and meticulous dissection is paramount. The ulnar nerve and artery lie immediately radial and deep to the FCU tendon in the distal forearm. As the FCU is harvested, particularly near its insertion on the pisiform, these vital structures must be definitively identified and protected. Furthermore, the Dorsal Ulnar Sensory Branch (DUSB) of the ulnar nerve typically arises 5 to 8 centimeters proximal to the pisiform, coursing dorsally around the ulnar neck to provide sensation to the dorso-ulnar aspect of the hand. The DUSB is exceptionally vulnerable in the distal aspect of the volar-ulnar incision. Iatrogenic injury to this branch results in a painful neuroma that can severely compromise the functional outcome of an otherwise technically perfect transfer.
Exhaustive Indications and Contraindications
Patient selection is arguably the most critical determinant of success in cerebral palsy upper extremity surgery. The decision to proceed with an FCU to ECRB transfer must be predicated on a comprehensive understanding of the patient's functional deficits, volitional control, and the absence of fixed structural barriers.
The ideal candidate is typically a child between the ages of 6 and 12 years with spastic hemiplegia. By this age, neurologic maturation has plateaued, and the child's spontaneous functional use patterns are well-established. The primary indication for this transfer is a dynamic wrist flexion and ulnar deviation deformity that impairs the patient's ability to position the hand in space for effective grasp and release. Crucially, the patient must demonstrate adequate passive range of motion of the wrist and digits. If the patient exhibits poor active wrist extension but demonstrates robust active digital extension when the wrist is passively held in a neutral or slightly extended position, they are an excellent candidate. This confirms that the extrinsic finger extensors are functional but are simply overpowered by the flexed posture of the wrist.
Conversely, absolute contraindications must be strictly respected to avoid catastrophic functional deterioration. The presence of a fixed, rigid radiocarpal contracture precludes an isolated tendon transfer; skeletal realignment or joint release must precede or accompany any transfer. The most treacherous contraindication is the "clenched fist" deformity. If a patient exhibits severe spasticity of the FDP and FDS, such that extending the wrist passively causes the fingers to curl tightly into the palm (a severe Volkmann sign), transferring the FCU to extend the wrist will only exacerbate the clenched fist, rendering the hand entirely useless. Furthermore, profound athetosis, dystonia, or a complete lack of volitional motor control (House classification 0) are strong contraindications, as the unpredictable muscle firing patterns will inevitably defeat the transferred tendon.
| Parameter | Indications for FCU to ECRB Transfer | Absolute Contraindications |
|---|---|---|
| Deformity Type | Dynamic wrist flexion/ulnar deviation | Fixed skeletal/articular contracture |
| Passive ROM | Full passive wrist and digital extension | Rigid joint stiffness |
| Digital Function | Good active finger extension with wrist neutral | "Clenched fist" when wrist is extended |
| Motor Control | Poor active wrist extension, but some volitional control | Severe athetosis, dystonia, or zero volitional control |
| Age | Typically > 6 years (established motor patterns) | Infants (spontaneous improvement possible) |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the bedrock upon which surgical success is built. Our assessment begins with a detailed, nuanced history from the caregivers regarding the child's spontaneous use of the affected limb in activities of daily living. We specifically look for signs of premature hand dominance and delays in bimanual skill acquisition. We utilize House’s classification of upper extremity functional use to establish a definitive baseline.
Our physical examination must be exhaustive. Passive Range of Motion (ROM) of every joint from the shoulder to the distal interphalangeal joints is documented. The Volkmann Angle Test is critical: we passively extend the wrist and measure the angle at which the fingers begin to flex due to FDP/FDS tightness. If the finger flexors are excessively tight, a concomitant fractional lengthening of the flexors will be mandatory to prevent a postoperative clenched fist. We meticulously assess Active ROM, looking for any volitional control of the ECRB and ECRL. Stereognosis and two-point discrimination are tested; while profound sensory deficits do not strictly contraindicate surgery, they heavily influence the ultimate functional ceiling the patient can achieve, and caregivers must be counseled accordingly.
Adjunct diagnostic modalities are frequently employed in complex cases. Fine-Needle dynamic Electromyography (EMG) is invaluable for assessing the phasic control of the FCU during grasp and release phases. An FCU that demonstrates continuous, non-phasic spastic firing is less ideal than one that retains some semblance of phasic activity. Furthermore, preoperative Botulinum Toxin Type A (Botox) injections into the FCU can serve as both a diagnostic tool and a therapeutic bridge. By temporarily paralyzing the FCU, we can simulate the postoperative state, allowing the therapist to assess the underlying strength of the wrist extensors and confirming whether neutralizing the FCU will unmask functional digital extension.
"Alright team, let's get our patient positioned." The patient is positioned supine on the operating room table. We ensure the affected upper limb is fully supported and extended on a dedicated radiolucent armboard. This provides optimal exposure for both the volar and dorsal aspects of the forearm. A pneumatic tourniquet is applied proximally on the brachium, well padded with cast padding. Following standard chlorhexidine preparation and sterile draping, the limb is exsanguinated with an Esmarch bandage, and the tourniquet is inflated to 250 mmHg to maintain a pristine, bloodless field, which is absolutely critical for the meticulous neurovascular dissection that follows.
Step-by-Step Surgical Approach and Fixation Technique
"Scalpel, please. Let's begin." Our surgical strategy utilizes a dual-incision approach, a modification of the classic Green technique, allowing for safe harvest, optimal routing, and precise tensioning of the transfer.
Volar-Ulnar Incision and FCU Harvest
"We will commence with our volar-ulnar approach. Nurse, please confirm our landmarks." A longitudinal incision is made along the volar-ulnar aspect of the forearm, originating at the junction of the proximal and middle thirds of the forearm and extending distally to the proximal pole of the pisiform. At the distal extent, we curve the incision slightly radially to avoid crossing the wrist flexion creases perpendicularly and to optimize exposure of the neurovascular bundle.
We incise through the skin and subcutaneous tissues, achieving hemostasis with bipolar electrocautery. The robust antebrachial fascia is identified and incised longitudinally with Metzenbaum scissors, immediately exposing the muscular belly of the FCU proximally and its stout tendon distally. The FCU tendon is the most ulnar structure on the volar forearm.
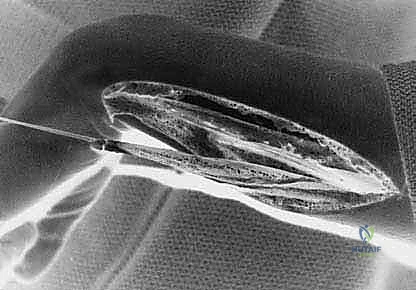
"This is the critical step, fellows. The ulnar nerve and artery lie immediately radial to the FCU tendon." Using blunt dissection and fine Senn retractors, we sweep the radial soft tissues to definitively identify the ulnar neurovascular bundle. A blunt nerve hook is used to gently mobilize the ulnar nerve, ensuring it is protected throughout the remainder of the harvest. We must also remain highly vigilant of the Dorsal Ulnar Sensory Branch (DUSB) in the distal-ulnar subcutaneous tissues. Once the neurovascular structures are secured, the FCU tendon is sharply transected directly at its insertion onto the pisiform to maximize graft length. A 1-0 braided non-absorbable locking whipstitch (e.g., Ethibond or FiberWire) is immediately placed into the distal stump of the FCU to secure control.
We then proceed with extensive proximal mobilization of the FCU muscle belly. Using a combination of sharp and blunt dissection, the muscle is freed from its dense fascial attachments to the ulna and surrounding flexor mass. Mobilization must extend into the proximal third of the forearm. This extensive release is non-negotiable; it ensures a straight line of pull when the tendon is routed dorsally and significantly enhances the muscle's secondary vector as a forearm supinator, counteracting the pronation deformity frequently seen in these patients.
Dorsal Radial Incision and Recipient Preparation
"With our FCU fully mobilized and prepared, let us transition to the dorsal aspect of the wrist."
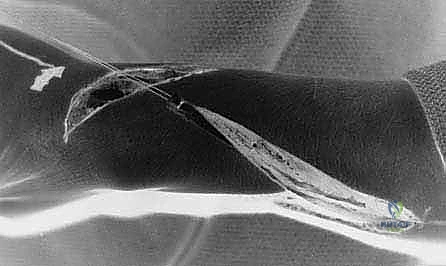
A second, oblique incision is made dorsally, centered over the second and third dorsal extensor compartments, roughly 3 to 4 centimeters proximal to the radiocarpal joint. We dissect through the subcutaneous tissues, taking care to preserve the superficial radial nerve branches. The extensor retinaculum is identified and longitudinally incised over the second compartment. We identify the ECRB and ECRL tendons. The ECRB is selected as the recipient due to its central insertion on the third metacarpal, providing pure wrist extension. The ECRL is retracted radially.
Subcutaneous Routing and the Pulvertaft Weave
We must now create a wide subcutaneous tunnel to route the FCU from the volar-ulnar aspect to the dorsal-radial aspect of the forearm. Using a large curved tendon passer, we develop a tunnel passing around the ulnar border of the forearm, superficial to the deep fascia but deep to the subcutaneous fat. The tunnel must be capacious to prevent any tethering or kinking of the FCU muscle belly, which would drastically reduce its excursion and power. The FCU tendon is drawn through the tunnel into the dorsal wound.
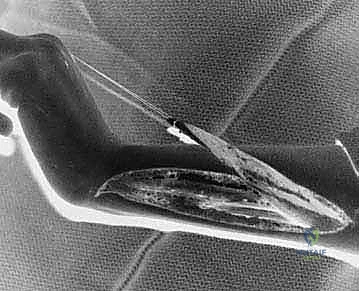
We now perform a classic Pulvertaft weave to secure the FCU to the ECRB. The ECRB tendon is split longitudinally with a #11 blade. The FCU tendon is passed through this slit. "Tensioning is the absolute crux of this procedure." The wrist is held in approximately 30 to 45 degrees of extension, with the forearm in maximal supination and the fingers allowed to rest in a natural cascade. The FCU is pulled distally to its resting physiological tension, and the first pass of the weave is secured with a 3-0 braided non-absorbable mattress suture.
We perform a "tenodesis check" on the table. When we passively flex the wrist, the fingers should extend; when we extend the wrist, the fingers should naturally fall into slight flexion. If the fingers remain extended when the wrist is extended, the transfer is too tight, or the finger flexors require lengthening. Once optimal tension is confirmed, two to three additional passes of the Pulvertaft weave are performed, weaving the FCU back and forth through the ECRB, securing each pass with multiple mattress sutures to ensure a biomechanically robust construct capable of withstanding early active rehabilitation. The wounds are irrigated and closed in layers, and a well-padded long-arm cast is applied with the wrist in 45 degrees of extension and the forearm in supination.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, complications following FCU to ECRB transfer in the spastic upper extremity are well-documented. The surgeon must navigate the delicate balance between under-correction (recurrent deformity) and over-correction (iatrogenic extension contracture). Because we are operating on a dynamic, neurologically abnormal system, the predictability of tendon transfers is inherently lower than in peripheral nerve palsy models.
Over-correction is a devastating complication, resulting in a fixed wrist extension and radial deviation deformity. This typically occurs due to excessive tensioning of the transfer intraoperatively, or mistakenly transferring the FCU into the ECRL instead of the ECRB, which introduces an overpowering radial deviation vector. An over-corrected wrist severely impairs the ability to bring the hand to the midline or to the mouth, devastating activities of daily living. Management of severe over-correction requires surgical revision, often involving lengthening of the transferred tendon or transferring it back to a more neutral position.
Under-correction, or recurrent wrist flexion deformity, is more common. This can result from inadequate initial tensioning, failure to recognize and release concomitant fixed volar capsular contractures, or the progressive nature of the underlying spasticity overpowering the transfer over time. If the transfer stretches out, the patient may require a secondary procedure, such as a fractional lengthening of the volar flexor mass or, in severe recurrent cases, a proximal row carpectomy and total wrist arthrodesis to achieve a stable, functional position. Neurologic complications, particularly injury to the Dorsal Ulnar Sensory Branch (DUSB), occur in up to 5% of cases if the distal volar incision is not carefully managed. DUSB neuromas are exquisitely painful and require prompt surgical excision and proximal transposition of the nerve stump into the pronator quadratus muscle belly.
| Complication | Estimated Incidence | Primary Etiology | Salvage / Management Strategy |
|---|---|---|---|
| Over-correction (Extension/Radial Deviation) | 5 - 10% | Excessive tension; Transfer to ECRL instead of ECRB | Surgical lengthening of transfer; Revision to ECRB |
| Under-correction (Recurrent Flexion) | 10 - 20% | Inadequate tension; Unaddressed joint contracture | Flexor mass lengthening; Wrist arthrodesis |
| DUSB Neuroma | 2 - 5% | Iatrogenic transection during FCU distal harvest | Neuroma excision and deep muscular implantation |
| Clenched Fist Exacerbation | < 5% | Failure to recognize severe FDP/FDS spasticity pre-op | Fractional lengthening of FDP/FDS |
Phased Post-Operative Rehabilitation Protocols
The surgical intervention is merely the first phase of treatment; the ultimate functional outcome is heavily reliant on a rigorous, phased postoperative rehabilitation protocol. In the cerebral palsy population, motor re-education is exceptionally challenging due to the underlying central nervous system pathology, requiring dedicated pediatric occupational therapy.
Phase I: Immobilization and Protection (Weeks 0-4)
Immediately postoperatively, the patient is immobilized in a long-arm bivalved cast. The wrist is strictly maintained in 30 to 45 degrees of extension, the forearm in full supination, and the elbow in 90 degrees of flexion. The digits are left free to encourage early active digital motion, which prevents extrinsic tendon adhesions. The primary goal during this phase is absolute protection of the Pulvertaft weave to allow for robust biological healing of the tendon-to-tendon interface. Caregivers are instructed on cast care and the importance of monitoring for neurovascular compromise.
Phase II: Early Active Motion and Cortical Re-mapping (Weeks 4-8)
At four weeks, the rigid cast is transitioned to a custom-molded thermoplastic volar wrist splint, maintaining the wrist in extension. The splint is removed multiple times daily for therapist-guided active range of motion exercises. This phase focuses on neuromuscular re-education. Because the FCU (a former flexor) is now acting as an extensor, the patient must undergo cortical re-mapping to learn how to actively fire the muscle in its new role. Biofeedback techniques, mirror therapy, and play-based functional tasks are heavily utilized. Passive stretching of the digits is performed to prevent secondary flexor tightness, but forceful passive wrist flexion is strictly avoided to protect the transfer.
Phase III: Strengthening and Bimanual Integration (Weeks 8 and Beyond)
By eight weeks, the tendon transfer is biologically secure. The daytime splint is gradually weaned, though nighttime splinting in extension is maintained for at least 6 to 12 months to counteract nocturnal spastic tone. Therapy shifts toward progressive resistance strengthening of the new wrist extensor complex. More importantly, therapy focuses on the integration of the corrected limb into bimanual activities of daily living—such as tying shoes, cutting food, and dressing. The ultimate success of the FCU to ECRB transfer is judged not by the radiographic angle of the wrist, but by the patient's improved score on the House functional classification scale and their enhanced independence in daily life.
Summary of Landmark Literature and Clinical Guidelines
The evolution of the FCU to ECRB transfer is deeply rooted in orthopedic history. Green’s landmark publication in the mid-20th century revolutionized the management of the spastic flexed wrist, establishing the FCU as the premier donor motor due to its expendability as a flexor and its robust cross-sectional area. Green's original description advocated for transferring the FCU to the ECRL; however, subsequent biomechanical studies demonstrated that this frequently led to an unacceptable radial deviation deformity.
Modern clinical guidelines, heavily influenced by the work of Manske and House, strongly advocate for routing the FCU to the centrally located ECRB. Furthermore, debate has historically existed regarding the routing of the tendon—subcutaneous versus interosseous (through the interosseous membrane). Contemporary consensus, supported by extensive outcome studies, favors the subcutaneous route around the ulnar border. The subcutaneous route avoids the high risk of adhesions associated with the interosseous membrane, preserves greater tendon excursion, and crucially, provides a secondary supination vector that addresses the concomitant pronation deformity often seen in these patients.
Long-term outcome literature confirms that while grip strength measured by dynamometer may not significantly increase postoperatively, the functional grip—the ability to appropriately position the hand in space to grasp an object—improves dramatically. Studies utilizing motion laboratory analysis have objectively quantified the improvement in dynamic wrist kinematics, showing a shift from a resting flexed posture to a functional neutral or extended posture during the reach-and-grasp phase. Ultimately, the literature underscores that surgical intervention must be embedded within a multidisciplinary framework, combining precise surgical execution with aggressive spasticity management and dedicated occupational therapy to achieve enduring functional gains.
