Rheumatoid Tendon Ruptures: An Intraoperative Masterclass in Hand and Wrist Reconstruction
Key Takeaway
This masterclass provides an exhaustive, real-time guide to managing rheumatoid tendon ruptures. We'll meticulously cover comprehensive anatomy, precise preoperative planning, and granular intraoperative execution of extensor and flexor tendon transfers. Emphasis is placed on identifying underlying pathology, meticulous dissection, and salvaging complications, ensuring fellows gain a deep understanding of this challenging reconstructive surgery.
Comprehensive Introduction and Patho-Epidemiology
Rheumatoid arthritis (RA) of the hand and wrist presents one of the most formidable reconstructive challenges in orthopedic surgery. Operating on these patients is not merely a mechanical exercise in tendon repair; it requires a profound understanding of a progressive, systemic autoimmune disease that relentlessly compromises the musculoskeletal system. If left poorly controlled, RA drives profound joint destruction, capsular attenuation, and the insidious attrition of adjacent tendinous structures. The pathophysiology is bipartite, driven simultaneously by enzymatic degradation and mechanical abrasion. Chronic synovitis results in a hyperplastic, highly vascularized pannus that releases a cascade of matrix metalloproteinases (MMPs), interleukins (particularly IL-1 and IL-6), and tumor necrosis factor-alpha (TNF-α). These biochemical agents directly degrade the collagenous matrix of the tendon and its mesotendon, compromising both its structural integrity and microvascular perfusion.
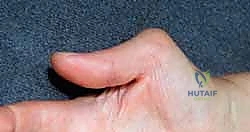
Concurrently, this synovial hypertrophy destabilizes critical ligamentous restraints, most notably the radioulnar ligaments and the triangular fibrocartilage complex (TFCC). This destabilization precipitates joint dislocation or subluxation, creating a harsh biomechanical environment. The classic dorsal manifestation is the Vaughan-Jackson syndrome, also known as caput ulnae syndrome. In this cascade, the distal radioulnar joint (DRUJ) becomes unstable, allowing the distal ulna to subluxate dorsally. The exposed, eroded ulnar head acts as a serrated osteophyte, mechanically grinding through the overlying joint capsule and subsequently the extensor tendons. Because of the anatomical proximity, the extensor digiti quinti (EDQ) is typically the first to succumb to this attritional wear. As the carpus supinates and subluxates volarly over time, the dorsal prominence of the ulna is accentuated, leading to a sequential, ulnar-to-radial rupture pattern involving the extensor digitorum communis (EDC) to the small, ring, and occasionally long fingers.
On the volar aspect of the wrist, a distinct but equally devastating pattern occurs, known as Mannerfelt syndrome. Here, the flexor pollicis longus (FPL) and the adjacent flexor digitorum profundus (FDP) to the index finger are the most vulnerable. These ruptures are typically attritional, secondary to bony erosion from osteophytes at the scaphotrapezial articulation or the volar lip of the radius. The FPL tendon, tethered tightly against these bony prominences within the proximal carpal canal, undergoes ischemic and mechanical necrosis. Beyond mechanical attrition, the hypertrophic tenosynovium directly infiltrates the substance of the flexor tendons, causing an intratendinous rupture that leaves a frayed, disorganized collagen stump that is entirely unsuitable for primary end-to-end repair.
Our surgical objective in these advanced cases is tripartite: complete eradication of the offending tenosynovial tissue, skeletal stabilization to eliminate the mechanical etiology of the rupture, and robust tendon reconstruction to restore functional kinematics. Without addressing the underlying bony deformity—such as performing a Darrach procedure or Sauvé-Kapandji procedure for caput ulnae—any tendon transfer is doomed to inevitable failure.
Detailed Surgical Anatomy and Biomechanics
A masterful reconstruction demands an absolute command of the regional anatomy and the altered biomechanics characteristic of the rheumatoid wrist. The extensor retinaculum, a focal point of pathology, is divided into six distinct fibro-osseous compartments. In the rheumatoid patient, the tenosynovial lining of these compartments undergoes massive proliferation, transforming these protective tunnels into restrictive, destructive choke-points.
The first compartment houses the abductor pollicis longus (APL) and extensor pollicis brevis (EPB). While primary ruptures here are less common, severe tenosynovitis can mimic De Quervain's disease and necessitate release. The second compartment contains the extensor carpi radialis longus (ECRL) and brevis (ECRB). These robust wrist extensors are frequently spared from rupture and serve as excellent, reliable donor tendons for transfers. The third compartment, containing the extensor pollicis longus (EPL), routes the tendon around Lister's tubercle. This sharp anatomical fulcrum makes the EPL highly susceptible to mechanical attrition, often resulting in an isolated rupture that presents with an inability to retropulse the thumb.
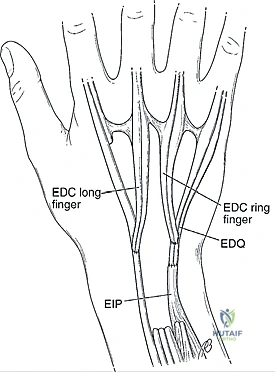
The fourth compartment is the most densely populated, containing the extensor indicis proprius (EIP) and the four slips of the EDC. The EIP is typically located ulnar and volar to the EDC of the index finger and possesses an independent muscle belly extending further distally, making it the premier workhorse for extensor tendon transfers. The fifth compartment, housing the EDQ, lies directly over the DRUJ. Its vulnerability to the subluxated ulnar head initiates the Vaughan-Jackson cascade. Finally, the sixth compartment contains the extensor carpi ulnaris (ECU). In advanced RA, attenuation of the ECU subsheath allows the tendon to subluxate volarly relative to the DRUJ axis. Paradoxically, the ECU transforms from a wrist extensor into a wrist flexor, exacerbating the volar subluxation and supination deformity of the carpus.
Volar anatomy is dominated by the carpal tunnel, a rigid space bounded by the carpal arch dorsally and the transverse carpal ligament volarly. The FPL lies most radially, traversing directly over the radiocarpal and scaphotrapezial joints. The nine flexor tendons (FPL, four FDPs, four FDSs) and the median nerve are tightly packed. Rheumatoid tenosynovitis here not only causes flexor tendon attrition but frequently induces severe, concomitant carpal tunnel syndrome due to elevated intracompartmental pressures. When dissecting volarly, the surgeon must meticulously protect the palmar cutaneous branch of the median nerve and the recurrent motor branch, which may have anomalous courses in chronically deformed wrists.
Clinical Evaluation and Differential Diagnosis
The clinical presentation of rheumatoid tendon ruptures can be deceptively subtle, often masked by pre-existing joint deformities, contractures, or pain-limited excursion. Patients typically report a sudden, painless loss of active digit motion. A meticulous physical examination is required to differentiate true tendon rupture from joint subluxation, extensor tendon subluxation into the intermetacarpal valleys, or neurological deficits.
When assessing dorsal ruptures, the inability to actively extend the metacarpophalangeal (MCP) joints is the hallmark sign. However, the examiner must be aware that the proximal and distal interphalangeal (PIP/DIP) joints may still extend via the intrinsic lumbrical and interosseous muscles. Isolated EDQ rupture can be easily missed if the patient possesses a robust EDC slip to the small finger or a strong junctura tendinum connecting the ring and small finger extensors. The most critical diagnostic maneuver is the tenodesis test. In a normal wrist, passive flexion of the wrist joint inherently increases tension on the extensor tendons, causing passive extension of the MCP joints.
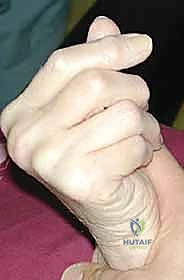
Conversely, passive wrist extension should result in passive finger flexion. In the setting of an extensor tendon rupture, this tenodesis effect is completely abolished; the affected digits remain flaccid and flexed despite passive wrist manipulation.
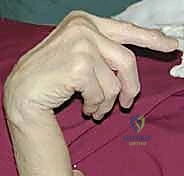
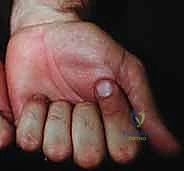
The primary differential diagnosis for an inability to extend the fingers in a rheumatoid patient is Posterior Interosseous Nerve (PIN) palsy. Synovial hypertrophy at the radiocapitellar joint can compress the PIN at the Arcade of Frohse. To differentiate the two clinically: a PIN palsy typically presents with an inability to extend the fingers and the thumb, but wrist extension is preserved and accompanied by radial deviation (due to intact ECRL/ECRB innervated by the radial nerve proper). In contrast, tendon ruptures are usually sequential (ulnar to radial), and the tenodesis effect is absent. If diagnostic ambiguity remains, electromyography (EMG) and nerve conduction studies (NCS) are mandatory. For volar ruptures (Mannerfelt syndrome), the absence of active flexion at the IP joint of the thumb or the DIP joint of the index finger is diagnostic. Direct compression of the FPL muscle belly in the proximal forearm should elicit passive thumb IP flexion; its absence confirms a structural discontinuity.
Exhaustive Indications and Contraindications
Surgical intervention in the rheumatoid hand is a delicate balance of timing, patient optimization, and realistic functional goals. The decision to proceed with tendon reconstruction must be made in the context of the patient's overall disease burden, medical comorbidities, and compliance capacity. Prophylactic tenosynovectomy is indicated when aggressive medical management fails to control local synovial proliferation after 3 to 6 months, particularly if there is impending tendon rupture or early signs of caput ulnae. Once a rupture has occurred, prompt surgical reconstruction is indicated to prevent secondary joint contractures and muscle belly retraction, which complicate delayed repairs.
| Parameter | Indications for Surgical Intervention | Absolute & Relative Contraindications |
|---|---|---|
| Tendon Status | Acute or chronic attritional tendon rupture causing functional deficit. | Intact tendons with medically manageable tenosynovitis (Relative). |
| Joint Status | Progressive DRUJ instability with impending Vaughan-Jackson syndrome. | End-stage, rigid MCP/radiocarpal joint destruction requiring arthrodesis instead (Relative). |
| Neurological | Concomitant compressive neuropathy (e.g., severe Carpal Tunnel Syndrome). | Untreated Cervical Myelopathy mimicking hand dysfunction (Absolute). |
| Medical/Systemic | Medically optimized RA with clearance from Rheumatology. | Active systemic infection, profound immunosuppression, or severe cardiopulmonary disease (Absolute). |
| Soft Tissue | Adequate dorsal/volar skin envelope to cover transferred tendons. | Severe skin atrophy, active vasculitic ulcers, or active local infection (Absolute). |
| Patient Factors | High motivation, cognitive ability to participate in complex post-op rehab. | Severe cognitive impairment or inability to comply with strict immobilization/rehab protocols (Absolute). |
Contraindications must be strictly respected. Performing a complex tendon transfer in a patient with an end-stage, destroyed radiocarpal joint is a technical misstep; such a patient requires a total wrist arthrodesis to establish a stable foundation before or concurrent with tendon reconstruction. Furthermore, the skin envelope in patients on chronic corticosteroids is notoriously fragile. Any sign of impending skin necrosis or active rheumatoid vasculitis is an absolute contraindication to elective tendon transfers, as wound dehiscence over a tendon weave invariably leads to infection and catastrophic failure.
Pre-Operative Planning, Templating, and Patient Positioning
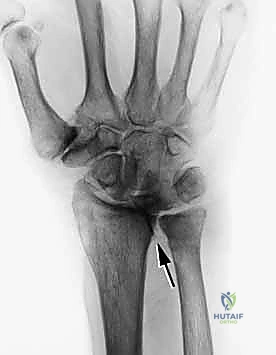
Thorough pre-operative planning is the cornerstone of a successful reconstruction. Radiographic evaluation must include standard posteroanterior, lateral, and oblique views of the wrist and hand to assess the degree of radiocarpal destruction, carpal translocation, and DRUJ subluxation. Advanced imaging, such as Ultrasound or MRI without contrast, is highly sensitive for assessing the extent of tenosynovitis and confirming the exact number of ruptured tendons, which directly dictates the choice of donor tendons.
Medical optimization is paramount. The surgeon must coordinate with the patient's rheumatologist regarding the perioperative management of Disease-Modifying Antirheumatic Drugs (DMARDs) and biologic agents. Current American College of Rheumatology (ACR) and American Association of Hip and Knee Surgeons (AAHKS) guidelines generally support continuing conventional synthetic DMARDs (like Methotrexate) through the perioperative period, while withholding biologic agents (e.g., TNF inhibitors) for one dosing cycle prior to surgery to mitigate infection risk, restarting them once wound healing is secure.
The patient is positioned supine with the operative extremity extended on a radiolucent hand table. A well-padded proximal arm tourniquet is applied. Regional anesthesia, specifically an axillary or supraclavicular brachial plexus block, is highly preferred. This not only provides excellent intraoperative anesthesia and a sympathectomy-induced vasodilation but also affords profound post-operative analgesia, which is critical for these chronic pain patients. The limb is exsanguinated, and the tourniquet is inflated to 250 mmHg. A sterile marking pen is used to map out the planned incisions, the path of the donor tendons, and the anticipated location of the DRUJ reconstruction.
Step-by-Step Surgical Approach and Fixation Technique
Dorsal Exposure and Extensor Retinaculum Management
The procedure commences with a longitudinal, slightly curvilinear dorsal incision centered over the wrist, extending from the distal third of the forearm to the mid-metacarpal level. Full-thickness fasciocutaneous flaps are meticulously elevated to preserve the delicate subdermal vascular plexus and the dorsal sensory branches of the radial and ulnar nerves.
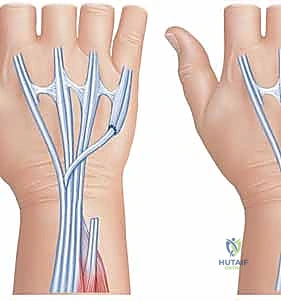
The extensor retinaculum is identified and step-cut or incised longitudinally over the third compartment.
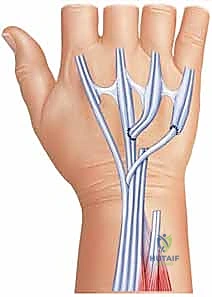
The retinaculum is elevated radially and ulnarly to unroof compartments one through six. A radical tenosynovectomy is performed. The hypertrophic, invasive pannus is meticulously dissected away from the surviving extensor tendons using sharp dissection and tenotomy scissors. Care must be taken to preserve the vincula and the intrinsic blood supply to the remaining intact tendons.
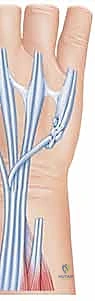
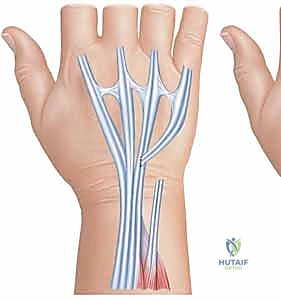
Once the tendons are cleared, the extent of the ruptures is definitively cataloged. The ruptured ends are typically frayed, attenuated, and encased in fibrotic scar tissue.
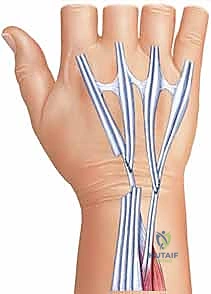
The diseased proximal and distal stumps are sharply debrided back to healthy, organized collagen.
Distal Radioulnar Joint (DRUJ) Reconstruction
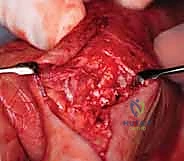
Addressing the underlying skeletal pathology is the most critical step in preventing re-rupture. The prominent, eroded ulnar head (caput ulnae) must be managed. The dorsal capsule of the DRUJ is opened.
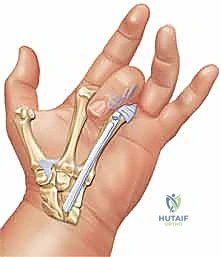
In older, lower-demand patients with severe destruction, a Darrach procedure (resection of the distal ulna) is performed. The distal ulna is transected approximately 1.5 to 2.0 cm proximal to the articular surface using an oscillating saw.
The ulnar stump is smoothed with a rongeur and rasp to ensure no sharp edges remain. To prevent painful radioulnar impingement (ulnar stump instability), the volar capsule and the pronator quadratus are mobilized and sutured dorsally over the resected stump.
In higher-demand patients, a Sauvé-Kapandji procedure (DRUJ arthrodesis with proximal ulnar pseudoarthrosis) may be preferred to maintain ulnar support of the carpus.

Tendon Transfer and Weaving Techniques
With the mechanical environment stabilized, attention turns to tendon reconstruction. The choice of transfer depends on the number of ruptured tendons.
For an isolated EPL rupture, the EIP is the gold standard donor. The EIP is identified at the level of the index MCP joint, transected, and withdrawn into the dorsal wrist wound. It is then rerouted subcutaneously towards the thumb.
For a Vaughan-Jackson presentation with two ruptured tendons (e.g., EDQ and EDC to the small finger), the EIP can be transferred to the distal stump of the EDC/EDQ. Alternatively, an end-to-side transfer (suturing the distal stump of the ruptured small finger EDC to the intact ring finger EDC) is highly effective and minimizes donor site morbidity.
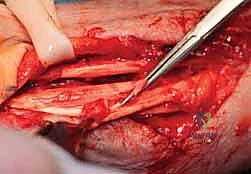
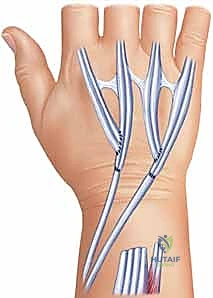
When three or more tendons are ruptured, a more robust donor, such as the Flexor Digitorum Superficialis (FDS) of the ring or long finger, must be harvested via a volar incision, routed through the interosseous membrane, and woven into the distal extensor stumps.
The anastomosis is performed using a Pulvertaft weave. The donor tendon is woven through the recipient tendon at perpendicular angles at least three times.
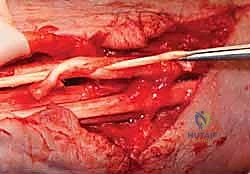
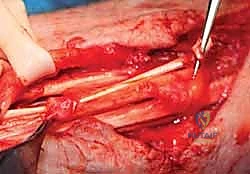
Tensioning is an art form: the transfer must be set slightly tighter than physiological resting tension, as all tendon transfers stretch post-operatively. The wrist is held in 30 degrees of extension, and the MCP joints are held in neutral. The weaves are secured with a non-absorbable, braided 3-0 or 4-0 core suture, supplemented by a running epitendinous 5-0 suture to smooth the junction and prevent adhesions.
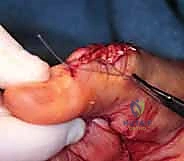
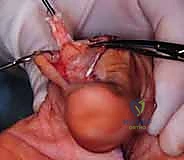
Following the weave, the extensor retinaculum is relocated deep to the reconstructed tendons. This crucial step provides a smooth gliding surface for the new tendons and prevents them from adhering to the raw dorsal carpal capsule or the resected ulnar stump.
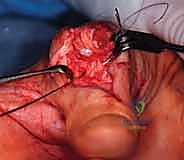
Volar Reconstruction for Mannerfelt Syndrome
For volar ruptures, an extended carpal tunnel incision is utilized. The transverse carpal ligament is released, and a thorough flexor tenosynovectomy is performed. The bony floor of the carpal canal is inspected, and any offending osteophytes from the scaphoid or trapezium are aggressively rongeured flat and covered with a local capsular flap. For an FPL rupture, primary repair is almost never possible. The FDS of the ring finger is the preferred donor. It is transected at the base of the ring finger, withdrawn into the palm
