Surgical Anatomy of Finger Flexor Tendons: Unlock Repair Success
Key Takeaway
Learn more about Surgical Anatomy of Finger Flexor Tendons: Unlock Repair Success and how to manage it. The anatomy of the finger flexor tendons is divided into specific zones, critical for understanding and treating injuries. These zones, like Zone 5 in the forearm, Zone 4 through the carpal tunnel, and Zone 3 (lumbrical origin), each possess unique anatomical characteristics. This zoning impacts injury pathology, repair strategies, and prognosis, making precise anatomical knowledge essential for surgical success.
Introduction and Epidemiology
Flexor tendon injuries of the hand represent a significant challenge in orthopedic surgery, demanding meticulous anatomical understanding and refined surgical technique to restore optimal function. These injuries predominantly affect young, active individuals and are often the result of sharp trauma, crush injuries, or avulsions. The epidemiology highlights a prevalence in industrial and domestic settings, with a disproportionate impact on the dominant hand. Functional outcomes are directly correlated with the precision of repair, the subsequent rehabilitation, and the avoidance of complications such as adhesion formation and rupture. The intricate anatomy of the finger flexor system, characterized by confined osteofibrous tunnels, complex pulley mechanisms, and delicate vascular supply, dictates the technical demands and prognostic implications for each injury zone. This review aims to delineate the critical surgical anatomy, biomechanical principles, and contemporary management strategies essential for optimizing outcomes in flexor tendon repair.
Historically, the management of flexor tendon lacerations, particularly within the digital fibro-osseous sheath, yielded notoriously poor outcomes characterized by severe stiffness or secondary rupture. The evolution of our understanding regarding tendon nutrition, intrinsic healing capabilities, and the biomechanics of multi-strand core sutures has revolutionized treatment paradigms. Modern flexor tendon surgery relies on a delicate balance between achieving a repair strong enough to withstand early active motion while maintaining a low-profile construct that glides smoothly through the native pulley system.
Surgical Anatomy and Biomechanics
The anatomy of the finger flexor tendons is central to understanding the pathology and guiding the treatment of injuries. The system is characterized by a series of distinct anatomical zones, originally devised by Verdan and modified by Milford, each presenting unique surgical challenges and implications for repair. We approach this system proximally to distally, from Zone 5 to Zone 1, alongside a detailed exploration of the associated structures and biomechanics.
Zone 5 Forearm Proximal to Carpal Tunnel
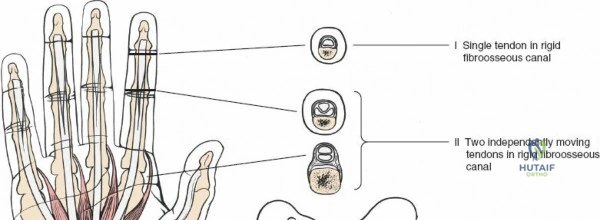
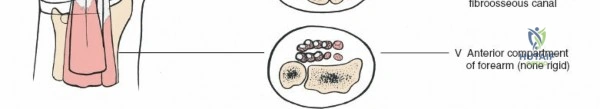
Zone 5 encompasses the anterior compartment of the forearm, extending proximally from the flexor retinaculum and the carpal tunnel. In this region, nine distinct tendons proceed towards the digits: four from the flexor digitorum superficialis (FDS), four from the flexor digitorum profundus (FDP), and one for the thumb, the flexor pollicis longus (FPL).
These tendons, while not constrained by tight osteofibrous canals, are enveloped by a common synovial sheath in the distal forearm, proximal to the carpal tunnel. This common sheath, along with individual FPL sheath, lubricates and nourishes the tendons. Injuries in Zone 5 generally carry a more favorable prognosis for independent digital flexion due to the relatively abundant space surrounding the tendons, minimizing the risk of restrictive adhesions often seen in more distal zones. However, multiple tendon injuries and associated neurovascular compromise are common due to the proximity of the median and ulnar nerves and the radial and ulnar arteries.
Zone 4 Carpal Tunnel
Zone 4 spans the carpal tunnel, a fibro-osseous canal formed by the carpal bones and the transverse carpal ligament (flexor retinaculum). Within this confined space, eight digital flexor tendons (four FDS, four FDP) and the FPL tendon pass alongside the median nerve. The tight anatomical arrangement within the carpal tunnel means that injuries in this zone frequently involve multiple tendons, the median nerve, and sometimes the ulnar nerve or artery depending on the exact location of the trauma.

The FDS tendons are superficial to the FDP tendons at this level. The median nerve lies anterior and radial to the FDS tendons. Tendon repairs in Zone 4 require careful attention to minimize postoperative adhesion formation to adjacent tendons and the median nerve, which can lead to carpal tunnel syndrome or restricted gliding. The integrity of the flexor retinaculum is crucial for preventing bowstringing, but overly tight repair can exacerbate adhesion formation.
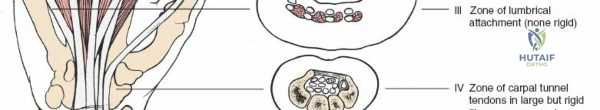
Zone 3 Lumbrical Origin and Palm
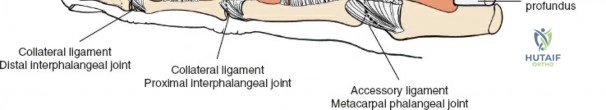
Zone 3 is situated in the palm, extending from the distal border of the carpal tunnel to the proximal aspects of the A1 pulleys. In this zone, the FDS and FDP tendons separate and dive toward their respective digital sheaths. The lumbrical muscles originate from the FDP tendons within this zone, serving a critical biomechanical role in coordinating interphalangeal joint extension and metacarpophalangeal joint flexion. Lacerations in Zone 3 frequently involve the superficial palmar arch and the common digital nerves. Because the lumbricals originate from the FDP tendons here, a severed FDP tendon in Zone 3 or distal Zone 2 may retract proximally, but its retraction is often arrested by the intact lumbrical origin, preventing it from retracting into the carpal tunnel.
Zone 2 Fibro Osseous Sheath
Zone 2, historically termed "No Man's Land" by Bunnell due to the historically dismal outcomes of primary repair, extends from the proximal edge of the A1 pulley to the insertion of the FDS at the middle phalanx. Within this tightly constrained fibro-osseous sheath, the FDS and FDP tendons are intimately associated.
At the level of the proximal phalanx, the FDS tendon bifurcates, allowing the FDP tendon to pass through it at Camper's chiasm. The two slips of the FDS then reunite dorsal to the FDP and insert onto the volar base of the middle phalanx. This intricate relationship requires absolute precision during surgical repair. Any bulkiness in the tendon repair or disruption of the gliding surface will lead to severe adhesions between the FDS, FDP, and the surrounding sheath, severely limiting digital excursion.
Zone 1 Distal to FDS Insertion
Zone 1 extends from the insertion of the FDS on the middle phalanx to the insertion of the FDP at the base of the distal phalanx. Injuries in this zone isolate the FDP tendon. The primary surgical challenge involves managing tendon retraction and ensuring secure fixation of the FDP to the distal phalanx, often requiring pull-out sutures or suture anchors if the distal stump is insufficient for a standard core suture.
Thumb Flexor Zones
The flexor pollicis longus (FPL) anatomy dictates a separate zonal classification. Zone T1 corresponds to the segment distal to the interphalangeal joint. Zone T2 encompasses the fibro-osseous sheath from the metacarpal neck to the interphalangeal joint, containing the A1, oblique, and A2 pulleys. Zone T3 covers the thenar eminence. Understanding the unique oblique pulley of the thumb is paramount, as its preservation or reconstruction is critical to prevent bowstringing and loss of interphalangeal flexion power.
The Pulley System
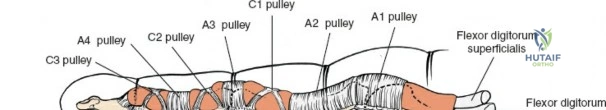
The digital flexor sheath is reinforced by localized thickenings of fibrous tissue that form the pulley system, essential for maintaining the tendons close to the axis of joint rotation and translating linear tendon excursion into angular joint motion. The system consists of five annular (A1 to A5) and three cruciform (C1 to C3) pulleys.
The A2 pulley (located over the proximal phalanx) and the A4 pulley (located over the middle phalanx) are the most critical biomechanical structures. They arise directly from the periosteum and are vital for preventing bowstringing. The A1, A3, and A5 pulleys arise from the volar plates of the metacarpophalangeal, proximal interphalangeal, and distal interphalangeal joints, respectively. The cruciform pulleys are thin, flexible structures that collapse to allow digital flexion without sheath buckling.
Vascular Supply and Tendon Nutrition
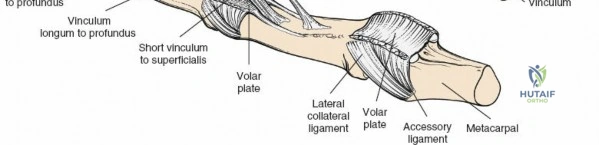
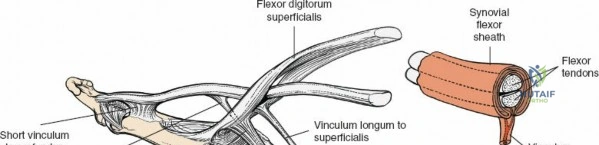
Flexor tendons receive nutrition through a dual mechanism consisting of vascular perfusion and synovial diffusion. The intrinsic vascular supply enters proximally from the muscle belly and distally from the osseous insertions. Segmental blood supply is provided by the vincula system within the digital sheath. The vincula brevia and longa carry vessels from the digital arteries to the dorsal aspect of the tendons.
Synovial diffusion is the primary source of nutrition for the volar aspect of the tendons, particularly within the relatively avascular "watershed" zones located in Zone 2. This physiological reality underscores the importance of preserving the volar surface of the tendon during repair and highlights the intrinsic healing capacity of the tendon, which relies heavily on synovial fluid diffusion when the vincula are compromised.
Indications and Contraindications
The management of flexor tendon injuries requires careful patient selection and timing. Primary repair, performed within the first few days post-injury, is the gold standard for clean, sharp lacerations. Delayed primary repair (within 10 to 14 days) is acceptable if the initial presentation is delayed or if wound conditions necessitate a brief period of observation.
| Clinical Scenario | Operative Indication | Non Operative Indication |
|---|---|---|
| Acute Sharp Laceration (< 14 days) | Primary end-to-end repair | None (unless medically unfit) |
| Partial Laceration (< 60 percent of tendon) | Debridement and epitendinous repair | Splinting and early protected motion |
| Delayed Presentation (3 to 6 weeks) | Delayed primary repair or single-stage graft | Conservative management if asymptomatic |
| Chronic Neglected Rupture (> 6 weeks) | Two-stage tendon reconstruction | Arthrodesis or tenodesis in low-demand |
| Severe Crush or Gross Contamination | Debridement, delayed repair or reconstruction | Wound care, secondary intention healing |
| Significant Soft Tissue Loss | Flap coverage prior to tendon reconstruction | Amputation or terminalization |
Contraindications to immediate primary repair include massive soft tissue avulsion requiring complex flap coverage, profound gross contamination where infection risk is prohibitive, and severe crush injuries resulting in non-viable tendon ends over a large segment. In such instances, thorough debridement and delayed reconstruction, often utilizing a two-stage approach with a silicone Hunter rod, is the indicated pathway.
Pre Operative Planning and Patient Positioning
Thorough preoperative clinical examination is mandatory to assess the integrity of both the FDS and FDP. The FDS is tested by holding the non-injured fingers in full extension (neutralizing the shared FDP muscle belly) and asking the patient to flex the injured finger at the proximal interphalangeal joint. The FDP is evaluated by stabilizing the proximal interphalangeal joint in extension and observing active flexion at the distal interphalangeal joint. Neurovascular status must be meticulously documented, employing two-point discrimination and capillary refill assessments.
While imaging is rarely indicated for sharp lacerations, plain radiographs are necessary to exclude associated fractures or retained radiopaque foreign bodies. Ultrasound or magnetic resonance imaging may be utilized in chronic cases to assess the degree of tendon retraction and the viability of the muscle belly, though physical examination remains the cornerstone of diagnosis.
Patient positioning is standardized to optimize surgical access. The patient is placed supine with the operative extremity extended on a radiolucent hand table. A well-padded pneumatic tourniquet is applied to the upper arm to ensure a bloodless surgical field, which is critical for identifying delicate anatomical structures and minimizing iatrogenic injury. Loupe magnification (minimum 2.5x to 3.5x) and precise microsurgical instrumentation are absolute requirements for modern flexor tendon surgery.
Detailed Surgical Approach and Technique
The surgical approach must provide adequate exposure for tendon retrieval and repair without compromising the vascularity of the skin flaps or creating restrictive scars.

Incision and Exposure
Bruner zigzag incisions or mid-lateral incisions are the standard approaches. The Bruner incision utilizes palmar apices that must not cross the flexion creases at right angles to prevent flexion contractures. The apices of the zigzag flaps should be placed at the lateral borders of the digit, taking extreme care to identify and protect the neurovascular bundles, which lie immediately deep to the subcutaneous fat at the apices of the flaps.
Once the skin flaps are elevated, the fibro-osseous sheath is exposed. The site of laceration is identified. If the tendon has retracted, retrieval must be performed with minimal trauma. Blind grasping with forceps within the sheath is strictly prohibited as it destroys the delicate synovial lining and the epitenon. Techniques for retrieval include milking the forearm and palm, utilizing a pediatric feeding catheter passed through the sheath to pull the tendon distally, or making a separate proximal counter-incision in the palm or distal forearm to identify the tendon and thread it back through the native pulley system.
Core Suture Biomechanics and Placement
The biomechanical goal of the core suture is to provide sufficient tensile strength to withstand the forces of early active motion rehabilitation while minimizing gap formation at the repair site. The normal work of flexion requires approximately 10 to 15 Newtons of force, but this can increase significantly in the presence of post-traumatic edema and joint stiffness.
Contemporary evidence strongly supports the use of multi-strand core repairs. A 2-strand repair (e.g., modified Kessler) provides approximately 20 to 30 Newtons of strength, which is generally insufficient for safe early active motion. A 4-strand repair (e.g., cruciate, Strickland, or double Kessler) yields 40 to 50 Newtons, while 6-strand repairs offer even greater strength at the cost of increased technical complexity and potential tendon bulk.
The core suture is typically performed using a 3-0 or 4-0 non-absorbable braided synthetic suture. The suture purchase should be approximately 0.7 to 1.0 centimeters from the cut edge of the tendon to ensure adequate grip within the longitudinal collagen fibers. The knots should be buried within the repair site to minimize friction against the sheath. Careful attention must be paid to tensioning the core suture; excessive tension causes bunching and increases gliding resistance, while insufficient tension permits gap formation, leading to repair elongation, weakness, and potential rupture.
Epitendinous Suture Application
The addition of an epitendinous suture is a critical step in modern flexor tendon repair. Utilizing a 5-0 or 6-0 monofilament suture in a running, running-locking, or cross-stitch pattern, the epitendinous repair serves multiple vital functions. It increases the overall tensile strength of the construct by 10 to 50 percent, minimizes gap formation, and most importantly, tucks in the frayed edges of the epitenon to create a smooth, low-friction surface that facilitates gliding within the fibro-osseous tunnel.
Pulley Management and Sheath Closure
The management of the flexor sheath and pulleys during repair remains a topic of nuanced surgical debate. Historically, absolute preservation of the A2 and A4 pulleys was mandated to prevent bowstringing. However, if a bulky tendon repair cannot glide smoothly through an intact pulley, the resulting friction will inevitably lead to adhesion formation or rupture.

Current paradigms support the judicious venting of pulleys to accommodate the repair. Up to 50 percent of the A2 pulley and 50 percent of the A4 pulley can be divided longitudinally without causing clinically significant bowstringing or loss of mechanical advantage, provided the remaining pulley structures and the A3 pulley are intact. If the sheath can be closed without restricting tendon glide, it may be repaired with fine absorbable sutures to restore the synovial environment, though routine sheath closure is not universally practiced and is secondary to ensuring unhindered tendon excursion.
Complications and Management
Despite meticulous surgical technique, flexor tendon repairs are fraught with potential complications. The biological response to tendon injury involves both intrinsic healing (from tenocytes within the tendon) and extrinsic healing (from fibroblasts migrating from the surrounding sheath). Excessive extrinsic healing leads to the formation of dense peritendinous adhesions.
| Complication | Estimated Incidence | Etiology and Risk Factors | Management and Salvage Strategies |
|---|---|---|---|
| Adhesion Formation | 15 to 30 percent | Immobilization, bulky repair, severe trauma | Intensive hand therapy; surgical tenolysis after 3 to 6 months of plateaued therapy |
| Tendon Rupture | 4 to 9 percent | Weak core suture, non-compliance, gap formation | Prompt re-exploration and primary repair if acute; two-stage reconstruction if delayed |
| Joint Contracture | 10 to 20 percent | Prolonged splinting, capsular scarring | Dynamic splinting, serial casting; surgical capsulotomy concurrent with tenolysis |
| Bowstringing | 1 to 5 percent | Excessive loss of A2 or A4 pulleys | Pulley reconstruction using extensor retinaculum or palmaris longus tendon grafts |
| Infection | 1 to 3 percent | Contaminated wound, compromised skin flaps | Aggressive surgical debridement, targeted intravenous antibiotics, delayed reconstruction |
Tenolysis is a demanding salvage procedure indicated only when the patient has reached a definitive plateau in therapy (typically 3 to 6 months postoperatively), soft tissues are supple, and passive range of motion significantly exceeds active range of motion. The procedure involves meticulous lysis of adhesions while preserving the critical pulleys and vascular supply. Patients must be prepared for immediate postoperative active motion to prevent the recurrence of adhesions.
In cases of chronic rupture or severe destruction of the fibro-osseous sheath, a two-stage tendon reconstruction is indicated. Stage one involves radical excision of the scarred tendon and insertion of a silicone rod to induce the formation of a biological pseudosheath, alongside pulley reconstruction. Stage two, performed 3 to 6 months later, involves replacing the silicone rod with an autologous tendon graft (e.g., palmaris longus or plantaris).
Post Operative Rehabilitation Protocols
The success of a flexor tendon repair is inextricably linked to the postoperative rehabilitation protocol. The primary objective is to apply controlled mechanical stress to the repair site, which stimulates intrinsic tenocyte proliferation and collagen realignment, while simultaneously promoting tendon excursion to prevent restrictive adhesions.
Historically, the Kleinert protocol utilized dynamic rubber band traction to passively flex the digit, while the patient actively extended against the traction within the confines of a dorsal blocking splint. The Duran protocol emphasized controlled passive motion programs, where the patient or therapist passively moved the digits through specific arcs of motion to ensure differential gliding between the FDS and FDP tendons.
Contemporary practice has largely shifted towards Early Active Motion (EAM) protocols, provided a robust multi-strand core repair (minimum 4-strand) has been achieved. Protocols such as the Manchester or St. John regimens initiate true active flexion within the first few days postoperatively. The patient is placed in a dorsal blocking splint with the wrist in slight extension or neutral, and the metacarpophalangeal joints flexed to 50 to 70 degrees. This positioning reduces the passive resting tension on the flexor tendons.
Patients are instructed to perform controlled "place-and-hold" exercises or light active flexion to a half-fist position. The application of early active force significantly improves ultimate tendon excursion and reduces adhesion formation. However, it requires a highly compliant patient and close supervision by a specialized hand therapist, as the risk of rupture is highest between the second and fourth postoperative weeks when the tendon is in its weakest, most inflammatory phase of healing.
Summary of Key Literature and Guidelines
The evolution of flexor tendon surgery is heavily rooted in biomechanical and clinical research. Strickland's seminal work established the biomechanical rationale for multi-strand repairs, demonstrating that a 4-strand core suture combined with an epitendinous repair provides the necessary tensile strength to safely withstand the forces of early active motion.
Further advancements by Tang and colleagues have refined the management of Zone 2 injuries, challenging the dogma of absolute pulley preservation. Their research supports the strategic venting of the A2 and A4 pulleys to accommodate tendon edema and repair bulk, proving that smooth gliding takes precedence over anatomical pulley restoration in the prevention of adhesions.
Current clinical guidelines from major hand surgery societies strongly advocate for a minimum of a 4-strand core repair utilizing non-absorbable synthetic suture, the routine use of a meticulously placed epitendinous suture, and the implementation of early active motion rehabilitation protocols under the guidance of certified hand therapists. The integration of these evidence-based principles has fundamentally transformed the prognosis of flexor tendon injuries, shifting the clinical expectation from salvage of function to the reliable restoration of complex digital kinematics.
