Hereditary Multiple Exostoses: Arab Board Orthopedic Review
Comprehensive Introduction and Patho-Epidemiology
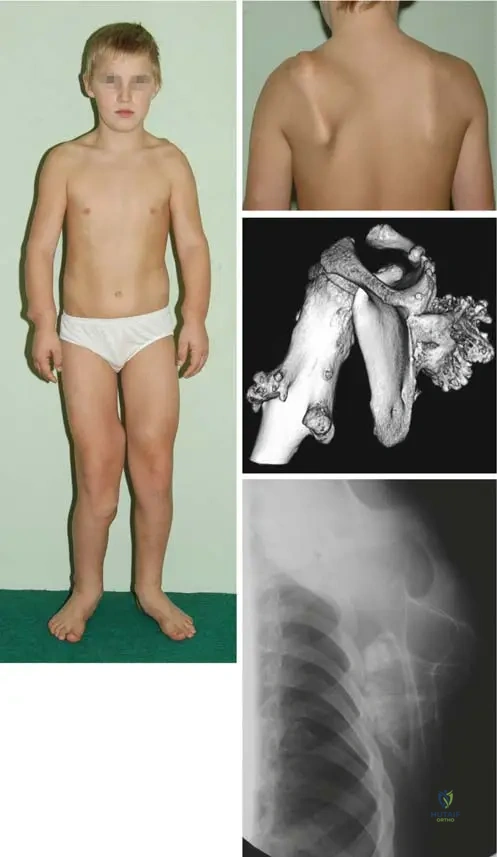
Hereditary Multiple Exostoses (HME), also known historically as diaphyseal aclasis or familial osteochondromatosis, is a complex, autosomal dominant skeletal dysplasia characterized by the development of multiple benign osteochondromas. These lesions typically arise from the juxta-epiphyseal regions of long bones, though they can manifest in any bone that develops via endochondral ossification. For the orthopedic surgeon preparing for the Arab Board Orthopedic Review, a profound understanding of the molecular pathogenesis, clinical manifestations, and long-term sequelae of HME is absolutely paramount. The classic presentation, often tested in board examinations, involves a young child presenting with palpable, painless bony prominences, frequently with a positive family history, such as the classic vignette of a 6-year-old boy and his 4-year-old cousin exhibiting scapular lesions.

The genetic basis of Hereditary Multiple Exostoses is rooted in loss-of-function mutations in the tumor suppressor genes EXT1 (located on chromosome 8q24.11) and EXT2 (located on chromosome 11p11.2). These genes are responsible for encoding Golgi-associated glycosyltransferases, which are critical for the biosynthesis of heparan sulfate. Heparan sulfate proteoglycans (HSPGs) are essential components of the extracellular matrix in the physeal growth plate. They play a vital regulatory role in the diffusion and signaling of Indian Hedgehog (Ihh), Parathyroid Hormone-related Protein (PTHrP), and Fibroblast Growth Factors (FGFs). When EXT mutations cause a deficiency in heparan sulfate, the highly orchestrated negative feedback loop between Ihh and PTHrP is disrupted. This disruption allows islands of physeal cartilage to escape the normal longitudinal growth vector, herniating through the perichondrial ring of LaCroix and proliferating outward to form exostoses.
Epidemiologically, HME has an estimated incidence of 1 in 50,000 live births, though isolated populations may exhibit higher prevalence due to founder effects. The penetrance of the EXT mutations is exceptionally high, approaching 100% by the age of 12. Consequently, nearly all individuals harboring the mutation will develop clinically or radiographically detectable osteochondromas by early adolescence. EXT1 mutations are generally associated with a more severe clinical phenotype, characterized by a higher burden of exostoses, more profound limb deformities, and a statistically higher risk of malignant transformation compared to EXT2 mutations. While the majority of these lesions remain benign, the systemic burden of the disease frequently leads to significant mechanical, cosmetic, and psychological morbidity for the patient.
Clinical manifestations typically begin in the first decade of life. Patients present with multiple palpable masses, most commonly around the knee (distal femur and proximal tibia), proximal humerus, distal radius and ulna, and the scapula. Beyond the mere presence of the masses, the aberrant growth of these lesions frequently tethers surrounding soft tissues and disrupts the normal growth of the host bone. This tethering effect leads to characteristic angular deformities, such as forearm bowing, ulnar deviation of the wrist, valgus deformities of the knee and ankle, and global limb length discrepancies. Furthermore, the lesions can cause mechanical impingement on adjacent neurovascular structures, tendons, and muscles, leading to chronic pain, peripheral neuropathies, and restricted range of motion.
The Phenomenon of Malignant Transformation
The most dreaded complication of Hereditary Multiple Exostoses is the malignant transformation of a benign osteochondroma into a secondary peripheral chondrosarcoma. While the risk of malignant transformation in a solitary osteochondroma is estimated to be less than 1%, patients with HME face a lifetime risk ranging from 1% to 5%. This transformation almost exclusively occurs in adulthood, after skeletal maturity has been reached and the normal physiological growth of the exostoses should have ceased.
The pathogenesis of malignant transformation is believed to involve a "second hit" mutation in the remaining wild-type EXT allele within the cartilage cap, coupled with subsequent genetic alterations in pathways such as p53 or retinoblastoma (Rb). Clinically, malignant transformation must be suspected in any adult patient with HME who presents with a newly enlarging mass, new-onset pain in a previously asymptomatic lesion, or neurovascular compromise. Radiographically, signs of malignancy include irregularity or destruction of the underlying bony stalk, a dispersed or "fluffy" calcification pattern in the cap, and most importantly, a cartilage cap thickness exceeding 1.5 to 2.0 centimeters as measured on T2-weighted Magnetic Resonance Imaging (MRI).
Detailed Surgical Anatomy and Biomechanics
An in-depth comprehension of the surgical anatomy of an osteochondroma is essential for safe and effective excision. An osteochondroma is not merely a tumor of bone; it is a developmental hamartoma characterized by a cartilage cap that overlies a bony stalk. The pathognomonic anatomical feature of an osteochondroma, which distinguishes it from other surface bone lesions, is the absolute continuity of its medullary cavity and cortical bone with the medullary cavity and cortex of the host bone.

Histologically, the cartilage cap of a growing osteochondroma mirrors the zonal architecture of a normal physeal growth plate. It contains resting, proliferative, and hypertrophic zones, which undergo endochondral ossification at the base of the cap to elongate the bony stalk. A distinct perichondrial layer covers the cartilage cap; this layer is continuous with the periosteum of the host bone. During surgical excision, it is biomechanically and anatomically critical to excise the lesion extra-periosteally at its base. Failure to completely remove the perichondrium and the entire cartilage cap leaves residual chondrogenic cells, which is the primary cause of post-operative recurrence, particularly in the skeletally immature patient.
Biomechanics of Forearm Deformities
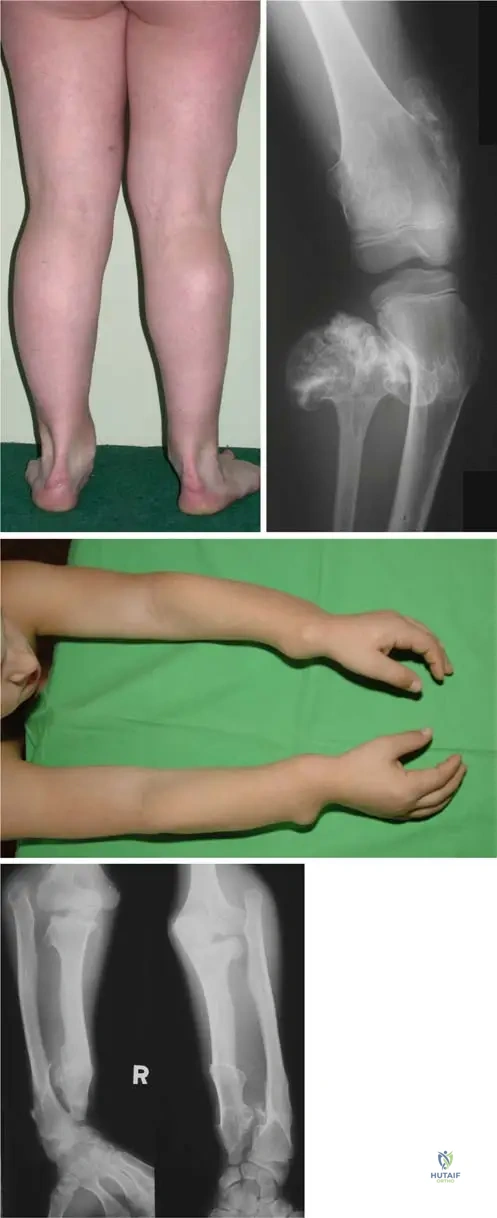
The forearm is one of the most frequently and severely affected anatomical regions in patients with HME, occurring in up to 60% of cases. The biomechanical interplay between the paired radius and ulna makes this region uniquely susceptible to deformity. The distal ulna, which contributes significantly to the longitudinal growth of the forearm, is disproportionately affected by osteochondromas. The presence of a distal ulnar lesion retards its growth, leading to absolute ulnar shortening.
Because the radius and ulna are tethered by the interosseous membrane and the proximal and distal radioulnar joints (PRUJ and DRUJ), the continued growth of the radius against a shortened ulna creates a profound biomechanical mismatch. This results in secondary bowing of the radius (usually convex laterally and dorsally), an increased ulnar tilt of the distal radial articular surface, and progressive ulnar deviation of the carpus. If left untreated, the severe radial bowing and relative ulnar shortening will eventually lead to the subluxation and frank dislocation of the radial head at the radiocapitellar joint, severely compromising elbow flexion, forearm pronosupination, and overall upper extremity function. This complex pathoanatomy is classified using the Masada classification system, which dictates the surgical reconstructive approach.
Biomechanics of Lower Extremity Deformities
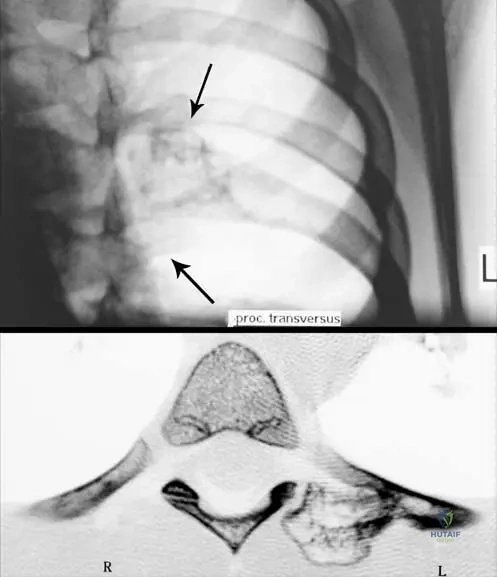
In the lower extremities, the biomechanical consequences of HME frequently manifest around the knee and the ankle. At the knee, asymmetric growth of the distal femoral or proximal tibial physes—often exacerbated by the tethering effect of large juxta-articular osteochondromas on the iliotibial band or medial collateral ligament—leads to progressive genu valgum.
At the ankle, a similar phenomenon occurs due to the differential growth between the tibia and the fibula. Osteochondromas of the distal fibula retard its longitudinal growth, leading to relative fibular shortening. The lateral malleolus fails to provide adequate buttressing for the talus within the ankle mortise. Consequently, the talus shifts laterally into valgus, creating a secondary valgus deformity of the distal tibial plafond. This altered biomechanical axis shifts the weight-bearing forces laterally, predisposing the patient to early-onset osteoarthritis of the tibiotalar joint and significant gait abnormalities.
Exhaustive Indications and Contraindications
The management of Hereditary Multiple Exostoses requires a highly individualized, patient-centric approach. Because patients possess dozens to hundreds of lesions, prophylactic excision of all osteochondromas is neither feasible nor indicated. The decision to intervene surgically must be based on a careful risk-benefit analysis, weighing the symptoms and progressive deformities against the risks of multiple anesthetic exposures and surgical complications.
The primary indication for surgical excision is pain. This pain may result from direct mechanical irritation of overlying soft tissues, bursitis (bursa formation over the cartilaginous cap is common), or compression of adjacent neurovascular structures. For example, a proximal fibular osteochondroma frequently compresses the common peroneal nerve, leading to foot drop and paresthesias, necessitating urgent decompression and excision. Another absolute indication is any clinical or radiographic suspicion of malignant transformation in an adult patient.
Deformity correction represents a more complex set of indications. In the growing child, progressive angular deformities (such as Masada type I and II forearm deformities, or significant genu valgum) should be addressed early to prevent permanent joint subluxation and irreversible articular damage. Prophylactic excision of lesions that are clearly tethering growth, even if currently painless, is often indicated to normalize the biomechanical axis.
| Parameter | Surgical Indications | Surgical Contraindications |
|---|---|---|
| Symptomatology | Intractable pain, mechanical block to joint motion, recurrent bursitis. | Asymptomatic lesions discovered incidentally. |
| Neurovascular | Compression neuropathies (e.g., peroneal nerve, radial nerve), vascular compromise or pseudoaneurysm formation. | Absence of neurovascular impingement; lesions in close proximity to critical structures without symptoms. |
| Deformity | Progressive angular deformity (genu valgum, ankle valgus), impending or actual radial head dislocation (Masada I/II). | Mild, non-progressive deformities that do not affect function or joint stability. |
| Oncologic | Suspicion of malignant transformation (growth after skeletal maturity, cap > 1.5-2 cm, new pain). | Stable lesions in adults with thin cartilage caps (< 1 cm) and no interval growth. |
| Patient Factors | Severe cosmetic distress (relative indication), limb length discrepancy > 2-3 cm. | Severe medical comorbidities precluding anesthesia; unrealistic patient expectations regarding cosmetic outcomes. |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough pre-operative planning is the cornerstone of successful surgical intervention in HME. Standard orthogonal radiographs of the affected limb are mandatory to confirm the diagnosis (demonstrating medullary continuity) and to assess the size, location, and base characteristics (sessile vs. pedunculated) of the osteochondroma. Full-length standing radiographs of the lower extremities are essential for evaluating the mechanical axis, joint orientation angles, and limb length discrepancies.
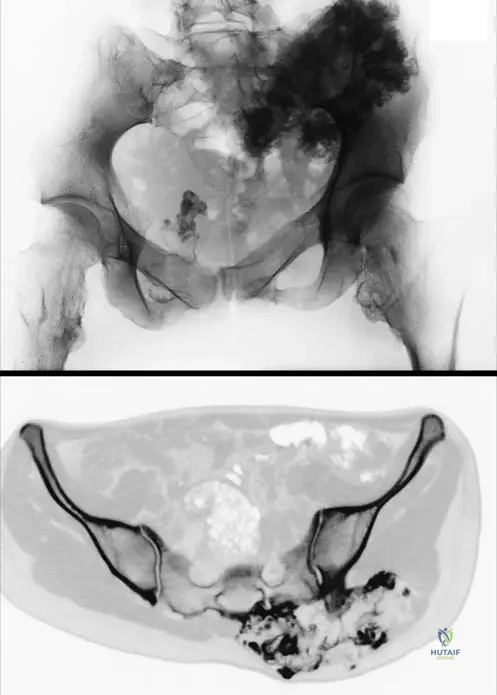
Advanced imaging is frequently required. Magnetic Resonance Imaging (MRI) is the gold standard for evaluating the thickness of the cartilage cap and assessing the relationship of the osteochondroma to adjacent soft tissues, particularly neurovascular bundles. In the context of suspected malignant transformation, a T2-weighted MRI is critical; a hyperintense cartilage cap thicker than 1.5 to 2.0 cm is highly suspicious for secondary peripheral chondrosarcoma. Computed Tomography (CT) with 3D reconstruction is invaluable for surgical planning in anatomically complex regions, such as the pelvis, scapula, and spine, where intra-operative orientation can be challenging.
Templating for Deformity Correction
When addressing complex deformities, such as those in the forearm, meticulous pre-operative templating is required. For a Masada Type I deformity (distal ulnar osteochondroma with ulnar shortening and radial bowing), the surgeon must calculate the exact amount of ulnar lengthening required to restore the DRUJ and pull the radial head back into alignment. This often involves templating for a uniplanar or multiplanar external fixator for gradual distraction osteogenesis. If an acute correction is planned, the size of the structural bone graft and the appropriate plate length must be determined. For lower extremity deformities in growing children, the calculation of remaining growth using the multiplier method or Bowen's charts is necessary to determine the optimal timing for hemiepiphysiodesis (guided growth).
Patient positioning is dictated by the anatomical location of the target lesion. For upper extremity and knee procedures, the patient is typically positioned supine with a radiolucent hand table or leg ramp. A sterile tourniquet is highly recommended to maintain a bloodless field, which is critical for identifying and protecting neurovascular structures that may be draped over or displaced by the exostosis. For posterior lesions of the scapula or proximal femur, the lateral decubitus or prone position may be required. Fluoroscopy should always be available in the operating room to confirm the complete excision of the base and to verify the alignment of any corrective osteotomies.
Step-by-Step Surgical Approach and Fixation Technique
The surgical management of HME ranges from simple extra-periosteal excision to highly complex, multi-staged deformity corrections. The fundamental principle of any exostosis excision is the complete removal of the cartilage cap and its overlying perichondrium to prevent recurrence.
Technique for Simple Exostosis Excision
- Incision and Dissection: A longitudinal incision is made over the most prominent aspect of the lesion, taking care to place the incision in a cosmetically acceptable line if possible. Deep dissection must proceed meticulously. The surgeon must be acutely aware that normal anatomical planes are often distorted by the mass.
- Neurovascular Protection: Identify and protect major neurovascular structures before addressing the bone. For example, when approaching a proximal fibular lesion, the common peroneal nerve must be identified proximally and traced distally as it winds around the fibular neck, often stretched tightly over the exostosis.
- Extra-Periosteal Exposure: The overlying muscle and fascia are split longitudinally. The bursa, if present, is incised. The surgeon must incise the periosteum of the host bone proximal to the base of the stalk and elevate it continuously over the exostosis.
- Resection: Using an osteotome, oscillating saw, or rongeur, the stalk is transected flush with the normal cortical contour of the host bone. It is imperative not to leave a prominent stump, but equally important not to create a massive cortical defect that could serve as a stress riser for a subsequent fracture.
- Defect Management: The medullary canal of the host bone will be exposed. Bone wax can be applied to control cancellous bleeding. The excised specimen must always be sent for routine histopathological examination to rule out malignant transformation, regardless of its benign clinical appearance.
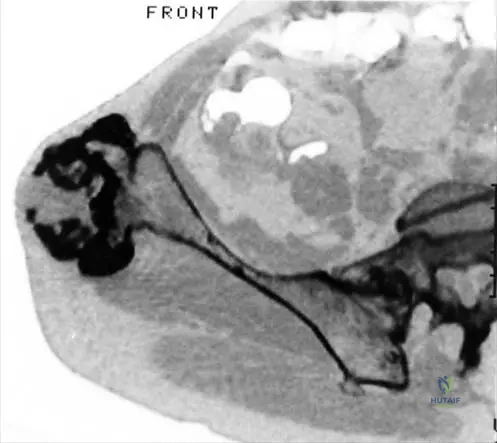
Forearm Reconstruction (Masada Classification Directed)
The surgical approach to the forearm is dictated by the Masada classification.
* Masada Type I (Mainly Ulnar Involvement): The primary goal is to restore ulnar length. The distal ulnar osteochondroma is excised. An ulnar osteotomy is performed, typically in the mid-diaphysis. Gradual lengthening using a monolateral external fixator is the preferred method, allowing for soft tissue stretching and precise restoration of the radioulnar variance. As the ulna is lengthened, the tethering effect on the radius is relieved, which often leads to spontaneous improvement of radial bowing and reduction of the radial head.
* Masada Type II (Radial Head Dislocation): In addition to ulnar lengthening, direct intervention at the radius is often required. If the radial head is chronically dislocated and deformed, a radial closing wedge osteotomy may be necessary to redirect the proximal radius into the radiocapitellar joint. In severe, neglected cases in adults, radial head excision may be the only salvage option to restore rotation.
* Masada Type III (Significant Radial Involvement): These present with distal radial osteochondromas causing severe relative radial shortening. Treatment involves excision of the radial lesion and, if necessary, radial lengthening or a corrective distal radial osteotomy to restore the articular tilt.
Lower Extremity Deformity Correction
For the valgus knee in a skeletally immature patient, guided growth using tension band constructs (e.g., eight-Plates) applied to the medial distal femoral or proximal tibial physis is highly effective and minimally invasive. The osteochondromas contributing to the deformity should be excised concurrently. In the skeletally mature patient, acute correction via a medial closing wedge or lateral opening wedge distal femoral osteotomy (DFO) or high tibial osteotomy (HTO) secured with rigid plate fixation is required.
Ankle valgus is addressed by excising the distal fibular osteochondroma. If the patient has significant remaining growth, a medial malleolar transphyseal screw can be used to tether the medial distal tibial physis, allowing the lateral side to "catch up" and correct the valgus. If the fibula is severely shortened, a distal tibial and fibular supramalleolar osteotomy, or a fibular lengthening procedure, may be indicated to restore the ankle mortise.
Complications, Incidence Rates, and Salvage Management
Surgical intervention in HME is fraught with potential complications, primarily due to the distorted anatomy and the extensive nature of the required reconstructions. The orthopedic surgeon must be prepared to identify and manage these complications promptly.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage Management & Prevention |
|---|---|---|---|
| Tumor Recurrence | 2% - 5% (Higher in children) | Incomplete excision of the cartilage cap or perichondrial ring; operating on very young, rapidly growing children. | Prevention: Meticulous extra-periosteal dissection; complete removal of the base. Salvage: Revision excision if symptomatic or causing new deformity. |
| Neurovascular Injury | 1% - 3% | Distorted anatomy; stretching of nerves over large lesions (e.g., peroneal nerve at proximal fibula, popliteal artery at distal femur). | Prevention: Wide exposure; routine nerve identification prior to resection. Salvage: Primary nerve repair, nerve grafting, or tendon transfers (e.g., posterior tibial tendon transfer for permanent foot drop). |
| Malignant Transformation | 1% - 5% (Lifetime Risk) | "Second hit" mutation; EXT1 genotype; advanced age. | Prevention: Annual clinical screening; MRI for any suspicious changes. Salvage: Wide en bloc oncologic resection; endoprosthetic reconstruction; amputation in advanced cases. |
| Post-Operative Fracture | < 1% | Excessive cortical resection creating a stress riser; aggressive physical therapy before bone remodeling. | Prevention: Flush resection with the host cortex; avoid deep cratering. Salvage: Standard fracture management (casting vs. internal fixation depending on location). |
| Nonunion / Premature Consolidation | 5% - 10% (In lengthening procedures) | Poor regenerate formation during distraction osteogenesis; overly rapid or slow distraction rates. | Prevention: Optimize distraction rate (typically 1 mm/day in divided doses); preserve periosteum during osteotomy. Salvage: Bone grafting and internal fixation for nonunion; osteoclasis for premature consolidation. |
| Loss of Joint Motion | Variable | Scar tissue formation; failure to achieve concentric reduction of the radial head in Masada II reconstructions. | Prevention: Early, aggressive post-operative rehabilitation. Salvage: Capsular release; radial head excision in mature, symptomatic patients. |
Phased Post-Operative Rehabilitation Protocols
Rehabilitation following surgery for HME is highly dependent on the specific procedure performed. A simple exostosis excision requires a vastly different protocol than a multi-planar forearm reconstruction utilizing distraction osteogenesis.
Phase I: Immediate Post-Operative (Weeks 0-2)
The primary goals in this phase are wound healing, pain control, and the prevention of joint contractures. For simple excisions, the limb is typically placed in a soft, bulky dressing. Immediate, gentle active and active-assisted range of motion (ROM) is encouraged to prevent soft tissue adhesions, particularly when the exostosis was adjacent to a joint capsule or major muscle group (e.g., quadriceps mechanism). Weight-bearing is usually tolerated for lower extremity simple excisions, provided a large cortical defect was not created. For osteotomies and external fixator placements, the limb is immobilized or protected, and pin-site care protocols are initiated.
Phase II: Early Rehabilitation and Distraction (Weeks 2-6)
For patients undergoing simple excision, this phase focuses on achieving full, symmetric ROM and initiating progressive resistance exercises. Return to normal activities of daily living is expected.
For patients undergoing distraction osteogenesis (e.g., ulnar lengthening), this phase coincides with the active distraction period. Physical therapy is absolutely critical during this time to maintain joint motion above and below the fixator. In forearm lengthening, aggressive therapy to maintain wrist flexion/extension, elbow ROM, and pronosupination is mandatory to prevent joint subluxation and contractures as the bone and soft tissues are placed under tension.
Phase III: Consolidation and Strengthening (Weeks 6-12+)
Once full ROM is achieved and radiographic evidence of bone remodeling (or regenerate consolidation in the case of lengthening) is observed, the focus shifts to advanced strengthening, proprioception, and return to sports. For external fixator patients, the frame is removed once three out of four cortices of the regenerate bone show solid consolidation on orthogonal radiographs. Following frame removal, the limb is often protected in a functional brace or cast for an additional 2-4 weeks to prevent fracture through the regenerate bone.
Long-term surveillance is a critical component of the post-operative protocol. All patients with HME require lifelong follow-up. Clinical examinations should be performed annually to assess for new deformities, monitor the growth of existing lesions, and screen for any signs of malignant transformation.
Summary of Landmark Literature and Clinical Guidelines
The management of Hereditary Multiple Exostoses has evolved significantly, guided by landmark genetic discoveries and refined surgical classification systems.
- Genetic and Molecular Foundations: The seminal work by Bovée et al. and Wuyts et al. established the critical role of EXT1 and EXT2 mutations in the pathogenesis of HME. Their research elucidated the function of these genes in heparan sulfate biosynthesis, providing the molecular explanation for the disruption of the Indian Hedgehog signaling pathway at the physeal level. This understanding has paved the way for potential future targeted medical therapies aimed at restoring HSPG function.
- Forearm Deformity Classification: The Masada et al. classification system (1989) remains the gold standard for evaluating and surgically planning the treatment of HME-related forearm deformities. By categorizing the deformities based on the primary site of involvement (ulna vs. radius) and the presence of radial head dislocation, Masada provided a reproducible algorithm that dictates whether ulnar lengthening, radial osteotomy, or a combination thereof is required.
- Malignant Transformation Guidelines: Current orthopedic oncology guidelines emphasize that routine, whole-body MRI screening for asymptomatic patients is not cost-effective and is generally not recommended. However, targeted MRI evaluation is the standard of care for any symptomatic lesion in an adult. The literature consistently supports that a cartilage cap thickness greater than 1.5 cm on T2-weighted MRI in a skeletally mature individual is the most reliable radiographic predictor of secondary peripheral chondrosarcoma, mandating prompt oncologic resection.
- Guided Growth Principles: The application of the tension-band principle (e.g., eight-Plates), popularized by Stevens, has revolutionized the management of angular deformities in the growing child with HME. This minimally invasive technique has largely replaced highly morbid acute osteotomies for the correction of genu valgum and ankle valgus in the pediatric population, allowing for gradual correction utilizing the child's remaining growth potential.
