Transfemoral Amputation: An Oncologic Masterclass for Fellows
Key Takeaway
This masterclass guides orthopaedic fellows through oncologic transfemoral amputation. We meticulously cover surgical anatomy, preoperative planning, and granular intraoperative steps from incision to closure. Learn precise techniques for neurovascular management, bone transection, myodesis, and nerve handling. Extensive pearls, pitfalls, and postoperative strategies for optimal patient outcomes and complication management are emphasized, ensuring a comprehensive understanding of this complex procedure.
Comprehensive Introduction and Patho-Epidemiology
The transfemoral amputation, commonly referred to as the above-knee amputation (AKA), represents a profound intersection of radical oncologic resection and reconstructive functional preservation. In the realm of orthopedic oncology, this procedure is rarely the first line of defense; rather, it is reserved for aggressive, high-grade sarcomas, massive soft tissue malignancies with critical neurovascular encasement, or cases of failed limb-salvage surgery complicated by recalcitrant infection or local recurrence. While the psychological and physiological impacts on the patient are immense, the primary directive remains absolute: the complete extirpation of the malignancy with definitively negative margins.
Historically, the pendulum of surgical management for extremity sarcomas swung heavily toward radical amputations, including hip disarticulations, to ensure survival. However, with the advent of neoadjuvant chemotherapy, advanced cross-sectional imaging, and sophisticated radiation protocols, limb-sparing procedures have become the standard of care for over 85% of extremity sarcomas. Yet, the transfemoral amputation remains an indispensable tool in the orthopedic oncologist’s armamentarium. It is a technically demanding procedure that requires a nuanced understanding of three-dimensional anatomy, tumor biology, and the biomechanics of prosthetic ambulation.
The patho-epidemiology of tumors necessitating transfemoral amputation typically involves large, extracompartmental lesions of the distal femur or popliteal fossa. High-grade osteosarcomas with massive extraosseous extension, undifferentiated pleomorphic sarcomas invading the sciatic nerve, or synovial sarcomas encasing the popliteal trifurcation often leave the surgeon with no viable reconstructive options that would yield a functional, sensate limb. Furthermore, the rising incidence of radiation-induced sarcomas and the complex management of periprosthetic joint infections following distal femoral replacements have expanded the indications for this ablative procedure.
As orthopedic surgeons, our responsibility extends beyond the mere removal of the afflicted limb. The construction of the residual limb—the "stump"—must be approached with the same meticulous care as a complex arthroplasty or free flap reconstruction. The residual limb is the patient's new interface with the world; its length, soft tissue envelope, myodesis integrity, and neuromatous pain profile will directly dictate the patient's future ambulatory capacity and overall quality of life. An inadequately constructed residual limb will lead to prosthetic intolerance, profound disability, and a failure of the ultimate rehabilitative goal.
Detailed Surgical Anatomy and Biomechanics
Before initiating any surgical incision, a masterful, three-dimensional understanding of the thigh's intricate anatomy is paramount. The goal is to achieve a wide oncologic margin while preserving functional musculature, optimizing the soft tissue envelope, and minimizing neurovascular complications. The spatial relationships of major structures change drastically along the longitudinal axis of the femur.
Proximal Thigh Anatomy and High Transfemoral Amputation
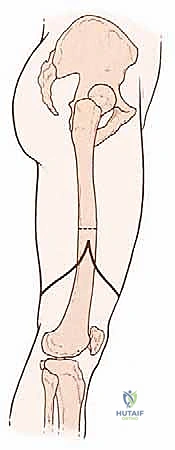
At the level of the proximal thigh, just distal to the lesser trochanter, the cross-sectional anatomy is particularly dense and critical. The musculature here is defined by the origins of the quadriceps femoris (rectus femoris, vastus lateralis, medialis, and intermedius), the adductor group (adductor longus, brevis, magnus, pectineus, and gracilis), and the hamstrings (semitendinosus, semimembranosus, and biceps femoris). The sartorius muscle courses obliquely, forming the lateral boundary of the femoral triangle, a critical landmark for initial vascular control.
Within the femoral triangle, the neurovascular bundle is superficial and easily accessible. The femoral artery lies just beneath the sartorius muscle, anterior to the adductor longus and the femur. Medial to the artery is the femoral vein, while the femoral nerve branches lie laterally, supplying the quadriceps and providing sensation to the anterior thigh. A critical structure at this level is the profunda femoris artery (deep femoral artery), which branches posteriorly and provides the primary blood supply to the thigh musculature via its perforating branches. Ligation of these vessels must be meticulous to prevent massive postoperative hematoma.
The sciatic nerve, the largest nerve in the body, is positioned deep within the posterior compartment at this high level. It lies posterior to the adductor magnus muscle and anterior to the long head of the biceps femoris. Due to its massive cross-sectional area, the sciatic nerve is accompanied by a significant vasa nervorum. High transection of this nerve requires careful ligation of its intrinsic vessels to prevent troublesome bleeding deep within the posterior muscular bed.
Midthigh Diaphyseal Anatomy
The midthigh represents the most common level for a standard transfemoral amputation, offering optimal bone length for prosthetic fitting while allowing for adequate oncologic margins for distal third tumors. The muscular compartments are well-defined by the thick fascial septa. The vastus medialis and adductor magnus become particularly relevant as they form the boundaries of the adductor canal (Hunter's canal).
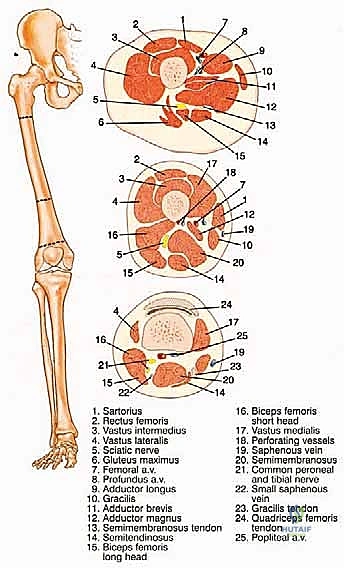
In the midthigh, the femoral artery has migrated medially and posteriorly to lie between the vastus medialis and the adductor magnus. Interestingly, at this level, the spatial relationship of the vein and artery often reverses from their proximal arrangement, with the femoral vein typically positioned lateral and posterior to the artery. The saphenous nerve accompanies these vessels within the canal and must be identified and managed to prevent a superficial neuroma.
The sciatic nerve maintains its vertical descent in the posterior compartment, situated between the short head of the biceps femoris and the semimembranosus. At this level, it may begin to bifurcate into the tibial and common fibular (peroneal) nerves, though they are usually still bound within a common epineural sheath. The surgeon must be prepared to manage these as separate entities if the bifurcation is high.
Distal Thigh and Biomechanical Considerations
While less common for primary bone sarcomas due to the risk of inadequate margins, supracondylar amputations may be indicated for specific soft tissue lesions or non-oncologic salvage. The anatomy here is dominated by the distal tendinous attachments of the thigh musculature and the transition of the neurovascular bundle into the popliteal fossa. The femoral artery passes through the adductor hiatus to become the popliteal artery, positioned directly posterior to the femur and deep to the sciatic nerve branches.
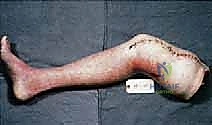
Biomechanically, the transfemoral amputee faces significant challenges. The loss of the anatomical knee joint eliminates the primary shock absorber and dynamic stabilizer of the lower extremity. Consequently, the energy expenditure required for ambulation increases by 40% to 65% compared to baseline. The preservation of the adductor magnus and its rigid fixation to the distal femur (myodesis) is the single most critical biomechanical step in this operation.
Without a secure adductor myodesis, the unantagonized hip abductors (gluteus medius and minimus) and flexors (iliopsoas) will drive the residual femur into a fixed abduction and flexion contracture. This malposition forces the distal tip of the femur laterally through the soft tissue envelope, creating severe pain, skin breakdown, and an inability to achieve a functional prosthetic socket fit. The adductor magnus, therefore, must be captured and tensioned appropriately to restore the anatomic mechanical axis of the femur.
Exhaustive Indications and Contraindications
The decision to proceed with a transfemoral amputation is complex, requiring a synthesis of oncologic staging, patient physiology, and functional prognosis. The primary directive is life preservation through local disease control. When limb salvage compromises oncologic margins or yields a non-functional, painful appendage, amputation becomes the superior choice.
| Category | Transfemoral Amputation Parameters |
|---|---|
| Absolute Indications | - High-grade bone or soft tissue sarcoma with extensive neurovascular encasement (e.g., popliteal trifurcation, sciatic nerve) precluding functional salvage. - Massive fungating or bleeding tumors of the distal extremity. - Failed limb-sparing surgery secondary to profound local recurrence. - Unsalvageable periprosthetic joint infection post-distal femoral replacement with massive bone loss. - Ischemic, non-viable distal limb due to tumor embolization or primary vascular invasion. |
| Relative Indications | - Severe, intractable pain from a distal extremity tumor in a palliative setting where amputation would significantly improve quality of life. - Pathologic fracture of the distal femur with massive soft tissue contamination in a patient who is a poor candidate for complex reconstruction. - Pediatric patients with massive distal femoral tumors where limb salvage would result in unacceptable leg length discrepancies at skeletal maturity. |
| Absolute Contraindications | - Patient refusal or lack of informed consent. - Tumors extending into the proximal femur or pelvis requiring hip disarticulation or hemipelvectomy for negative margins. - Severe, uncorrectable systemic coagulopathy or hemodynamic instability precluding major surgery. |
| Relative Contraindications | - Palliative scenarios where the patient's life expectancy is measured in mere weeks, and the surgical trauma would outweigh the benefits of local tumor control. - Severe contralateral limb pathology that would render the patient entirely non-ambulatory post-amputation, though amputation may still be required for pain/infection control. |
The surgeon must carefully weigh these factors in a multidisciplinary tumor board setting. The presence of pulmonary metastases, for instance, is not an absolute contraindication if the primary tumor is a source of severe pain, hemorrhage, or systemic sepsis. In such cases, a "palliative amputation" can drastically improve the patient's remaining quality of life. Conversely, if a patient has a highly functional limb despite a large tumor, and margins can be achieved via complex vascular grafting and nerve transfer, limb salvage should be aggressively pursued.
Pre-Operative Planning, Templating, and Patient Positioning
Pre-operative planning for an oncologic amputation is an exhaustive process that dictates the entire surgical execution. It relies heavily on advanced imaging to define the exact proximal extent of the tumor, its relationship to the neurovascular structures, and the presence of any skip metastases within the medullary canal.
Advanced Imaging and Level Determination
Our planning begins with a comprehensive review of all imaging modalities. Plain radiography, while providing the initial macroscopic view of bony destruction, is notoriously insensitive to early cortical invasion and marrow replacement. Orthogonal views of the femur, tibia, and fibula are mandatory to assess the gross structural integrity of the limb.
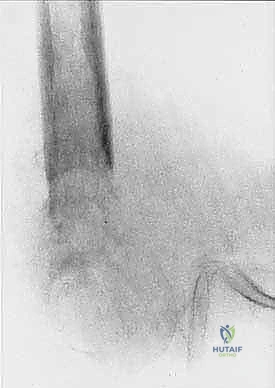
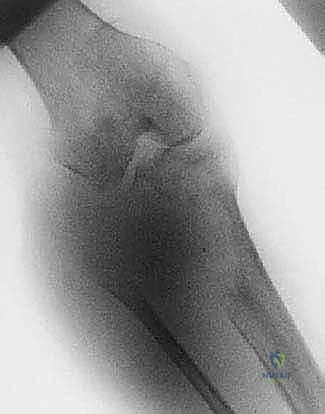
Magnetic Resonance Imaging (MRI) is the undisputed gold standard for preoperative planning in orthopedic oncology. A total femur MRI is required to evaluate the intraosseous extent of the tumor and to rule out skip lesions. The T1-weighted coronal and sagittal sequences are critical for defining the exact level of marrow replacement, which directly dictates the level of the femoral osteotomy. The T2-weighted axial sequences provide unparalleled detail regarding extraosseous soft tissue extension and neurovascular encasement.
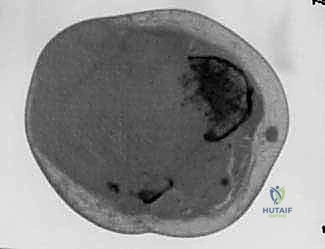
Computed Tomography (CT) is utilized for detailed assessment of cortical bone destruction and is highly sensitive for detecting subtle periosteal reactions or pathologic micro-fractures. In older patients or those with large tumors compressing the vascular tree, CT angiography is vital. It confirms the patency of the proximal vessels and helps the surgeon anticipate the level of vascular ligation required to ensure a well-perfused residual limb.
The biopsy tract must be meticulously planned and documented. The absolute oncologic rule is that the biopsy tract is considered contaminated with tumor cells and must be excised en bloc with the amputation specimen. If the biopsy was performed poorly, extending proximally into the planned surgical field, the level of amputation must be raised to encompass this contaminated tissue.
Patient Positioning and Operating Room Setup
Patient positioning must facilitate 360-degree access to the thigh while protecting pressure points. The patient is positioned supine on a standard radiolucent operating table. A bump is placed under the ipsilateral hip to slightly internally rotate the leg, bringing the entire thigh into a neutral, accessible position.
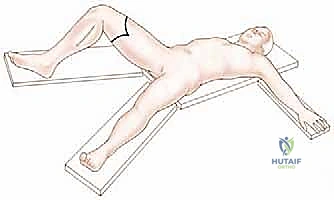
The operative extremity is prepped and draped freely to allow for manipulation, flexion, and abduction during the procedure. In oncologic cases, the use of an exsanguinating Esmarch bandage is strictly contraindicated, as it may force tumor emboli into the systemic circulation. Instead, the limb is elevated for 3 to 5 minutes to allow for gravity exsanguination before inflating a sterile pneumatic tourniquet placed as proximally as possible on the thigh. If the tumor extends too proximally to allow for a tourniquet, the procedure must be performed under direct vascular control, isolating and ligating the femoral vessels as the very first step of the operation.
Step-by-Step Surgical Approach and Fixation Technique
The execution of a transfemoral amputation requires a deliberate, methodical approach. The surgeon must balance the need for wide oncologic margins with the creation of a robust, well-padded, and biomechanically sound residual limb.
Flap Design and Incision
The standard approach utilizes equal anterior and posterior "fish-mouth" flaps. The apex of the flaps should be positioned at the level of the planned bone resection. The anterior flap is designed to be slightly longer and thicker, incorporating the fascia and a portion of the quadriceps muscle to provide a robust cushion over the distal end of the cut femur.
However, in oncologic surgery, flap design is entirely dictated by tumor geometry. If the tumor extends anteriorly, an atypical long posterior flap (incorporating the hamstrings and gastrocnemius) may be required. The incisions are carried sharply through the skin and subcutaneous tissue down to the deep fascia. Hemostasis is meticulously maintained using electrocautery.
Deep Dissection and Vascular Control
The deep dissection begins anteriorly. The deep fascia is incised in line with the skin incision. The quadriceps muscle is transected, beveling the cut proximally so that the muscle is slightly longer than the planned bone cut, allowing it to easily drape over the distal femur.
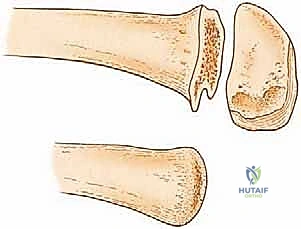
Attention is then turned to the medial neurovascular bundle. The femoral artery and vein are identified within the adductor canal (or femoral triangle, depending on the level). These vessels must be isolated individually. The artery is double-ligated with heavy non-absorbable sutures (e.g., #0 silk) and transected. The vein is managed similarly. It is critical to avoid mass ligation, as this can lead to arteriovenous fistula formation.
Nerve Management and Targeted Muscle Reinnervation (TMR)
The management of major peripheral nerves is a critical step in preventing debilitating postoperative neuroma pain. The sciatic nerve is identified in the posterior compartment. Historically, the nerve was simply placed under gentle traction, ligated to control the vasa nervorum, sharply transected, and allowed to retract deep into the muscular bed.
In contemporary orthopedic oncology, Targeted Muscle Reinnervation (TMR) or Regenerative Peripheral Nerve Interfaces (RPNI) should be strongly considered. TMR involves identifying redundant motor nerve branches in the residual limb (e.g., motor branches to the transected quadriceps or hamstrings) and coapting the cut ends of the major sensory/mixed nerves (sciatic, saphenous) to these motor targets. This provides the regenerating axons with a functional target, drastically reducing the incidence of symptomatic neuromas and phantom limb pain.
Bone Resection and Myodesis
Once the soft tissues have been circumferentially divided, the periosteum of the femur is incised circumferentially at the planned osteotomy level. The periosteum should not be stripped proximally, as this devascularizes the bone and promotes ring sequestrum formation. The femur is transected using an oscillating saw, with continuous saline irrigation to prevent thermal necrosis.
The linea aspera (the posterior ridge of the femur) is notoriously sharp and must be aggressively rasped and beveled to prevent it from eroding through the posterior soft tissues.

The most critical reconstructive step is the myodesis. Drill holes are placed through the distal lateral and anterior cortices of the femur. The adductor magnus tendon is identified, pulled under physiologic tension across the distal end of the femur, and sutured securely to the lateral drill holes using heavy, non-absorbable sutures (e.g., #2 FiberWire or #5 Ethibond). This anchors the adductors, counteracting the abductor pull. The quadriceps tendon is then brought down and sutured to the posterior femur or the adductor fascia, completing the soft tissue envelope.
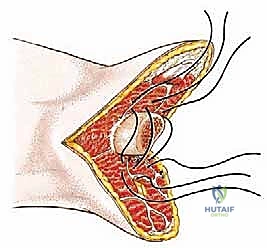
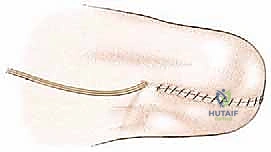
The wound is irrigated copiously. A deep subfascial drain is placed. The deep fascia is closed meticulously to ensure a watertight seal over the bone, followed by layered closure of the subcutaneous tissue and skin using non-absorbable sutures or staples.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, transfemoral amputations in the oncologic population carry a significant risk of complications. The systemic effects of neoadjuvant chemotherapy, local radiation, and profound malnutrition inherently impair wound healing and immune response.
| Complication | Estimated Incidence | Pathophysiology and Salvage Management |
|---|---|---|
| Wound Dehiscence / Infection | 15% - 25% | Driven by poor tissue perfusion, prior radiation, and immunocompromise. Management: Aggressive operative debridement. Superficial infections may heal secondarily or with negative pressure wound therapy (NPWT). Deep infections involving the bone require revision amputation at a higher level to achieve clean, well-vascularized tissue |
