FHL Tendon Augmentation for Insertional Achilles Tendinosis: An Intraoperative Masterclass
Key Takeaway
Join us in the OR for an immersive masterclass on Flexor Hallucis Longus (FHL) tendon augmentation for insertional Achilles tendinosis. We'll meticulously cover preoperative planning, precise patient positioning, and a granular, real-time breakdown of surgical execution, from initial incision to graft fixation and wound closure. Learn critical anatomical considerations, pearls, pitfalls, and comprehensive postoperative rehabilitation strategies to ensure optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
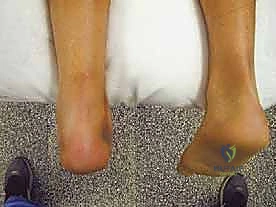
The surgical management of end-stage insertional Achilles tendinopathy represents one of the most demanding challenges in foot and ankle orthopedics. The term "insertional Achilles tendinitis" has historically misled clinicians into treating a fundamentally degenerative process as an acute inflammatory one. In reality, we are confronting a profound tendinosis—a chronic, non-inflammatory, degenerative cascade characterized by angiofibroblastic hyperplasia, mucoid degeneration, and the loss of normal parallel collagen fiber orientation. This pathology accounts for approximately 10% to 20% of all Achilles disorders and presents a unique biomechanical dilemma at the bone-tendon interface.
The pathophysiologic cycle is driven by repetitive microtrauma that outpaces the tendon's intrinsic healing capacity. This leads to tenocyte apoptosis and the deposition of disorganized, mechanically inferior type III collagen in place of the robust type I collagen normally found in the Achilles. As the disease progresses, neovascularization occurs, bringing with it unmyelinated nociceptive nerve fibers that contribute to the patient's recalcitrant pain. Furthermore, the mechanical impingement from a prominent posterosuperior calcaneal tuberosity (Haglund’s deformity) exacerbates the mechanical wear on the anterior aspect of the tendon, frequently leading to intratendinous calcification and macroscopic tearing.
When conservative measures fail and the structural integrity of the Achilles insertion is compromised—specifically when thorough débridement necessitates the resection of more than 50% of the tendon's cross-sectional area—simple reattachment is biomechanically insufficient. It is in these advanced cases that Flexor Hallucis Longus (FHL) tendon augmentation becomes the gold standard. The FHL transfer not only provides robust mechanical augmentation but also introduces a highly vascularized muscle belly to a relatively hypovascular region, thereby fundamentally altering the biologic environment and promoting durable healing.
Detailed Surgical Anatomy and Biomechanics
To execute this procedure with precision, a profound understanding of the posterior heel's "enthesis organ" and the deep posterior compartment of the leg is non-negotiable. The Achilles tendon is the confluence of the gastrocnemius and soleus muscles, functioning as the primary plantarflexor of the ankle. Its insertion is not a single point but a broad, complex expansion over the posterior calcaneal tuberosity. The tendon fibers rotate approximately 90 degrees externally as they descend, meaning the medial gastrocnemius fibers insert posterolaterally, a crucial detail when mapping the topography of partial tears and degenerative nodules.
The enthesis organ itself comprises the tendon insertion, the underlying fibrocartilage, the retrocalcaneal bursa, and the Kager fat pad. The retrocalcaneal bursa sits in the recess between the anterior Achilles tendon and the posterosuperior calcaneus. In chronic tendinosis, this bursa becomes hypertrophied and fibrotic, acting as a space-occupying lesion that increases tension and friction. The Kager fat pad, which normally acts as a dynamic neurovascular supply to the anterior tendon, often becomes fibrotic and retracts, further starving the diseased tendon of essential nutrients and progenitor cells.
The Flexor Hallucis Longus is the ideal candidate for augmentation due to its synergistic function, proximity, and favorable anatomic characteristics. Originating from the distal two-thirds of the posterior fibula, the FHL is a powerful, bipennate muscle. Crucially, its muscle belly extends very distally—often to the level of the tibiotalar joint—which allows the vascular muscle to be draped directly over the repaired Achilles tendon, enhancing the local biologic milieu. Furthermore, the FHL fires in phase with the Achilles during the gait cycle, ensuring a seamless biomechanical transition.
However, the harvest of the FHL is fraught with potential neurovascular hazards. The FHL lies in the deep posterior compartment, immediately adjacent to the tibial nerve and posterior tibial vessels. As the FHL courses distally toward its fibro-osseous tunnel beneath the sustentaculum tali, the neurovascular bundle sits directly medial to it. Meticulous dissection and positive identification of these structures are imperative. Inadvertent traction or direct injury to the tibial nerve during FHL harvest can result in devastating sensory and motor deficits to the plantar aspect of the foot, turning a successful mechanical repair into a clinical failure.
Exhaustive Indications and Contraindications
The decision to proceed with FHL augmentation hinges on a careful preoperative assessment of tendon viability. The primary indication for FHL transfer in the setting of insertional Achilles tendinosis is the anticipation—or intraoperative realization—that adequate débridement of the diseased tendon and calcific deposits will leave less than 50% of the native tendon intact. Attempting to repair a severely compromised, thinned Achilles tendon under high tension invariably leads to failure, elongation, or catastrophic rupture.
Patient selection is equally critical. The ideal candidate is a patient with chronic, recalcitrant insertional pain who has exhausted a minimum of six months of comprehensive non-operative management. This includes eccentric loading protocols, immobilization in a controlled ankle motion (CAM) boot, orthoses, and activity modification. Older, low-demand patients with severe degenerative changes, or patients presenting with chronic, neglected insertional ruptures, are also prime candidates for primary FHL augmentation.
Conversely, absolute contraindications include active local or systemic infection, severe peripheral vascular disease that precludes adequate wound healing, and active Charcot neuroarthropathy. Relative contraindications involve younger, elite athletes where the loss of terminal hallux flexion strength—despite the compensatory action of the flexor hallucis brevis—might marginally impact explosive sprinting or jumping mechanics. In such highly specific athletic populations, alternative augmentation strategies might be considered, though the FHL remains the most robust option.
| Category | Specific Clinical Scenarios |
|---|---|
| Primary Indications | Degenerative tendon requiring >50% resection; Chronic insertional Achilles rupture; Failed prior Achilles repair; Severe intratendinous calcification compromising structural integrity. |
| Secondary Indications | Older/sedentary patients with poor tissue quality; Revision surgery for recurrent Haglund's syndrome with tendon compromise. |
| Absolute Contraindications | Active deep space infection; Severe peripheral arterial disease (ABI < 0.5); Active Charcot neuroarthropathy; Medically unstable for anesthesia. |
| Relative Contraindications | Elite jumping/sprinting athletes (due to potential loss of terminal hallux push-off); Severe peripheral neuropathy (impacting post-op rehab). |
Pre-Operative Planning, Templating, and Patient Positioning
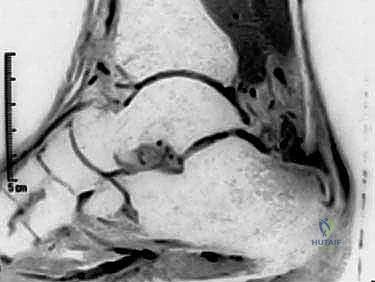
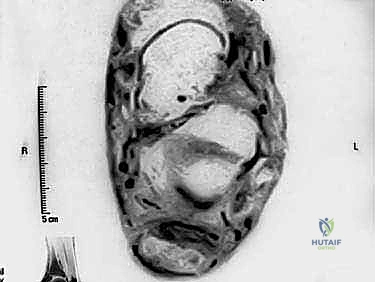
Preoperative planning relies heavily on advanced imaging. While weight-bearing radiographs are essential for evaluating the Haglund's deformity, calcaneal pitch, and the presence of intratendinous calcifications, Magnetic Resonance Imaging (MRI) is the definitive modality. Sagittal and axial T2-weighted sequences allow the surgeon to map the exact extent of mucoid degeneration, partial tearing, and the percentage of cross-sectional area involved. This dictates the surgical strategy before the first incision is ever made.
Patient positioning is a critical step that dictates the flow of the entire operation. The patient is placed prone on the operating table. It is absolutely vital that the patient is positioned such that the feet hang freely over the distal edge of the table. This allows for unrestricted intraoperative assessment of ankle dorsiflexion and plantarflexion, which is required for accurate tensioning of the FHL graft. All bony prominences must be meticulously padded to prevent positioning-related neuropathies.
A crucial, often overlooked step in the setup is the preparation of the surgical field. Both the operative and the contralateral non-operative lower extremities must be prepped and draped into the sterile field up to the level of the mid-calf. This bilateral exposure allows the surgeon to directly compare the resting equinus tension of the operative ankle with the normal contralateral side. Recreating this resting tension is the single most important factor in preventing postoperative overlengthening or excessive stiffness. A thigh tourniquet is applied and inflated after exsanguination to ensure a bloodless field, maximizing visualization of the delicate neurovascular structures.
Step-by-Step Surgical Approach and Fixation Technique
Incision and Exposure
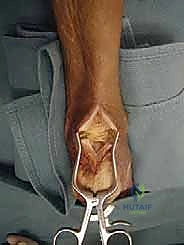
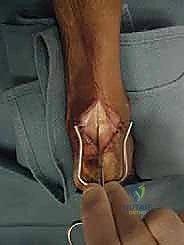
The procedure begins with a robust, longitudinal midline incision directly over the Achilles tendon, starting approximately 4 to 5 cm proximal to the insertion and extending distally to the distal aspect of the calcaneal tuberosity. The midline approach is favored over medial or lateral incisions because it avoids the sural nerve laterally and respects the angiosomal vascular supply to the posterior skin, minimizing the risk of ischemic wound breakdown.
Once the skin is incised, it is imperative to develop full-thickness fasciocutaneous flaps. The surgeon must dissect directly down to the paratenon and elevate the skin, subcutaneous tissue, and paratenon as a single, contiguous layer. Undermining the skin or creating thin flaps will inevitably lead to marginal necrosis and devastating postoperative wound complications. Retraction should be gentle, utilizing blunt self-retaining retractors to minimize tissue trauma.
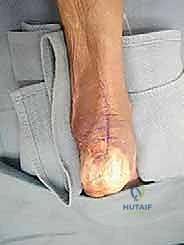
Retrocalcaneal Debridement and Ostectomy
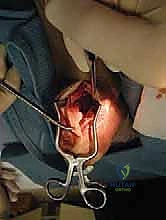
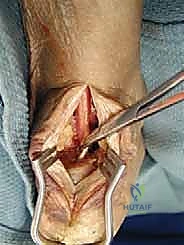
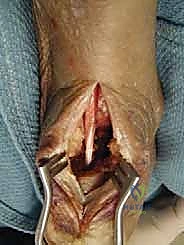
With the Achilles tendon exposed, a longitudinal, midline tendon-splitting incision is made through the substance of the tendon. This incision is carried down to the bone of the posterior calcaneus. The medial and lateral halves of the tendon are sharply elevated off the calcaneal footprint using a scalpel or an elevator. This exposes the underlying pathology: the hypertrophied retrocalcaneal bursa, the Haglund's prominence, and the degenerative intratendinous calcifications.
The retrocalcaneal bursa is excised in its entirety using rongeurs and sharp dissection. Following bursectomy, the Haglund's deformity is addressed. Using an oscillating saw or an osteotome, a generous posterosuperior ostectomy is performed. The cut should be angled from posterior-superior to anterior-inferior, ensuring that no bony prominence remains to impinge on the anterior aspect of the reconstructed tendon during maximal dorsiflexion. Following the ostectomy, the Achilles tendon itself is aggressively débrided of all necrotic, fibrotic, and calcified tissue until healthy, bleeding tendon margins are achieved. If this leaves less than 50% of the tendon, we proceed to FHL harvest.

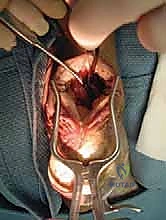
Flexor Hallucis Longus Harvest
The FHL is accessed through the same midline incision by dissecting deep to the Achilles tendon and the deep fascia of the posterior compartment. The FHL muscle belly is identified by its low-lying nature, extending distally toward the ankle joint. The investing fascia of the FHL is incised longitudinally. At this juncture, the surgeon must exercise extreme caution. The tibial nerve and posterior tibial vessels lie immediately medial to the FHL. Blunt dissection is used to gently sweep the neurovascular bundle medially, protecting it with a smooth retractor.
The FHL tendon is traced distally as it enters its fibro-osseous tunnel beneath the sustentaculum tali. While a double-incision technique (harvesting from the midfoot) yields a longer graft, a single-incision posterior harvest provides ample length for insertion into the calcaneus and significantly reduces morbidity. The ankle and hallux are maximally plantarflexed to deliver as much tendon proximally as possible. The tendon is then transected sharply as far distally into the tunnel as visualization allows. A locking whipstitch using a high-strength non-absorbable suture is immediately placed into the distal end of the harvested FHL tendon to maintain control and prepare it for passage.
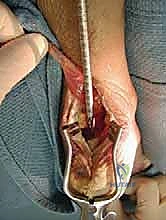
Tendon Routing and Calcaneal Fixation
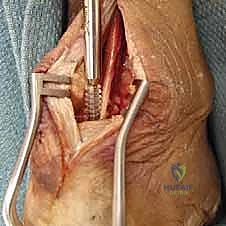
Attention is now turned to the calcaneus for graft fixation. A guide pin is placed into the superior aspect of the calcaneal tuberosity, directing it anteriorly and slightly inferiorly toward the plantar aspect of the calcaneus, taking care to avoid penetrating the subtalar joint. The position is confirmed with fluoroscopy. A reamer, sized to match the diameter of the harvested FHL tendon (typically 7 to 9 mm), is used to create a blind-ended bone tunnel of approximately 25 to 30 mm in depth.
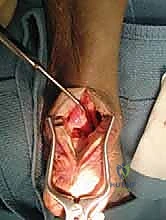
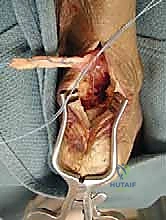
The whipstitched FHL tendon is then passed into the calcaneal tunnel. Fixation is achieved using a bio-tenodesis interference screw. The critical step here is establishing the correct tension. The ankle is placed in resting equinus, matching the exact resting posture of the prepped contralateral limb. The FHL tendon is pulled taut into the tunnel, and the interference screw is advanced over a guidewire to secure the tendon within the bony socket. This achieves robust, aperture-level fixation that allows for early postoperative rehabilitation.
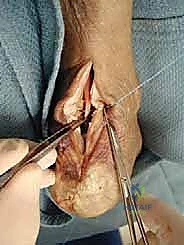
Augmentation and Closure
Following rigid fixation of the FHL, the low-lying muscle belly of the FHL is draped over the calcaneal insertion site, providing a rich vascular bed to the healing area. The remaining native Achilles tendon is then repaired over the FHL graft. Suture anchors may be utilized to reattach the medial and lateral slips of the Achilles back to their anatomic footprint on the calcaneus, effectively wrapping the native tendon around the FHL graft in a "pants-over-vest" or side-to-side fashion.
Hemostasis is meticulously achieved after tourniquet deflation. The wound is irrigated copiously. The paratenon, subcutaneous tissues, and skin are closed in a layered fashion. Given the high risk of wound complications in this region, a tension-free closure is paramount. Deep dermal sutures followed by a running nylon or staples for the skin are standard. The limb is then placed in a well-padded, short-leg splint in 15 to 20 degrees of plantarflexion to protect the repair.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, FHL augmentation for insertional Achilles tendinosis carries a distinct risk profile. The most feared complication is posterior wound breakdown and deep infection. The soft tissue envelope over the posterior heel is notoriously thin and possesses a tenuous blood supply, making it highly susceptible to necrosis, particularly in diabetic patients or smokers. Superficial dehiscence can often be managed with local wound care and oral antibiotics, but deep infection threatening the graft or hardware necessitates aggressive surgical débridement, hardware removal, and potentially complex soft tissue coverage (e.g., reverse sural artery flap).
Neurologic complications are also a significant concern. Sural nerve injury can occur during lateral dissection or retractor placement, leading to painful neuromas or lateral foot numbness. More devastating is the iatrogenic injury to the tibial nerve during the deep posterior compartment dissection for FHL harvest. Retraction injury or direct laceration can cause profound plantar numbness and intrinsic muscle paralysis. Salvage management for nerve injuries is notoriously difficult, often requiring neurolysis or nerve grafting if a complete transection is identified.
Functionally, patients may experience weakness in terminal hallux plantarflexion. While the flexor hallucis brevis preserves basic toe flexion, the loss of the FHL can lead to a slight decrease in push-off power during high-demand activities. Interestingly, clinical studies show that this deficit is rarely symptomatic in non-elite athletes, and the hallux often compensates remarkably well over time. Overlengthening of the Achilles-FHL complex due to improper intraoperative tensioning is a disastrous biomechanical complication, resulting in a persistent calcaneus gait and profound push-off weakness, which may require revision shortening osteotomies or complex reconstructive procedures to salvage.
| Complication | Estimated Incidence | Prevention Strategy | Salvage / Management |
|---|---|---|---|
| Wound Dehiscence / Necrosis | 5% - 10% | Full-thickness flaps; meticulous hemostasis; tension-free closure; smoking cessation. | Local wound care; VAC therapy; rotational flaps for deep defects. |
| Deep Infection | 1% - 3% | Strict sterility; prophylactic antibiotics; minimizing operative time. | Aggressive I&D; hardware removal; prolonged IV antibiotics. |
| Tibial Nerve Injury | < 1% | Direct visualization during FHL harvest; gentle retraction. | Gabapentinoids; surgical neurolysis; nerve grafting if transected. |
| Sural Nerve Injury | 2% - 5% | Midline incision; avoiding blind lateral dissection. | Neuroma excision; burying nerve stump in muscle. |
| Graft Overlengthening | 2% - 4% | Bilateral prep to match resting equinus tension intraoperatively. | Revision surgery with tendon shortening or advancement. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following FHL augmentation is a delicate balance between protecting the structural integrity of the bio-tenodesis repair and preventing restrictive arthrofibrosis. A rigidly adhered-to, phased protocol is essential for optimal functional outcomes. The initial phase focuses on wound healing and graft protection.
Phase I: Protection and Healing (Weeks 0-2)
Immediately postoperatively, the patient is placed in a strict non-weight-bearing short-leg splint in 20 degrees of plantarflexion. Elevation is critical to minimize edema, which directly threatens wound perfusion. At the two-week mark, sutures are removed provided the incision is fully coapted. The patient is then transitioned to a rigid CAM boot with heel wedges to maintain the plantarflexed position.
Phase II: Early Mobilization (Weeks 2-6)
During this phase, patients remain non-weight-bearing but begin active and active-assisted range of motion exercises out of the boot. Dorsiflexion is strictly limited to neutral to prevent excessive tension on the FHL graft and the Achilles repair. Gentle inversion and eversion, as well as toe flexion/extension, are encouraged to prevent adhesions of the FHL in its new tunnel.
Phase III: Progressive Weight-Bearing (Weeks 6-12)
At six weeks, assuming clinical evidence of healing, progressive partial weight-bearing is initiated in the CAM boot. Heel wedges are sequentially removed over a 4-week period to gradually stretch the gastroc-soleus-FHL complex. Physical therapy focuses on concentric strengthening, proprioception, and normalizing the gait pattern. Transition to a regular shoe with a small heel lift usually occurs around 10 to 12 weeks.
Phase IV: Return to Function (Months 3-6+)
The final phase involves aggressive eccentric strengthening, plyometrics, and sport-specific training. Patients are counseled that maximal medical improvement, particularly regarding calf circumference and explosive push-off strength, may take up to 12 to 18 months. The hypertrophic adaptation of the transferred FHL muscle belly continues well into the first postoperative year, providing increasing power and stability to the posterior ankle.
Summary of Landmark Literature and Clinical Guidelines
The evolution of FHL augmentation for Achilles pathology is grounded in robust clinical literature. Wapner et al. were pioneers in popularizing the FHL transfer for chronic Achilles ruptures and severe tendinosis, demonstrating excellent functional outcomes and highlighting the in-phase firing of the FHL as a critical biomechanical advantage. Their seminal work established the foundation for utilizing the FHL over other potential transfers like the flexor digitorum longus (FDL) or peroneus brevis.
Subsequent studies by Coull et al. evaluated the morbidity of FHL harvest, specifically addressing concerns regarding hallux function. Their research conclusively showed that while measurable weakness in terminal hallux flexion exists on dynamometry, it rarely translates into subjective clinical impairment or altered gait mechanics in the general population. This validated the safety profile of the procedure.
More recent biomechanical and MRI-based studies have focused on the fate of the transferred FHL muscle. Hunt and colleagues demonstrated significant hypertrophy of the FHL muscle belly following transfer, proving that the muscle not only acts as a passive vascular graft but actively adapts to its new role as a primary ankle plantarflexor. Current clinical guidelines strongly support single-incision FHL augmentation with interference screw fixation as the procedure of choice for insertional Achilles tendinosis requiring >50% tendon resection, offering a reliable, durable solution for a historically devastating clinical problem.
